Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
By the end of this chapter the reader should:
Understand the different research settings and the phases of clinical trials
Understand how to develop a research question and frame this as a null hypothesis to be tested
Be aware of the regulatory bodies and processes involved in trial design and approval
Understand how to enrol a young person on a clinical trial
Be aware of the monitoring requirements of a clinical trial
Have a basic understanding of the approach to statistical analysis of trial data
Appreciate the importance of trial reporting and the role that prospective registration of clinical trials has in this process
There has been a dramatic improvement in child health outcomes throughout the second half of the last century. This has mainly been driven by improvements in nutrition, housing, child safety, immunization and other public health developments. These important approaches are now being complemented by clinical trials, fuelled by the development of new ‘omics’ technologies, which are uncovering the molecular origins of disease faster than we are able to translate the new information into clinical benefit for patients.
Paediatricians are a critical component in ensuring that our ever improving understanding of fundamental molecular biology is translated into improvements in the health of young people. This requires that paediatricians continue to identify, and seek to answer, key questions relating to childhood development and disease. Identifying clinically relevant problems, framing questions and searching for answers from established research is at the heart of evidence-based medicine (see Chapter 39 , Evidence-based paediatrics). However, many questions remain unanswered. Designing studies to address these unanswered questions is the essence of clinical research.
This chapter aims to introduce clinical research, providing a broad understanding of what underpins clinical trial conception and design, the process of obtaining trial approval, recruitment, monitoring and finally reporting of trial findings.
Research into the causes of childhood illness underpins much of what we see as standard practice today. However, many of our daily clinical decisions are based on little or no evidence. Children are not small adults, but individuals with specific and ever changing developmental and physiological needs. Despite this, paediatricians are often obliged to extrapolate from adult trials and to prescribe drugs for which there is little clinical trial data from children. Challenges facing the development of research for young people are listed in Box 37.1 .
Disease rarity – compounded by the increasing molecular subcategorization of conditions
Consent required on child's behalf, often during distressing periods
Consideration of risk vs benefit by parents on child's behalf
Multiple sampling of blood or tissues
Repeated follow-up causing intrusion into family/school life
Concordance with therapy and follow-up amongst young people
Transition to adult services research setting affecting long-term follow-up
How, then, can child-specific clinical research be driven and what does it offer to the health of young people? Paediatricians involved in caring for children with rare, complex or life-threatening disorders are constantly looking to understand that disease in more detail, to identify high-risk patients and to develop innovative management strategies. For example, molecular characterization of malignant diseases has opened the door to the application of targeted therapies in place of non-selective cytotoxic chemotherapies. Cellular and genetic manipulation of donated stem cells will reduce the risk of rejection or graft-versus-host disease in recipients. Delivery of the wild type CFTR gene will reconstitute normal function, preventing the destructive lung disease seen in cystic fibrosis. However, the majority of child health research seeks to provide evidence relevant to the clinical management of children with more common diseases, forming the evidence base from which we develop our practice, and is the domain of all paediatricians ( Box 37.2 ). Whether as chief investigator, recruiting patients or as someone committed to improving local practice, understanding the principles and practice of clinical research is a key component of paediatrics. All paediatricians have a responsibility to improve children's healthcare and research is an essential part of this process.
Framed PICO question: In a pre-school child with wheeze associated with a viral upper respiratory tract infection [patient], is oral steroid (e.g. prednisolone) [intervention] more effective than placebo [comparison] in terms of time to resolution of symptoms, likelihood of admission and deterioration and side effects [outcomes]?
Study design: Randomized, double-blind, intention-to-treat, placebo-controlled trial comparing the role of a short (5-day) course of prednisolone in 700 children aged between 10 months and 60 months. The stated primary outcome measure was duration of hospitalization, with secondary outcome measures assessing symptom severity and salbutamol use.
Results: The study demonstrated that there was no difference in any of the stated outcome measures between children who received prednisolone and those who received placebo. This included a sub-analysis of children at risk of asthma (e.g. previous wheeze, dermatitis or parental asthma).
Discussion: Whilst this trial was conducted in a tertiary paediatric environment, it sought to address a common problem where there was no evidence to inform practice. Its impact comes not from high technological science, but from the importance of the clinical problem across tertiary, secondary and primary care settings. The use of a randomized, blinded and placebo-controlled design was critical in this study as many of the measured endpoints included a degree of subjectivity. This included the primary outcome measure, duration of hospitalization, as the decision to discharge a patient is based on clinician decision and therefore open to bias if the supervising clinician knew the allocated treatment. Equally, secondary outcome measures of degree of respiratory distress, total dose of inhaled β 2 agonist administered in hospital and following discharge and mean 7-day symptom score all include an element of subjectivity. The use of a placebo was essential to maintain blinding.
