Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Congenital melanocytic nevi (CMN) are composed of clusters of nevomelanocytes that are generally present at birth but occasionally arise as late as several years of age. These lesions arise from melanocytic stem cells that migrate from the neural crest to the embryonic dermis and upward into the epidermis. They may also migrate into the leptomeninges.
Although the bulk of these lesions are small and benign, some cover large portions of the body or can be in conspicuous locations, and may create an aesthetically displeasing appearance, resulting in psychological issues. Furthermore, their potential for malignant degeneration causes anxiety for the parent, primary care physician, and surgeon alike.
Small, pigmented nevi are present in 1 in 100 births; large nevi are present in only 1 in 20,000 births; and the giant lesions are even less common. As a result, most surgeons have little experience with them and little opportunity to develop a rational protocol for their treatment.
Congenital melanocytic nevi (CMN) consist of clusters of nevomelanocytes that develop in utero . Although many congenital nevi are visible at birth, some are “tardive”, probably because they are too small to be detected at birth or do not have sufficient melanin. CMN are one of the risk factors for eventual development of cutaneous and extracutaneous melanoma, with larger nevi having greater risk. Based on that, CMN are historically classified according to their estimated largest diameter in adulthood. Small nevi are up to 1.5 cm; medium are 1.5–19.9 cm; and large nevi are those with estimated diameter of more than 20 cm. Giant nevi are 40 cm or larger. Newer classification takes into account size, location, various phonotypical features, and presence of satellite nevi. Congenital nevi present in approximately 1% of births ; large CMN occur in approximately 1:20,000 births; and giant lesions (>40 cm) are 1 in 500,000.
While most surgeons are familiar with treating the small and intermediate-sized nevi, it is difficult to gain enough experience when approaching more extensive lesions.
Many strategies have been tried for removal and reconstruction of large and giant nevi. When direct excision and primary closure are not a possibility, then tissue expansion is the “workhorse” treatment modality for many medium to large nevi. Facial nevi that cross multiple aesthetic units as well as involving the periorbital area may require expansion in combination with full-thickness skin graft (expanded or non-expanded). Some unique cases may benefit from a free flap and tissue expansion as an adjunct procedure to close the donor site.
The etiology of CMN remains unclear. The development of CMN is determined in utero between 5 and 24 weeks’ gestation. One of the theories of melanocyte differentiation is that, as the neural tube develops during early embryogenesis, melanoblasts migrate from the neural crest along the leptomeninges to the embryonic dermis. From the embryonic dermis, the progenitor melanocytic cells migrate into the epidermis, where they differentiate into dendritic melanocytes.
Dysregulated migration, proliferation, and differentiation of melanocytes in the skin and leptomeninges are implicated in the pathogenesis of CMN and neurocutaneous melanosis (NCM).
Several molecular signaling pathways have been associated with the pathogenesis of CMN. Melanocyte development appears partially under the control of c-met and c-kit proto-oncogenes, which encode met and kit proteins, respectively. Hepatocyte growth factor (HGF), also known as scatter factor (SF), is a multifunctional regulator of epithelial cells expressing the tyrosine kinase receptor encoded by c-met. Overexpression of HGF/SF, which is a ligand for the met protein receptor, is implicated in perturbations of melanocyte proliferation, differentiation, survival, and migration. Transgenic mice overexpressing HGF/SF are born with cutaneous and leptomeningeal melanocytosis. HGF/SF also functions in regulating the migration and differentiation of premyogenic cells during embryogenesis. It has been shown that overexpression of this signaling molecule in mice may lead to rhabdomyosarcoma, a tumor that on rare occasions also arises in patients with large CMN. Furthermore, studies with met null mice suggest that met plays a role in NCM, because met knockout mice do not develop NCM. Overexpression of HGF/SF and/or met, and sustained activation of met, could explain the mechanism of cutaneous and leptomeningeal melanoma and rhabdomyosarcoma development in individuals with CMN. C-kit, a proto-oncogene that encodes the kit tyrosine kinase receptor for the ligand known as SCF, also plays a role in melanocyte development. In tissue cell culture, c-kit-expressing neural crest cells give rise to clones containing only melanocytes. Proliferative nodules, consisting of aggregates of epithelioid or spindled immature benign melanocytes in the dermis of CMN, highly express c-kit. Moreover, kit can activate N-RAS, which is an oncogene that is mutated in some cases of nodular melanoma.
