Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Non-Hodgkin lymphoma (NHL) refers to all malignancies of the lymphoid system with the exception of Hodgkin lymphoma. Development of the lymphoid system is a highly regulated process, characterized by differential expression of a number of cell-surface and intracytoplasmic proteins and antigen receptor gene rearrangements, somatic hypermutation, and class switching. Dysregulation of this orderly process can result in humoral deficiency, autoimmunity, or malignancy. The indolent B-cell lymphomas are mature peripheral B-cell neoplasms that exclude those diseases associated with an aggressive clinical course. Despite differences in cell of origin, molecular biology, clinical presentation, and clinical course, the indolent lymphomas share common features, including frequent localization to the principal lymphoid organs, a propensity for bone marrow (BM) infiltration and leukemic presentation, and generally an indolent clinical course. The classification of NHLs has been a challenge for pathologists as well as practicing physicians. A number of classifications have been proposed over the years, leading to considerable confusion and difficulty in comparison of outcomes of clinical trials performed using different pathologic classifications. The World Health Organization (WHO) lymphoma classification lists nearly 100 different types of lymphoid neoplasm. This classification uses all available information such as morphology, cytochemistry, immunophenotype, and molecular genetics as well as clinical features. The WHO classification does not include the terminology indolent lymphoma . This is a clinical and not a pathologic term and defines those lymphomas that tend to grow and spread slowly and produce few symptoms. Indolent lymphomas represent 35% to 40% of NHLs, and follicular lymphoma (FL) is the most common of the indolent lymphomas.
It is estimated that in 2021 more than 81,560 cases of NHL will be diagnosed in the United States, and more than 20,000 patients will die of their disease, making NHL the seventh most common cancer and the fifth most common cause of death from cancer ( http://seer.cancer.gov/statfacts/html/nhl.html ). NHL is extremely heterogeneous in its molecular pathophysiology, histology, and clinical course, and there are major differences in the incidence of subtypes in different geographic locations and among different racial and ethnic populations. This difference in geographic distribution is particularly striking for FL, and in the Western world, FL is the second most common lymphoma, comprising approximately 20% of all NHLs. The incidence of FL in the United States is approximately 2.7 cases per 100,000 persons per year and in Europe is approximately 3.14 cases per 100,000 persons per year. The incidence appears stable over time. There is no strong sex preponderance. The incidence in Whites is approximately twice that of Black and Asian populations, and the disease appears less common in Central and South America. The incidence increases with age, and the median age of diagnosis is 65 years. Although most cases are sporadic, there is an increased incidence in family members of affected individuals, with a relative risk of 2.3 for siblings of patients. Genome-wide association studies provide evidence for FL inheritance susceptibility. Multiple independent single nucleotide polymorphisms in human leucocyte antigen (HLA) class I and II genes on chromosome 6p21.3 were associated with susceptibility to FL. Furthermore, FL risk was significantly associated with homozygosity at the HLA class II gene loci.
Differentiation of complex environmental factors from true inherited factors remains difficult. The complexity of the epidemiology of NHL mirrors the complexity of the disease and that of the host immune system. Since lymphomas do not constitute a single disease, it should come as no surprise that there is no single etiologic factor. The influence on immune dysregulation of viruses, chemicals, radiation, diet, and aging remains unclear. Immune suppression leads to increased incidence of aggressive lymphomas but not usually indolent lymphomas.
FL ( Fig. 82.1 ) is a neoplasm derived from germinal center (GC) B cells underlined by: histological appearance, with predominant follicular morphology and maintenance of GC architecture; immunophenotypical behavior, with FL B cells expressing GC markers BCL6 + and CD10 + ; and molecular characteristics, with evidence of ongoing class-switch recombination and somatic hyper mutation. Bulk gene expression profiles of FL are closely related to normal GC light zone B cells, therefore, the cell of origin is assumed to correspond with a GC B cell blocked at that stage of differentiation. A recent study examining cells at single cell resolution has shown that a continuum of dynamic cell states coexist within single patient biopsies, suggesting that FL cells have functional plasticity as opposed to being “frozen” at a particular GC stage.