Reference: Panickar J, Lakhanpaul M, Lambert PC, et al . Oral prednisolone for preschool children with acute virus-induced wheezing. New England Journal of Medicine 2009:360;329–38. ISRCTN58363576.
Medical research is conducted across a number of different settings. Different components flow, in two directions, with clinical problems and observations being investigated at a fundamental level and improved fundamental knowledge being applied to the patient setting. The resultant interplay of ideas, questions, solutions and new questions ( Fig. 37.1 ) is what defines the process of medical research. Whilst the boundaries can merge, broadly the style of research being conducted allows these different settings to be considered separately.
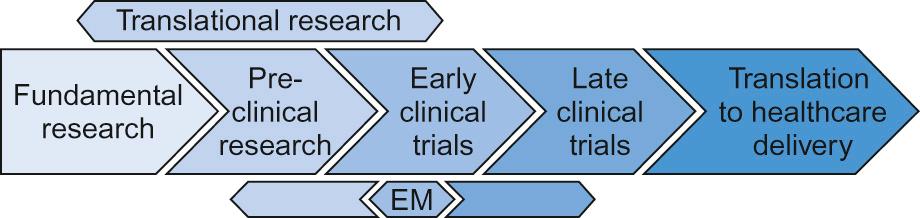
Fundamental (or basic) research seeks to develop our understanding and knowledge of the genetic, molecular, environmental or societal basis of disease. Examples include:
Investigating the cardiovascular or central nervous system effects of neonatal asphyxia using an animal model
Identifying the cytogenetic abnormalities found in acute lymphoblastic leukaemia
Identifying the underlying cause of the abnormally thick secretions seen in cystic fibrosis – characterization of the cystic fibrosis transmembrane regulator provided a pathological mechanism which, in turn, may offer a therapeutic option.
Translational research is a two-way process which provides a bridge between fundamental research and applied clinical research and adheres to the philosophy of ‘bench-to-bedside and back again’. In the forward direction, it aims to generate supportive pre-clinical data prior to investigating the clinical application of:
One of a number of different potential clinical biomarkers ( Box 37.3 )
Prognostic biomarkers – used to stratify patients according to the prognosis of their disease subtype. In childhood acute lymphoblastic leukaemia, cytogenetic analysis identifies patients at a higher risk of treatment failure, e.g. t(9;22) Philadelphia chromosome.
Predictive biomarkers – predict a patient's response to a particular treatment. Mutations of Kir6.2 causing infant-onset diabetes insipidus predicts sensitivity to sulphonylureas ( Box 37.4 ).
Scenario : You are a paediatrician working in a tertiary paediatric diabetes service.
Framed PICO research question : In children with diabetes due to mutations within the ATP-sensitive potassium channel [population], is oral sulphonylurea [intervention] as effective as standard insulin replacement [control] in maintaining or reducing HbA1c [primary outcome measure] without additional episodes of hypoglycaemia [secondary outcome]?
Method: This interventional cohort study looked at the management of 49 patients with diabetes due to mutations within the ATP-sensitive potassium channel.
Results : A significant reduction in HbA1c from 8.1% pre-treatment to 6.4% after 12 weeks of treatment (p < 0.001). Forty-four patients were able to stop insulin treatment.
Discussion: This collaborative multinational study demonstrated a safe and more effective way of managing diabetes resulting from this rare group of mutations. The patients in this study, all of whom were established on ‘standard’ insulin therapy, provided their own controls for the primary outcome measure of HbA1c, which can be assessed using a paired Student's t-test (see Chapter 38 , Statistics). Assuming that all patients were stable prior to enrolment, this approach provided the most appropriate trial design. It maximized the opportunity to study the effect of an intervention in a small population of patients with a rare condition. Randomizing patients between ongoing standard care and investigational treatment would have reduced the number in the trial therapy arm, thereby reducing the power of the study. However, a crossover approach would clearly not be appropriate in acute conditions where baseline measurements cannot provide stable data, or in life-threatening conditions where two interventions – usually current standard of care and new intervention – must be directly compared. In that situation, randomization would be most appropriate.
Reference: Pearson ER, Flechtner I, Njølstad PR, et al. Switching from insulin to oral sulfonylureas in patients with diabetes due to Kir6.2 mutations. New England Journal of Medicine 2006:355;467–77.
Response biomarkers – provide a surrogate measure of a patient's disease status and response to the chosen therapy – fever or C-reactive protein in infection.
Pharmacokinetic biomarkers – used to assess the therapeutic or toxic effects of a drug. Antibiotics such as gentamicin or vancomycin will commonly have drug levels monitored.
Imaging biomarkers – non-invasive imaging, e.g. CT or MRI, may provide prognostic or response biomarker information.
