Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The pancreas is a glandular organ that lies on the posterior abdominal wall in the retroperitoneum (see Chapter 2 ) It is an exocrine gland that secretes enzymes of digestion into the duodenum; it also performs endocrine functions, producing insulin, glucagon, somatostatin, pancreatic polypeptide, and ghrelin (see Chapter 3 ) Congenital abnormalities of the pancreas may be so severe that they are diagnosed in utero or in the neonatal period, such as pancreatic agenesis. However, many congenital conditions go undetected until adulthood, when the patient comes to medical attention with nonspecific symptoms or an abnormality is discovered incidentally. This chapter describes the diagnosis, investigation, and treatment of the various congenital pancreatic abnormalities.
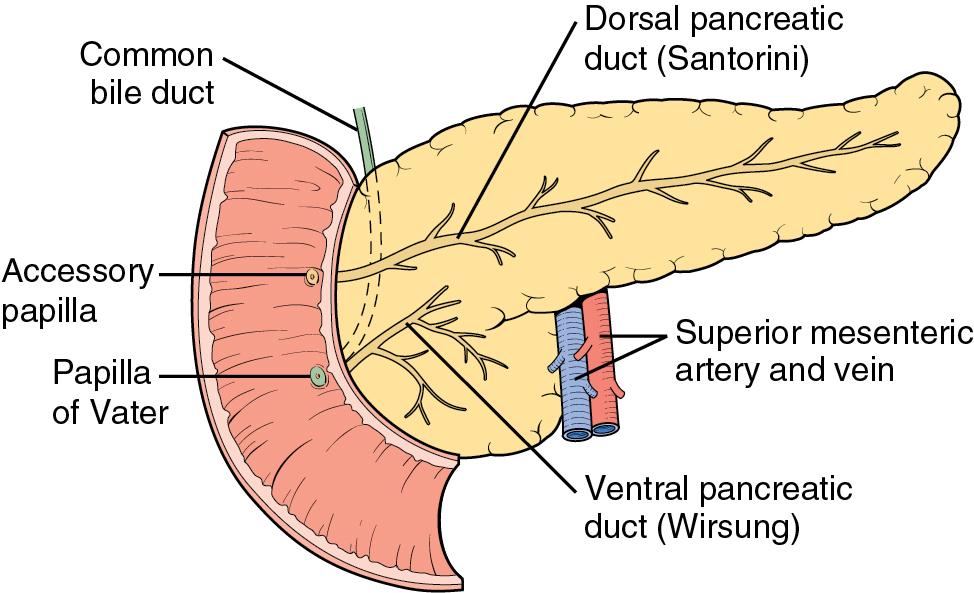
The basis for the understanding of congenital abnormalities of the pancreas is the embryologic development of the organ (see Chapter 1 ). The pancreas develops from two buds originating from the endodermal lining of the duodenum. The dorsal bud forms posteriorly within the mesentery, whereas the ventral bud is associated with the hepaticopancreatic duct ( Fig. 53.1 ). During the second month of development, the stomach rotates and the duodenum becomes C-shaped. As part of this process, the ventral pancreatic bud migrates dorsally, coming to lie posteroinferiorly to the dorsal bud, forming what will become the inferior head/uncinate process of the pancreas. In the majority of individuals, the main pancreatic duct (of Wirsung) is formed by the entire ventral duct and the distal dorsal duct and enters the duodenum at the major papilla. Persistence of the proximal part of the dorsal duct occurs in approximately 25% and results in an accessory duct (of Santorini), which enters the duodenum by the minor papilla (see Fig. 53.1 ). There is no known pathologic consequence of this normal variation. Failure of fusion of the ductal system occurs in roughly 10% of the normal population, , resulting in the entire dorsal pancreas—superior head, body, and tail—draining through the minor papilla, and ventral pancreas—the inferior head and uncinate process—draining through the major papilla ( Fig. 53.2 ). This abnormality is termed pancreas divisum (PD) and is described in more detail later (see Chapter 1 ).
