Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Clinical electromyography is a distinct medical discipline that plays a pivotal role in the diagnosis of peripheral nerve and neuromuscular disorders ( ). The designations clinical electromyography (EMG) , electrodiagnostic (EDX) examination , and electroneuromyography (ENMG) are used interchangeably to encompass the electrophysiological study of peripheral nerve, neuromuscular junction, and muscle; the terms needle electromyography and needle electrode examination are reserved for the specific testing that involves needle electrode evaluation of muscle. Although many still refer to all such testing as simply electromyography , use of the word without a descriptor is discouraged because it can be confusing, often implying only the needle electrode part of the evaluation. For clarity, the terms clinical EMG refers to the entire EDX study whereas needle EMG refers to the needle electrode component. These terms are used in this chapter.
The clinical EMG examination is an important diagnostic tool that helps localize a neuromuscular problem at the motor or sensory neuron cell body, nerve root, peripheral nerve, neuromuscular junction, muscle membrane, or muscle. It also helps to establish the underlying process in these disorders and assess their management and prognosis. EDX testing provides the most valuable diagnostic information when the clinical assessment suggests a short list of differential diagnoses. The clinician should perform a detailed or focused neurological examination before referring the patient for a clinical EMG, which in turn serves as an independent procedure to provide an objective assessment of the peripheral nervous system (PNS; ). Patients with complex clinical pictures are best served by neurological consultations prior to performing EDX testing.
The clinical EMG examination is composed of two main tests: nerve conduction studies (NCSs) and needle EMG. These tests complement each other, and both are often necessary for a definite diagnosis. Additional EDX procedures include assessment of F waves, H reflexes, and blink reflexes; repetitive nerve stimulation (RNS); and single-fiber EMG (SFEMG). A focused history and examination will help the electromyographer design the most appropriate EDX study Katirji, 2018; ( ). The electromyographer must be proficient in using modern EDX equipment and applying EDX techniques, know the normal values for commonly and uncommonly examined NCSs and for motor unit action potentials (MUAPs) in different muscles, and be familiar with the specific and nonspecific EDX findings in different neuromuscular disorders.
Electrical stimulation of nerve fibers initiates impulses that travel along motor, sensory, or mixed nerves and evoke a compound action potential. The three types of NCSs are motor, sensory, and mixed. Analysis of the compound muscle action potential (CMAP), which is evoked by stimulating a nerve while recording from a muscle, indirectly assesses the conduction characteristics of motor fibers. Analysis of the sensory nerve action potential (SNAP) assesses the sensory fibers by stimulating a nerve and recording directly from a cutaneous nerve. Mixed NCSs directly assess the sensory and motor fibers simultaneously by stimulating and recording from a mixed nerve and analyzing the mixed nerve action potential (MNAP). Use of standard NCSs enables the precise localization of a lesion and accurate characterization of peripheral nerve function.
NCSs use two different kinds of surface (percutaneous) electrical stimulators. Constant voltage stimulators regulate voltage output so that current varies inversely with the impedance of the system, including the skin and subcutaneous tissues. Constant current stimulators change voltage according to impedance so that the amount of current that reaches the nerve is within the limits of skin resistance. As the current flows between the cathode (negative pole) and the anode (positive pole), negative charges accumulate under the cathode and positive charges under the anode, depolarizing and hyperpolarizing the nerve, respectively. In bipolar stimulation, both electrodes are over the nerve trunk, with the cathode closer to the recording site. Anodal conduction block of the propagated impulse may occur with inadvertent reversal of the cathode and anode of the stimulator. The cause of the block is hyperpolarization at the anode. This may prevent the nerve impulse evoked by the depolarization occurring under the cathode from proceeding past the anode.
Supramaximal stimulation of a nerve that results in depolarization of all available axons is a paramount prerequisite to accurate and reproducible NCS measurements. To achieve supramaximal stimulation, one slowly increases the current (or voltage) intensity until it reaches a level at which the recorded potential does not increase in size. Then, increasing the current an additional 20%–30% ensures that the potential does not change further.
Surface electrodes record the CMAP, SNAP, or MNAP. The advantages of surface recording are reproducible evoked responses that change only slightly with the position of the electrodes in relation to the recording muscle or nerve. In contrast, needle electrode recording registers only a small portion of the muscle or nerve action potentials; as a result, the evoked responses are variable and not reproducible, although they have less interference from neighboring discharges. Needle recordings improve the recording from small atrophic muscles or a proximal muscle that is not excitable in isolation. Most recording electrodes used in clinical practice are disk electrodes; ring electrodes are convenient for recording the antidromic sensory potentials from digital nerves over the proximal and distal interphalangeal joints.