Novel therapeutic targets – if the CFTR gene is mutated in cystic fibrosis, is it possible to deliver a functionally normal gene to the airways and does this result in a biological improvement, e.g. in secretions?
In reverse, it aims to identify:
Important clinical problems and frame them as research questions for fundamental investigation
Unusual clinical situations as routes for fundamental science to develop learning of basic processes – identification of recessive genetic causes of primary immunodeficiency can identify critical elements in development of immunity.
A phase III clinical trial is opened to assess the benefit offered by a new insulin pump in children. The primary endpoint of the trial is the change in their HbA1c.
In this setting, HbA1c is an example of what kind of biomarker? Select ONE answer only.
Imaging biomarker
Pharmacokinetic biomarker
Predictive biomarker
Prognostic biomarker
Response biomarker
E. Response biomarker.
The HbA1c is being used as a surrogate measure of glycaemic control, and thus provides a measure of the effectiveness of the intervention.
Clinical research seeks to take potential diagnostic, monitoring or therapeutic strategies, identified by basic and translational research, into a representative clinical setting. It asks the question, ‘Does this treatment improve the health and well-being of real patients?’ Addressing this question will normally be performed in steps, or phases, with different aims at each phase ( Table 37.1 ). Phase I and II ( early phase ) trials recruiting small numbers of patients and focusing on determining the safety and appropriate dosing/schedule of a new intervention, may be combined so that patients are initially recruited to a phase I element followed by progression to a phase II element. This is increasingly common in trials of new therapies designed specifically for rare patient or disease/molecular subgroups.
| Phase | Numbers recruited | Primary aims | Secondary aims |
|---|---|---|---|
| Phase I | <30 |
|
Understand the pharmacology of the drug in humans |
| Phase II | <100 |
|
Determine optimal supportive care for side effects |
| Phase III | Hundreds to thousands |
|
Ongoing safety monitoring |
| Phase IV | Hundreds to thousands |
|
Increasingly, experimental medicine (see Fig. 37.1 , and see Personalized medicine, below) seeks to use human trials to generate pre-clinical data. For example, a clinical trial with a primary aim of investigating the effect of a new cytotoxic anti-cancer drug can also be used to assess the pharmacokinetics/pharmacodynamics of that drug and the effect on similar, potentially cross-reactive pathways in normal tissue. These studies cannot be performed in vitro or in healthy human volunteers due to the potential toxicity.
A number of different quantitative trial designs exist. Whilst the randomized controlled trial is frequently seen as the gold standard approach for investigating a new treatment intervention, each of the designs can be the most appropriate choice for a given clinical setting ( Table 37.2 ).
The following (A–E) is a list of trial designs:
Case-control study
Cohort study
Cross-sectional study
Ecological study
Randomized controlled trial
Match the trial designs to each of the studies below:
Given current theories on autism and the finding of low-level contamination of drinking water specifically from surface sources with pharmaceuticals including oestrogenic compounds and other pollutants, researchers questioned whether drinking water might be a common source of exposure for numerous potential risk factors for autism spectrum disorders. The study collected secondary data on county-level autism prevalence in the USA and data on the percentage of drinking water derived from surface water sources for each county from publicly available data sources.
Mothers were identified during attendance at an antenatal clinic and divided into two groups: those who smoked during pregnancy and those who did not. Following delivery, all children were followed up for five years with yearly questionnaires about a variety of developmental outcomes.
Families attending an outpatient clinic were approached and asked to complete a survey regarding the amount of physical exercise undertaken each week. During the same clinic, each child was weighed and their height measured in order to evaluate associations between physical activity and overweight.
Following recruitment, children were allocated to receive either the current standard first-line treatment for epilepsy or a new medication under review. Investigators, unaware of which medication each participant was taking, analysed the outcome data with regards to seizure control.
To assess and compare the oral health status of pre-school children with and without cerebral palsy (CP), pre-school children with CP were recruited from special child care centres and a gender-matched sample of pre-school children from mainstream pre-schools were recruited as the control group.
D. Ecological study.
B. Cohort study.
C. Cross-sectional study.
E. Randomized controlled trial.
A. Case-control study.
See Table 37.2 for discussion.
