Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Congenital muscular torticollis (CMT) must be distinguished from acquired torticollis and underlying structural torticollis (from cervical vertebral anomalies).
It is most often caused by a congenital asymmetry in the length and/or strength of the sternocleidomastoid (SCM) muscles.
CMT may be caused by early fetal head descent, an abnormal fetal position, venous occlusion, and/or trauma to the muscle during difficult deliveries.
Other postnatal causes of torticollis include muscle spasms, trauma, infection, inflammation, neoplasm, dystonic syndromes, drug reactions, and ocular malalignment.
Most cases of CMT are treated conservatively using active and passive neck-stretching exercises with repositioning.
Untreated CMT can result in progressive facial asymmetry and positional plagiocephaly.
The term torticollis is derived from the Latin terms torus , meaning “twisted,” and collum , meaning “neck,” and it refers to the posture that results from the head being twisted and turned to one side.
Torticollis is most often caused by a congenital asymmetry in the length and/or strength of the SCM muscles on each side of the neck, often characterized by the shortening or stiffness of the SCM muscle resulting in ipsilateral lateral flexion of the head with contralateral rotation. CMT must be distinguished from acquired torticollis from structural torticollis resulting from cervical vertebral anomalies ( Fig. 24.1 ). Other postnatal causes of torticollis include muscle spasms, trauma (fracture, dislocation, spinal hematoma, atlantoaxial rotary subluxation), infection, inflammation, neoplasm, dystonic syndromes, drug reactions, and ocular malalignment. Prenatally acquired congenital muscular torticollis (CMT) is the most common type of torticollis, and when it remains partly or completely untreated, it results in progressive facial asymmetry and positional plagiocephaly (see Chapter 25 ), which may be a presenting feature for the recognition of CMT. The incidence of CMT ranges from 3.9% to 16% of newborns. CMT is thought to be the third congenital orthopedic anomaly requiring intervention/referral, more frequently following congenital hip dysplasia and calcaneovalgus feet. Cheng and colleagues reported a 1.3% incidence of torticollis among Chinese children and studied the outcomes in 1086 children with CMT who were classified into three groups according to severity (from most severe to least severe): 42.7% with a sternomastoid tumor, 30.6% with SCM tightness but no tumor, and 22.1% with postural torticollis who presented with CMT but no demonstrable tightness or tumor.
CMT may occur as a consequence of early fetal head descent or an abnormal fetal position with the head tilted and turned, thereby resulting in muscle imbalance, with occasional associated deformations involving the back, hips, and feet (the intrauterine theory). More severe shortening and fibrosis of the muscle may be caused by venous occlusion from persistent lateral flexion and rotation of the neck (the vascular theory) and/or trauma to the muscle during difficult deliveries (the birth trauma theory). Because there is no difference in the severity of CMT when infants delivered vaginally are compared with those delivered by cesarean section, CMT appears to arise before birth. Among 178 subjects with CMT, there was no significant difference in the rate of surgical release according to the method of childbirth, and for 132 patients less than 6 months of age there was also no significant difference in the rate of stretching exercises. Many clinical studies have demonstrated that infants with CMT are more commonly first born and have an increased incidence of other deformations, such as metatarsus adductus, developmental dysplasia of the hip (DDH), and talipes equinovarus.