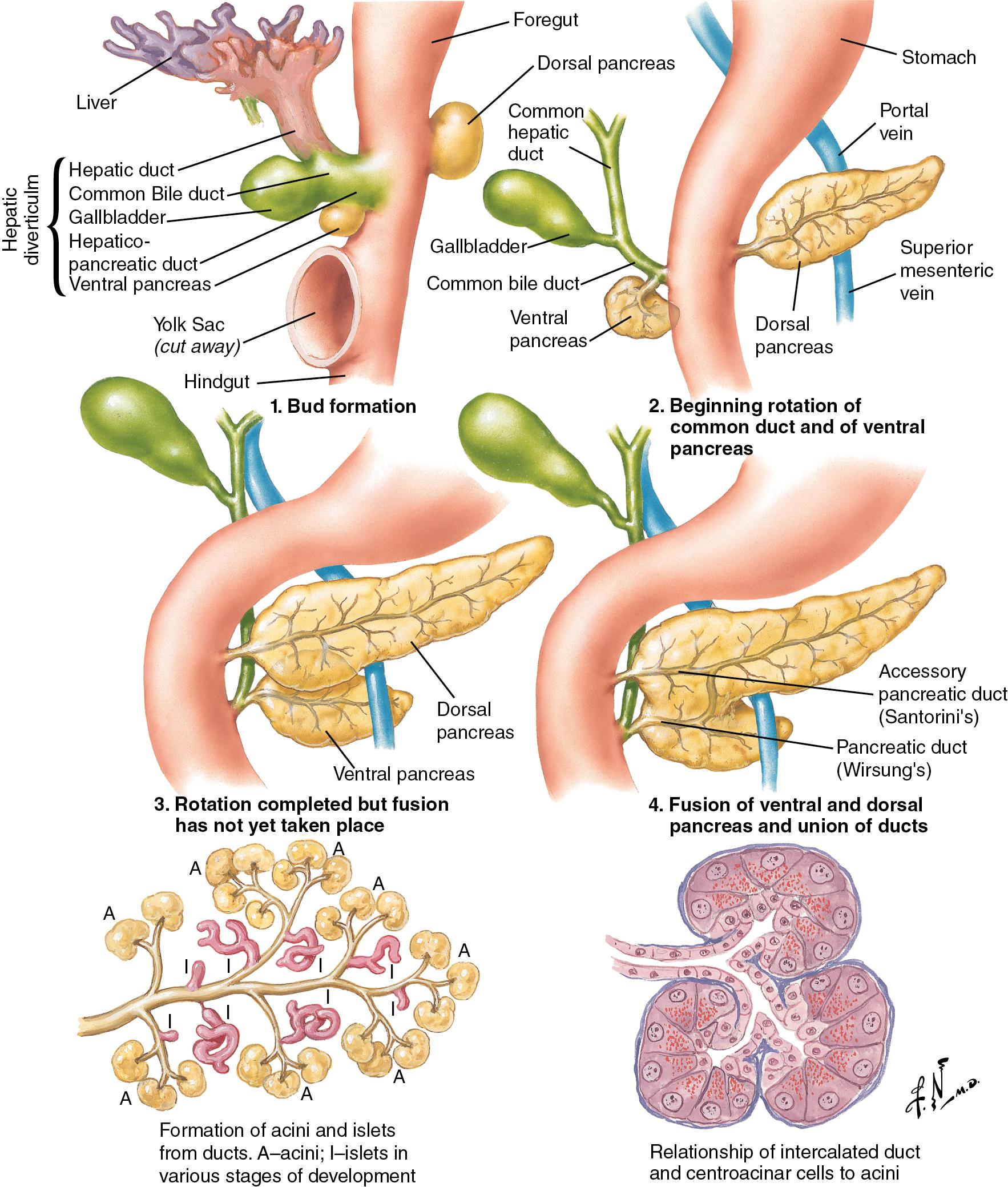
Annular pancreas is a rare congenital abnormality, the embryologic basis of which is poorly understood. A complete or incomplete ring of pancreatic tissue is found around the second part of the duodenum, and it may cause symptoms (also discussed later). A number of published studies have identified mechanisms by which the pancreas is specified from the early endoderm. Retinoic acid and bone morphogenic peptide both appear to have important roles in defining early endodermal compartments in the embryo. , The origins of the signaling mechanisms involved in the specification of the dorsal and ventral pancreas are different. In the dorsal pancreas, signals arising from the notochord and dorsal aorta are required; in the ventral pancreas, the lateral plate mesoderm is important. The specific identity of these signals has not yet been established, although the Hedgehog family of signaling molecules appears to be significant.
PD results from incomplete fusion of the dorsal and ventral pancreatic ducts toward the end of the second month of embryogenesis. The distal dorsal pancreatic duct typically fuses with the ventral pancreatic duct to drain the entire pancreas into the duodenum by the major papilla (see Chapter 1 ). The proximal dorsal duct can persist as an accessory pancreatic duct and may drain by the minor papilla. Complete PD exists when there is no communication between the dorsal and ventral systems and the majority of the pancreas drains by the dorsal duct through the minor papilla (see Fig. 53.2 ). Variations exist in which a small branch may connect the two ducts, termed incomplete pancreas divisum. The prevalence of this variant is about 15%.
Investigations have suggested that PD may be explained by distinct patterns of incomplete fusion of branches of the dorsal and ventral pancreatic ducts, confirming theories first proposed in early anatomic studies. The first description of PD is from 1865, attributed to Josef Hyrtl (1810–1894), Professor of Anatomy at the Universities of Prague and Vienna. However, as discussed by Stern (1986), a number of anatomists were aware of it much earlier than this, including Regnier de Graaf, who described the finding in 1664.
In postmortem studies performed throughout the 20th century, the prevalence of PD is reported to be approximately 8% (range, 4%–14.5%). With the development of endoscopic retrograde cholangiopancreatography (ERCP, see Chapters 20 and 30 ) in the late 1960s, however, the considerable congenital variation in the ductal system of the pancreas became more widely appreciated. The prevalence of PD is lower in published ERCP series than in anatomic series for reasons that are not clear, although it may be due to referral bias, difficulty in the interpretation of pancreatograms, or inability to cannulate the minor papilla. The reported prevalence is particularly low in Asian series (0.3%–0.6%) compared with Western populations (∼5%).
PD is clinically relevant for three reasons. First, the small ventral duct seen in PD must be differentiated from a similar appearance seen in some cases of pancreatic cancer. It is important for those performing ERCP to be aware of the anomaly to become proficient in interpreting pancreatograms ( Fig. 53.3 ). Other forms of imaging, such as computed tomography (CT) or magnetic resonance imaging, must be used if there is uncertainty whether a mass lesion is present and causing a ductal abnormality (see Chapter 17 ). Second, on cannulating the major papilla at ERCP, only the ventral portion of the pancreas may be visualized in PD, which risks missing important pathologic conditions if a pancreatogram is not performed via the minor papilla (see Chapters 20 and 30 ). It is important to become proficient at cannulation of the minor papilla, which is considerably more challenging than cannulating the major papilla. The minor papilla commonly sits in a superior, more ventral position. Cannulation may be facilitated by having the duodenoscope in the “long position” and by administration of intravenous secretin. Third, PD may be associated with pancreatitis or other pathologic condition (see Chapters 54 and 55 ), and this will be discussed in detail in this chapter.
