Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
In the open eye, contact lenses are primarily in physical apposition with the cornea. Well-fitted rigid lenses generally reside almost exclusively on the cornea and only occasionally impinge on the limbus. The situation is different with soft lenses. If the surface area of the cornea is taken to be 132 mm , and the surface area of a soft lens – with typical dimensions of 14.5 mm diameter and 8.7 mm back optic zone radius – is 213 mm , then 38% of the surface area of a soft lens lies in apposition with the bulbar conjunctiva. It is therefore apparent that changes in the conjunctiva may be seen as a result of the physical presence of soft lenses.
Contact lenses can also affect the conjunctiva via mechanisms that do not involve direct physical contact. Thick rigid lenses can ‘bridge’ the upper lid away from the conjunctiva, preventing blink-activated wetting and causing drying of the region of corneal and conjunctival tissue adjacent to the lens. Contact lens solutions can cause toxic or immunological reactions of the conjunctiva. Lens-induced changes in the volume and composition of the pre-ocular tear film may also indirectly lead to changes in the conjunctiva.
For the reasons outlined, careful inspection of the bulbar conjunctiva must be considered to constitute an essential part of the external ocular examination of contact lens wearers. An important technique that can be employed to reveal conjunctival tissue damage is to use vital dyes, so this chapter will review the various conjunctival staining techniques that have been proposed.
‘Conjunctival staining’ is used here as a catch-all phrase that encompasses a wide range of normal and abnormal appearances that are revealed after instillation of a vital dye – typically fluorescein – into the eye.
Fluorescein sodium (more commonly referred to as ‘fluorescein’) is generally adopted as the stain of first choice in ophthalmic diagnosis. It is assumed that fluorescein provides information about the surface quality of the conjunctiva by pooling in natural creases, folds and ridges in the conjunctiva and entering inter-epithelial spaces where an abrasion has occurred. To optimise visualisation of conjunctival staining, a cobalt blue ‘excitation’ filter, incorporated into the illumination system of the slit lamp biomicroscope, is required to limit illumination to wavelengths of light that maximally absorb fluorescein. When viewing the conjunctiva illuminated in this way, the sclera scatters the incident blue light and desaturates the fluorescent light. (This problem is not encountered when viewing fluorescent staining over a transparent cornea). A yellow ‘barrier’ filter ( Fig. 11.1 ) interposed within the observation system is therefore essential to limit light transmission back through the eyepieces and maximise contrast between the stained and unstained areas of conjunctiva.

Even with the use of excitation and barrier filters, the blue incident light excites a natural greenish fluorescence in the conjunctiva and sclera, which causes a general background glow that may diminish the contrast of any additional fluorescence present. A similar problem that is encountered is that the contrast of subtle corneal staining can be diminished as a result of the background fluorescent glow emanating from the crystalline lens. An added difficulty with respect to the observation of fluorescein staining of the conjunctiva is that fluorescein is slightly lipid soluble and stains the entire conjunctiva to some extent, further reducing contrast. In view of these limitations of fluorescein, researchers have sought to investigate the utility of other agents for staining the conjunctiva. Sulphorhodamine B, which has an orange fluorescence that can be separated from the green natural fluorescence of the ocular tissues, gives a greater contrast compared with fluorescein.
Lissamine green is a vital stain with dyeing quality similar to that of rose bengal, but causes less discomfort. It stains dead or degenerated epithelial cells green and is used to facilitate the diagnosis of dry eye conditions. It has a molecular weight of 577. Guillon and Maissa demonstrated that lissamine green shows greater specificity for symptomatic subjects, particularly in contact lens wearers. They recommended that the examination of patients complaining of dry eyes – in particular, those wearing contact lenses – should include observation of the conjunctiva stained with lissamine green. Within- and between-grader reliability of lissamine green staining seems to be at least substantial to moderate.