A prepulse preceding the stimulus triggers the sweep on a storage oscilloscope. The amplifier sensitivity determines the size (amplitude) of the potential. Overamplification truncates the response and underamplification prevents accurate measurements of the exact takeoff point from baseline. Digital averaging is very useful in recording low-amplitude SNAPs. Signals that are time locked to the stimulus summate with averaging at a constant latency and appear as an evoked potential distinct from the background noise. The signal-to-noise ratio increases in proportion to the square root of the trial number. For example, four trials give twice as big a response as a single stimulus, and nine trials give three times the amplitude. Most current instruments digitally indicate the latency and amplitude by cursors when the desired spot on the waveform is marked. The operator can override these cursors if needed.
The performance of motor NCSs requires stimulating a motor or mixed peripheral nerve while recording the CMAP from a muscle innervated by that nerve. Ideal muscles to record from are well isolated from neighboring muscles, which eliminates volume conduction. A pair of recording electrodes consists of an active lead, G1, placed on the belly of the muscle, and a reference (indifferent or inactive) lead, G2, placed on the tendon (belly-tendon recording) . The propagating muscle action potential, originating under G1 located near the motor point, gives rise to a simple biphasic waveform with an initial negativity. Initial positivity suggests incorrect positioning of the active electrode away from the motor end-plate zone or a volume-conducted potential from distant muscles activated by anomalous innervation or by accidental spread of stimulation to other neighboring nerves, thus generating potentials from distant muscles
The nerve is usually stimulated, whenever technically feasible, at two or more points along its course. Shorter nerves—such as the axillary, femoral, and facial nerves—are stimulated at only one point, because the more proximal portions of the nerves are inaccessible. Otherwise, the nerve is typically stimulated distally near the recording electrode and more proximally to evaluate one or more proximal segments. Motor NCSs evaluate several measurements ( Fig. 36.1 ):
CMAP amplitude: The usual measure of amplitude is from baseline to negative peak and is expressed in millivolts. When recorded with surface electrodes, CMAP amplitude is a semiquantitative measure of the number of axons conducting between the stimulating and recording points. CMAP amplitude also depends on the relative conduction speed of the axons, the integrity of the neuromuscular junctions, and the number of muscle fibers that are able to generate action potentials.
CMAP duration: This measurement is usually the duration of the negative phase of the evoked potential and is expressed in milliseconds. It is a function of the conduction rates of the various axons forming the examined nerve and the distance between the stimulation and recording electrodes. As a result of physiological temporal dispersion and phase cancellation, the CMAP generated from proximal stimulation is slightly longer in duration and lower in amplitude than that obtained from distal stimulation (see forthcoming section).
CMAP area: This is usually limited to the negative phase area under the waveform and shows linear correlation with the product of amplitude and duration. Measurement is in millivolts per millisecond and requires electronic integration using computerized equipment. The ability to measure CMAP area has practically replaced the need to record its duration.
Latencies: Latency is the time interval between nerve stimulation (shock artifact) and the CMAP onset. Expression of latency is in milliseconds and reflects the conduction rate of the fastest conducting axon. Whenever technically possible, the nerve is stimulated at two points: a distal point near the recording site and a more proximal point. The measures obtained are the distal latency and proximal latency , respectively. Both latencies depend mostly on the length of the nerve segment and, to a much lesser extent, on neuromuscular transmission time and propagation time along the muscle membrane. When anatomically feasible, several proximal stimulation points may be done, generating several proximal latencies; in these situations the exact site of stimulation should be specified—for example, below elbow, above elbow, or axilla—referring to stimulation sites while the ulnar nerve is being tested.
Conduction velocity: This is a computed measurement of the speed of conduction expressed in meters per second. Measurement of conduction velocity allows comparison of the speed of conduction of the fastest fibers between different nerves and subjects irrespective of the length of the nerve. The calculation requires measurement of the length of the nerve segment between distal and proximal stimulation sites. Measuring the surface distance along the course of the nerve estimates the nerve length; it should be more than 10 cm to improve the accuracy of surface measurement.