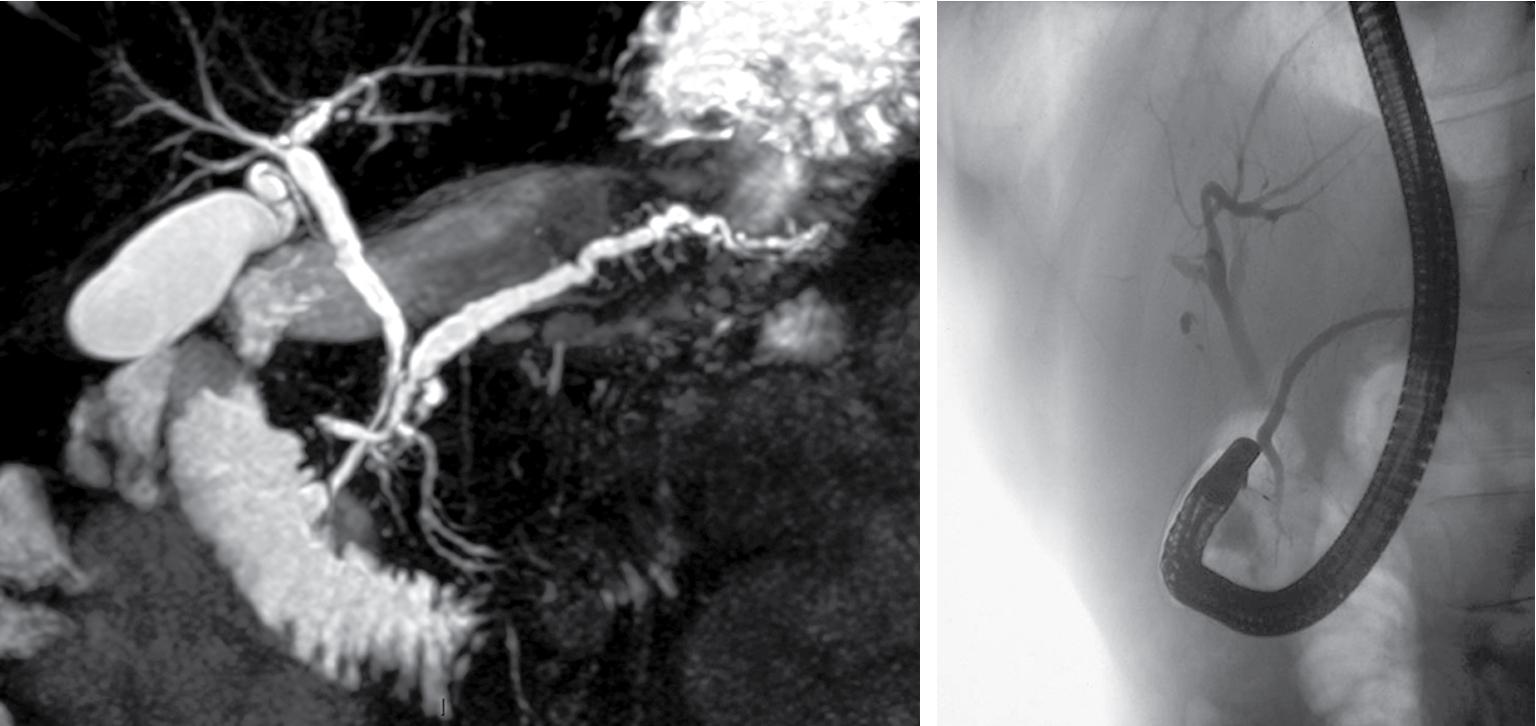
The question of whether PD causes recurrent acute pancreatitis, chronic pancreatitis, or pancreas-related pain has been debated for many years ( Table 53.1 ). An up-to-date systematic review concluded that “current research fails to define a clear association between PD and pancreatic disease.” Those proposing the theory suggest that obstruction occurs at the level of the minor papilla, the caliber of which is too narrow to provide adequate drainage of the pancreatic secretions. This is supported by an early study reporting an elevated pressure in the dorsal duct (23.7 ± 1.3 mm Hg) compared with the ventral duct (10.8 ± 1.9 mm Hg) in six patients with PD, whereas pressures were similar when two ducts existed in patients without PD. This was contradicted in a subsequent study in which no difference in duct pressures was observed between the major and minor papillae in four patients with recurrent acute pancreatitis and PD. Another early report demonstrated chronic pancreatitis confined to the dorsal pancreas in two pancreatoduodenectomy specimens from patients with PD who had resections for symptom control (see Chapters 54 and 55 ).