Norn reported that rose bengal stains degenerated epithelial cells and mucus; however, Tseng subsequently disputed this long-held belief by demonstrating that rose bengal also rapidly stains healthy epithelial cells when there is insufficient surface mucus. Contrast between rose bengal–stained and unstained areas is usually poor, although the appearance can be enhanced by viewing the stained region in ‘red-free’ light by interposing a green filter in the illumination system. Patients find this staining agent to be uncomfortable.
Lissamine green has similar properties to rose bengal in that it stains degenerated and dead epithelial cells and mucus. Manning et al. reported that lissamine green is better tolerated by patients compared with rose bengal and is equally as effective as rose bengal in evaluating the ocular surface. However, Emran and Sommer believed that lissamine green is an inadequately sensitive test for practical use.
Numerous other stains, including Congo red, neutral red, trypan blue, bromothymol blue and fluorexon, have been investigated. Foster cited 34 different chemical substances that have been used for ocular surface staining. In addition, the possible benefits of using various mixtures of staining agents, such as fluorescein–rose bengal and tetrazolium–alcian blue, have been examined. The key characteristics of various staining agents that have been applied clinically are outlined in Table 11.1 .
| Stain | Features revealed/highlighted |
|---|---|
| Fluorescein sodium | ‘Colours’ the tear film. Fills gaps, thus revealing ‘missing’ cells. Enters and lightly stains living epithelial cells. Enters and reversibly stains hydrogel lenses. |
| Fluorexon (Calcein) | Essentially high-molecular-weight fluorescein. ‘Colours’ the tear film. Fills gaps, thus revealing ‘missing’ cells. Enters and lightly stains living epithelial cells. Does not fluoresce as brightly as fluorescein Cannot enter and stain hydrogel lenses. |
| Sulphorhodamine B | Essentially fluorescein with different fluorescent characteristics. ‘Colours’ the tear film. Fills gaps, thus revealing ‘missing’ cells. Enters and lightly stains living epithelial cells. Does not fluoresce as brightly as fluorescein. |
| Lissamine green | Stains degenerated and dead cells. Stains mucus. Similar to rose bengal but better tolerated. |
| Rose bengal | Stains dead cells. Stains mucus (e.g. mucus threads). Reveals absence of surface mucus by staining normal surface tissue unprotected by mucus |
| Neutral red | Stains epithelial inclusion bodies and granulocytes. Essentially a less efficient version of rose bengal. |
| Congo red | Stains dead and degenerate cells. Stains mucus. Similar staining effect as rose bengal and lissamine green, but less effective and less intense. |
| Alcian blue | Only stains mucus. Acts as a counter-stain to rose bengal to help differentiate between dead cells and mucus. |
| Trypan blue | Stains dead cells. Stains mucus (e.g. mucus threads). |
| Bromothymol blue | Stains dead cells. Stains degenerate cells. Stains mucus (e.g. mucus threads). |
| Methylene blue | Stains nerve tissue. Clearly outlines areas of corneal ulceration. |
| Tetrazolium | Stains degenerate cells, but not live or dead cells. The red stain colouration only appears about 4 minutes after the stain has entered the cell. |
| Iodonitrotetrazolium | Stains degenerate cells, but not live or dead cells. The red stain colouration only appears about 4 minutes after the stain has entered the cell. |
Although the usefulness of some of the alternative staining agents described has been demonstrated, the general utility, safety, broad acceptance and ready availability of fluorescein mean that it is the staining agent of first choice for investigating ocular surface compromise. Rose bengal may be used as a supplementary stain to check for degenerated epithelial cells and mucus, and lissamine green may be especially useful in assessing contact lens-related dry eye. Thus, throughout the remainder of this textbook, reference will only be made to the use of fluorescein, rose bengal and lissamine green, which can be considered to be the primary ocular surface staining agents.
Fluorescein can be administered in liquid form from an eye dropper, but this method is seldom used today because of the propensity of Pseudomonas aeruginosa to flourish in liquid fluorescein sodium and hence the high risk for Pseudomonas corneal infection. This problem can be overcome by using sterile single-dose units; however, the standard method of introducing fluorescein dye into the eye is via sterile, single-use fluorescein-impregnated paper strips ( Fig. 11.2 ).