Recently, mutations within two oncogenes have been identified within most CMN. Mutations within N-RAS and B-RAF appear to be mutually exclusive to one another and present in up to 85% of lesions. Identical mutations in N-RAS codon Q61 have been found in affected skin lesions and the CNS of patients with NCM, but not in their non-lesional skin. This mosaicism might indicate a somatic versus a germline mutation of these oncogenes. BRAF may be more prevalent in patients with NCM. The identification of these mutations and further research show promise for the development of gene therapies.
The exact risk for development of melanoma in CMN is not clear. While the absolute lifetime risk for the incidence of melanoma in patients with large CMN is reported to be lower than previously thought, around 2%–5%, the highest risk group in this population is children with giant nevi, as high as 8% in CMN >60 cm. Patients with small and medium-sized CMN have a lower risk of melanoma, with a reported absolute risk of <1%. Many articles discussing the issue of relative risk of melanoma do not differentiate between cutaneous and extracutaneous melanoma and the presence of NCM may be the greater risk factor in subsequent risk of malignancy.
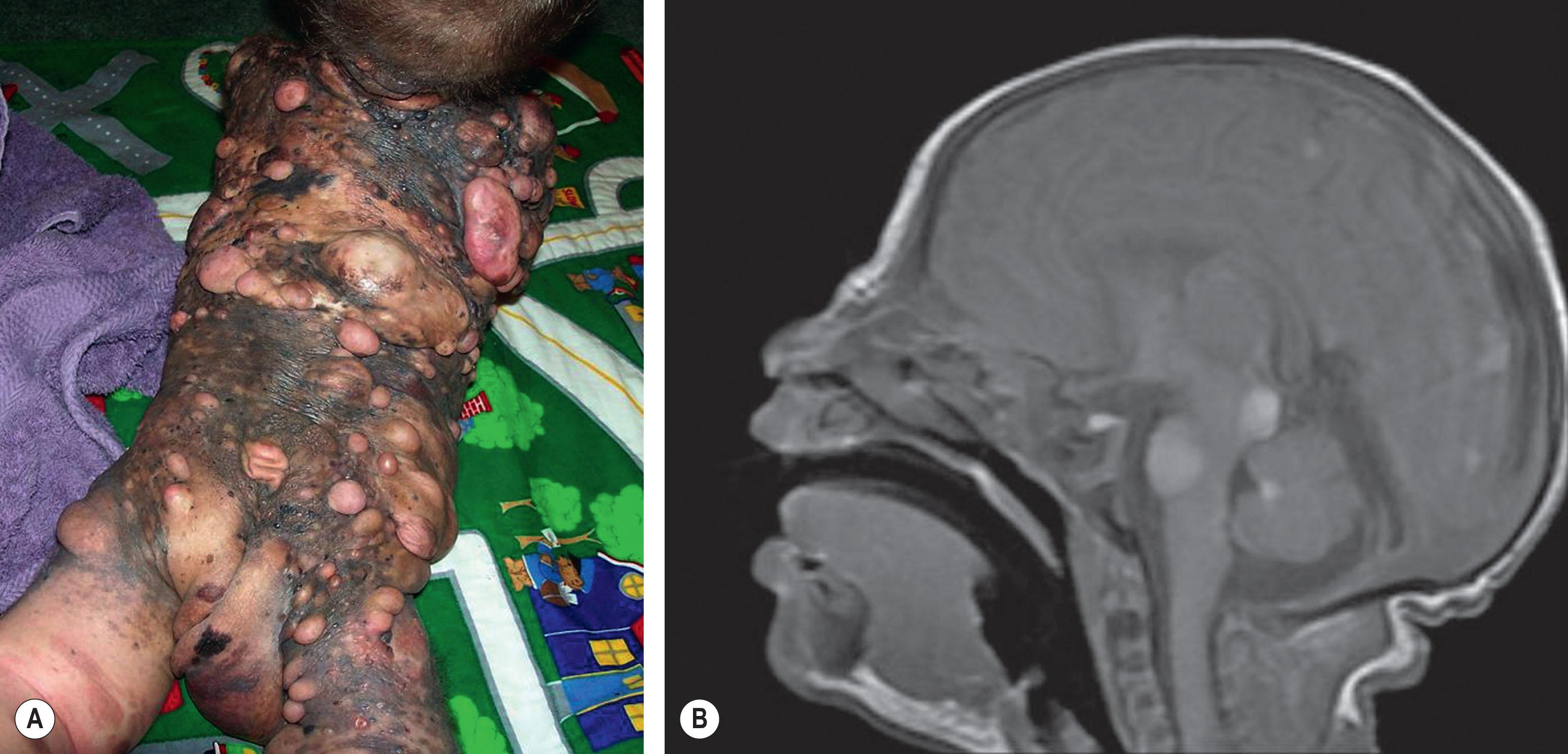
NCM is characterized by an excess deposition of melanocytes along the leptomeninges ( Fig. 30.1 ). It can occur in both patients with large CMN and those with multiple small or medium-sized CMN. Patients with large CMN located on the posterior axis are thought to have greater risk for NCM but, on a multivariate analysis, the only risk factor for NCM in patients with large congenital nevi is having multiple satellite nevi: >20 satellites had a 5.1-fold increased risk for NCM compared with patients with fewer satellites. Patients with multiple nevi and positive evidence of NCM on screening MRI may have the highest incidence of malignant melanoma (12%). The true incidence is not known, but symptomatic