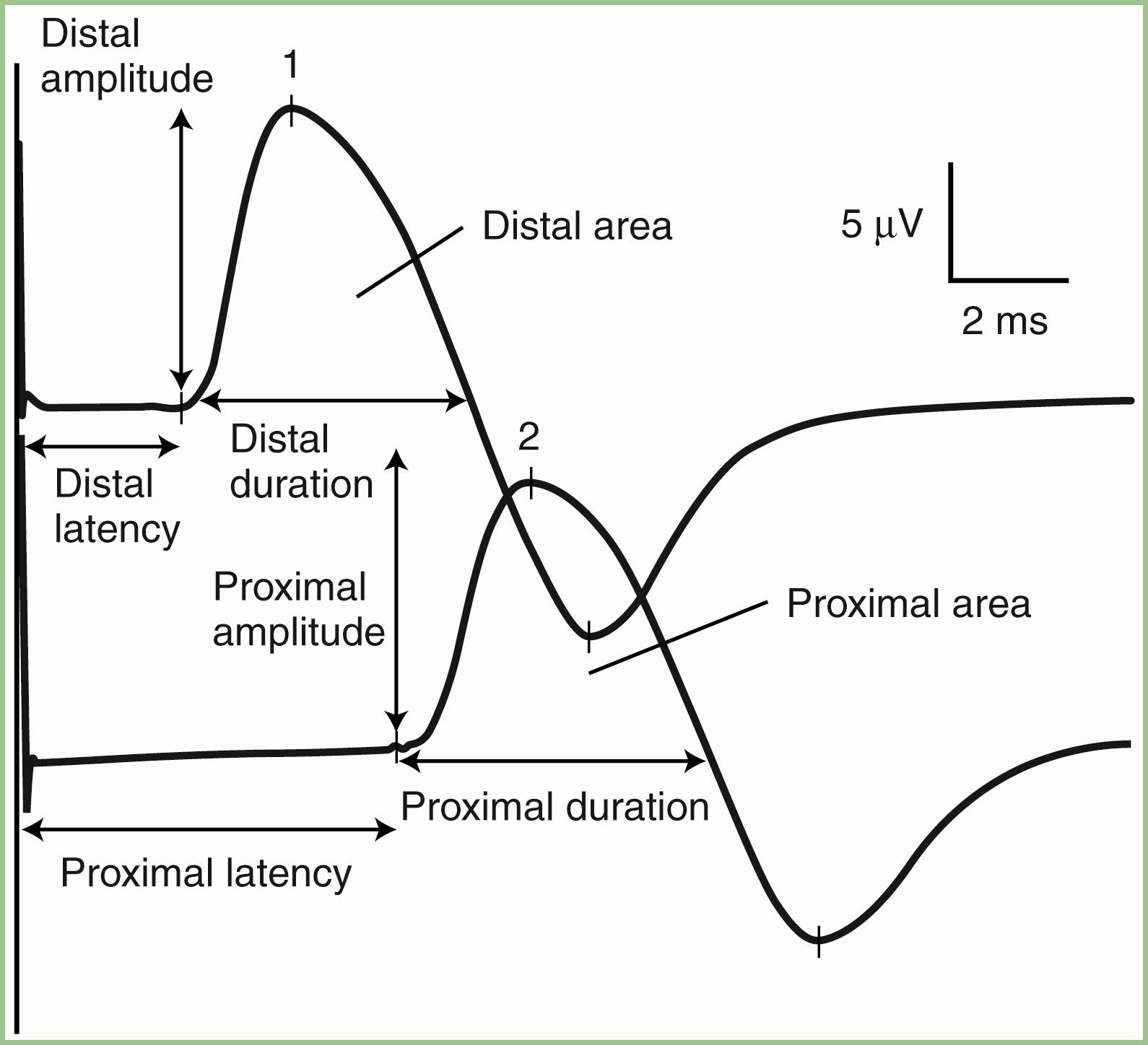
As with latencies, motor conduction velocity measures the speed of conduction of the fastest axon. In contrast with motor latency, however, motor nerve conduction velocity is a pure nerve conduction time because neuromuscular transmission time and muscle fiber propagation time are common to both stimulation sites, and the latency difference between two points is the time required for the nerve impulse to travel from one stimulus point to the other. When the nerve is stimulated at multiple proximal sites, several proximal conduction velocity segments may be calculated, such as above-elbow to below-elbow segment and below-elbow to wrist segment when the ulnar nerve is being tested.
Sensory axons are evaluated by stimulating a nerve while the transmitted potential from the same nerve is recorded at a different site. Therefore SNAPs are true nerve action potentials. The measurement of antidromic sensory NCSs requires recording potentials directed toward the sensory receptors, whereas obtaining orthodromic responses requires recording potentials directed away from these receptors. Sensory latencies and conduction velocities are identical with either method, but SNAP amplitudes are generally higher in antidromic studies. Orthodromic responses are sometimes low in amplitude, necessitating the use of averaging techniques. Action potentials from distal muscles may obscure antidromic responses because the thresholds of some motor axons are similar to those of large myelinated sensory axons. Fortunately, accurate measurement of SNAPs is still possible because the large-diameter sensory fibers conduct 5%–10% faster than motor fibers. This relationship may change in disease states that selectively affect different fibers.
