Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Lower gastrointestinal (GI) bleeding is defined as bleeding that occurs distal to the ligament of Treitz. Patients typically complain of hematochezia or blood clots per rectum. However, lower GI bleeding can be further subdivided into small bowel and colonic bleeding for diagnostic and therapeutic purposes. Colonic bleeding, whether acute or chronic, is associated with increased health care use, thus resulting in considerable cost to society.
Lower GI bleeding is a relatively common condition, with approximately 35.7 per 100,000 adults in the United States requiring hospitalization annually. In-hospital mortality rate is between 2% and 4%, usually related to comorbidities and nosocomial infections. Risk factors of mortality include advanced age (>70 years), intestinal ischemia, and comorbid illness. Although spontaneous resolution of bleeding occurs in the majority of cases, a proportion of patients will have ongoing bleeding requiring therapeutic intervention. Predictors of severe bleeding include tachycardia (heart rate >100 beats/min), hypotension (systolic blood pressure <115 mm Hg), syncope, nontender abdominal examination, rectal bleeding in the first 4 hours of evaluation, aspirin use, and more than two comorbid conditions.
Colonic bleeding may be due to multiple factors, including diverticulosis, neoplasia, vascular ectasias, ischemic or infectious colitis, inflammatory bowel disease, and postpolypectomy. Sources of anorectal bleeding include hemorrhoids, neoplasm, pressure necrosis from indwelling rectal tubes, and radiation proctitis. Anorectal sources are usually readily identified with a thorough history, rectal exam, and proctoscopy or flexible sigmoidoscopy; however, a full colonoscopy may be required to rule out a more proximal lesion. The most common etiology of lower intestinal bleeding is carcinoma. However, the most common etiology of acute symptomatic lower GI bleeding is diverticulosis. The American College of Gastroenterology (ACG) Bleeding Registry reported that patients with colonic bleeding were more likely to be male, more likely to use alcohol and tobacco, and were more likely to be on aspirin or other nonsteroidal antiinflammatory agents.
Diverticulosis is the most common cause of lower GI bleeding requiring hospitalization, accounting for 20% to 40% of cases. The prevalence of colonic diverticula is approximately 30% in patients older than 50 years, increasing to 60% in patients older than 80 years. Clinically significant bleeding occurs in 3% to 15% of patients with diverticulosis. Patients typically present with painless hematochezia. Bleeding is usually self-limited, and 70% to 80% of patients will have spontaneous resolution. Only 20% of patients obtain a definitive diagnosis of a diverticular bleed based on colonoscopic findings of active bleeding or a visible vessel or clot. Most diagnoses are presumptive and based on the presence of diverticula in the absence of another identified bleeding source identified at time of colonoscopy.
Colorectal neoplasia (either polyps or cancer) is a common cause of lower GI bleeding, with polyps accounting for 13% and cancers accounting for 8% of patients requiring hospitalization. However, the majority of patients with colorectal neoplasia have occult blood loss and are more likely to present with iron deficiency anemia. A history of weight loss and change in bowel habits is suspicious for neoplasia in a patient with lower GI bleeding. Digital rectal exam should be performed because this will identify rectal carcinoma in a significant proportion of patients. Acute lower GI bleeding due to neoplasia is usually due to ulceration of the tumor surface.
Vascular ectasias of the colon, synonymous with angiodysplasia, arteriovenous malformation, and angioectasia, are the most common vascular lesions of the colon and are a frequent cause of recurrent lower intestinal bleeding after age 60 years. These lesions arise from age-related degeneration of previously normal colonic blood vessels. Vascular ectasias are predominantly located in the cecum or ascending colon. These lesions typically are less than 5 mm in diameter and are multiple in number. Vascular ectasias are usually diagnosed with endoscopy and have a characteristic finding of a small flat red lesion with ectatic vessels radiating from the central lesion. Other less common vascular lesions of the colon may be congenital or related to other syndromes or diagnoses ( Box 156.1 ).
Vascular ectasia
Hemangioma
Congenital arteriovenous malformation
Colonic varices
Telangiectasia
Syndrome-related lesions (e.g., Klippel-Trénaunay-Weber syndrome, Maffucci syndrome)
Others
Vascular spiders and venous stars of liver disease
Degenerative phlebectasia of older adults
Vasculitic lesions
Focal hypervascularity of ulcerative, Crohn, and ischemic colitis
Neovascularity of radiation colitis
Angiosarcoma (e.g., Kaposi sarcoma)
The prevalence of vascular ectasia in the general population is not known, largely because most patients remain asymptomatic. It is estimated that up to 6% of the population older than age 50 years have some form of a colonic vascular lesion. There is no gender predilection, and the majority of symptomatic patients are older than 50 years of age.