| Trial design | Approach offered | Most suitable if | Example research question |
|---|---|---|---|
| Randomized controlled trial (RCT) | Patients are randomly assigned to receive either standard intervention or the new ‘trial’ intervention | A new treatment is being compared with the current best treatment. This approach allows comparison of efficacy and toxicity whilst minimizing sources of bias. |
PREDNOS trial: In children with new onset nephrotic syndrome [patient], is 16 weeks of prednisolone [intervention] more effective than 8 weeks [control] at reducing relapse rate [outcome]? |
| Crossover trial (variation of RCT) | Individuals provide both control and experimental arm by sequentially receiving multiple/all interventions at different time-points, interspersed with periods for drug ‘wash-out’ | The new treatment is for a chronic condition and aims to control symptoms Is unsuitable/unethical for an acute condition where life-saving treatment is on trial |
In adolescents with type 1 diabetes [patient], does pump therapy [intervention] compared to basal-bolus [control] reduce nocturnal hypoglycaemic episodes [outcome]? |
| Cohort | Children and young people in a particular setting are recruited and followed up to determine outcome | The effect of an exposure or predisposition to a condition is being investigated May be used, especially in early phase trials, where controlling with a placebo is unfeasible or unethical ( Box 37.4 ) |
In infants [population], does supine sleeping [cohort 1] compared to prone sleeping [cohort 2] lead to a high frequency of sudden infant death [outcome]? (See Box 37.4 .) |
| Case-control | Children and young people with a condition are compared with a control group without that condition. Controls must be matched for a number of characteristics, such as age, gender, socio-economic group | Predisposing/risk factors are being investigated. By comparing well-matched cases and controls, the differences between the groups can identify such risk factors. | In infants with pyloric stenosis [case group] compared with a matched group [control group], is there higher exposure to erythromycin? |
| Ecological | Data generated from a geographically (usually) defined population to identify risk-modifying factors on health outcomes | Able to identify available information about the population | Map of skin cancer deaths and sun exposure |
Whilst most clinical research revolves around the quantitative analysis of clinical criteria, the interrogation of qualitative information can also play an important role, both in informing quantitative clinical trial design and in influencing clinical service design and provision.
Qualitative research ( Box 37.5 ) aims to develop understanding of a defined area by collecting information, analysing it and using the output to generate new ideas or hypotheses which may or may not then be suitable for quantitative analysis. Information collecting may involve:
Review of documented evidence
Observational approaches – recording uninfluenced behaviours
Interviews – usually open-ended and defined by topics rather than specific questions
Group discussion – specifically focusing on the interactions of the group setting
Scenario : A 14-year-old boy is admitted to your ward. He has ‘brittle’ asthma. It is the fourth time he has been admitted this year. GP records demonstrate that he is not collecting his prescriptions. You have a large number of similar adolescent patients and want to investigate compliance in adolescent patients.
Research question : ‘To understand better the reasons for non-compliance in adolescents with asthma.’
Research study : In-depth interviews with a sample of 49 adolescents, aged between 14 and 20 years. All adolescents were diagnosed as asthmatic more than a year previously and were attending a hospital asthma clinic. The interviews focused around the adolescents' feelings about their illness and their illness-related behaviour, including self-management.
Key results : Reasons given for non-compliance with prescribed medication in the past or at present were: forgetfulness, belief that the medication is ineffective, denial that one is asthmatic, difficulty using inhalers, inconvenience, fear of side effects, embarrassment and laziness.
Research implementation : The results are implemented by improved education, including a peer education initiative.
Reference : Buston KM, Wood SF. Non-compliance amongst adolescents with asthma: listening to what they tell us about self-management. Family Practice 2000;17(2):134–8.
Whilst outcome measures are not restricted in qualitative studies, the study aim, methodology and analysis are no less rigorously defined and validated than in quantitative approaches. Commonly a number of validated methods will be used independently by more than one researcher and the results triangulated to identify areas of agreement, providing a high degree of validity.
The following (A–J) is a list of research settings:
Experimental medicine
Fundamental research
Phase I clinical trial
Phase I/II clinical trial
Phase II clinical trial
Phase III clinical trial
Phase IV clinical trial
Pre-clinical research
Research for patient benefit
Translational research
Choose the most appropriate research setting to address each of the problems below:
Regular intravenous antibiotics form an important part of the management of children with cystic fibrosis. However, these require frequent admissions to hospital and therefore disrupt children's social development and education. Your local respiratory team would like to assess a new home antibiotic delivery programme aimed at improving the experience of this element of care for children and their family.
A new targeted anti-cancer therapy has shown good efficacy in early phase trials in adults with relapsed non-Hodgkin's lymphoma. You wish to determine the tolerability of this agent alongside/in addition to your current standard therapy for aggressive mature B-cell lymphoma. Initially this study will identify the maximum tolerated dose in cohorts of three patients, followed by an extended cohort to look at short-term toxicity.
In order to gain the most information from your Phase II trial of a novel immune-modulatory agent in severe Crohn's disease, you design a number of associated studies to identify the pharmacokinetics of the drug in young people (existing data are derived from adult studies) and the effect on serum markers of inflammation (response biomarkers), and you require that fresh biopsy specimens are collected at subsequent endoscopic procedures for laboratory investigations of immune cell function. This way you aim to get the most information about the efficacy of the new drug.
I. Research for patient benefit. This approach looks at optimizing healthcare service delivery in a patient-centred manner.