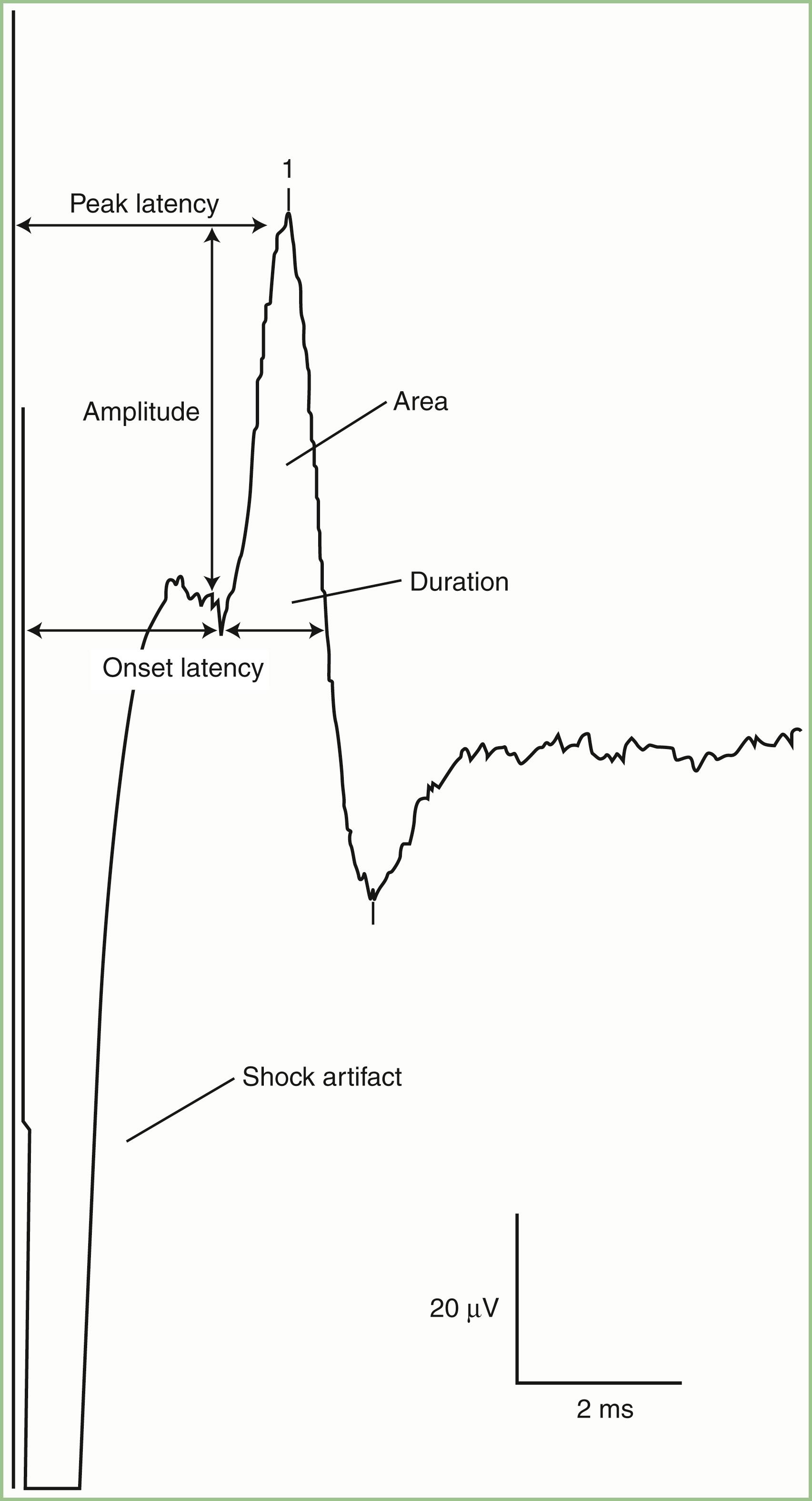
SNAPs may be obtained by several methods: (1) stimulating and recording a pure sensory nerve (such as the sural and radial sensory nerves); (2) stimulating a mixed nerve while recording distally over a cutaneous branch (such as the antidromic median and ulnar sensory responses); or (3) stimulating a distal cutaneous branch while recording over a proximal mixed nerve (such as in orthodromic median and ulnar sensory studies). Similar to their motor counterparts, sensory NCSs record several measurements ( Fig. 36.2 ):
SNAP amplitude: This semiquantitatively measures the number of sensory axons that conduct between the stimulation and recording sites. The calculation is from the baseline to negative peak or from positive peak to negative peak and is expressed in microvolts. SNAP duration and area may be measured, but such measurements are not useful because of significant temporal dispersion and phase cancellation (see later discussion).
Latencies: Sensory distal latencies are measured (in milliseconds) from the stimulus artifact to the peak of the negative phase (peak latency) or from the stimulus artifact to the onset of the SNAP (onset latency). A large shock artifact, a noisy background, or a wavy baseline may obscure onset latency. Although peak latency does not reflect the fastest conducting sensory fibers, it is easily defined and more precise than onset latency.
Conduction velocity: This requires stimulation at a single site only because the latency consists of just the nerve conduction time from the stimulus point to the recording electrode. As with motor velocity, the calculation may also be done using both distal and proximal stimulations. Only onset latencies (not peak latencies) are useful for calculating velocities to assess the speed of the fastest conducting fibers.
