Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The incidence of congenital heart disease (CHD) is estimated at approximately 8 per 1000 live births, with up to 25–30% of CHD deemed critical congenital heart disease (CCHD). CCHD includes lesions that necessitate early interventions to avoid significant morbidity and mortality and require multidisciplinary subspecialty care from pediatric cardiologists, cardiothoracic surgeons, neonatologists, anesthesiologists, and pediatric cardiac intensivists. Detection of CCHD by prenatal ultrasound (fetal anomaly screening) or by newborn pulse oximetry screening can occur prior to decompensation resulting in decreased morbidity and mortality in this vulnerable population. Although there have been remarkable advances in ultrasound technology and an increasing prenatal detection rate, a significant proportion of cases of CCHD are diagnosed postnatally. In the United States prenatal detection of CCHD has gradually increased from 26% in 2006 to 43% in 2012, with improvements in ultrasound technology and improved screening guidelines that emphasized the outflow tract views. As such, CCHD may be unknown at birth, and neonatologists or non-cardiologist acute care physicians may be the “first call” for a decompensating infant with CCHD. Congenital heart disease remains the leading cause of infant mortality at approximately 40% of all deaths resulting from birth defects; thus familiarity with signs and symptoms of CCHD is paramount in the care of a decompensating neonate. This chapter focuses on understanding the presenting pathophysiology and outlines the initial management by neonatal providers for a patient with CCHD.
Critical congenital heart disease traditionally refers to lesions deemed necessitating surgical or catheter-based procedures in the first year of life, with the focus of this chapter on those requiring life-sustaining intervention within the first postnatal month. Neonates with CCHD can manifest with varying degrees of severity due to several different pathophysiologic states: (1) increasing cyanosis due to lack of mixing of systemic and pulmonary venous return or inadequate pulmonary arterial blood flow; (2) signs of hypoperfusion resulting from inadequate systemic blood flow due to obstruction of systemic blood flow or decreased systolic function; and (3) respiratory distress due to excessive pulmonary flow or pulmonary venous obstruction. Neonates may have features of more than one presentation type depending on the lesion type and postnatal age. Timing of presentation will depend on type of CHDs ([i] ductal-dependent lesions and timing of PDA closure, e.g., hypoplastic left heart syndrome [HLHS], [ii] patent foramen ovale [PFO]/central mixing dependent, e.g., transposition of the great arteries [TGA], and [iii] obstructed pulmonary venous return), the rate of drop in pulmonary vascular resistance (PVR), and associated heart lesions (see Table 30.1 ). In a study assessing time to significant hemodynamic compromise in the setting of undiagnosed CCHD, defined as severe metabolic acidosis, cardiac arrest, lab evidence of renal or hepatic insult, or seizure, most (83%) occurred after 12 postnatal hours. Aortic arch obstructive lesions comprised approximately 90% of cases with significant hemodynamic compromise that were potentially preventable. In addition, early diagnosis of CCHD (antenatal or pre-discharge) had a higher survival advantage versus late diagnosis following discharge from hospital (16% vs. 27%).
| Soon After Birth (Severe Cyanosis With Inadequate Mixing or Respiratory Compromise * ) | Within First Days to 1–2 Weeks (PDA Dependent Perfusion) | Within First Month (PVR Decreases, CHF, Cyanosis, Hypoperfusion) |
|---|---|---|
|
|
|
* Corresponds to respiratory compromise with TOF with absent pulmonary valve in column. CAVC , complete atrioventricular canal CCHD , critical congenital heart disease; CHF , congestive heart failure; HLHS , hypoplastic left heart syndrome; PDA , patent ductus arteriosus; PS , pulmonary stenosis; PVR , pulmonary vascular resistance; TAPVC , total anomalous pulmonary venous connection; TGA , transposition of the great arteries; TOF , tetralogy of Fallot; VSD , ventricular septal defect.
Within CCHD, five categories of lesions prevail, with recognition that many lesions can fall into more than one category: (1) ductal-dependent pulmonary blood flow, (2) ductal-dependent systemic blood flow, (3) pulmonary venous obstruction, (4) mixing lesions, and (5) transposition of the great arteries (see Table 30.2 ). CCHD lesion types with inadequate aortic or pulmonary outflow are termed “ductal dependent”, given the need for flow through the ductus arteriosus to provide or augment either pulmonary or systemic blood flow.
| DD Pulmonary Flow | DD Systemic Flow | Obstructive Lesions | Mixing Lesions | Parallel Circulation |
|---|---|---|---|---|
|
|
|
|
* Pulmonary flow may be supplied by multiple aortopulmonary collaterals (MAPCAs) and not be DD.
