Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Neuromodulation, defined by the International Neuromodulation Society as “the alteration of nerve activity through targeted delivery of a stimulus, such as electrical stimulation or chemical agents, to specific neurological sites in the body” ( ), has been in use for thousands of years ( ). The pain-relieving effect of stimulation delivered via electric fish was a serendipitous finding, based on anecdotal experience that was passed along verbally and then engraved onto stone tablets. As artificial means of generating and controlling electricity were developed, the use of stimulation for a host of maladies, still based on anecdotal evidence and testimonials, was publicized widely, thanks to the invention of the printing press. Justifiable skepticism accompanied enthusiastic marketing, but just as little good evidence existed to support extravagant claims, there was little good evidence to refute them.
Publication of the “gate theory” of pain transmission 50 years ago provided a rationale for the use of electrical stimulation for the treatment of pain ( ), and concurrent development of solid-state electronics made implanted electronic devices feasible. Spinal cord stimulation (SCS) was introduced in this context in the 1960s, supported by published case reports and small case series ( ), and in a wave of technological zealotry this form of stimulation became the most common application of neuromodulation. By the 1990s, the published scientific literature included reports of case series with hundreds of patients accumulated over two decades with favorable results ( ).
Today, the standards for clinical studies demand much more than case series. Indeed, in the current era of evidence-based medicine (EBM), clinical studies are expected to adhere to certain standards. Entire textbooks and university courses are devoted to this subject, about which this chapter will set forth some general principles, along with special considerations for the design of neuromodulation studies.
Clinical study design should support the inference that treatment and response are, in fact, cause and effect. Mere observation of an association, such as relief of pain by exposure to electricity, does not demonstrate a causal relationship but can generate a hypothesis amenable to testing by clinical trial. A preliminary trial might take the form of a consecutive case series, which can yield useful data on safety and efficacy but can logically demonstrate only associations, not cause and effect. Observed effects might be nonspecific (placebo) or might simply result from regression to the mean ( ). Thus, a study design comparing treatment with control, in parallel groups, is more convincing, that is, of higher quality than observational studies. Early controlled trials relied on alternate allocation of subjects to treatment groups; potential sources of bias using this method, however, were recognized and addressed, first by randomization and then by blinding study subjects and investigators ( ).
Fig. 4.1 shows a typical contemporary randomized controlled trial (RCT) design in the form recommended by the Consolidated Standards of Reporting Trials (CONSORT) group, as developed since the 1990s ( ). The CONSORT checklist includes “25 items selected because empirical evidence indicates that not reporting the information is associated with biased estimates of treatment effect, or because the information is essential to judge the reliability or relevance of the findings” ( ). This chapter highlights those items with particular relevance to neuromodulation.
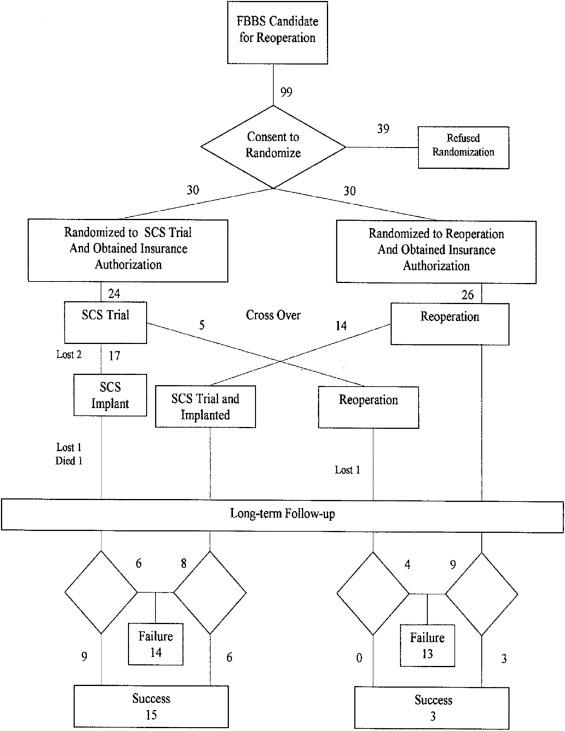
Fig. 4.2 presents the flow chart of a representative RCT ( ), which, although begun before publication of the CONSORT statement, is nonetheless in general conformity with its guidelines. Sample sizes and patient flow as the study progressed are shown plainly within the diagram. An obvious feature of this study, superimposed on basic RCT design, is the inclusion of crossover. In this study, crossover was optional; patients who were not satisfied with the results of their randomized treatment assignment were allowed, after 6 months, to cross over to the alternative, and this decision on their part was considered one of the study outcome measures. This study compared a neuromodulation procedure with a surgical alternative; in general, however, neuromodulation studies lend themselves to crossover trials, comparing one neuromodulation protocol with another delivered via the same device, without any need for additional procedures apart from programming changes. Crossover studies in general can increase statistical power for a given sample size and address some ethical concerns, while potentially raising others ( ).