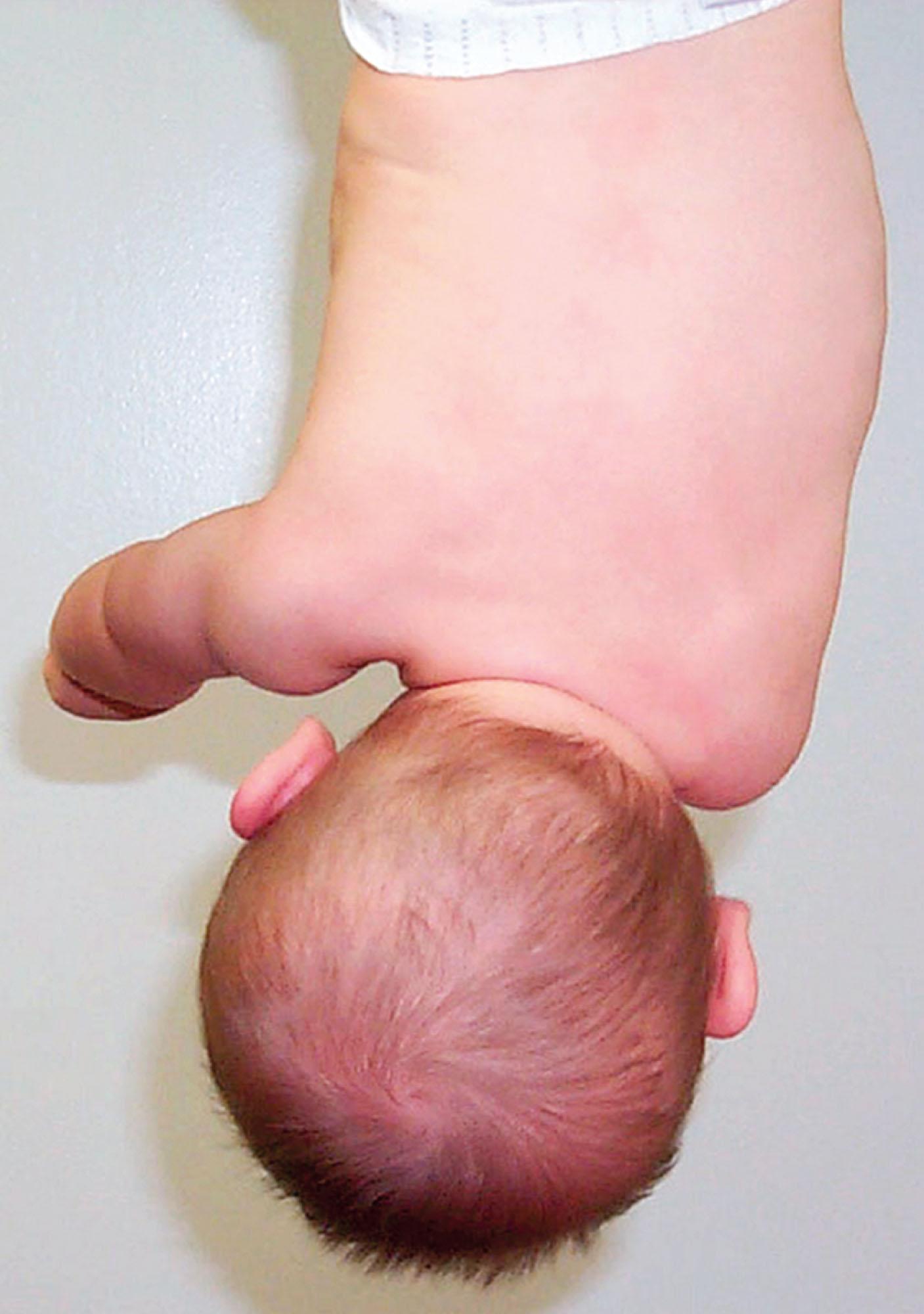
Histology of affected muscles has demonstrated replacement of muscle by dense fibrous tissue, and when magnetic resonance imaging (MRI) was used to study the affected muscle, altered signal characteristics suggested that severe CMT might be caused by sequelae of an intrauterine or perinatal sternomastoid compartment syndrome. In cadaver studies, flexion with lateral bending and rotation of the head and neck caused the ipsilateral sternomastoid muscle to kink on itself, resulting in ischemia and edema within the sternomastoid muscle compartment. When birth histories were compared with MRI findings, the side of birth presentation correlated with the side of the torticollis. The intrauterine theory concerning the etiology of CMT (which does not exclude the compartment theory) is based on epidemiologic data and emphasizes the concept that CMT is one of a group of associated deformations arising from malposition of the fetus during the third trimester. This theory relies on the association between unilateral hip dislocation, CMT, infantile idiopathic scoliosis, and plagiocephaly. (See Chapter 20 for a discussion of scoliosis and the molded baby syndrome.) It is suggested that the ipsilateral coexistence of a t urned head, a dduction contracture of the hip, and truncal c urvature (TAC syndrome) indicates a specific intrauterine asymmetric posture of the fetus (see Figs. 20.1 and 24.2 ). Based on a study of 108 infants born with the clinical triad of TAC syndrome, this condition occurs in 5.8 per 1000 live births with a preponderance of females, first born children, winter births, low birth weights, children born to mothers with a small abdominal circumference, and first born children of mothers of advanced maternal age. The frequency of change of position in utero was significantly reduced from that in normal fetuses, and the side of fetal position correlated with the side of the TAC syndrome. When the left side of the fetus in vertex presentation was against the mother’s spine, with the back on the left, the hip adduction contracture and torticollis were also on the left; the reverse was true when the back of the fetus was on the mother’s right side.