Aside from lower intestinal bleeding, vascular ectasia of the colon is predominantly asymptomatic. Patients typically present with recurrent, self-limited episodes of bleeding of varying volumes, ranging from gross hematochezia to occult blood in the stool. Iron deficiency anemia alone occurs in 10% to 15% of patients. This spectrum reflects the varied rate of bleeding from the ectatic capillaries, venules, and, in advanced lesions, arteriovenous communications. Greater than 90% of patients have spontaneous resolution of bleeding. Approximately 15% of patients present with massive hemorrhage. Although the risks of bleeding are not well defined, the number of lesions and presence of coagulopathy or bleeding disorders may predispose to bleeding episodes.
Other causes of colonic bleeding include inflammatory bowel disease, infectious colitis, postpolypectomy bleeding, and ischemic colitis. A careful history will help guide the diagnostic work-up for these etiologies. Severe abdominal pain and bloody diarrhea raises suspicion for inflammatory bowel disease or infectious colitis in a young patient. A similar presentation in an older patient, especially with comorbid cardiovascular disease, would be more consistent with ischemic colitis. Ischemic colitis is the underlying etiology in approximately 7% of patients hospitalized for lower GI bleeding, and will be discussed at the end of the chapter. A history of recent colonoscopy and lower GI bleeding suggests postpolypectomy bleeding. Postpolypectomy bleeding occurs in 8.7 per 1000 procedures and accounts for between 2% and 8% of acute lower GI bleeding.
The diagnostic approach used in patients with evidence of lower GI bleeding varies with age, presence or absence of active bleeding, and the severity of hemodynamic compromise. All patients with lower GI bleeding require a physical examination, including a digital rectal exam. Laboratory testing is performed to evaluate the level of anemia and rule out coagulopathy. In addition, medications should be reviewed looking for nonsteroidal antiinflammatory drug (NSAID), antiplatelet, or anticoagulant use. Further evaluation is dependent on the rate of blood loss. To provide a framework for the diagnosis and treatment of lower GI bleeding based on bleeding severity, the American Society for Gastrointestinal Endoscopy (ASGE) classifies bleeding as occult bleeding, melena, scant intermittent hematochezia, and severe ( Fig. 156.1 ).
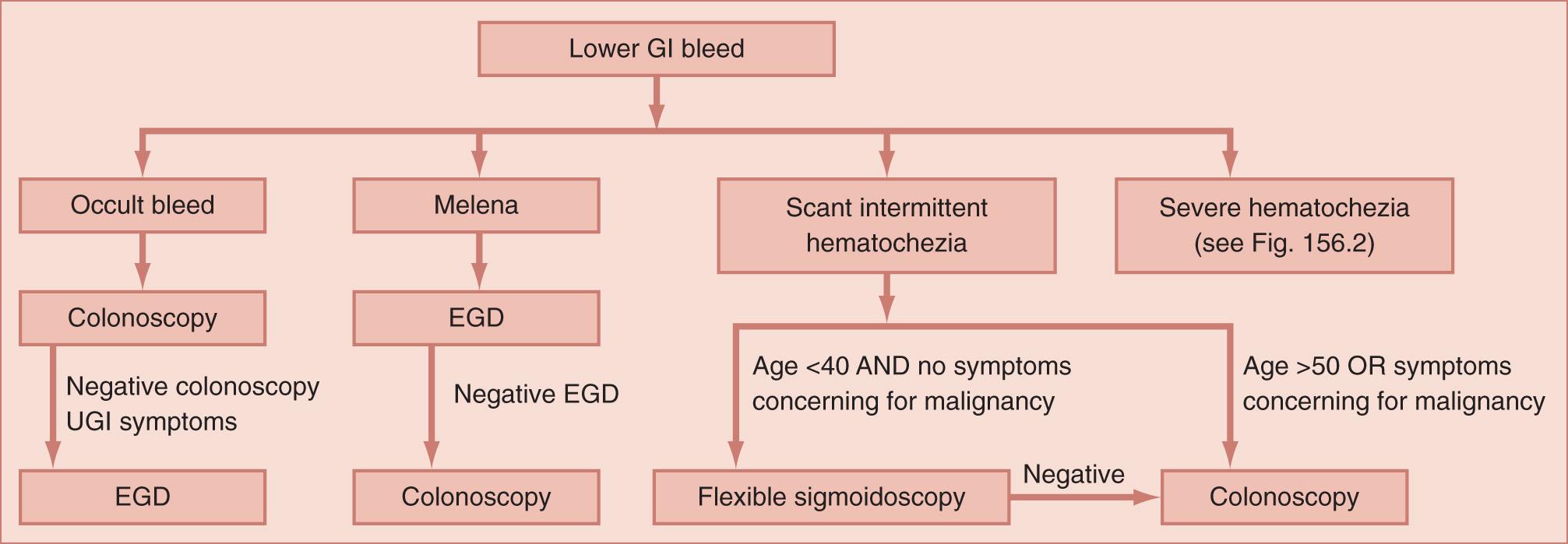
GI bleeding is defined as occult when there is no evidence of gross bleeding. Rather, the patient presents with heme-positive stools or as iron deficiency anemia suspected to be caused by GI blood loss. Patients with occult GI bleeding should undergo colonoscopy to evaluate for colorectal neoplasia. Patients with negative colonoscopy and upper GI symptoms, iron deficiency anemia, or NSAID use should be further evaluated by upper endoscopy. Evaluation of the small bowel may be necessary in patients with continued occult bleeding after negative upper and lower endoscopy.