| STUDY | PANCREATITIS RELATIONSHIP SUPPORTED | STUDY TYPE | N (%) | INTERVENTION |
|---|---|---|---|---|
| Phillip J et al., 1974 | — | ERCP series | 18/911 (2.0) | — |
| Rösch et al., 1976 | — | ERCP series | 63/1850 (3.4) | — |
| Gregg, 1977 | — | ERCP series | 33/1100 (3.0) | — |
| Heiss & Shea, 1978 | Yes | ERCP series | 4 | — |
| Mitchell et al., 1979 | No | ERCP series | 21/449 (4.7) | — |
| Cotton, 1980 | Yes | ERCP series | 47/810 (5.8) | — |
| Tulassay & Papp, 1980 | Yes | ERCP series | 33/2410 (1.4) | Unspecified surgery in 11; no outcomes described |
| Richter et al., 1981 | Yes | ERCP series | 519 | Open sphincteroplasty to minor ampulla in 9; decreased pain: 5/6 with acute pancreatitis, 0/3 with chronic |
| Thompson et al., 1981 | Yes | ERCP series | 11/850 (1.3) | — |
| Cooperman et al., 1982 | Case series | 21/314 (6.7) | Open sphincteroplasty to minor ampulla in 5; 4 of 6 “have done well” at 28 months | |
| Sahel et al., 1982 | Yes | ERCP series | 41/812 (5.0) | — |
| Blair et al., 1984 | Yes | Case series | 14 | 15 operative resections in 14 patients with RAP; 7/14 had no pain after surgery |
| Delhaye et al., 1985 | No | ERCP series | 304 total; 6.9% AP/CP, 5.7% no pancreatitis | — |
| Sugawa et al., 1987 | No | ERCP series | 55/1529 (3.6) | Open sphincteroplasty to minor ampulla in 3; no improvement in symptoms |
| Bernard et al., 1990 | Yes | ERCP series | 137/1825 (7.5) | — |
Difficulty exists in demonstrating an epidemiologic relationship between PD and pancreatitis because early anatomic studies were small and confounded by differing definitions, and ERCP series have been skewed by selection bias. In the largest early series of 1850 successful major ampulla cannulations, Rösch et al. identified PD in 63 cases (3.4%). The indications for pancreatography were not given, but pathologic findings were seen in 13 of 63 patients with PD; changes were consistent with pancreatitis in 12 patients and tumor in 1 patient. In another large series by Gregg, 33 patients with PD were identified among 1100 patients (3%) referred primarily for investigation of pancreatic-type pain or pancreatitis. Documented pancreatitis was present in 15 patients, and another 11 had recurrent episodes of pain typical of pancreatitis.
These and similar studies have been used as evidence of a link between PD and pancreatitis, but the absence of a suitable control group makes the assertion weak. Mitchell et al. identified this problem and performed a retrospective analysis of patients who had undergone ERCP and observed that 21 (4.7%) of 449 patients had PD, whereas 4 (3.3%) of 120 patients with pancreatitis defined by clinical and/or ERCP criteria had PD. Thus, in this series, the prevalence of PD in patients with pancreatitis was the same as the prevalence of PD in the series as a whole. This has been supported by the largest published ERCP series of 304 patients, in which the dorsal duct was visualized in 97 patients. The frequency of PD was similar in patients with acute or chronic pancreatitis (6.9%) as in all patients in the series undergoing ERCP (5.7%).
Contrary to these findings, Cotton reported that in patients with primary biliary disease who had an incidental pancreatogram at the time of endoscopic retrograde cholangiogram, the prevalence of PD was 3.6%. This was compared with a prevalence of 16.4% in those with chronic or recurrent acute pancreatitis. In 83 patients with idiopathic pancreatitis, recurrent pancreatitis with no clear cause, such as gallstones, alcohol, or trauma, the prevalence of PD was 25.6%.
This controversy is a good example of the difficulty in determining a causal relationship between two factors that appear to have a clinical association. The traditional criteria used in evaluating such a relationship can be applied: (1) strength of association, (2) consistency across studies, (3) dose-response relationship, and (4) biologic plausibility. The relationship is not a strong one, and many of those in the general population who have PD (∼10%) never have any related symptoms, and many with pancreatitis do not have PD; less than 5% of individuals with PD are estimated to ever develop pancreatic symptoms. Furthermore, those with a more pronounced form of the anomaly (i.e., complete PD) do not seem to be more likely to develop pancreatitis than those with incomplete PD.
One of the difficulties in relating PD to pancreatitis is the inconsistency in making the diagnosis. ERCP studies of patients without pancreatitis consistently report a lower prevalence of PD when compared with magnetic resonance cholangiopancreatography (MRCP) or postmortem studies ( Table 53.2 ) (see Chapters 17 , 20 , and 30 ). The use of secretin during MRCP (S-MRCP) improves the detection rate of PD, yet it is still reported to be missed on S-MRCP, possibly because of suboptimal magnetic resonance techniques or inexperience of those reporting the MRCP (see Chapter 19 ). A more recent study reported the use of portal venous phase 64–multidetector-row CT (MDCT). Of 93 patients, 5 had PD diagnosed on MRCP or ERCP. Of these, one observer detected three cases, and a second observer found four cases by reviewing the MDCT images. ERCP features of the minor papilla that suggest the presence of PD include an enlarged papilla or open orifice and are thought to moderately predict the presence of PD; however, a significant number of patients with PD do not have these features. , Endoscopic ultrasound (EUS) as an alternative imaging modality has gained in popularity and is reported to be useful in the investigation of patients with acute recurrent pancreatitis of unknown cause (see Chapter 22 ). The sensitivity of EUS for PD has been shown to be 85% to 95%, which was superior to the sensitivity of CT (50%–60%) , or MRCP (50%–70%), , but similar to that with secretin-enhanced MRCP (83%–86%). ,
| INVESTIGATION TYPE | N | % PREVALENCE OF PD (95% CI) |
|---|---|---|
| Postmortem | 2895 | 7.8 (6.8-8.8) |
| MRCP | 505 | 9.3 (6.8-11.8) |
| S-MRCP | 156 | 17.9 (11.9-24.0) |
| ERCP | 16,078 | 4.1 (3.8-4.4) |
If PD is associated with obstruction at the minor papilla, which in turn contributes to the occurrence of pancreatitis and pancreatic pain, it follows that surgical intervention to relieve this obstruction would be beneficial. As has been discussed, significant controversy exists as to whether this assumption is correct. A number of studies have been performed examining whether therapy to the minor papilla is beneficial in patients with acute recurrent pancreatitis, chronic pancreatitis, or pancreas-related pain; however, little in the way of randomized controlled data has been generated ( Table 53.3 ) (see Chapter 54, Chapter 55, Chapter 56, Chapter 57, Chapter 58 ). Interventions examined include endoscopic dilation; papillotomy; sphincterotomy of the minor papilla, with or without stent placement; and surgical sphincteroplasty.
