Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
■ Cryotherapy
A minimally invasive surgical technique that utilizes subzero temperatures to destroy benign, premalignant, and malignant lesions
Compared to standard excisional surgery, cryosurgery requires less time to perform but healing time is often longer
Damage to surrounding structures is predictable and can be limited, with the preserved underlying stroma providing a structural framework for wound repair
The preferred and most commonly used cryogen is liquid nitrogen due to its low boiling point (−196°C) which allows for freezing of skin lesions at temperatures needed for their destruction
Cryosurgery often leads to an excellent cosmetic result, but in patients with darker skin phototypes, hypopigmentation due to destruction of melanocytes can occur
Reducing the temperature of the skin was initially done to provide anesthesia or lessen inflammation. With the advent of potent cryogens, selective destruction of tissue became possible. James M. Arnott is credited with first describing the use of controlled freezing in the 1800s to destroy tumors . Liquefied oxygen and carbon monoxide preceded the introduction of liquid nitrogen (LN) by Carl von Linde. By 1898, James Dewar had developed a flask that permitted storage and transportation of LN thereby rendering LN accessible to physicians.
In 1899, Archibald Campbell White reported on the successful treatment of multiple benign and malignant skin conditions with liquefied air applied via an adapted bottle; the latter could be considered the prototype of modern hand-held units. Herman V. Allington then described how LN-saturated cotton swabs could be used to treat various skin conditions. In 1961, the neurosurgeon Irving S. Cooper, together with Arnold Lee, developed the first cryosurgical probe. Increased use of LN for medical purposes followed the design of safe portable hand-held units.
Cryosurgery is defined as a surgical procedure that utilizes very low temperatures to destroy living tissue. The term cryotherapy should be reserved for when living tissue is exposed to cold in order to induce physiological changes but not tissue destruction. Cryobiology represents the study of the effects of low temperatures on living tissues.
Heat transfers from hot to cold , and the larger the temperature difference between a cryogen and its target tissue, the faster the heat transfer. The ideal cryogen has the lowest possible temperature and is capable of producing the parameters necessary to destroy both benign and malignant cells; for the latter, the tissue temperature needs to be below −50°C. The most commonly used cryogen is LN which boils at −196°C (77°K; −321°F). LN is relatively safe to handle, non-toxic, and commercially available. For the treatment of actinic keratosis (AKs) and cutaneous carcinomas, LN is the recommended cryogen whereas for benign lesions, cryogens with lower boiling points (e.g. nitrogen gas [−88°C], carbon dioxide gas [−78°C], other compressed organic gases [−55 to −75°C]) can also be employed.
The mechanisms of injury in cryosurgery can be attributed to both direct and indirect effects ( Table 138.1 ). Such events are the result of fast freezing followed by slow thawing, i.e. a freeze–thaw cycle. Repeated freeze–thaw cycles increase the destructive effects of cryosurgery. The material interposed between the cryogen and the lesion to be treated is also a determinant of the speed of freezing. Air is a poor conductor of cold such that the greater the distance between the skin lesion and the source of the cryogen, the lesser the freezing. Water is a better conductor than air (e.g. a presoaked verruca freezes faster) and ice is better than water as in the intra- and extracellular ice crystals present after the first freeze–thaw cycle. Finally, metal is an excellent conductor explaining the deep and fast freezing of tissue that results from frozen metal probes placed in direct contact with the skin .
| CRYOSURGERY – MECHANISMS OF INJURY | ||||
|---|---|---|---|---|
| Type of injury | Type of cell death | Phase | Location | Type of response |
| Direct injury | Necrosis: direct freezing of water leads to extracellular then intracellular ice crystal formation with edema and cellular lysis Temperature required: −20°C for most cells −50°C or lower for malignant cells |
Freeze period | Center of treated lesion | Inflammation: +++ Short-lived, nonspecific innate immune response |
| Vascular injury | Ischemia due to secondary damage of vascular endothelium | Thawing (~1 hour after freezing) |
Periphery of treated lesion and normal surrounding skin | Inflammation: +/++ |
| Apoptosis | Occurs at sublethal temperatures, e.g. approx. −20°C for malignant cells | 4–8 hours after thawing | Periphery of treated lesion | Inflammation: −/+ |
| Immunologic | Antigens released from damaged cells lead to immune response | Late event | Lymph nodes | Acquired immune response |
Different cells and tissues have different sensitivities to cold. The lethal temperature for melanocytes is around −4°C, while that for keratinocytes is −35°C. Keratinocyte cancer cells require around −50°C, sarcoma cells −60°C, and vascular endothelium −15°C to −40°C. Of note, cartilage and bone are quite resistant to freezing.