Melena is defined as passage of dark, black, or tarry stools. Patients with melena should first be evaluated by upper endoscopy because the majority will have an upper GI source of bleeding. A negative upper endoscopy should be followed by colonoscopy to evaluate for a colonic source. Evaluation of the small bowel may be necessary in patients with continued melena after negative upper and lower endoscopy.
The most common pattern of lower GI bleeding is scant intermittent hematochezia, defined as intermittent passage of small amounts of usually bright red blood per rectum. Bleeding may occur daily or weekly, may be associated with bowel movements, and resolves spontaneously. Bleeding in this setting is typically from an anorectal or colonic source. In healthy patients younger than 40 years of age, digital rectal examination, anoscopy, and flexible sigmoidoscopy may be sufficient to identify the source of bleeding. However, complete colonoscopy should be performed in patients aged 50 and older, in patients with symptoms concerning for malignancy (iron deficiency anemia, weight loss, or change in bowel habits), or in patients without a definitive source identified on anoscopy or flexible sigmoidoscopy.
Severe hematochezia is defined as large-volume bright red blood per rectum. Patients with evidence of active bleeding require prompt resuscitation and hemodynamic stabilization with crystalloid intravenous (IV) fluids, blood products, and correction of coagulopathy. This should be followed by maneuvers aimed at the diagnosis and treatment of the causative lesion ( Fig. 156.2 ). A nasogastric tube should be inserted to exclude an upper GI etiology. Bloody aspirate from the nasogastric tube indicates an upper GI source, whereas absence of blood and presence of bile in the aspirate exclude bleeding proximal to the ligament of Treitz. However, a clear, nonbilious aspirate is an indication for upper endoscopy in actively bleeding patients because there may be a lesion distal to a closed pylorus. Patients with evidence of an upper GI source should undergo urgent upper endoscopy.
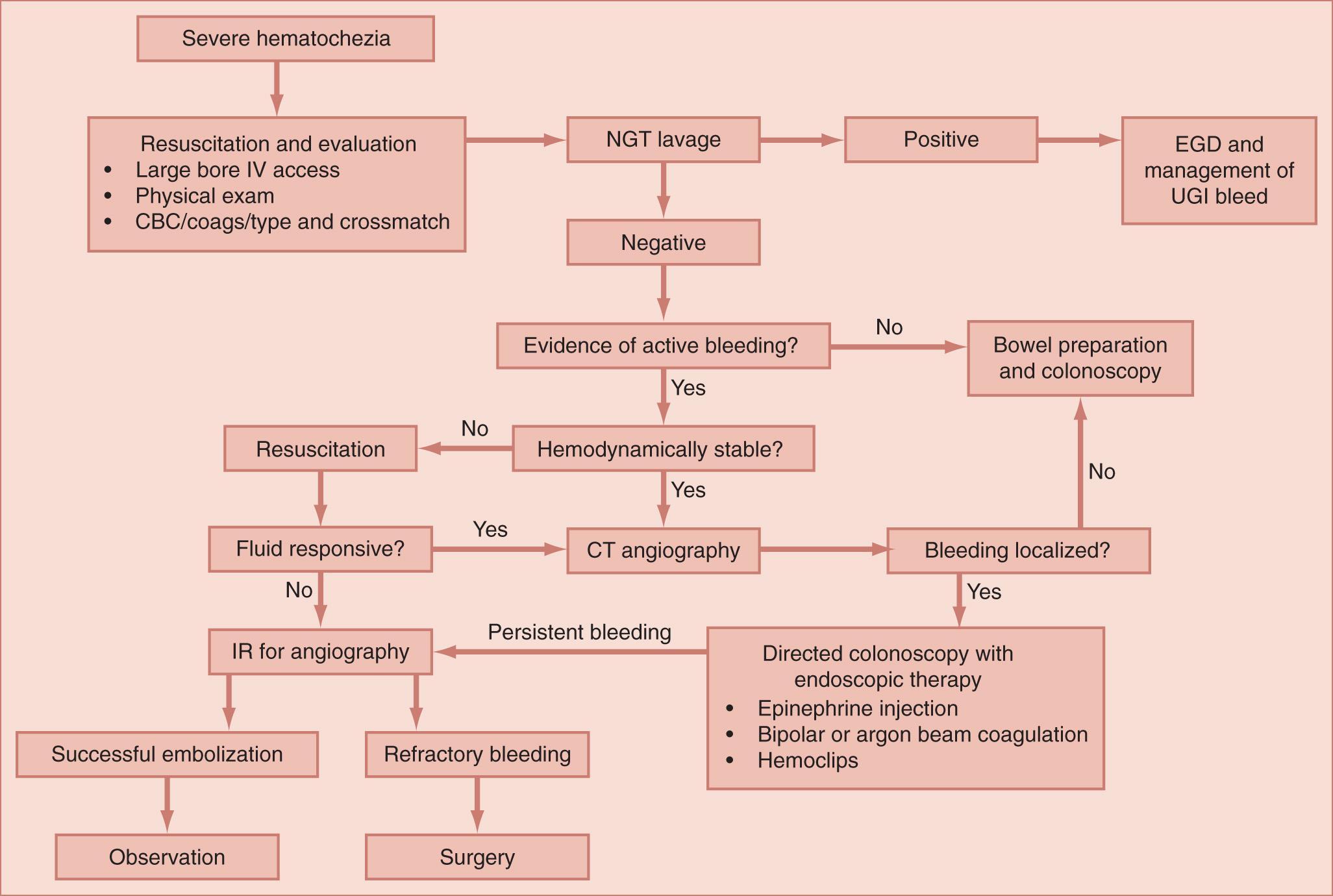
One of the most widely accepted algorithms for management of active lower GI bleeding is urgent colonoscopy following nonbloody return on nasogastric lavage. Colonoscopy has the clear advantage over other diagnostic testing in that it can provide both a diagnosis and potential for therapeutic intervention. Colonoscopy identifies the bleeding source in 45% to 100% of patients, depending on the series. However, the optimal timing of colonoscopy remains controversial in the literature. Most authors agree that early colonoscopy (within 8 to 24 hours of presentation) is likely to provide the most benefit in terms of enhanced diagnostic yield and therapeutic success. In addition, early colonoscopy is associated with shorter length of hospital stay and decreased hospital costs as compared with delayed (>24 hours after presentation) colonoscopy. Prior to colonoscopy, colon preparation is imperative to allow for adequate visualization of the bleeding source.