Many choices of comparator or control are possible: first and most fundamental is incorporating a placebo to show that treatment effects are more than nonspecific. The use of placebo as a treatment arm can raise ethical concerns ( ), but such concerns are substantially addressed by a neuromodulation study in which all patients are implanted and placebo therapy is delivered via a mere programming change. As will be detailed here, however, the usefulness of placebo control diminishes in forms of neuromodulation, producing paresthesia, where blinding is impossible.
Second, an active comparator, such as “optimal medical management” or conventional medical management (CMM) ( ), is commonly used as a control for “comparative efficacy studies.” A comparator that simply continues a previously failed therapy, albeit with that treatment “optimized,” is subject to criticism, even on ethical grounds, but ethical concerns can be addressed substantially by crossover. A comparator that is more invasive and potentially more morbid (e.g., reoperation) than the treatment in the other study arm might also be criticized, except when it is the prevailing norm and the effectiveness of the neuromodulation procedure as an alternative therapy remains to be proved. In general, comparative efficacy studies that include treatments in common use are particularly relevant clinically; they help to address risk-benefit and cost issues. On the other hand, some studies of novel waveforms have used traditional/conventional waveforms as comparators to achieve regulatory objectives; as will be detailed here, the implications of these studies for clinical practice are complex, particularly if the same device can be used to deliver more than one waveform.
A clinical study is only as meaningful as its design, and the composition of a study population is of particular importance, as this determines external validity (the population to which the study results may be generalized). Criteria for patient inclusion and exclusion may be broad; for example, to support a regulatory goal for SCS, a study might simply include subjects with intractable pain of the trunk and limbs, consistent with current labeling requirements. On the other hand, to address a specific clinical question, selection criteria might be narrow; for example, patients with persistent leg (not exceeded by low back) pain following one or more lumbosacral spine surgeries, now candidates for another surgery, with a confirmatory second opinion. Many nuances exist for selection of patients with specific conditions, for example, in the case of pain, minimum pain intensity, pain rating variability, or lack thereof on serial baseline ratings ( ). Details of these fine points are beyond the scope of this chapter. In general, we should not presume that study results may be generalized or extrapolated beyond the study population or design, but in clinical practice this is often done, especially if the ratio of potential benefit to risk is favorable. High-quality clinical studies simply cannot be performed to address all possible clinical scenarios.
Implantation and adjustment of neuromodulation devices involve technical skill, which is important in clinical practice ( ) and must be considered in clinical study design and interpretation. Favorable results of a study by the inventors and developers of a new procedure, demonstrating its safety and efficacy, cannot be applied to the general population as readily as, for example, a pharmacologic trial that is not dependent on procedural skill ( ). Even multicenter trials, which are assumed to be more representative of general clinical practice than single-center trials, are not necessarily so.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here