| STUDY | THERAPY | MEAN FOLLOW-UP (mo) | RAP | PAIN ONLY | CP | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| N | % IMPROVED | N | % IMPROVED | N | % IMPROVED | |||||
| Soehendra et al., 1986 | MiES | 3 | 2 | 100 | 0 | — | 4 | 75 | ||
| Liguory et al., 1986 | MiES | 24 | 8 | 63 | 0 | — | 0 | — | ||
| McCarthy et al., 1988 | Stent | 21 | 19 | 89 | 0 | — | 0 | — | ||
| Lans et al., 1992 | Stent | 30 | 10 | 90 | 0 | — | 0 | — | ||
| Lehman et al., 1993 | MiES | 22 | 17 | 76 | 23 | 26 | 11 | 27 | ||
| Coleman et al., 1994 | MiES/stent | 23 | 9 | 78 | 5 | 0 | 20 | 60 | ||
| Sherman et al., 1994 (RCT) | MiES | 28 | 0 | — | 16 | 44 | 0 | — | ||
| Kozarek et al., 1995 | MiES/stent | 20 | 15 | 73 | 5 | 20 | 19 | 32 | ||
| Ertan, 2000 | Stent | 24 | 25 | 76 | 0 | — | 0 | — | ||
| Heyries et al., 2002 | MiES/stent | 39 | 24 | 92 | 0 | — | 0 | — | ||
| Linder et al., 2003a | Stent | NG (range, 3-36) | 83 | 66 | 48 | 23 | 38 | 38 | ||
| Bierig et al., 2006 | MiES | 19 | 16 | 94 | 7 | 43 | 16 | 38 | ||
| Linder et al., 2003b | MiES | NG (range, 1-120) | 38 | 58 | 12 | 0 | 4 | 25 | ||
| Vitale et al., 2007 | Stent | 59.6 | — | — | — | — | 24 * | 58 | ||
| Borak et al., 2009 | MiES/stent | 43 | 62 | 71 | 29 | 62 | 22 | 46 | ||
| Total | 25 | 328 | 77 | 145 | 33 | 135 | 41 | |||
The first minor papilla endoscopic sphincterotomy was described by Cotton in 1978, and the majority of publications since have been small case series with short follow-up times. Lehman et al. described the effects of minor papilla sphincterotomy in 52 PD patients with chronic pancreatic pain ( n = 24), recurrent acute pancreatitis ( n = 17), or chronic pancreatitis ( n = 11) with long-standing symptoms refractory to conservative management. Minor papilla sphincterotomy was performed with a needle knife over a previously placed dorsal pancreatic duct stent, with a mean follow-up of 1.7 years. When compared with the chronic pain and chronic pancreatitis groups, patients with recurrent acute pancreatitis had a significant reduction in mean symptom score (recurrent acute pancreatitis, 76.5%; chronic pancreatitis, 27.3%; chronic pain, 26.1%) and inpatient hospital stay. This reflects other published results that consistently show less benefit after minor papilla therapy in those with chronic pancreatitis or chronic pain compared with those with recurrent acute pancreatitis. Complications were seen in 15%, and one patient died of a pancreatic abscess after a failed cannulation. Of concern, 50% of patients evaluated at the time of stent removal had stent-induced dorsal duct changes.
In a prospective, randomized, controlled trial published by Lans et al., 19 patients with PD and recurrent acute pancreatitis—two or more episodes of abdominal pain associated with a rise in amylase greater than twice the upper limit of normal—and no other identified cause were recruited. Of these, 10 patients were randomized to stent placement, and 9 were randomized to no treatment, with observation for 1 year. In the stent group, no patients subsequently came to the hospital with pain, but 5 patients in the control group were admitted, and 2 more came in with pain. Furthermore, 9 patients in the stent group rated their pain improved by 50% or more, but only 1 patient in the control group reported a similar improvement.
It has been pointed out that a significant period of time can exist between attacks of pancreatitis in this patient group, and this study has since been criticized on the basis of the short duration of observation. Sherman et al. have also published randomized data in abstract form in which patients with chronic abdominal pain thought to be pancreatic in origin and PDs were randomized to minor papilla sphincterotomy ( n = 16) or no intervention ( n = 17). Mean observation time in the treated and untreated groups was 2.1 and 1.2 years, respectively. Although an improvement in pain was seen in 43.8% of treated patients compared with 23.5% in the control group, this trend did not reach statistical significance.