The preferred technique is to introduce a small drop of sterile un-preserved saline onto the fluorescein-impregnated tip of the paper strip and then to lightly apply the end of the strip to the surface of the eye. Although the strip should not be applied directly onto the cornea, it can be applied to any part of the bulbar or palpebral conjunctiva. If conjunctival staining is to be assessed, the strip should be applied to a region of the conjunctiva of ‘least interest’, such as the lower palpebral conjunctiva. This is because a very high fluorescein concentration will usually be deposited at the point of contact of the fluorescein strip (irrespective of how delicately the fluorescein is applied), leaving an intense discrete region of iatrogenic ‘pseudo-staining’ ( Fig. 11.3 ).
For instillation of fluorescein into the lower lid, the patient should be asked to look up, the lower lid should be gently retracted by a small amount, and the moistened fluorescein strip should be allowed to lightly touch the inner surface (palpebral conjunctiva) of the lower lid. Physical touch may not be required if a sufficient amount of fluorescein in the form of a ‘hanging drop’ remains suspended from the moistened strip.
The amount of fluorescein that enters the eye using the described technique will usually provide the correct balance between there being enough fluorescein to effect useful staining but insufficient to ‘flood’ the eye, which, in the latter case, could reduce contrast and possibly induce false staining. Abdul-Fattah et al. have demonstrated that the amount of fluorescein entering the eye can be controlled by using paper strips in which the fluorescein-impregnated portion varies in area, whereby a smaller area will result in less fluorescein entering the eye. Reduced amounts of fluorescein are usually only required for assessing the tear film in suspected dry eye patients. Korb et al. reported that a specially modified fluorescein strip with a substantially reduced area of fluorescein impregnation (the ‘dry eye test’) provided a significant reduction in sensation upon application, improved single measurement reliability and enhanced measurement precision, compared with a conventional fluorescent strip.
When fluorescein is instilled into a normal eye, patterns of small, thin, curved lines of staining can be observed on the bulbar conjunctiva ( Fig. 11.4 ). The curved lines of staining in regions of conjunctiva close to the cornea are generally concentric with the limbus, whereas they tend to run parallel to the lid margins in regions of the bulbar conjunctiva further away from the cornea. Although the term ‘staining’ is used to describe these patterns, this is not true staining in that there is no tissue disruption. This appearance is best described as ‘normal furrow staining’. What is being observed is pooling of fluorescein in the normal concertina folding or furrows of the conjunctiva. Because the conjunctiva is essentially fixed to the globe at the region of the limbus and to the eyelids, this normal pattern of ‘staining’ will therefore vary at different gaze directions, as the conjunctiva stretches and folds up in a concertina-like manner to accommodate the position of the globe.
Schwallie et al. have characterised the day-to-day variability in normal conjunctival staining by instilling fluorescein into the eyes of 16 subjects who were monitored for 1 week. All eyes showed some degree of normal conjunctival staining over the week of observation. In total, normal conjunctival staining was noted in 71% of ocular evaluations. Overall, staining ranged from grade 0 to 3, with a mean of 0.5. There was a significant variation in the average grade of staining between subjects. Furthermore, there was a significant variation in the range of grading observed between subjects; the day-to-day range of conjunctival staining varied by 0.5 grading scale units in six subjects, by 1.0 grading scale units in six subjects and by 1.5 grading scale units in four subjects.
A high correlation of staining grade was observed between the two eyes. This finding has important clinical implications in the evaluation of uniocular ‘abnormal’ conjunctival staining, in that the contralateral (unaffected) eye can be used for comparison with the appearance of the eye that is suspected to have suffered conjunctival compromise.