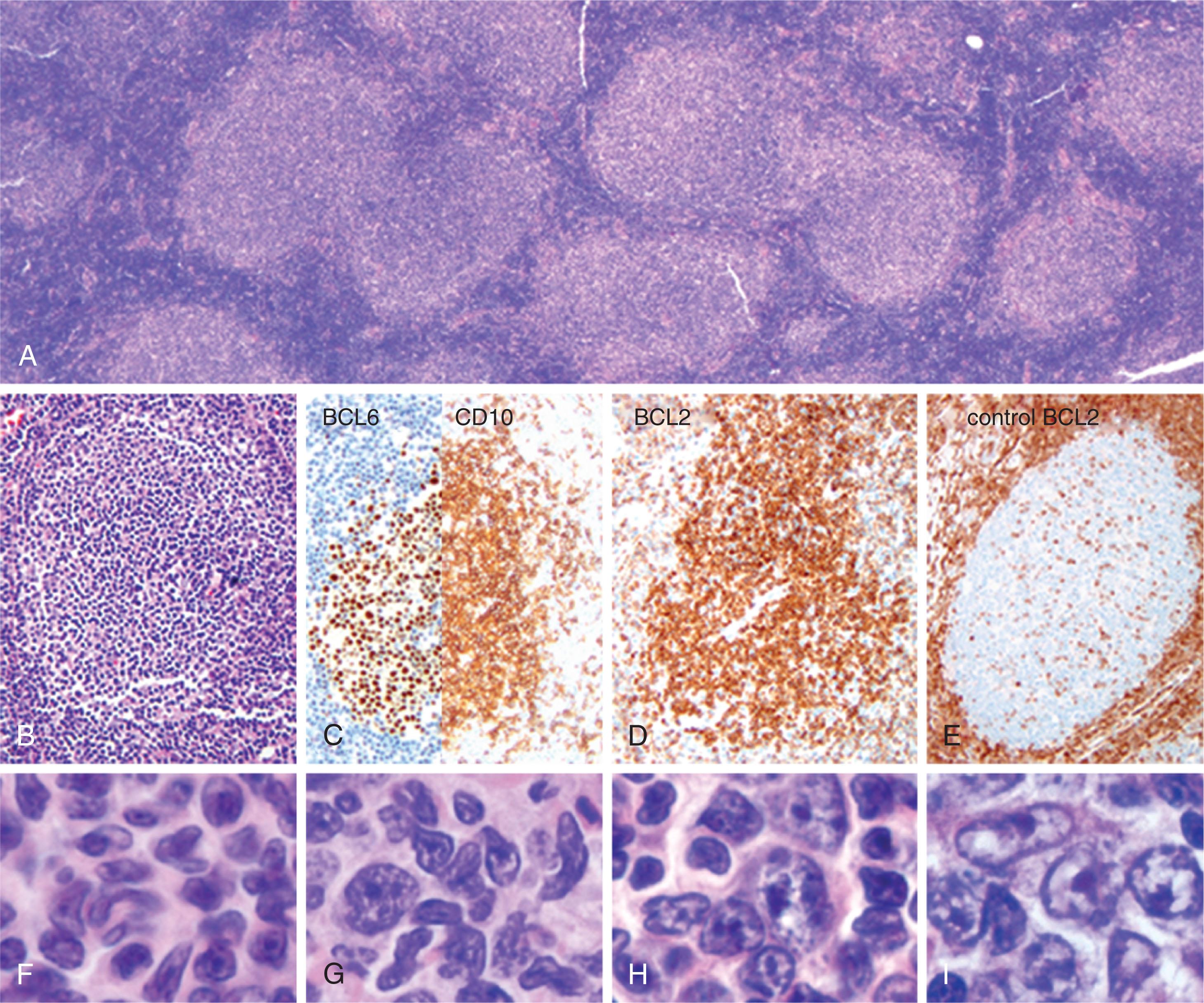
A hallmark of FL is the chromosomal translocation t(14;18)(q32;q21), placing the B cell lymphoma 2 ( BCL2 ) gene under transcriptional control of the immunoglobulin heavy chain ( IGH) locus, resulting in overexpression of the antiapoptotic protein BCL2. Morphologically, the disease is composed of a mixture of centrocytes and centroblasts. The WHO third edition pathologic classification recommends grading in grades 1 to 3 according to the number of centroblasts (0 to 5, 6 to 15, and >15 per high-power field, respectively). Grade 3 was further subdivided into 3 A (centrocytes still present) and 3B (sheets of centroblasts). An increased percentage of centroblasts is predictive of poor outcome. A problem with this classification is that it is poorly reproducible among pathologists. It is also clear that there are no major biologic or clinical differences between grades 1 and 2—whereas grade 3B FL is biologically distinct from grades 1 to 3 A—and clinical and histological features suggest a closer relationship to diffuse large B cell lymphoma (DLBCL). These considerations led to the recommendation to group together FL grade 1 to 3 A as FL and create a new category called FL3B. The final classification combined FL grades 1 and 2 into one category (FL1–2 of 3) and made the distinction between FL3A and FL3B optional rather than mandatory. The fourth edition of the WHO classification recognizes some distinctive clinical and genetic subtypes of FL, including primary duodenal FL and the pediatric type of FL that lacks the t(14;18) translocation and usually presents with localized disease. Several variants of FL that lack the t(14;18) have some distinctive features, e.g., predominantly diffuse FL with deletions of 1p36 that presents with localized bulky disease in the inguinal region. A characteristic finding of t(14;18)-negative FL is an enrichment of activating mutations in the transcription factor STAT6 , occurring in 57% of cases, compared with 10% in t(14;18) positive FL. STAT6 mutations co-occurred with mutations in TNFRSF14 and/or CREBBP , potentially revealing alternative oncogenic mechanisms in this FL subtype.