Stimulating and recording from nerve trunks containing sensory and motor axons constitute mixed NCSs. Often these tests require stimulating a nerve trunk distally and recording more proximally because large CMAPs contaminate the reverse by obscuring the lower-amplitude MNAPs. The MNAP may be of low amplitude or not elicitable when the nerve is deeply situated (as at the elbow or knee) because of tissue interposed between the nerve and the recording electrode. Therefore MNAPs are limited to assessing mixed nerves in distal nerve segments in the hand or foot, such as the mixed palmar and mixed plantar studies used to evaluate carpal tunnel syndrome and tarsal tunnel syndrome, respectively.
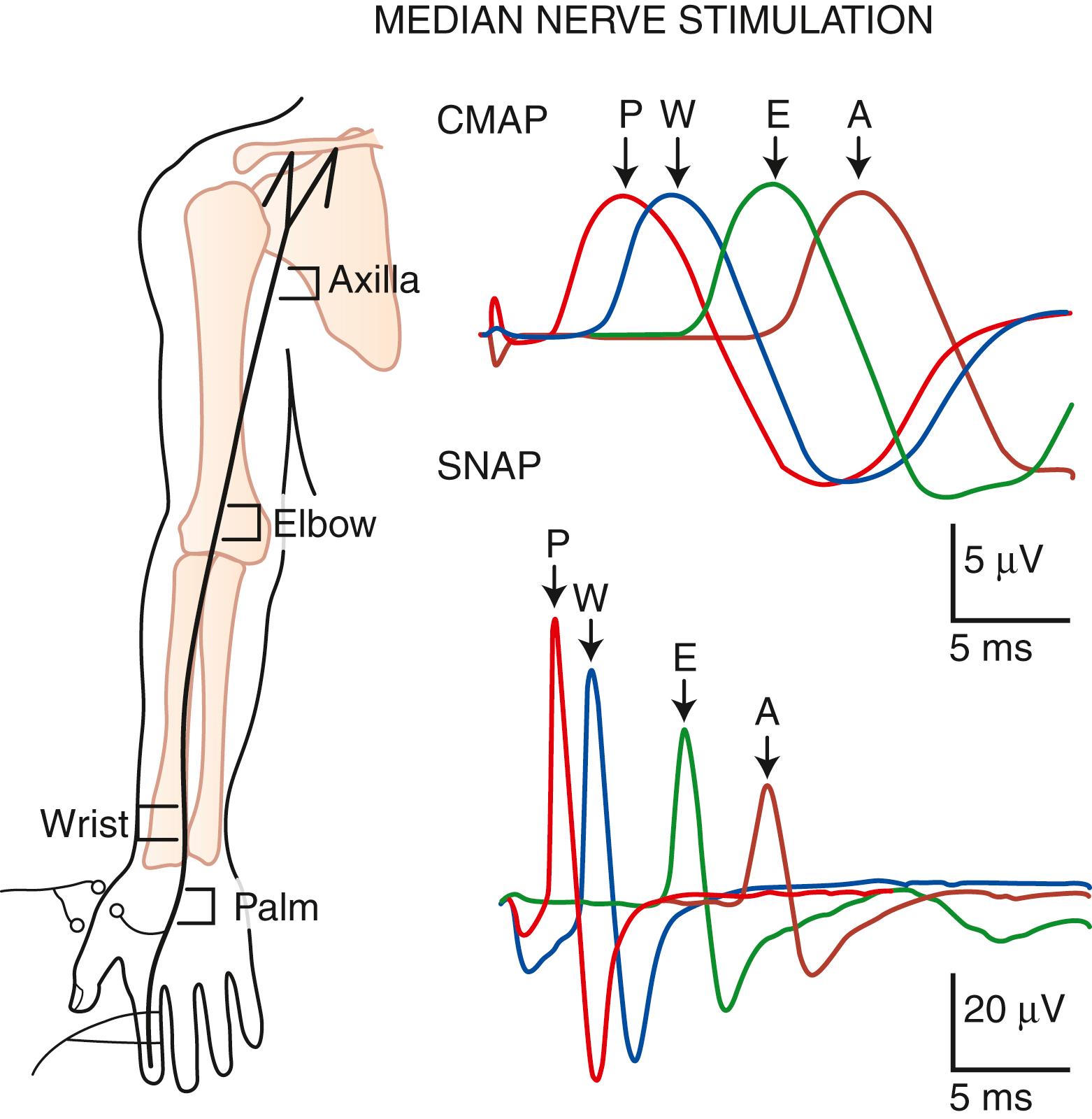
Routine NCSs are usually sufficient to localize the site of involvement in most patients with entrapment neuropathies. During the evaluation of a focal demyelinating lesion, however, inclusion of the unaffected nerve segment in a relatively long distal latency or conduction velocity calculation dilutes the effect of slowing at the injured site and decreases the sensitivity of the test. Therefore incremental stimulation across a shorter nerve segment is useful to help localize an abnormality that might otherwise escape detection. Localization that is more precise entails “inching” the stimulus in short increments along the course of the nerve. The study of short segments provides better resolution of restricted lesions. For example, a nerve impulse may be found to conduct at a rate of 0.2 ms per 1.0 cm (50 m/sec). For a 1-cm segment, then, demyelination would double the conduction time to 0.4 ms/cm. In a 10-cm segment, normally covered in 2.0 ms, a 0.2-ms increase would constitute a 10% change, or approximately 1 standard deviation, or well within the normal range of variability. However, the same 0.2-ms increase would represent a 100% change in latency if it were measured over a 1-cm segment. The large per-step increase in latency more than compensates for the inherent measurement error associated with stimulating multiple times in short increments.