+ Most commonly normally related great vessels, with DD pulmonary flow; can be transposed great vessels and reliant on ductus to augment systemic flow.
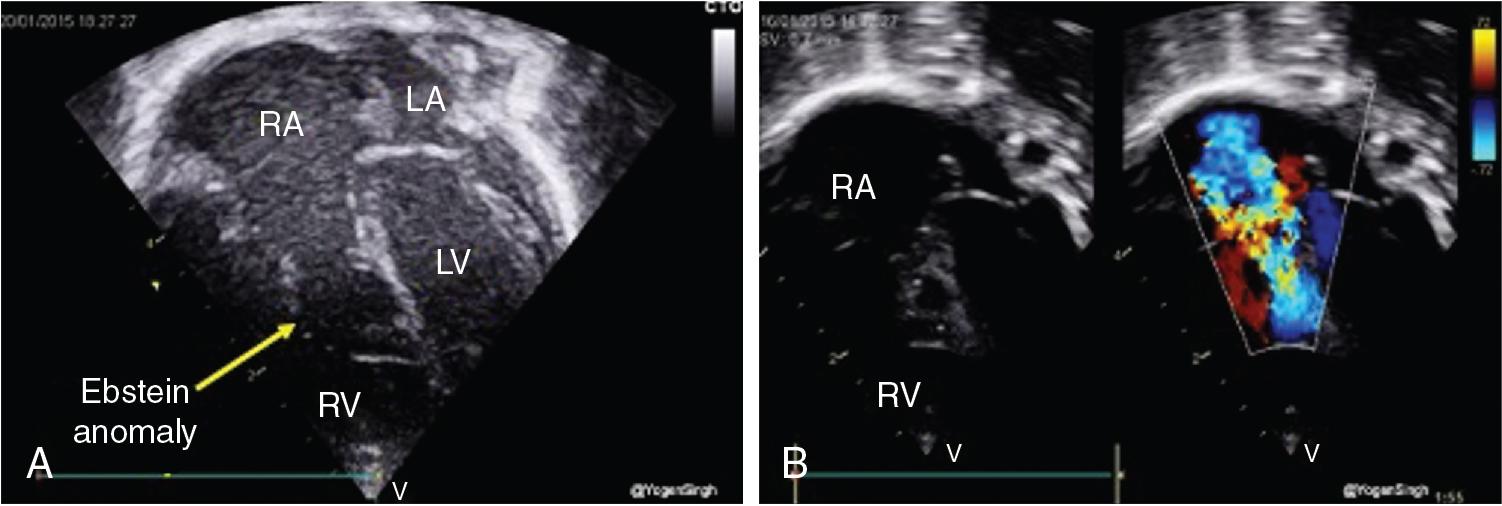
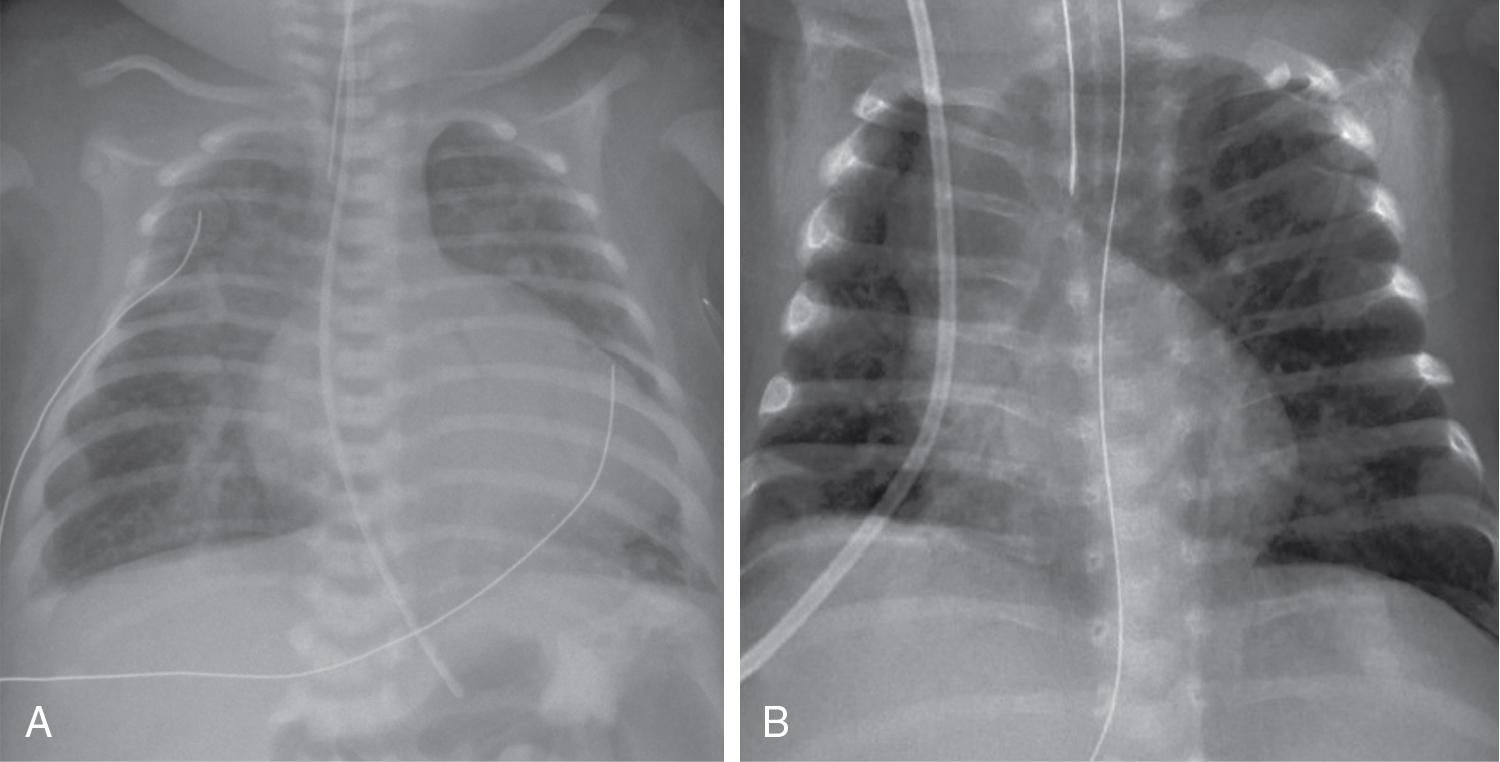
In patients with ductal-dependent pulmonary circulation, prostaglandin E1/E2 is required to augment pulmonary blood flow. This obstruction can occur anywhere along the path through the right side of the heart, from the tricuspid valve (e.g., tricuspid atresia with normally related great vessels) to the right ventricular outflow tract (RVOT, e.g., pulmonary atresia with intact septum, and tetralogy of Fallot with severe pulmonary stenosis or Ebstein’s anomaly with pulmonary obstruction) (see Figure 30.1 ). Flow through the ductus arteriosus in patients with ductal-depending pulmonary circulation will traverse from the aorta to the lungs, with increasing pulmonary flow (Qp) over time as the PVR decreases. In patients with ductal-dependent pulmonary circulation, decreased saturations could be a result of decreased Qp if pulmonary flow decreases, such as in the case of ductal closure or restriction, or due to decreased pulmonary venous saturation related to pulmonary edema in cases with increased Qp as PVR decreases. Chest radiograph can help differentiate between these conditions with oligemic lung fields as pulmonary flow decreases and plethoric lung fields in patients with increased Qp (see Figure 30.2 ).
Lesions with ductal-dependent systemic flow include left-sided obstructive lesions that limit systemic blood flow without the presence of a patent ductus arteriosus (PDA), such as interrupted aortic arch, severe coarctation, and hypoplastic left heart syndrome (HLHS) (see Table 30.1 ). As the ductus arteriosus (DA) restricts, patients may present in cardiogenic shock with hypoperfusion and acidosis and in extreme cases with death when the PDA closes. As the DA starts to constrict, pulses and perfusion can diminish, and if it continues to close completely, the patient may present in extremis with shock. Even once the DA is reopened successfully, the cardiac function may need time to recover as a result of the acidosis and the increased afterload on the left ventricle prior to surgical intervention. Early intervention may be necessary in patients with ductal-dependent systemic circulation. While most cases of HLHS can be stabilized by maintaining DA patency to provide adequate systemic flow, approximately 6–20% of HLHS cases have an intact or restrictive atrial septum, with pulmonary venous obstruction. These infants present with early decompensation after birth, similar to d-TGA with restrictive atrial septum/intact atrial septum (RAS/IAS) though related to different pathophysiologic mechanism related to lack of egress from the left atrium and pulmonary vasculopathy. Infants with a restrictive or intact atrial septum present a time-critical emergency as they require immediate intervention for the creation of mixing at the atrial level despite successfully keeping the DA patent. Catheter-based or surgical intervention on the atrial septum is imperative to ensure adequate atrial mixing; nevertheless, even with intervention, mortality is still high.
Catheter-based fetal septal interventions have been attempted in these infants, and while a recent analysis demonstrated procedural successes, this has not yet translated into improved survival when compared with a cohort of HLHS with patent atrial septum.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here