Cryoimmunology is the study of the innate and humoral immune responses to antigens derived from frozen skin lesions. Development of new immunologic assays has made it possible to measure such responses, which can be either immunostimulatory or immunosuppressive .
One major difference between conventional excisional surgery and cryosurgery is that the latter leaves the tumor “ in situ ”. At the center of the frozen lesion, there will be necrosis due to direct cellular damage (see Table 138.1 ), leading to release of tumor-specific antigens capable of stimulating an immune response. Once the innate immune system is activated, antigen-presenting cells (APCs) engulf tumor fragments and present processed antigens to T cells, initiating both T- and B-cell immune responses (see Ch. 4 ). At the periphery, the temperatures are higher and therefore sublethal, with the cells undergoing apoptosis rather than necrosis. This leads to a “silent death” associated with minimal inflammation (see Table 138.1 ) .
The response to cryosurgery can be enhanced by the addition of adjuvants . These include agents that augment ice injury (e.g. glycine), inducers of apoptosis (e.g. topical 5-fluorouracil) , and immunomodulators (e.g. topical imiquimod). Such combinations have been termed immunocryosurgery , with promising results having already been published . However, further studies are needed in order to better understand the underlying mechanisms of cryoimmunology, define the ideal parameters for tumor destruction, and determine the precise interactions between cryosurgery and potential adjuvants.
Cryosurgical techniques can be subdivided into several types ( Table 138.2 ):
Open ( spray , open-spray ; Fig. 138.1A ). This is the most frequently used technique. The instrument consists of a metal container with spraying tips or openings through which the cryogen is released. This technique is suitable for lesions that are flat or elevated as well as benign or malignant; the key factor is achieving the correct freezing temperature. Larger openings deliver more LN and freeze tissue faster so they are ideal for larger, bulky lesions. Smaller openings freeze tissue slower and allow for a more precise freezing. Therefore, they are ideal for small benign lesions, such as lentigines or seborrheic keratoses.
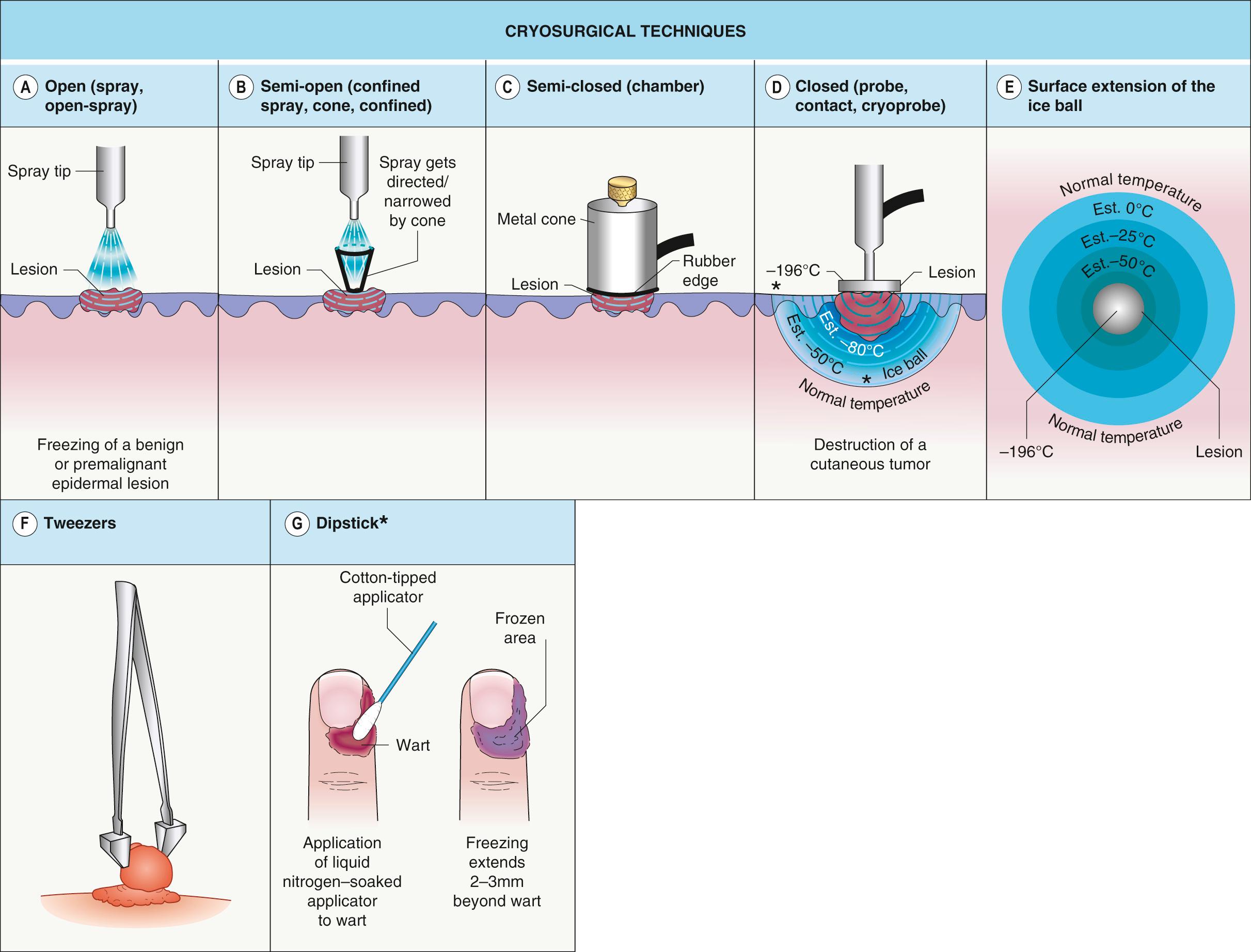
Semi-open ( confined spray , cone , confined ; Fig. 138.1B ). This refers to the use of a polycarbonate plate with holes, neoprene cones, or otoscope specula to restrict sprayed LN to a limited area. All of these items are composed of non-conducting materials. Freezing is faster than with the open technique as the LN is confined to the lesion. Splattering of LN is avoided and normal surrounding tissue is spared.