NCM may affect 6%–11% of patients with multiple or large CMN. Symptomatic NCM has a poor prognosis. Symptoms frequently present in early childhood. Neurologic symptoms can manifest themselves as seizures, developmental delay, hydrocephalus, and delayed motor development. NCM cells may display a more aggressive growth pattern having recently been associated with several cases of disseminated visceral nevocytoma, related to ventriculo-peritoneal shunting in patients with NCM-associated hydrocephalus.
Rarely, other tumors, such as rhabdomyosarcoma and liposarcoma, are associated with CMN.
Initial reports of giant congenital nevus were descriptive: in 1832, a giant nevus was first mentioned in Alibert’s Monograph of Dermatology , where it was described as a “waist coat and drawers type naevus”. In 1861, the great Austrian pathologist Karl Rokitansky described a patient with giant congenital nevi in the Allgemeine Wiener medizinische Zeitung ( Vienna General Medical Journal ).
Many techniques were described for treating congenital nevi. Since the introduction of tissue expansion, it has become invaluable in the reconstruction of extensive skin defects after surgical excision of giant congenital nevi.
Historically, CMN have been categorized by predicted adult diameter as the lone criterion to differentiate one lesion from another. With increasing understanding of the genetic make-up of these lesions and improved, prospective data collection techniques, efforts have been made to standardize the classification system. In 2012, Krengel et al . proposed a schema that has been widely accepted by many experts in the field of CMN ( Table 30.1 ). By using a standardized system to describe these highly variable lesions, clinicians and researchers will be able to correlate morphogenic features with the molecular genetics underlying this rare disease, eventually leading to improved treatment methods, targeted gene therapy, and better understanding of nevomelanocyte behavior.