FL lymphomagenesis is a complex multistep process with the acquisition of the t(14;18)(q32.3;q21.3) reciprocal translocation, generally considered the first hit in the oncogenic cascade. However, there are multiple lines of evidence that t(14;18) in isolation is insufficient to propagate malignant transformation. Firstly, the translocation can be detected at low levels in the blood of healthy individuals. Secondly, BCL2 overexpression mouse models require additional genetic hits to promote overt tumor formation. Finally, t(14;18) negative FL patients follow similar clinical trajectories. Together, these data suggest GC B cells must acquire further oncogenic hits for complete transformation to FL.
The advent of next generation sequencing (NGS) technologies in the last decade—including whole genome, exome and targeted sequencing—have advanced our understanding of the genomic and epigenomic lesions that occur alongside the t(14;18) translocation. Compared to other malignancies, FL has an apparent “addiction” to epigenetic mutations, with greater than 90% of patients carrying one or more mutation in chromatin-modifying genes. The histone lysine methyltransferase KMT2D is the most frequently mutated gene in FL affecting approximately 70% to 80% of cases. Aberrations in the histone acetyltransferase (HATs) enzymes CREBBP and EP300 occur in approximately 70% and 15% of FL cases, respectively. The histone methyltransferase EZH2 forms the catalytic component of the polycomb repressive complex 2 (PRC2) and is mutated in 20% to 25% of FL cases. Other epimutations occur in genes involved in chromatin remodeling, such as ARID1A and the linker histones ( HIST1H1B-E ), occurring in approximately 10% and 30% of cases, respectively.
Inactivating mutations of KMT2D , CREBBP , and EP300 , and gain-of-function mutations of EZH2 , result in a shift from active to repressive marks, leading to aberrant repression of gene transcription and interference with transcriptional programs that regulate normal GC B cell selection, differentiation, and exit. Consequently, aberrant GC B cells accumulate resulting in exposure to further genomic instability, favoring their malignant transformation. Epimutations alone do not appear sufficient to initiate FL but require dysregulation of BCL2 expression to induce lymphomagenesis. Other recurrently disrupted pathways in FL include: immune recognition ( TNFRSF14, CTSS ); BCR-NFκB ( CARD11, TNFAIP3, MYD88 ); mTOR ( RRAGC, ATP6AP1, ATP6V1B2, SESTRIN1 ); and JAK-STAT signaling ( STAT6, SOCS1 ). The major mutations seen in FL are listed in Table 82.1 .
| Gene Name | Abbreviation | Location | Frequency at Diagnosis (%) |
|---|---|---|---|
| Lysine methyltransferase 2D | KMT2D | 12q | 80–90 |
| CREB binding protein | CREBBP | 16p | 33–70 |
| Enhancer of zeste 2 polycomb repressive complex 2 subunit | EZH2 | 7q | 7–25 |
| TNF receptor superfamily member 14 | TNFRSF14 | 1p | 20–50 |
| B cell lymphoma 2 | BCL2 | 18q | 80–90 |
| TNF alpha induced protein 3 | TNFAIP3 | 6q | 5–26 |
| Ras related GTP binding C | RRAGC | 1p | 17 |
| ATPase H+ transporting V1 subunit B2 | ATP6V1B2 | 8p | 11 |
| ATPase H+ transporting accessory protein 1 | ATP6AP1 | Xq | 9.9 |
| H1 linker histone, cluster member | HIST1H1A-E | 6p | 44 |
| AT-rich interaction domain 1A | ARID1A | 1p | 15 |
| Myocyte enhancer factor 2B | MEF2B | 19p | 13–15 |
| Caspase recruitment domain family member 11 | CARD11 | 7p | 8–10 |
| Signal transducer and activator of transcription 6 | STAT6 | 12q | 10 |
| Suppressor of cytokine signaling 1 | SOCS1 | 16p | 8–27 |
| MYD88 innate immune signal transduction adaptor | MYD88 | 3p | 2–9 |
| B cell lymphoma 6 | BCL-6 | 3q | 6–47 |
| Cyclin D3 | CCND3 | 6p | 10–15 |
| Interferon regulatory factor 8 | IRF8 | 16q | 10 |
| Forkhead box O1 | FOXO1 | 13q | 8-15 |
| Tumor protein P53 | TP53 | 17p | 8-31 |
| MYC proto-oncogene, BHLH transcription factor | MYC | 8q | 17 |
| REL proto-oncogene, NF-κB subunit | REL | 2p | 10–33 |
| Cyclin dependent kinase inhibitor 2 A | CDKN2A | 9p | 46 |
| Early B cell factor 1 | EBF1 | 5q | 7–17 |
FL cells exist within a tumor microenvironment milieu of non-malignant immune, stromal, and extracellular components, and are critically reliant on micro-environmental signals for proliferation and survival. A detailed review of the complex interactions between these components is beyond the scope of this chapter, but readers can refer to specific reviews on this topic.