D. Phase I/II. Initial identification of maximum tolerated dose (Phase I) is followed by an extended early phase approach to confirm tolerability alongside otherwise very intensive therapies using an extended early phase trial approach.
A. Experimental medicine. This concept aims to maximize the experimental and learning opportunities available during clinical trials of new treatments by use of pharmacokinetic, pharmacodynamic, biomarker and fundamental science studies on patient-derived samples.
The origins of all clinical trials should be an identifiable clinical need . This is one of the key contributions to be made by translational researchers. The process of identifying a need, investigating potential solutions and then validating them is key to framing the clinical research question ( Fig. 37.2 ). Whilst basic scientists may follow lines of investigation which appear interesting in the hope of further defining fundamental elements of biology, a greater expectation must be placed on the question underpinning a clinical trial. Asking a child and their family to consent to novel intervention or additional investigations can only be ethically justified if the benefit outweighs the risk. There must therefore be a reasonable expectation of patient benefit, identified as a clinical need. It may not, for example, be justified to randomize children between two established therapies with an equivalent, well-documented success rate, simply to demonstrate equivalence in a formal setting. If, however, there was a rationale for one treatment being more effective or less toxic than the other, then this would need to be investigated in a randomized clinical trial. The research question becomes:
Treatment effect:
Is treatment A more effective than treatment B in this clinical setting (e.g. In pre-school children with mild chronic asthma [patient], is oral leukotriene antagonist [intervention] as or more effective then inhaled steroid [control] in preventing chronic symptoms and/or acute exacerbations [outcomes])?
Side effects/toxicity:
Is treatment A less toxic than treatment B in this clinical setting (e.g. in pre-school children with mild chronic asthma [patient], does oral leukotriene antagonist [intervention] lead to greater height potential [outcome] than inhaled steroid therapy [control])?
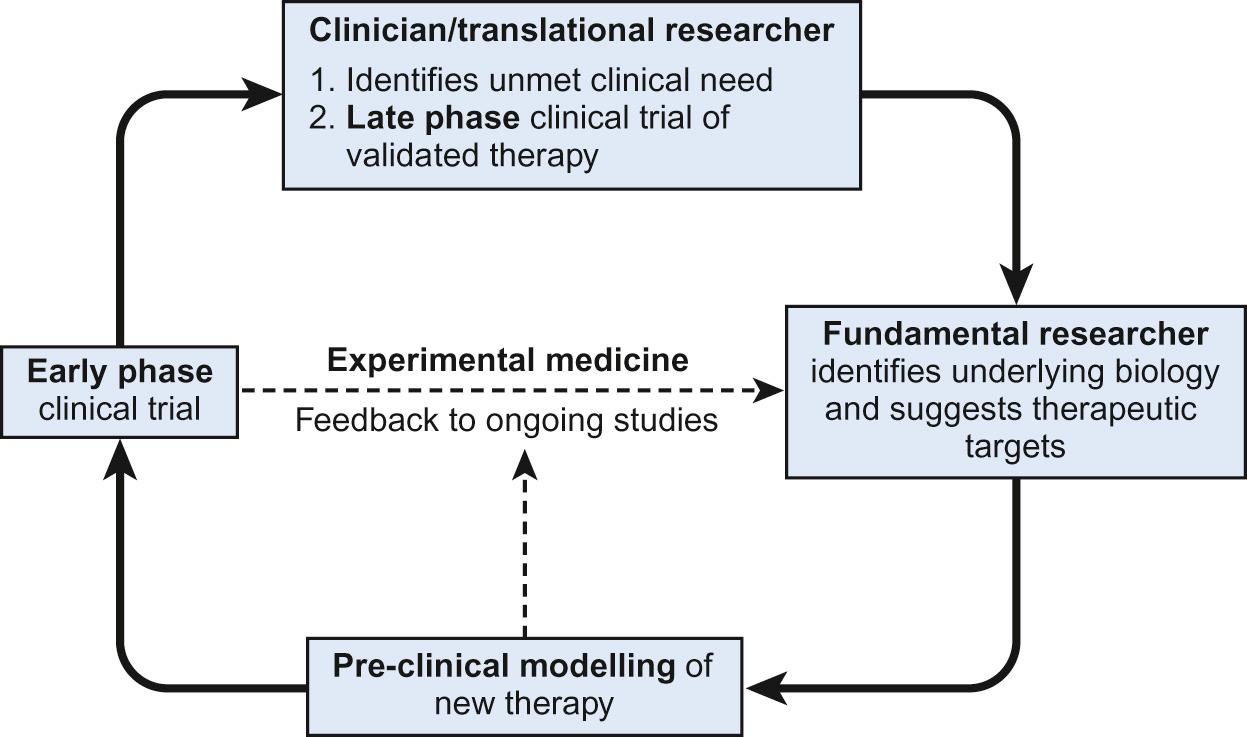
Having identified a clinical need, basic evidence must be sought on how to approach meeting that need. Frequently this will involve a thorough search of the published literature, as there is often a large body of evidence in existence. However, this will often need to be complemented by additional studies investigating the relevance of published knowledge in the specific disease setting. Combined, these data will hopefully provide possible routes for a new intervention aimed at meeting the clinical need.