Semi-closed ( chamber ; Fig. 138.1C ). One end of a metal cone is attached to the cryogen unit while the rubber-protected distal end is held firmly against the skin. This system generates potent freezing and should be reserved for skin cancers and cutaneous metastases.
Closed ( probe , contact , cryoprobe ; Fig. 138.1D,E ). The cryogen is delivered via a closed system, i.e. from the unit through metal probes in contact with the skin; the remaining cryogen exits by way of a rubber releasing hose. Probes come in different shapes and diameters and to ensure homogeneous freezing, they need to be applied to flat tumor surfaces. In addition, when treating hemangiomas, the probe should be applied with pressure in order to “press out” the blood and lower the final temperature. For delicate procedures, such as freezing cysts near the eye, ultrasound gel or water can be placed on the skin under the probe.
Tweezers . ( Fig. 138.1F ) A previously frozen Teflon® forceps is used to grasp pedunculated lesions. The freezing front is then allowed to advance just to the skin surface. This is an ideal technique for filiform lesions, with sparing of normal surrounding skin as well as minimization of post-treatment hypo- or hyperpigmentation.
Intralesional . While originally developed for keloids, this technique consists of passing a thick, curved needle through the tumor. One end of the needle is attached to the cryogen unit (screwed on or via a Luer lock), while the other end that has exited the skin allows for LN release. It can be used for large nodular tumors and has an advantage of the freezing originating from the center of the mass.
Dipstic k ( Fig. 138.1G ). A LN-saturated, cotton-tipped applicator is placed directly onto the lesion. This technique is still used by some physicians for verrucae and solar lentigines.
| CRYOSURGICAL TECHNIQUES | ||||||
|---|---|---|---|---|---|---|
| Technique | Common uses | Advantages | Disadvantages | |||
| Type | Synonym | B | PM | M | ||
| Open | Spray Open-spray |
√ | √ | √ | Any size or surface | For small lesions, care should be taken regarding normal surrounding skin Not appropriate for pedunculated lesions |
| Semi-open | Confined spray Cone Confined |
√ | Relative sparing of normal surrounding skin | Appropriate size of cone required | ||
| Semi-closed | Chamber | √ | Deep and fast freezing | Indicated for cutaneous malignancies Appropriate size of chamber required |
||
| Closed | Contact Probe Cryoprobe |
√ | √ | Deep and fast freezing | Appropriate size and shape of probe required | |
| Tweezers | Cryotweezers | √ | Spares normal surrounding skin | Limited to superficial lesions | ||
| Intralesional | − | √ | √ | Freezing occurs from the center of the lesion | Invasive | |
| Dipstick | Cotton-tipped applicator | √ | May be less frightening for children | Variability in amount of LN within cotton tip Loss of temperature control Dripping of LN may lead to unintended sites of freezing |
||
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here