The majority of patients present with lymphadenopathy in one or more sites. Patients with FL often present with a long history of having noted painless, enlarged lymph nodes (LN) over a number of years before presentation. Lymphadenopathy may wax and wane, and spontaneous remissions can occur, albeit rarely. Disease transformation to a more aggressive histologic type confers inferior outcomes, especially early transformation.
Extranodal disease is relatively common and can affect any organ. The most common sites of extranodal disease include the BM, skin, gastrointestinal tract, and bone. Symptoms may be nonspecific or related to the site of disease involvement. Many patients are asymptomatic, but some, particularly those with bulky disease may present with B symptoms defined as fever, drenching sweats, or weight loss of more than 10% of body weight. Patients may present with evidence of bowel obstruction from intraabdominal lymphadenopathy, and retroperitoneal disease may manifest as obstructive uropathy. Inguinal disease may cause compression of the venous system with deep venous thrombosis. Central nervous system involvement can occur, but is uncommon in FL.
Suggested guidelines for the diagnosis of lymphomas have been outlined by the National Comprehensive Cancer Network (guidelines available at http://www.nccn.org/ ) and by the European Society for Medical Oncology. In all cases possible, diagnosis should be confirmed by excisional biopsy of an accessible LN and review by an expert hematopathologist with expertise in lymphoma diagnosis. In patients without easily accessible peripheral nodes, computed tomography (CT) or ultrasound-guided biopsies are typically well tolerated. Fine-needle aspiration is not appropriate for diagnosis in these conditions, and sufficient material must be obtained for adequate histologic review, immunophenotyping, and genetic studies, as required for diagnosis and assay for prognostic markers. When possible, consent should be obtained for the procurement, storage, and use of excess tissue from LN biopsies—at the time of presentation and at each subsequent relapse of disease—for research purposes to investigate the molecular biology of these diseases as new technologies and findings become available.
Initial essential investigations are shown in Table 82.2 . Physical examination should include careful examination of all peripheral LN groups, including the cervical, supraclavicular, axillary, and inguinal chains, and examination of the Waldeyer ring. Abdominal examination should focus on evaluation of any intraabdominal masses, with particular attention paid to detection of enlargement of the liver or spleen. The skin should be carefully examined. Patients may present with pleural or pericardial effusions, although this is less common than in the aggressive lymphomas. Laboratory investigations should include a complete blood count to evaluate for cytopenias, which may be evidence of BM infiltration or of autoimmunity. A white blood count with differential and examination of the peripheral blood smear may elucidate leukemic involvement with disease. Baseline electrolytes including calcium and phosphate, creatinine, and liver function tests are important to determine organ dysfunction that may be related to direct infiltration by lymphoma. Elevation of lactate dehydrogenase (LDH) is an important prognostic factor and may be a useful indicator of transformation from indolent to aggressive lymphoma. Hepatitis B testing is essential as patients will likely require subsequent treatment with anti-CD20 monoclonal antibody therapy.
Whole body PET-CT
|
Staging workup also includes a CT scan of the chest, abdomen, and pelvis, and increasingly, a positron emission tomography (PET) scan to direct sites for biopsy and to help rule out histologic transformation. Particular attention should be paid to sites of bulk disease and to the number of involved sites. BM biopsy provides essential information and should be performed routinely as it is required for staging and prognostication of disease. Liver biopsy may be indicated based upon abnormal imaging or laboratory testing.
The staging of NHL uses the Ann Arbor Classification ( Table 82.3 ). Functional and anatomical imaging with positron emission tomography-computed tomography (PET-CT) is recommended as the standard for staging fluorodeoxyglucose (FDG)-avid nodal lymphomas. FDG PET-CT may detect additional nodal and extranodal sites of disease when compared with standard CT. In a retrospective study, more nodal and extranodal lesions were detected by FDG PET-CT when compared with CT, and 11% of patients were upstaged from limited stage disease (I/II) to advanced stage disease (III/IV). PET-CT staging may also contribute to improved prognosis in patients with stage I disease treated with involved field radiotherapy, likely due to more accurate identification of stage I disease. However, PET-CT is of limited value for the detection of BM involvement. A BM aspirate and biopsy is still required if documentation of BM involvement by FL will have an impact on clinical management.