| CMN parameter | Terminology | Definition |
|---|---|---|
| CMN projected adult size | Small | <1.5 cm |
| Medium | ||
| M1 | 1.5–10 cm | |
| M2 | 10–20 cm | |
| Large | ||
| L1 | >20–30 cm | |
| L2 | >30–40 cm | |
| Giant | ||
| G1 | >40–60 cm | |
| G2 | >60 cm | |
| Multiple | >3 medium CMN without a single predominant CMN | |
| CMN localization | ||
| Head | Face, scalp | |
| Trunk | Neck, shoulder, upper/mid/lower back, breast/chest, abdomen, flank, gluteal region, genital region | |
| Extremities | Upper arm, forearm, hand, thigh, lower leg, foot | |
| Number of satellites (refers to the number within the first year of life) | S0 | No satellites |
| S1 | <20 satellites | |
| S2 | 20–50 satellites | |
| S3 | >50 satellites | |
| Additional morphologic features | C0, C1, C2 | None, moderate, marked color heterogeneity |
| R0, R1, R2 | None, moderate, marked surface rugosity | |
| N0, N1, N2 | None, scattered, extensive nodules | |
| H0, H1, H2 | None, notable, marked hypertrichosis | |
Small to medium-sized CMN usually present as round to oval homogeneous pigmented lesions, light to dark brown in color, with sharply demarcated borders, rugous surface, and hypertrichosis. However, larger CMN, in particular, may show asymmetry, irregular borders, multicolored pigment pattern, rugous texture, and a nodular surface. In addition, large CMN are often associated with many smaller satellite nevi. As the child grows, especially at puberty, the CMN may change color becoming lighter or darker, develop hair, and become more heterogeneous or more homogeneous. CMN may spontaneously regress, and some patients may develop vitiligo. Nodular proliferation may be present from birth or develop at a later age. CMN are usually asymptomatic; however, patients with larger lesions may present with pruritus, xerosis, skin fragility, erosions, or ulcerations and decreased ability to sweat from the involved skin ( Fig. 30.2 ).
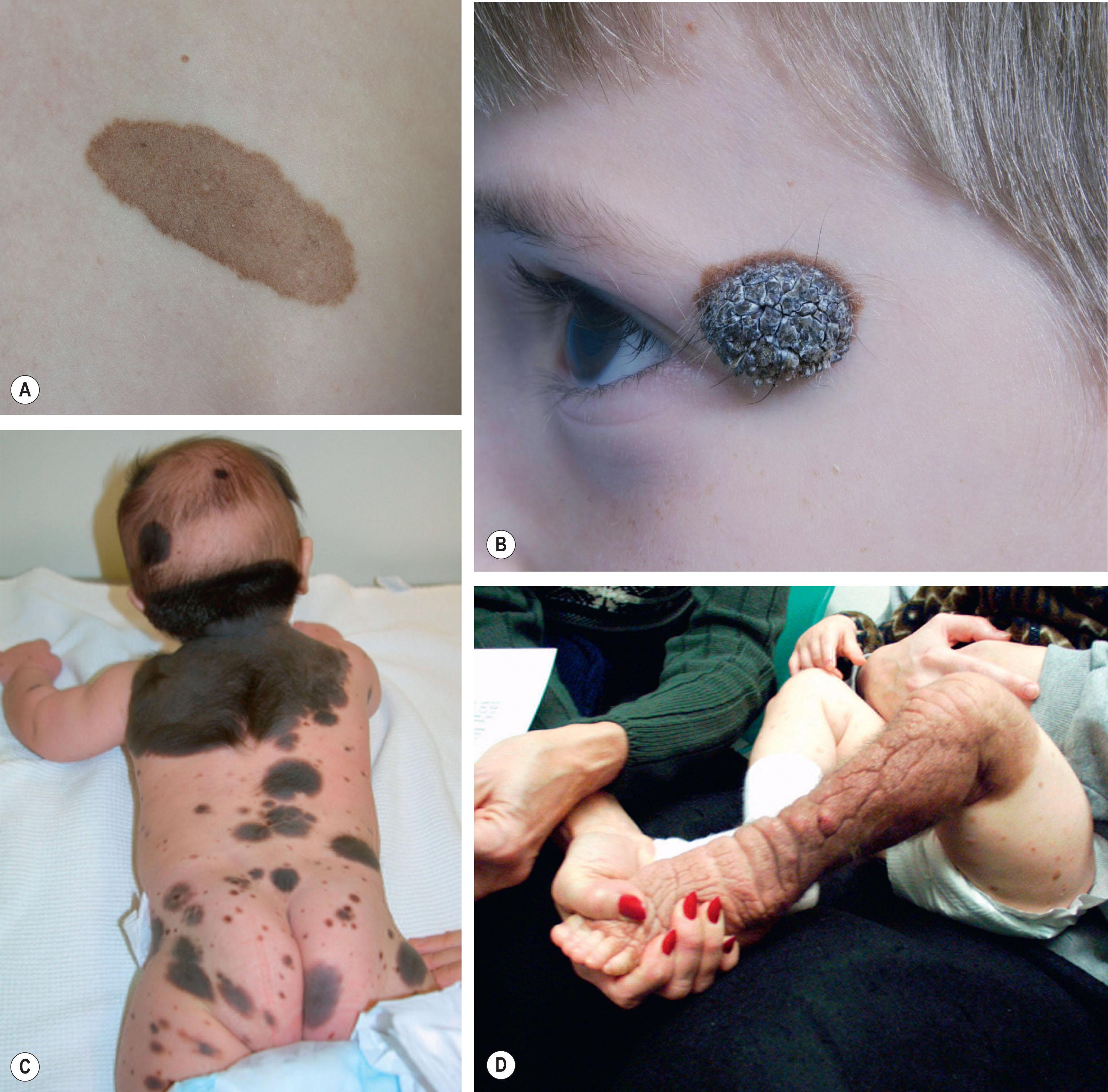
A review of dermoscopy patterns in congenital nevi found that most nevi demonstrate a reticular, globular, or reticuloglobular pattern. The findings varied with age and the anatomic location of the nevus, with the globular pattern found more often in younger children and the reticular pattern found in patients aged 12 years or older.
Because of the increased risk of melanoma associated with congenital nevi, attempts have been made to distinguish congenital nevi from acquired nevi on the basis of histology. Distinguishing histologic features include: (1) involvement by nevus cells of deep dermal appendages and neurovascular structures (including hair follicles, sebaceous glands, arrector pili muscles, and within walls of blood vessels); (2) extension of nevus cells to deep dermis and subcutaneous fat; (3) infiltration of nevus cells between collagen bundles; and (4) a nevus cell-poor subepidermal zone. In contrast to congenital nevi, acquired nevi are usually composed of nevus cells that are limited to the papillary and upper reticular dermis and do not involve the appendages.