Having identified a potential novel intervention, this needs to be validated in the most appropriate pre-clinical model available. Some models will involve in vitro drug testing using derived cell lines. However, more complex models, often involving animal research, are frequently used as these may be felt to be more representative of the clinical setting with, for example, metabolism of drugs and complex tissue microenvironments in place of homogeneous single cell cultures ( Box 37.6 ). However, the animal model may only partially represent the human disease and the metabolism and cellular effects of a drug may differ between animal model and humans. One approach to solving this, predominantly relevant to malignancies, is to use human diseases engrafted into immunodeficient animal models – xenografts. It should be noted that even then, factors such as differential metabolism of a drug or the altered microenvironment may limit the clinical relevance of the model.
Scenario: You are a community paediatrician who regularly cares for children with neurodisability secondary to hypoxic–ischaemic encephalopathy (HIE). You want to engage in pre-clinical research to identify new interventions to reduce neurodevelopmental complications in the population.
Research question: In newborn pigs that have undergone induced hypoxia, is there evidence of secondary brain insult despite normal pH, oxygen and glucose levels?
Background: Understanding the mechanism of injury in neonatal asphyxia requires a complex model including developing neural tissue, oxygenation, glucose metabolism and metabolic by-products. Such modelling could not be produced in vitro and therefore animal models of asphyxia were developed to investigate not only the pathological mechanisms, but also the response to a number of treatment options.
Using both newborn pig and newborn rat models of asphyxia, an initial insult to cerebral metabolism is following by a period of normal metabolism. However, a late or secondary cerebral energy failure was identified, despite normal blood pH, oxygen and glucose levels. As the severity of this secondary insult was shown to correlate with the risk of death, severe neurological disability or microcephaly (Roth et al 1992), it provided a possible therapeutic opportunity – a second period during which a protective therapy could be implemented.
Further research stemming from this finding : Further analysis of these models found that whilst infusing magnesium sulphate did not prevent the secondary energy failure (Penrice et al 1997), inducing moderate hypothermia did (Thoresen et al 1995), reducing neuronal apoptosis (Edwards et al 1995) and infarct size (Bona et al 1998). These studies paved the way for further optimization of cooling strategies using models prior to early phase trials investigating the safety of both selective head cooling or whole body hypothermia in newborn infants (Gunn et al 1998, Azzopardi et al 2000, Eicher et al 2005), see Box 37.13 .
References :
Azzopardi D, Robertson NJ, Cowan FM, et al. Pilot study of treatment with whole body hypothermia for neonatal encephalopathy. Pediatrics 2000;106(4):684–94.
Bona E, Hagberg H, Løberg EM, et al. Protective effects of moderate hypothermia after neonatal hypoxia-ischemia: short- and long-term outcome. Pediatr Res 1998;43(6):738–45.
Edwards AD, Yue X, Squier MV, et al. Specific inhibition of apoptosis after cerebral hypoxia-ischaemia by moderate post-insult hypothermia. Biochem Biophys Res Commun 1995;217(3):1193–9.
Eicher DJ, Wagner CL, Katikaneni LP, et al. Moderate hypothermia in neonatal encephalopathy: efficacy outcomes. Pediatr Neurol 2005;32(1):11–7.
Gunn AJ, Gluckman PD, Gunn TR, et al. Selective head cooling in newborn infants after perinatal asphyxia: a safety study. Pediatrics 1998;102(4 Pt 1):885–92.
Penrice J, Amess PN, Punwani S, et al. Magnesium sulfate after transient hypoxia-ischemia fails to prevent delayed cerebral energy failure in the newborn piglet. Pediatr Res 1997;41(3):443–7.
Roth SC, Edwards AD, Cady EB, et al. Relation between cerebral oxidative metabolism following birth asphyxia, and neurodevelopmental outcome and brain growth at one year. Dev Med Child Neurol 1992;34(4):285–95.
Thoresen M, Penrice J, Lorek A, et al. Mild hypothermia after severe transient hypoxia-ischemia ameliorates delayed cerebral energy failure in the newborn piglet. Pediatr Res 1995;37(5):667–70.