| Stage | Criteria |
|---|---|
| I | Involvement of 1 lymph node (I) or 1 extralymphatic organ or site (IE) |
| II | Involvement of ≥2 lymph nodes on same side of diaphragm (II) or localized extralymphatic organ or site and ≥1 involved lymph node on same side of diaphragm (IIE) |
| III | Involvement of lymph nodes on both sides of diaphragm (III) or same side with localized involvement of extralymphatic site (IIIE), spleen (IIIS), or both (IIIS+E) |
| IV | Diffuse or disseminated involvement of 1 extralymphatic organ or tissues with or without lymph node enlargement |
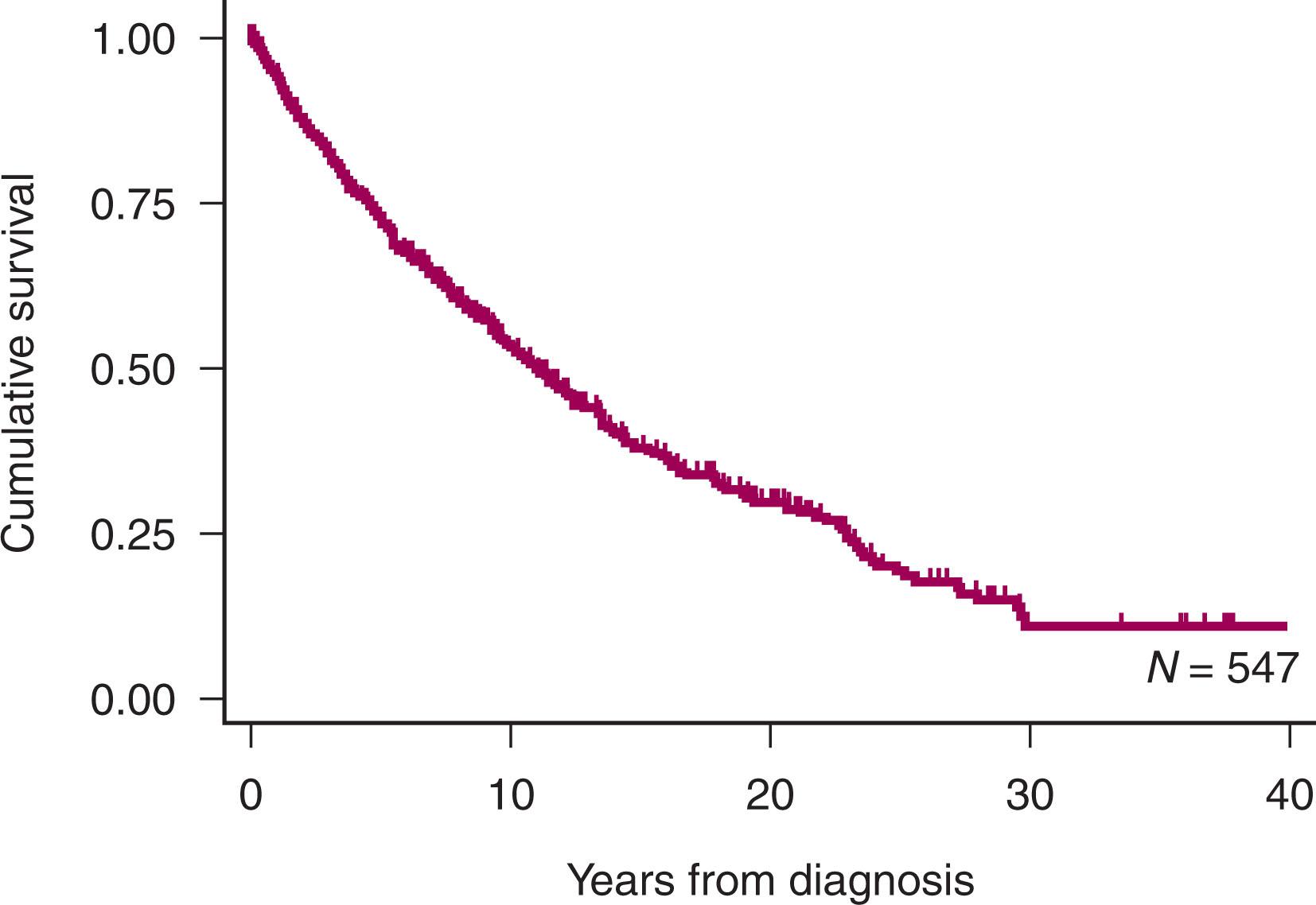
Until recently, there was little evidence that the natural history of FL had changed over the last 30 years from the median survival of 10 years from diagnosis. The survival of patients with FL presenting at St. Bartholomew’s Hospital in London is shown in Fig. 82.2 . The introduction of monoclonal antibodies, in combination with chemotherapy, has led to an improvement in survival. The median overall survival (OS) for FL now extends to 15 to 20 years. However, it is still considered as an incurable malignancy. The natural history of FL is characterized by a protracted, relapse-remitting course, with each disease-free period becoming progressively shorter, leading to eventual treatment refractoriness. Importantly, significant clinical heterogeneity exists among patients, which poses dilemmas for treatment decision-making. Progression of disease within 24 months of initial immunochemotherapy (POD-24) occurs in 10% to 20% of patients and is associated with poor outcomes, with only 50% of patients alive after 5 years. Similarly, transformation to high-grade lymphoma confers inferior outcomes, especially early transformation.