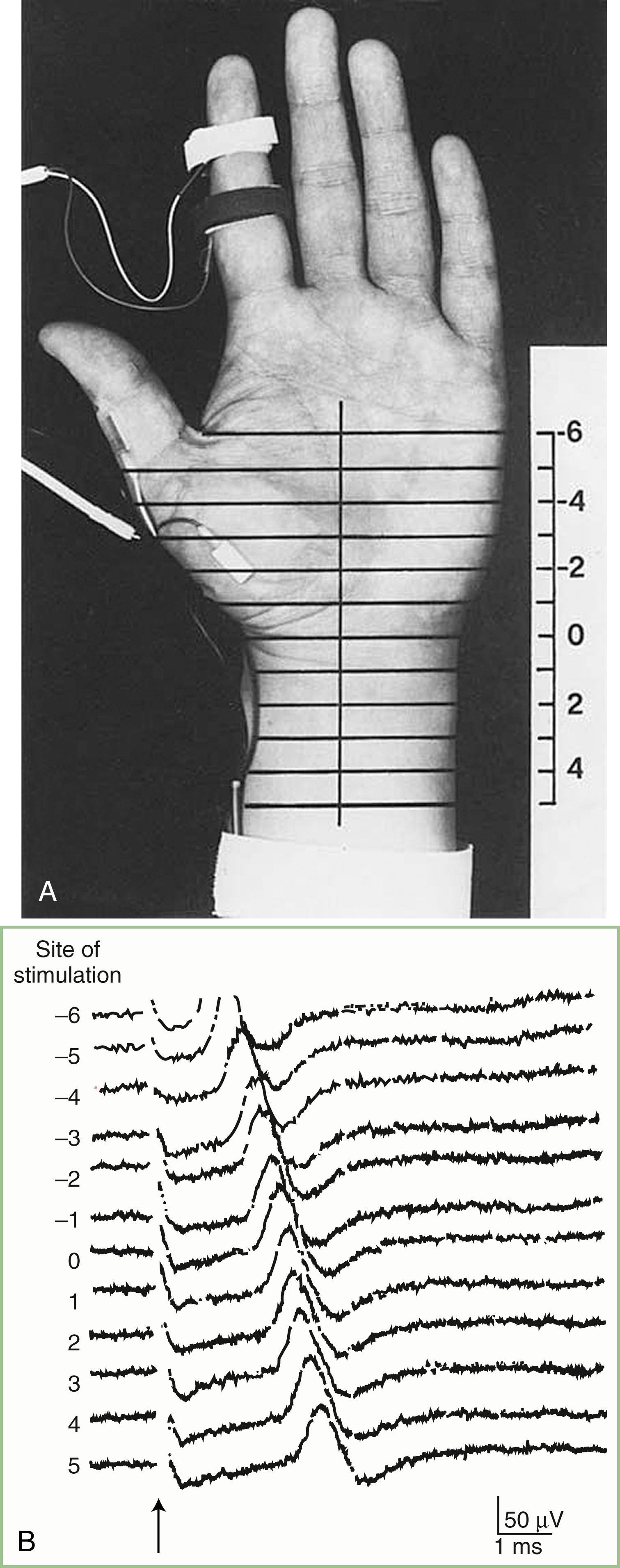
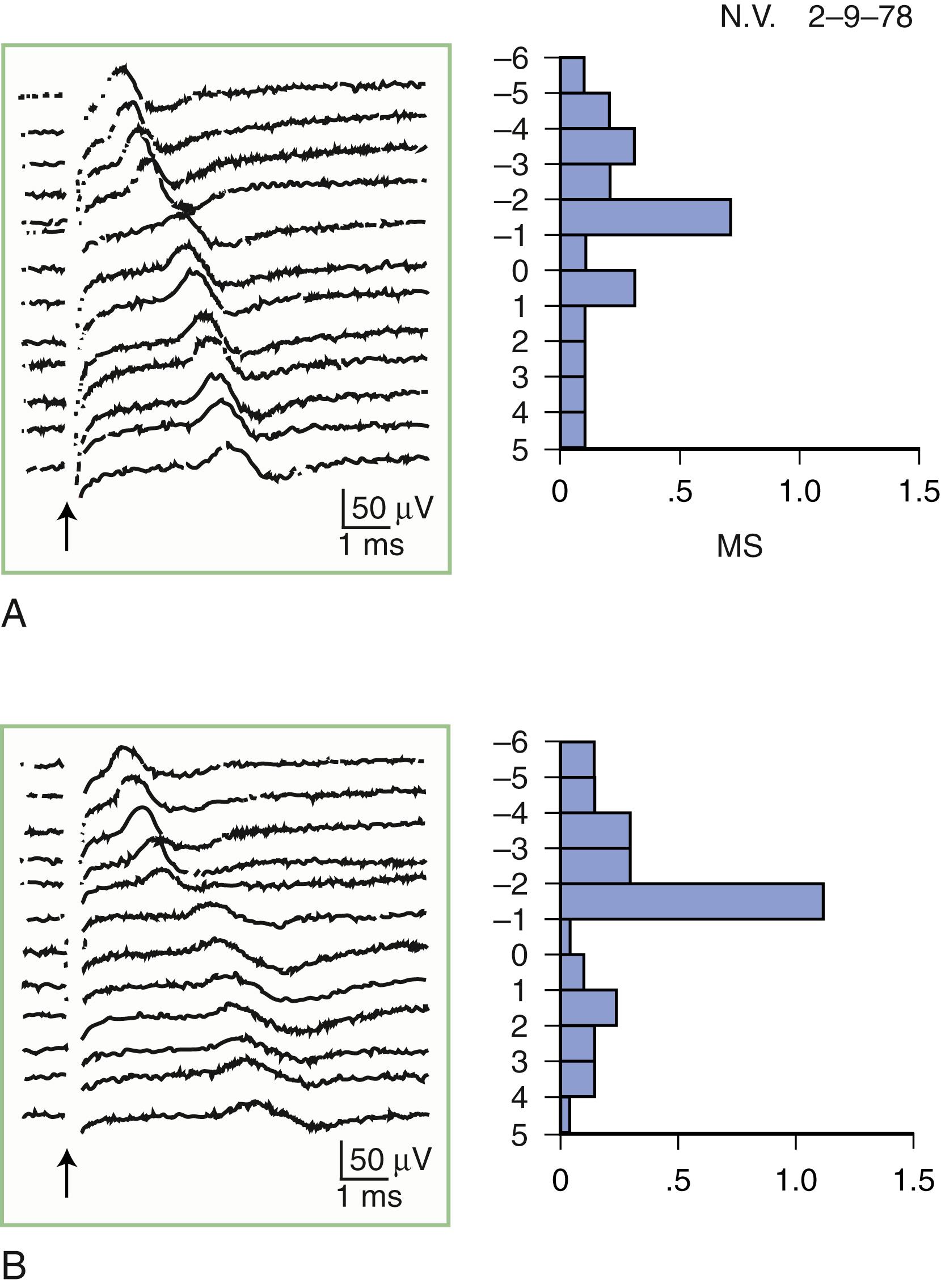
The inching (or actually “centimetering”) technique is particularly useful in assessing nerve conduction in patients with carpal tunnel syndrome or an ulnar neuropathy at the elbow or wrist ( ). For example, stimulation of a normal median nerve in 1-cm increments across the wrist results in latency changes of approximately 0.16–0.21 ms/cm from midpalm to distal forearm ( Fig. 36.3 ). A sharply localized latency increase across a 1-cm segment indicates a focal abnormality of the median nerve ( Fig. 36.4 ). An abrupt change in waveform usually accompanies the latency increase across the site of compression.
The major pitfalls in NCS usually involve technical errors in the stimulating or recording system ( ). Common errors include large stimulus artifact, increased electrode noise, submaximal stimulation, costimulation of an adjacent nerve not under study, eliciting an unwanted potential from distant muscles, recording or reference electrode misplacement, and errors in measurement of nerve lengths and conduction times. Other errors are attributable to intertrial and physiological variability, including the effects of temperature, age, the length of the studied nerve, anomalous innervation, and temporal dispersion.
Nerve impulse propagation slows by 2.4 m/sec, or approximately 5%, per degree centigrade from 38°C to 29°C of body temperature. Also, cooling results in a higher CMAP and SNAP amplitude and longer response duration, probably because of accelerated and slowed sodium channel inactivation ( ). Therefore a CMAP or SNAP with high amplitude and slow distal latency or conduction velocity should raise the suspicion of a cool limb. To reduce this type of variability, a plate thermistor is used to measure skin temperature. This measurement correlates linearly with the subcutaneous and intramuscular temperatures. If the skin temperature falls below 33°C, the limbs are warmed by immersion in warm water or by the application of warming packs or a hydrocollator. Adding 5% of the calculated conduction velocity for each degree below 33°C theoretically normalizes the result. The use of such conversion factors is based on evidence obtained in healthy persons; however, this may not be applicable in patients with abnormal nerves.