Having identified a clinical need, defined the research question and examined the basic and pre-clinical evidence available, the approach to answering the question and resolving the need must be expressed as a hypothesis ( Box 37.7 ). A research hypothesis aims to state a prediction about the outcome of a research study, which can then be experimentally tested. Conventionally this hypothesis is based on the outcome of literature searches and previous, pre-clinical, studies. However, it is not statistically possible to prove the research hypothesis, as the results observed within the study might be due to chance occurrence. Instead, a study's results must be compared against the opposite situation, which is known as the null hypothesis . Evidence which argues against the null hypothesis is evidence in favour of the original research hypothesis, which now becomes known as the alternative hypothesis . Deciding what size of effect the study is looking to identify is an essential component of defining the alternative hypothesis, providing the clinically relevant outcome with which power calculations can be made and data analyses can be performed. Defining the research question and subsequently the research hypothesis is key to determining how the resultant study will be structured, performed and analysed.
Scenario: You are a respiratory paediatrician who regularly treats children with cystic fibrosis (CF).
Framed PICO research question : In children with CF [population], is retroviral delivered normal CFTR gene [intervention] compared to placebo [control] effective in reducing the development of chronic airway damage [outcome]?
Hypotheses:
Research hypothesis : ‘Retroviral delivery of a normal CFTR gene to the airways of children with cystic fibrosis will reduce the development of chronic airways damage.’
Null hypothesis : ‘ Retroviral delivery of a normal CFTR gene to the airways of children with cystic fibrosis will not reduce the development of chronic airways damage.’
Alternative hypothesis : ‘Retroviral delivery of a normal CFTR gene to the airways of children with cystic fibrosis will delay the development of chronic airways disease by X years.’
Consideration must be given to the ability of the study team to deliver a successful trial within a suitable timeframe. The key factors to consider are shown in Box 37.8 . If the expected duration of the study, including necessary follow-up, risks making the delivery of results so delayed that they may no longer be clinically relevant (if, for example, other approaches to management have improved substantially), then it may not be appropriate to initiate the trial. Options for increasing recruitment should be considered, especially widening the trial setting to include national ( Box 37.9 ) or, commonly, international recruitment, the use of alternative trial designs and the sharing of data across trials by prospective or retrospective meta-analysis.
Incidence – including genetic/molecular subclassification
Number of patients (sample size) required to give the study adequate power (see Chapter 38 , Statistics)
Predicted eligibility rates – pathways of referral, additional inclusion/exclusion criteria
Predicted rates of consent – affected by trial design, complexity, additional impact on family and potential risks
Widespread recruitment to clinical trials within paediatric oncology has seen a stepwise improvement in outcome for many childhood malignancies. Improvements in supportive care have been an important part of this development, including:
Criteria for identification of febrile neutropenia and the use of broad-spectrum antibiotics
Improved prophylaxis for Pneumocystis jiroveci and population immunity for measles
Improved imaging – CT, MRI, CT/PET, isotope imaging
Improved surgical/anaesthetic techniques – operating microscopes, coagulation diathermy, peri/intraoperative imaging
More advanced intensive care facilities
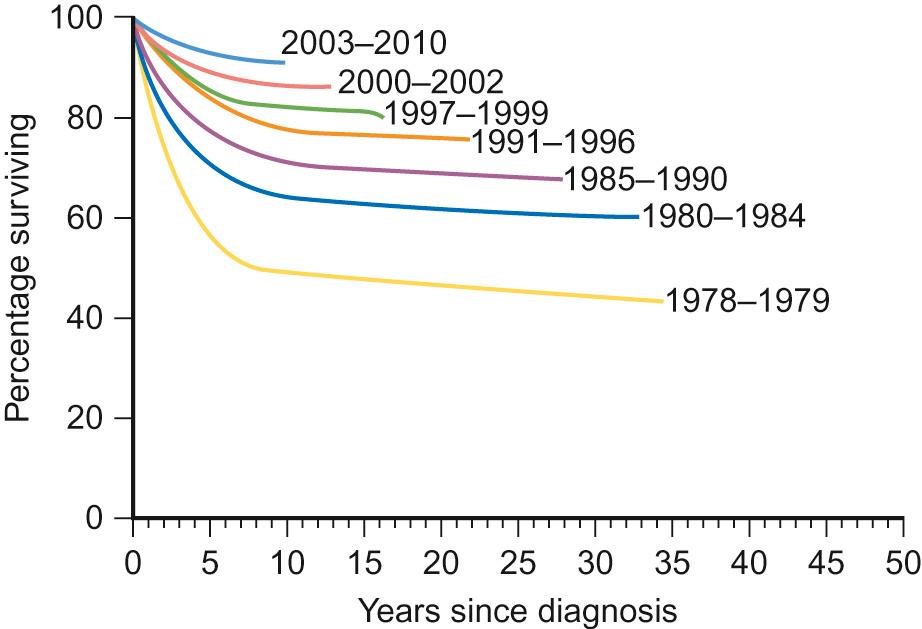
However, many of the clinical trials conducted in childhood malignancy have provided a clear improvement in outcome. This is well demonstrated by the results of UK trials in acute lymphoblastic leukaemia ( Fig. 37.3 ), which have recruited over 8500 children between 1980 and 2011, with the most recent complete trial, UKALL 2003, recruiting over 95% of eligible children:
UKALL VIII – Attempted to reproduce the superior results seen in the US by using the US CCG162 protocol. Inclusion of daunorubicin in induction improved disease control, albeit at the cost of higher treatment related mortality. Extension of maintenance therapy from two to three years improved survival, although again at the cost of increased toxicity. Overall, similar results were achieved compared to the US, but neither randomization demonstrated a clear overall benefit.