Borak et al published the long-term outcomes after endoscopic minor papilla therapy in 145 patients with PD over a 6-year period. Follow-up data were available for 113 patients (78%), and the median observation time was 43 months. The majority of patients had a needle-knife sphincterotomy (82%) and temporary stent placement (90%). Primary success, defined as the patient being better or cured after a single ERCP session, was seen in 53.2% of patients with recurrent acute pancreatitis, 18.2% of those with chronic pancreatitis, and 41.4% of those with chronic/recurrent epigastric pain. Two or more ERCP sessions were required in 41.6%, with success in 71% of patients with recurrent acute pancreatitis, 46% of those with chronic pancreatitis, and 55% of those with chronic, recurrent epigastric pain. In a multivariate analysis that corrected for age, sex, symptom frequency/duration, and length of observation, chronic pancreatitis and younger age both independently predicted failure of improvement after treatment. Complications occurred in 13% after ERCP, including mild and moderate pancreatitis, mild bleeding, and anesthetic complications.
Surgical sphincteroplasty to the minor papilla, usually combined with cholecystectomy and major papilla sphincteroplasty, has been used in the treatment of recurrent acute pancreatitis, chronic pancreatitis, and chronic pancreatic-type pain associated with PD (see Chapter 117D ) ( Table 53.4 ). In the largest published series by Warshaw et al., 88 patients with recurrent acute pancreatitis (49%) or “pancreatic pain” (51%) had minor papilla sphincteroplasty with a mean observation of 53 months; 70% of patients were reported to show improvement, 85% if the minor papilla was stenotic at surgery, compared with 27% if it was not ( P < .01). Of those with recurrent acute pancreatitis, 82% were reported to have improved compared with 56% in the chronic pain group ( P < .01). Preoperative ultrasonography (US) with secretin stimulation was compared with examination of the minor papilla and had a sensitivity of 78% and a specificity of 97%. Thus preoperative US with secretin stimulation was judged to be a good predictor of surgical success (92% success if positive, 40% success if negative). Seven patients were documented to have developed a restenosis at the minor papilla, six of whom had further surgery. The study concluded that a demonstrable stenosis at the minor papilla was a necessary cofactor in the development of recurrent acute pancreatitis or pancreatic pain in patients with PD.
| STUDY | TOTAL (N) | PATIENTS IMPROVED (%) * | RAP | PAIN ALONE † | CP ‡ | RESTENOSIS | MAJOR COMPLICATIONS | DEATHS | MEAN FOLLOW-UP (mo) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N | PATIENTS IMPROVED (%) | N | PATIENTS IMPROVED (%) | N | PATIENTS IMPROVED (%) | ||||||||||
| Warshaw et al., 1990 | 88 | 77 | 43 | 82 | 45 | 56 | 0 | 0 | 7/88 | 1/88 | 0 | 53 | |||
| Brenner et al., 1990 | 13 | 54 | 10 | 70 | 3 | 0 | 0 | 0 | 0/13 | 0/13 | 0 | 18 | |||
| Cooperman et al., 1982 | 4 | 75 | 4 | 75 | 0 | 0 | 0 | 0 | 1/4 | NG | 0 | 14 | |||
| Bragg et al., 1988 | 4 | 100 | 3 | 100 | 1 | 100 | 0 | 0 | 0/4 | NG | 0 | 21 | |||
| Rusnak et al., 1988 | 4 | 75 | NG | — | NG | — | NG | — | 1/4 | 0/4 | 0 | ≈ 48 | |||
| Madura, 1986 | 32 | 75 | 11 | 82 | 19 | 68 | 2 | 0 | NG | 3/32 | 0 | 31 | |||
| Britt et al., 1983 | 5 § | 60 | 4 | 75 | 0 | 0 | 1 | 0 | 1/5 | NG | 0 | 21 | |||
| Russell et al., 1984 | 7 | 71 | NG | — | NG | — | NG | — | 1/7 | NG | 0 | 8 | |||
| Gregg et al., 1983 | 19 | 53 | NG | — | NG | — | NG | — | 1/19 | 1/19 | 1 | NG | |||
| Keith et al., 1989 | 22 § | 86 | 13 | 100 | 8 | 75 | 1 | 0 | 1/22 | 2/22 | 0 | 53 | |||
* Includes patients graded excellent, good, and fair (if more than 50% improved and off opioids).
† Pain suggestive of pancreatic origin (generally epigastric with back radiation) without serologic, ultrasound, computed tomographic scan, or ductographic evidence of pancreatitis.
‡ Some patients had a history of heavy alcohol ingestion.
§ Plus one patient who did not respond to minor papilla identification at laparotomy.
Surgical sphincteroplasty is not without risk, however. In a recent large series of 446 patients, complications were reported in 34.8% of patients; pancreatitis (8.8%), asymptomatic hyperamylasemia (6.0%), and wound/abdominal infection (7.1%) were the most common morbidities. One death occurred after a duodenal leak.
A recent study evaluated the question of whether children with acute recurrent pancreatitis or chronic pancreatitis should be treated differently? In this report, 38 pediatric patients were presented in whom 74 ERCPs were performed. The frequency of pancreatitis episodes decreased significantly (from 2.31 to 0.45) during a median 41-month observations period. The authors concluded that therapeutic ERCP may be useful in this population.