There is, therefore, a need for prognostic markers that can help identify those patients who will have a good or poor prognosis. A number of prognostic scoring systems have been developed, with the aim of dichotomizing patients into groups to identify high-risk patients. The follicular lymphoma international prognostic index (FLIPI) is a five-factor prognostic index based upon the clinical characteristics age, stage, number of nodal sites, hemoglobin, and LDH level ( Table 82.4 ). The index defines three prognostic risk groups of an almost equal numbers of patients. This tool is useful in assessing the likely need for early treatment of patients and potential outcome, as well as in comparing the outcomes of different clinical trials. A revised FLIPI2 (incorporating β 2 -microglobulin, diameter of largest LN, BM involvement, and hemoglobin level) may better discriminate the outcome for patients requiring treatment, with 5-year OS of 96% for low-risk patients, 80% for intermediate-risk patients, and 50% for high-risk patients. Therefore, it is important to include the features required for calculation of FLIPI and FLIPI2 at the time of diagnosis.
| FLIPI | ||
|---|---|---|
| Parameter | Adverse Factor | HR |
| Age | ≥60 years | 2.38 |
| Ann Arbor stage | III–IV | 2.00 |
| Hemoglobin level | <120 g/L | 1.55 |
| Serum LDH level | >ULN | 1.50 |
| Number of nodal sites | >4 | 1.39 |
| FLIPI2 | ||
|---|---|---|
| Parameter | Adverse Factor | HR (in Final Model) |
| β 2 -microglobulin | >ULN | 1.5 |
| BM involvement | BM involvement with disease | 1.59 |
| Hemoglobin level | <120 g/L | 1.51 |
| Largest diameter of LN | >6 cm | 1.66 |
| Age | >60 years | 1.38 |
The m7-FLIPI score combines clinical features (FLIPI and performance status) with the mutation status of seven genes (EZH2, ARID1A, MEF2B, EP300, FOXO1, CREBBP, CARD11) . It was validated for patients receiving rituximab with CHOP or CVP chemotherapy. However, it was not prognostic for patients receiving immunotherapy in combination with bendamustine. In