Interstitial fibrosis of the muscle is sometimes palpable as a fusiform fibrous mass or “tumor” that becomes evident within the first 3 weeks after birth and reaches maximum size by 1 month of age. This mass is composed of myoblasts, fibroblasts, myofibroblasts, and mesenchyme-like cells that usually mature and differentiate, resulting in disappearance of the mass over the first 4–8 months of life with variable sequelae. Such masses usually involve the lower two-thirds of the muscle, and their evolution can be followed by ultrasound, which also allows for simultaneous ultrasound screening for hip dysplasia. In a study, DDH occurred in 17% of 47 infants confirmed to have CMT by ultrasound, although only 8.5% of the infants required treatment with a Pavlik harness or surgery. Subsequent studies have confirmed a 12% (12 of 97 consecutive patients with CMT, all requiring orthopedic intervention) to 15% (20 of 133 patients reviewed retrospectively, of whom half required orthopedic intervention) association between CMT and DDH. The difference and ratio of SCM thickness between the normal and abnormal side was significantly greater in DDH patients, and when the SCM ratio was greater than 2.08 and the SCM difference was greater than 6.1 mm, the efficiency of ultrasound for diagnosis of DDH was found to be the best. Because physical examination showed poor sensitivity, hip ultrasound was recommended in 4–6-month-old infants for the diagnosis of DDH associated with CMT when physical examination was positive, or in patients with a large SCM difference.
If myoblasts in the mass differentiate in a normal way, the mass will disappear and conservative treatment with neck physical therapy will have a high likelihood of success; however if the myoblasts degenerate and the remaining fibroblasts produce excess collagen, the result may be a scar-like band that is visible by ultrasound and causes muscular contracture. Myoblasts can be mechanically stimulated to undergo hypertrophy and hyperplasia in vitro by intermittent stretching and relaxation, and the proper orientation of skeletal fibers during myogenesis is maintained by rhythmic contractions. Among 28 SCM muscles from 23 subjects with CMT and 5 normal SCMs, gene expression studies showed that 269 genes were differentially expressed within five protein networks relating to fibrosis and collagen and elastin fibrillogenesis, and expression of 8 CMT signature genes showed good correlation with the preoperative severity of the CMT, as determined by MRI. Thus, there is good physiologic justification for the early initiation of neck physical therapy to prevent muscle fibrosis.
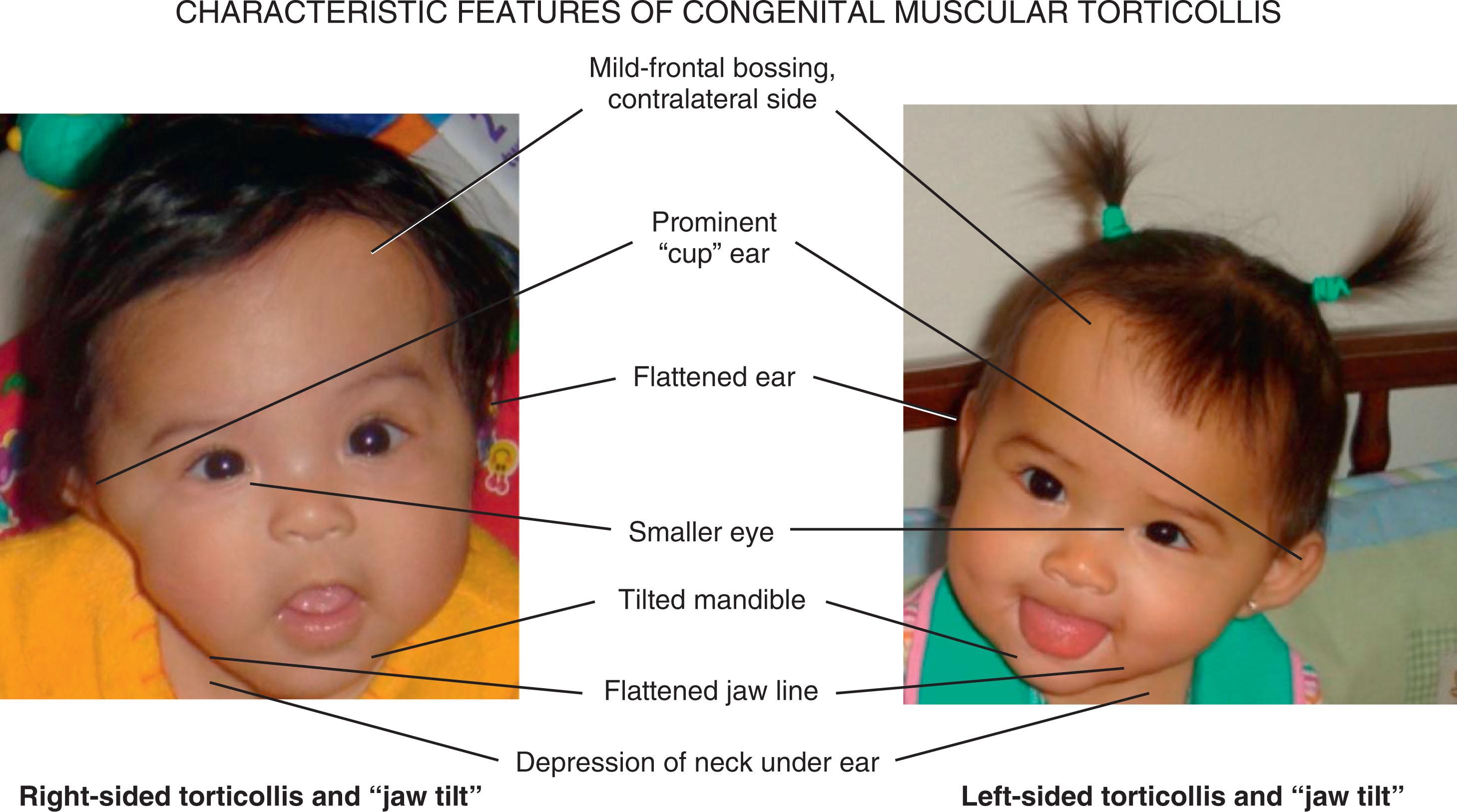