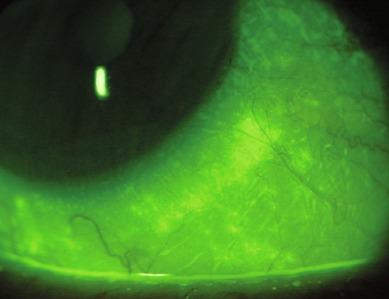
A diffuse punctate region of staining (or ‘stipple staining’) may be observed near the limbus, corresponding to the region of conjunctiva covered by a soft lens ( Fig. 11.5 ). This takes the form of stippling, which refers to a region of numerous, fine, punctate spots. In some areas, these punctate spots tend to coalesce. Confluent areas of staining representing total coalescence, can sometimes be observed in the cornea. Stipple staining is usually only observed in soft lens wearers.
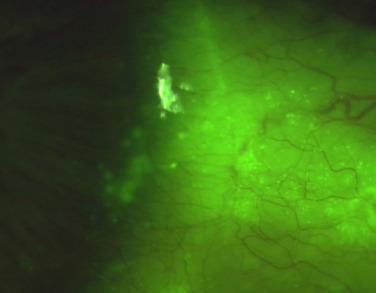
Lakkis and Brennan assessed the extent of conjunctival staining in 48 hydrogel lens wearers and 50 non-lens-wearing control subjects. Ignoring the normal furrow staining described previously, these authors observed that only 12% of controls displayed conjunctival staining greater than grade 1.0, whereas 62% of lens wearers had staining above this level. The mean difference in the grade of staining varied between 0.12 and 0.15 for overall conjunctival staining, and contact lens wearers displayed most staining temporally. Neither Lakkis and Brennan nor any of the other authors referred to later specifically described the type of staining they were observing. It is therefore presumed that these authors were observing stipple staining, as defined previously.
Brautaset et al. retrospectively examined the records of 338 adapted hydrogel contact lens wearers and found conjunctival staining to be present in 32.5% of the subjects. Brennan et al. reported an increase in conjunctival staining in patients wearing silicone hydrogel lenses, from baseline grades of 0.39 ± 0.53 and 0.44 ± 0.61 with Acuvue Advance (Johnson & Johnson Vision Care) and Focus Night & Day (CIBA Vision) silicone hydrogel lenses, respectively, to 2-week results of 0.60 ± 0.55 and 0.52 ± 0.69.
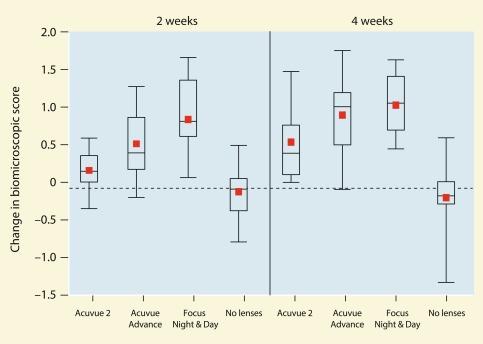
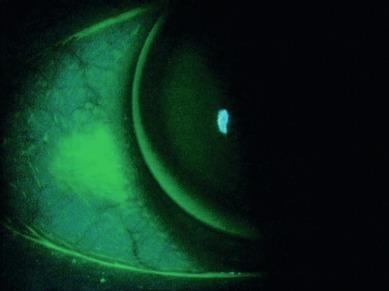
Maldonado-Codina et al. investigated the extent of conjunctival staining 2 weeks and 4 weeks after fitting 43 neophytes with one hydrogel lens (Acuvue 2 (Johnson & Johnson Vision Care)) and two silicone hydrogel lenses (Acuvue Advance and Focus Night & Day). Nineteen non-lens-wearing control subjects were also assessed. More conjunctival staining was seen with the silicone hydrogel lenses compared with the conventional hydrogel lenses ( Fig. 11.6 ).
The effect of contact lens preservative system on the ocular surface was investigated by Young et al. They observed that nasal and temporal conjunctival staining was significantly higher after 2 years' use of solutions containing polyhexamethylene biguanide compared with solutions containing polyquaternium-1 (p < 0.05).