It should be noted that the patient must be hemodynamically stable to undergo endoscopy. Hemodynamically unstable patients or patients with persistent bleeding despite endoscopic intervention may be better served by mesenteric angiography, with or without a preceding diagnostic radiologic study.
Scintigraphy is a noninvasive nuclear medicine study capable of identifying bleeding over a 24-hour period. However, it cannot anatomically localize a bleed (i.e., distinguish between retroperitoneum, duodenum, or colon) and is unable to treat identified lesions. The radionuclide commonly used to detect intestinal bleeding is technetium 99m ( 99m Tc)-labeled red blood cells (RBCs). 99m Tc-RBCs can reliably detect active bleeding even at rates below 0.1 mL/min, and serial studies can be obtained for up to 36 hours after a single injection of the radionuclide, thus detecting lesions that bleed intermittently. However, with the increasing availability, speed, and sensitivity of computed tomography angiography (CTA), the utility of scintigraphy in the acutely bleeding patient is declining.
Multidetector contrast-enhanced CTA is increasingly being used as a diagnostic tool for the active lower GI bleeding. CTA is available in most hospitals and may be rapidly performed in the emergency department. In addition, CTA can determine whether a patient is actively bleeding and identify the anatomic source, thus guiding therapeutic interventions, such as directed colonoscopy, angiographic embolization, or surgery. CTA is capable of reliably detecting GI bleeding rates of 0.5 mL/min. Chua and Ridley pooled data from eight studies conducted between 1997 and 2007 comparing CT scan with other diagnostic imaging for identification of lower GI bleeding source. The authors estimated a pooled sensitivity of 86% and specificity of 95%. CTA also provides information regarding etiology of the bleed, such as diverticula, tumors, or vascular ectasias. Limitations of CTA include ability to identify only active bleeding, lack of therapeutic capability, and risks of contrast allergy or contrast-induced nephropathy. The utility of CTA in the diagnosis of lower GI bleeding may be maximized by incorporation of CTA into an early branch point of a diagnostic algorithm. In such an algorithm, hemodynamically stable patients with active bleeding in whom an upper GI source has been excluded would undergo CTA. A “positive” CTA reveals the location and possible etiology of lower GI bleeding, leading to a better selection of the most appropriate therapeutic step (angiography, expedited colonoscopy, or surgery). However, a “negative” CTA suggests that the bleeding may be self-limited, allowing for thorough bowel preparation prior to colonoscopy.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here