addition, the m7-FLIPI was not prognostic for FL patients who received rituximab monotherapy. GEP 23, a model derived from the expression profile of 23 genes, appears to be able to risk stratify FL patients, although this requires prospective validation. . However, it remains uncertain by what means these prognostic tools can be used to inform clinical decisions on how and when to treat patients. These scores need to be revalidated for use with new therapies, in an ever evolving treatment landscape. In addition, these scores cannot necessarily identify high-risk patients more reliably than simpler baseline assessments, such as total metabolic tumor volume (TMTV) by PET-CT or PRIMA-PI, which uses serum B2M and the presence/absence of BM involvement.
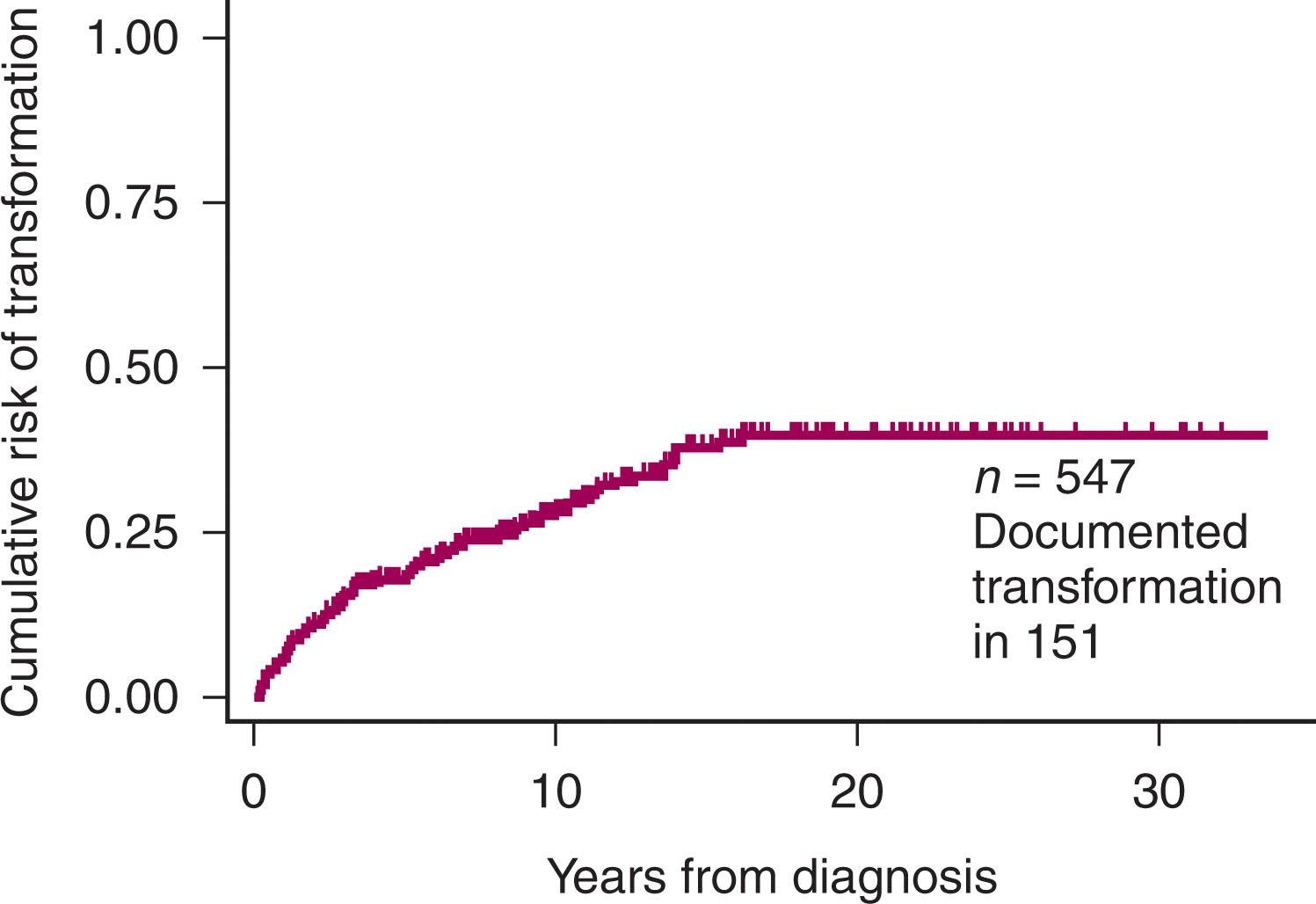
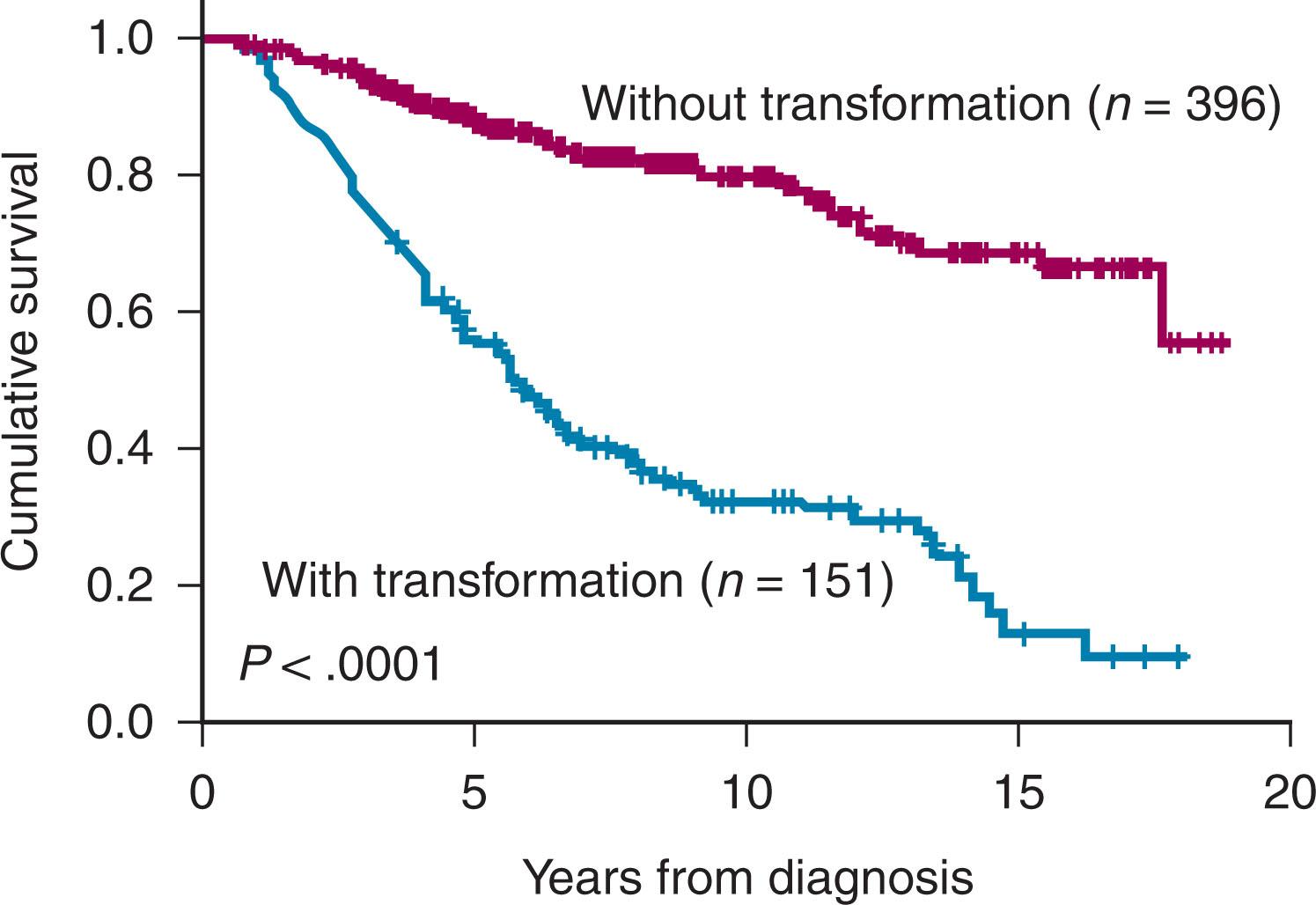
An important factor for the prognosis of FL is whether patients undergo histologic transformation. The actuarial risk of FL undergoing histologic transformation in the database of the patients treated at St. Bartholomew’s Hospital is shown in Fig. 82.3 .The survival of patients with and without transformation is shown in Fig. 82.4 . Despite a considerable body of information on the pathologic and molecular events associated with histologic transformation, the pathogenesis of transformation remains elusive, and the molecular events that have been identified have not been translated into changes in clinical practice. Our current, “one approach treats all” particularly underserves the high-risk FL population. A major research focus is attempting to identify patients at high risk of histologic transformation early in their clinical course, but this is not yet possible. The outcome of patients who undergo transformation after already having received multiple lines of therapy remains poor, but for those patients who undergo transformation and receive their first therapy for the transformed disease, the use of chemo-immunotherapy has led to a significant improvement in prognosis.