A number of associated features suggest that CMT is related to late gestational constraint. The laterality of the birth head position usually matches the laterality of the torticollis. Among 202 infants assessed between 2002 and 2003 for deformational plagiocephaly at around 6 months of age in a craniofacial clinic, 68% were male, 74% were flat on the right occiput, 14% were from a multiple gestation, and 27% were premature. Most parents (93%) did not notice any flattening at birth, but many (92%) noticed a preferential head turn after birth, which tended to improve in 95% of cases. Only 24% of cases had been diagnosed or treated for torticollis, and the mean difference between the cranial diagonal measurements was 12.5 mm (range 8–25 mm), with 97% of infants showing rotational asymmetry of 15 degrees or greater, and younger infants showing more asymmetry, which was correlated with the degree of cranial asymmetry. Because this condition improves rapidly during early infancy, the findings are often subtle and only detected through an unusual head shape with a history of preferential head rotation. Without treatment, the shortened muscle tends to maintain the aberrant posture of the head and neck, with the head tilted toward the affected side and the chin rotated toward the opposite side. There can be associated mild frontal bossing and flattening of the ear on the contralateral side and a prominent ear, flattened jawline, and smaller appearing eye on the side of the torticollis (see Fig. 24.1 ). This results in deformational posterior plagiocephaly with a rhomboid head shape that can progress in severity if the infant’s head remains turned toward one side. Early physical therapy, frequent “tummy time” when the infant is awake and under adult observation, and head repositioning onto the more prominent occiput can prevent many of these secondary positional craniofacial effects.
Among the 52 infants with CMT who were 3 months old or less, 46 (88.5%) were successfully managed with only stretching exercises for 1–6 weeks, and 6 infants (11.5%) were managed with botulinum toxin injection, surgical release, or both in addition to stretching exercises. The difference in the SCM thickness between the affected and normal sides was significantly greater in the six children requiring more extensive management, and there was a strong correlation between the total duration of stretching exercise and the thickness of the SCM, with a thicker SCM requiring a longer duration of stretching exercises and other therapeutic interventions. More limited cervical range of motion also corresponds to longer treatment duration, and infants with CMT who were diagnosed earlier and had an earlier intervention had a shorter duration of treatment. Eventually uncorrected torticollis has the potential to result in permanent craniofacial asymmetry, but surgical correction of torticollis need not be attempted until after at least 6 months of neck physical therapy has failed to correct the torticollis, or the muscle has clearly become fibrotic. Among 50 infants less than 3 months of age with a palpable neck mass who were classified by ultrasound according to the severity of fibrosis in the SCM and underwent physical therapy, this treatment was successful in 49 patients (98%). Thus in young infants with CMT, ultrasound can document severity of fibrosis, and an early physiotherapy is effective, even in those with severe fibrosis.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here