The place of duodenum-preserving pancreatic head resection and longitudinal pancreaticojejunostomy (Frey procedure) for painful chronic pancreatitis associated with PD has been presented elsewhere (see Chapter 58 ). In this study, 14 patients (6 PD, 5 alcohol, and 3 idiopathic) who underwent a Frey procedure for chronic pancreatitis and disease-related intractable pain were compared. The series is clearly small, but outcomes for those with PD treated with the Frey procedure were equivalent to those with other etiologies. The authors commented on the potential advantage of this approach over minor duct sphincteroplasty as lateral pancreaticojejunostomy is associated with removal of the fibrotic tissue of the head of the pancreas, the potential origin of the pain. It was pointed out in the accompanying editorial that PD patients with abdominal pain but no evidence of chronic pancreatitis were unlikely to benefit from this approach.
Those making the case against an association between PD and pancreatitis begin by highlighting problems with the epidemiologic studies. It is argued that in ERCP series, the true incidence of PD is underdiagnosed. This is supported by autopsy studies that demonstrate the prevalence of PD to be approximately 10%, whereas ERCP series show it to be less than 5%. It is argued that if ERCP were able to accurately diagnose PD, then its prevalence in control and pancreatitis groups would be the same. Patients in groups used as controls often only have cholangiography and do not undergo intentional pancreatography. Pancreatitis patients, on the other hand, have the pancreatic ducts imaged intentionally; therefore PD may be diagnosed more accurately. Furthermore, the ERCP prevalence of PD in pancreatitis patients is the same as the prevalence of PD in the general population in autopsy studies (7.6% vs. 7.8%). The argument therefore follows that if ERCP accurately diagnosed PD in the general population, no difference would exist.
An argument is also made against the theory of minor papilla obstruction. It is argued that patients with pancreatitis secondary to PD should have a dilated ductal system, but this has not been shown to be the case in the majority of studies. Similarly, if pancreatitis were associated with PD, only the dorsal pancreas should be affected, yet ventral duct pancreatitis is present in up to 11.8% of patients with PD and is the only duct involved in 4.2% of patients.
Cohn et al. and Sharer et al. first described the link between cystic fibrosis gene mutations and idiopathic pancreatitis (see Chapters 3 and 54 to 57 ). More recently, Choudari demonstrated that mutations of the cystic fibrosis transmembrane conductance regulator (CFTR) gene were present in 8 (22%) of 37 patients with PD and pancreatitis compared with 0 of 20 patients with PD and no history of pancreatitis (odds ratio, 11.8; confidence interval [CI], 8.9 to 14.7; P = .02). The authors concluded that in patients with PD, CFTR mutations increased the risk for pancreatitis. Furthermore, Gelrud et al. directly measured CFTR gene function in nasal epithelium in response to isoproterenol and demonstrated that those with PD and recurrent acute pancreatitis had results somewhere between those observed for healthy controls and those for classic cystic fibrosis patients. Thus CFTR dysfunction may explain why some PD patients have recurrent acute pancreatitis but others with PD do not. Moreover, of 12 patients with PD and recurrent acute pancreatitis, 10 had undergone therapy to the minor papilla, and only 2 had resolution of their symptoms. Finally, Bertin et al. evaluated the presence of PD with MRCP in 40 patients with idiopathic pancreatitis. The frequency of PD was 7% in those without pancreatic disease, 7% in those with alcohol-induced pancreatitis, and 5% in those with idiopathic pancreatitis. However, PD was seen in 47% of those with CFTR -associated pancreatitis, and further associations between other functional genetic anomalies were demonstrated (SPINK1, PRSS1). The authors propose that although the frequency of PD was not different in patients with idiopathic pancreatitis compared with control patients, the frequency of PD was higher in those with genetic pancreatitis, suggesting a cumulative effect of these two factors.
Despite the association of pancreatitis and PD being described in the 1970s, significant controversy continues regarding whether a causal relationship exists. Although many case series report the benefit of therapy to the minor papilla in patients with PD and pancreatitis, a lack of randomized, controlled data means interpretation of these studies must be guarded. At best, careful case selection is paramount before embarking on any such therapy. Patients most likely to benefit are older, have documented recurrent acute pancreatitis, and have been thoroughly investigated for a recognized cause of pancreatitis. Those with chronic pancreatitis or pancreas-type pain are much less likely to benefit. Recurrence of symptoms is common, and repeat therapy involving the minor ampulla may be required. More recent data showing an association between functional genetic anomalies and PD may begin to explain why few patients with PD experience pancreatitis. Genetic testing in those with symptomatic PD is likely to become more commonplace.
Annular pancreas is a rare congenital abnormality in which the head of the pancreas completely encircles the second part of the duodenum ( Fig. 53.4 ) (see Chapter 1 ). It was first described in 1862 by Ecker, who reported a “ring derived from the head of the pancreas which surrounded the descending portion of the duodenum and was formed by uninterrupted glandular tissue.” , The prevalence in autopsy studies is approximately 3 in 20,000 but is higher in patients undergoing ERCP, at approximately 3 in 1000. Given the difficulty in making the diagnosis during autopsy and the highly selected patient population undergoing ERCP, the true prevalence likely lies somewhere in between.