UKALL X – Examined the role of post-induction intensification. Inclusion of both early and late intensification phases was better than one or no intensification phase. Five year event-free survival was 71% with 2 intensification phases, 62–63% with a single intensification and 57% without intensification.
UKALL 97/99 – This trial demonstrated the superiority of dexamethasone over prednisolone, stratified patients according to white cell count and age at diagnosis: standard risk – age <10 years, white cell count >50 × 10 9 /L; high risk – age >10 years or white cell count >50 × 10 9 /L. The UK trial again adopted an apparently superior US approach with longer intensification blocks. Introduction of more extensive and sensitive cytogenetic analysis to identify high risk groups.
UKALL 2003 – Stratified children according to minimal residual disease analysis (MRD), a molecular test for low levels of residual disease not detectable by traditional bone marrow analysis. Low risk MRD allowed for reduction of treatment intensity whilst high risk MRD resulted in escalation of treatment.
Further reading :
Hargrave DR, Hann II, Richards SM, et al. Medical Research Council Working Party for Childhood Leukaemia. Progressive reduction in treatment-related deaths in Medical Research Council childhood lymphoblastic leukaemia trials from 1980 to 1997 (UKALL VIII, X and XI). Br J Haematol 2001;112;293–9.
Mitchell C, Richards S, Harrison CJ, Eden T. Long-term follow-up of the United Kingdom medical research council protocols for childhood acute lymphoblastic leukaemia, 1980–2001. Leukemia 2010:24;406–18.
One critical component of developing a clinical trial is to give consideration to defining the outcomes to be assessed and the number of patients required to demonstrate whether those outcomes are affected by the intervention on trial. By defining the outcome which the intervention is hoped to produce, statistical analyses can be tailored to address that specific question. Collecting large quantities of data and then asking multiple questions is statistically unsound and will both risk identifying effects resulting from chance and reduce the likelihood of producing a statistically significant result for the most important clinical outcome as statistical significance needs to be adjusted from when performing multiple tests (Bonferroni calculation). Ideally, outcomes should be defined as:
Primary outcome measures – the main outcome(s) under investigation
Secondary outcome measures – additional important impacts of the intervention
Once the nature of the outcome to be assessed is defined, preliminary data on the clinically relevant size and distribution of the effect within the study population can be used to determine the number of patients (sample size) which need to be recruited to the study. This is the power calculation , which will usually be required by regulatory/funding bodies as one part of demonstrating that the study is practicable. The factors required to produce a power calculation are:
The significance level to be used in the analysis – the value against which the likelihood of incorrectly rejecting the null hypothesis will be judged. This will be calculated from the trial data as the p-value .
The magnitude of effect in the study population (either a direct measurement or, preferably, standardized according to the spread of the effect within the population)
The power – the probability of rejecting the null hypothesis when the alternative hypothesis is correct ( Box 37.10 ). Frequently set to be 0.8, this represents an 80% chance of correctly identifying the pre-determined clinically significant effect in the study population.
Scenario: You are a general paediatrician who regularly treats children with pneumonia (see Table 39.6 ).
Framed PICO research question : In a pre-school aged child with pneumonia [patient], are oral antibiotics [intervention] as effective as intravenous antibiotics [comparison] for time to resolution of symptoms, rate of hospital admission, length of stay and rate of complications [outcomes]?
Power calculation: You work with a steering group who advise a difference of more than 20% could not be considered clinically equivalent (magnitude of effect) .
With a 5% level of significance (statistical significance) , 80% power and equivalence defined as no more than a 20% difference ( magnitude of effect ) between treatments of the proportion meeting the primary outcome measure at any time, 98 children would be required in each arm of the trial.
Reference: Atkinson M, Lakhanpaul M, Smyth A, et al. Comparison of oral amoxicillin and intravenous benzyl penicillin for community acquired pneumonia in children (PIVOT trial): a multicentre pragmatic randomised controlled equivalence trial. Thorax 2007;62:1102–6.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here