Zhao et al. reported that in a cohort of patients wearing four different silicone hydrogel lenses and using four different multi-purpose solutions, the amount of protein extracted from lenses was inversely correlated with the extent of conjunctival staining (R = − 0.23; p < 0.01); that is, protein deposition may be protective against conjunctival staining.
Lakkis and Brennan reported that temporal conjunctival staining in soft lens wearers was related to the symptom of dryness and superior conjunctival staining was related more to the symptom of itchiness.
Nichols and Sinnott observed that among 413 contact lens wearers, increased fluorescein corneal staining was associated with increased lissamine green conjunctival staining (p = 0.002).
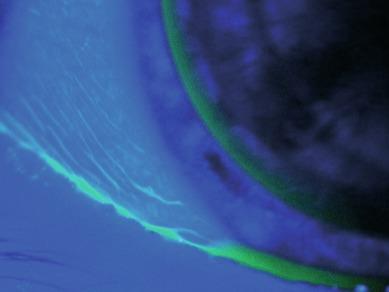
An arcuate band of staining corresponding to the lens edge may be seen 2 to 3 mm from the limbus. This may form as a continuous band or as a broken line and can vary from being just visible to being an intense ring of fluorescence up to about 0.5 mm wide. Arcuate conjunctival staining is usually observed in soft lens wearers ( Fig. 11.7 ) but is sometimes seen after overnight wear of rigid lenses that have become decentred during sleep ( Fig. 11.8 ). Covey et al. reported that high levels of conjunctival edge staining occur with silicone hydrogel lenses; specifically, they reported a mean grade of conjunctival staining of 2.4 ± 0.6 in silicone hydrogel lens wearers versus 0.9 ± 0.7 in non-lens-wearing controls (the latter being ‘normal furrow staining’).
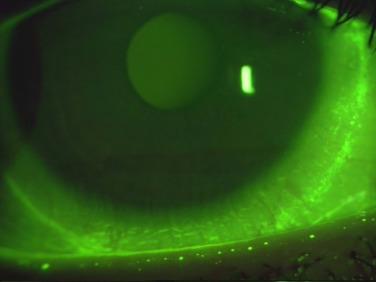
Maissa et al. assessed arcuate conjunctival staining (using lissamine green) caused by silicone hydrogel contact lenses with different edge designs: Acuvue Oasys (knife edge design), Air Optix, Biofinity (chisel edge–rounded edge combination) and PureVision (rounded edge design), and one hydrogel contact lens, Acuvue 2 (knife edge design). The experiment was conducted on a cohort population of 27 established soft contact lens wearers, with each wearing a different contact lens type, in a random order, for a period of 10 ± 2 days.
The results obtained showed that contact lens edge design was the primary factor in controlling circumlimbal staining for silicone hydrogel lenses: a rounded edge away from the ocular surface produced the lowest staining (average 0.19%) and a knife edge in close apposition to the ocular surface produced the highest staining (average 1.34%). Contact lens material rigidity impacted arcuate staining. An inverse association between arcuate staining and contact lens comfort was demonstrated, with the rounded edge design producing the lowest comfort (72 of 100) and the knife edge design producing the highest (87 of 100). The authors concluded that soft contact lens wear induces arcuate staining, with the level of staining being influenced by the contact lens edge design. However, they observed that high levels of circumlimbal staining were not associated with decreased comfort.
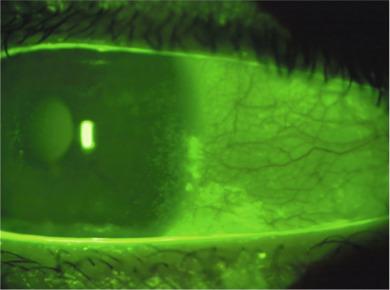
This is a common form of ocular surface staining observed in association with rigid contact lens wear ( Fig. 11.9 ). Although it is often described in the literature as 3 & 9 o’clock corneal staining, the staining patterns observed usually span the limbus at the temporal and nasal aspects of the cornea and also affect the adjacent conjunctiva.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here