For most cases of FL, the goal of therapy has been to maintain the best quality of life and treat only when patients develop symptoms. Any alteration to this approach requires demonstration of improved survival with early institution of therapy, or identification of criteria that define patients sufficiently “high-risk” to merit early therapy. There are many available therapies and no consensus on an optimal first-line or relapse treatment. Despite the lack of any data demonstrating benefit for early therapy, patients are being treated earlier in their disease course. There is no clear-cut treatment pathway for patients with FL, and although we have a good evidence base to decide on a particular treatment, there are little or no data regarding the optimal sequencing of treatment approaches in this disease. In the absence of such data, treatment choices remain empiric and should always involve discussion regarding patient choice and the goal of therapy. This is becoming even more complicated because many novel agents are either approved or are in clinical studies, particularly novel monoclonal antibodies and agents that alter the B cell receptor signaling pathways or anti-apoptotic pathways. The impact of these new agents on practice will be dependent upon the results of the ongoing clinical trials.
Multiple treatment approaches exist for advanced stage low-grade lymphomas, and these patients are best treated in the setting of clinical trials. Options range from a “watch and wait” expectant management approach, to a single agent chemotherapy (rarely used today) or a monoclonal antibody therapy with anti-CD20 antibodies, to a combination chemo-immunotherapy, with use of autologous or allogeneic stem cell transplantation (SCT) or chimeric antigen receptor (CAR) T cells ( Table 82.5 ). Patients remaining on an expectant course should be documented every 3 to 6 months for 5 years, and then annually if stable, with history, physical exam, and blood counts including LDH. Special attention should be paid to any change in symptoms that might be suggestive of transformation, as these are an indication for a repeat biopsy to examine for histologic evidence to confirm transformation. Repeat scanning is not routinely performed unless this is indicated by symptoms or signs.
| Localized Disease |
|
| Advanced Stage Disease |
|
Since there is no clearly defined treatment algorithm for most patients with FL, eligible patients should be included whenever possible in clinical trials. This ensures delivery of optimal care and helps inform design of subsequent trials, hopefully leading to a cure. Information on available clinical trials can be found at http://www.clinicaltrials.gov . (See box on Management of Follicular Lymphoma .)
Patients most often present with asymptomatic lymphadenopathy. The diagnosis should be made by excisional biopsy and reviewed by an expert hematopathologist. In the absence of symptoms requiring treatment or bulky lymphadenopathy, an expectant “watch and wait” approach is the treatment of choice. While in this phase of treatment, patients should be followed every 3–6 months for history, physical, and laboratory examination with radiologic restaging as clinically indicated. Once a decision to treat has been made, there is no clear treatment algorithm and a number of treatment options are available. The treatment goal, whether palliative or potentially with curative intent, is dependent upon the age and performance status of patients. Enrollment in a clinical trial should be the treatment of choice. For younger patients in whom high-dose therapy may be indicated later in their disease course, it is best to avoid profoundly myelotoxic regimens. Rituximab maintenance therapy in first remission given every 2 months for 2 years has been demonstrated to improve progression-free, but not overall survival. The role of maintenance therapy in first remission using interferon-α remains controversial. The choice of therapy after first relapse is also dependent upon the goal of therapy, but is also dependent upon the previous therapy given, response, and duration of response. Autologous or allogeneic stem cell transplantation has a role to play in selected younger patients with this disease, and chimeric antigen receptor (CAR) T cells are now licensed for treatment of multiply relapsed FL.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here