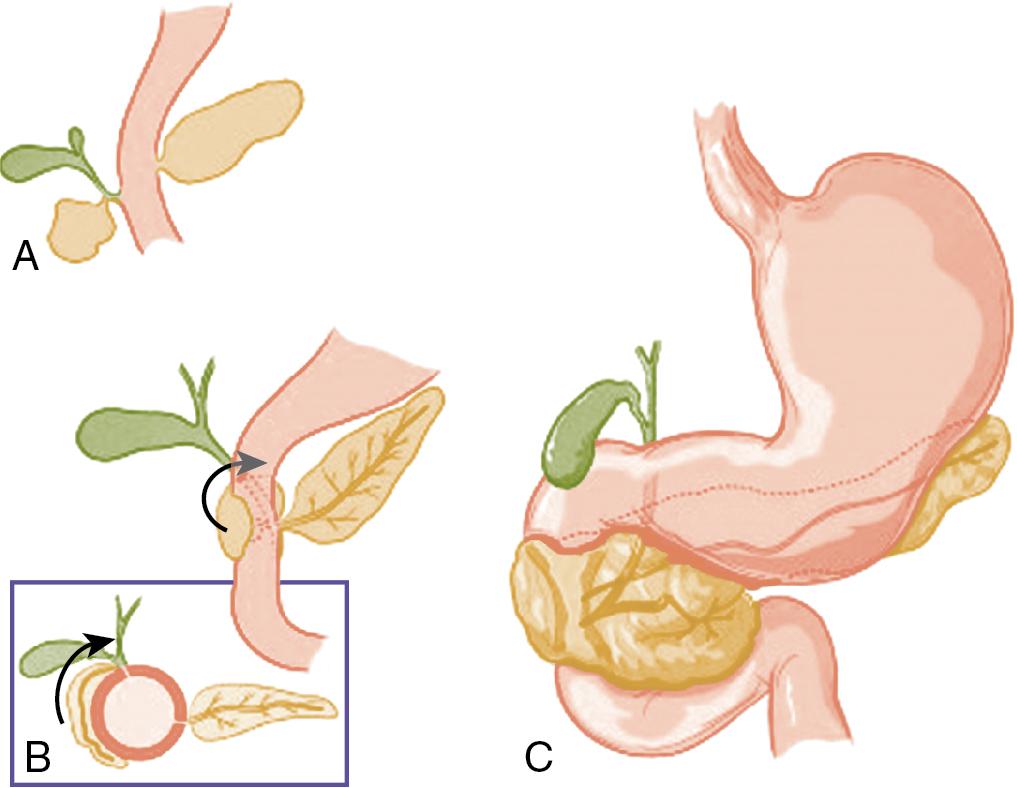
In early development, the ventral pancreas is formed by two buds (see Chapter 1 ). In mammals, the left ventral pancreatic bud is thought to regress, , and the larger right ventral pancreatic bud migrates to take up the position seen in the adult. A number of hypotheses have been proposed to explain the occurrence of annular pancreas. The most persistent is that of Lecco, who suggested that adherence of the right ventral pancreatic bud to the duodenum before gut rotation resulted in a partial or complete ring of pancreatic tissue around the duodenum. A competing hypothesis offered by Baldwin postulates that the left ventral bud persists to form the annulus. Although in most annular pancreas specimens, only one ventral lobe is apparent, reports of a bilobar ventral pancreas have surfaced that support Baldwin’s theory. Neither of these theories explains the variation in the position of the annular duct seen in various specimens, and Kamisawa et al. proposed a new theory that the tip of the left ventral bud adheres to the duodenum and stretches to form a ring. The exact location of this attachment in relation to the bile duct determines the final arrangement of the annular duct.
Reports of familial annular pancreas support a genetic basis for the disease. Annular pancreas has been described in siblings, a mother and three of her children, a mother and son, a mother and daughter, , and a father and son, and a recent report describes the anomaly in monozygotic twins.
Clues as to the genetic basis of the disease are beginning to emerge. Only recently has it been shown that the cells that form the annulus are derived entirely from the ventral pancreas. In the same report, it was demonstrated that in Xenopus embryos, inactivation of transmembrane 4 superfamily member 3 (TM4SF3), a member of the tetraspanin family, inhibited fusion of the dorsal and ventral pancreatic buds. Overexpression of the same gene promoted development of annular pancreas. This would suggest that the product of this gene directly regulates the migration of ventral pancreatic bud cells, although whether it plays a role in the formation of annular pancreas in humans remains unknown.
Another recent study has demonstrated a link between the Hedgehog signaling pathway and the development of annular pancreas. Members of the Hedgehog family of genes promote growth and differentiation of organs, and defects are associated with congenital malformations of the foregut. In 42% of mice embryos with a targeted inactivation of the Indian Hedgehog (IHH) gene, a member of the mammalian Hedgehog family, changes in the morphology of the ventral pancreatic bud similar to annular pancreas were seen. In most, the annulus was complete, although pancreas cells were not seen within the muscularis of the duodenum, as has been described in humans. Annular pancreas is also frequently seen after inactivation of Sonic Hedgehog (SHH), although the incidence of the anomaly is closely related to the background strain of the transgenic mouse, suggesting interaction with other genetic modifiers. SHH is not expressed in pancreatic tissue; therefore it has been suggested that a defect in duodenal Hedgehog signaling may lead to the anomaly because both SHH and IHH are expressed in the developing gut. , SHH knockout mice embryos also demonstrate a number of other abnormalities, including gut malrotation (100%), intestinal transformation of the stomach (100%), duodenal stenosis (67%), and imperforate anus (100%), strikingly similar to congenital malformations seen in humans with annular pancreas (see later).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here