In cases associated with a high index of suspicion for the presence of NCM, magnetic resonance imaging of the central nervous system is a useful diagnostic tool (see Fig. 30.1B ).
The treatment of large and giant nevi is controversial. Although the risk of malignant transformation in congenital pigmented nevi is well established, many feel that the risk of developing melanoma is too low to warrant the unsightly scars or grafts that may follow treatment. There is no evidence in the literature that demonstrates decrease in occurrence of melanoma after excision of large congenital melanocytic lesions. Furthermore, these patients have an increased risk of extracutaneous melanoma. Others feel that, in the presence of NCM, the greatest risk lies within the central nervous system, so the excision of the cutaneous lesion can only have limited benefits. However, the appearance of these lesions clearly produces a stigma with significant psychological implications. The challenge for the surgeon involved in treating these often-complex lesions is to develop treatment modalities that not only accomplish the excision of all or most of the nevus but also lead to an optimal aesthetic and functional outcome.
Although the lifetime risk of malignant melanoma for small and medium congenital pigmented nevi is reported to be 0–4.9%, the risk of melanoma is nearly nil prior to puberty for small nevi, and so one may comfortably wait until the child is old enough to excise the lesion under local anesthesia. If the lesion is located in an area where the excision and reconstruction may not likely be accomplished under local anesthesia, or where there may be the possibility of a better final scar with earlier excision, then early excision under general anesthesia may be warranted. Certainly, many nevi positioned in prominent parts of the face may present as a significant source of peer ridicule starting quite early in the school years, and delaying the excision in an effort to avoid a general anesthetic is not in the child’s best interest.
The authors advocate treatment of large and giant nevi beginning at 6 months of age in most cases. Although many of the tissue expansion procedures used in the treatment of large nevi can be applied to older children and adults, the intolerance for repeated procedures and the decreased elasticity of the skin may make the excision of extensive lesions impractical in older patients. Also, for larger nevi the greatest risk for malignancy is in the first few years.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here