Because myelination is incomplete at birth, nerve conduction velocities are half the adult values in full-term newborns; in 23- to 24-week premature newborns, velocities are one-third the values for term newborns. They attain adult values at 3–5 years. Motor and sensory nerve conduction velocities tend to increase slightly in the arms and decrease in the legs during childhood up to the age of 19 years. Conduction velocities slowly decline after age 50, so the mean conduction velocity is reduced by approximately 10% at 60 years of age.
Aging also diminishes SNAP and CMAP amplitudes, which decline slowly after age 60. SNAP amplitudes are affected more prominently, so much so that normal upper limb SNAP amplitude drops to 50% by age 70 and lower limb SNAPs in many healthy persons older than 60 are low in amplitude or unevokable. Therefore the absence of lower extremity SNAPs in older adults must be interpreted with caution; the finding is not necessarily abnormal without other confirmatory data.
An inverse relationship between height and nerve conduction velocity suggests that longer nerves conduct more slowly than shorter nerves. For example, the nerve conduction velocities of the peroneal and tibial nerves in the lower extremities are 7–10 m/sec slower than those of the median and ulnar nerves in the upper extremities. The slightly lower temperature of the legs compared with the arms is not the entire explanation. Possible factors accounting for the length-related slowing include abrupt distal axonal tapering, progressive reduction in axonal diameter, and shorter internodal distances. For similar reasons, nerve impulses propagate faster in proximal than in distal nerve segments. Adjustments of normal values are necessary for patients of extreme height; this usually is no more than 2 m/sec below the lower limit of normal.
Several anomalous peripheral innervations may influence interpretation of the EDX study. Two of these variants, the Martin-Gruber anastomosis and the accessory deep peroneal nerve, have a significant effect on NCSs.
In the Martin-Gruber anastomosis, anomalous fibers cross from the median to the ulnar nerve in the forearm. The communicating branches usually consist of motor axons supplying the ulnar innervated intrinsic hand muscles, particularly the first dorsal interosseous muscle, the hypothenar muscles (abductor digiti minimi), and the thenar muscles (adductor pollicis, deep head of flexor pollicis brevis), or a combination of these muscles ( ). The Martin-Gruber anastomosis occurs in approximately 15%–20% of the population and is sometimes bilateral. This anomaly manifests as a drop in the ulnar CMAP amplitude between distal and proximal stimulation sites (simulating the appearance of conduction block on ulnar NCS recording from the abductor digiti minimi or first dorsal interosseous). With distal stimulation (at the wrist), the CMAP reflects all ulnar motor fibers, whereas proximal stimulation activates only the uncrossed fibers, which are fewer in number. This anomaly can be confirmed by median nerve stimulation at the elbow, which evokes a small CMAP from the abductor digiti minimi or first dorsal interosseous, which is not present on median nerve stimulation at the wrist. Although in the majority of subjects the abnormal decline in CMAP amplitude occurs across the forearm segment, this pseudo-conduction block may occasionally be encountered across the elbow segment, resembling partial conduction block in a patient with ulnar neuropathy at the elbow ( ). When anomalous fibers innervate the thenar muscles, stimulation of the median nerve at the elbow activates the nerve and the crossing ulnar fibers, resulting in a large CMAP, often with an initial positivity caused by volume conduction of action potential from the ulnar thenar muscles to the median thenar muscles. By contrast, distal median nerve stimulation evokes a smaller thenar CMAP without the positive dip because the crossed fibers are not present at the wrist. In addition, the median nerve conduction velocity in the forearm is spuriously fast, particularly in the presence of carpal tunnel syndrome, because the CMAP onset represents a different population of fibers at the wrist than at the elbow. Collision studies obtain an accurate conduction velocity by using action potentials of the crossed fibers ( ).
About 20%–30% of subjects have an anomalous accessory deep peroneal nerve. It is a branch of the superficial peroneal nerve and usually arises as a continuation of the muscular branch that innervates the peroneus longus and brevis muscles. It passes behind the lateral malleolus and terminates in the extensor digitorum brevis (EDB) on the dorsum of the foot. During peroneal motor NCS recording from the EDB, the peroneal CMAP amplitude is larger-stimulating proximally than distally because the anomalous fibers are not present at the ankle. Stimulation behind the lateral malleolus confirms this anomaly, which yields a small CMAP that approximately equals the difference between the CMAP amplitudes evoked with distal and proximal peroneal nerve stimulations. Complete innervation of the EDB by the accessory deep peroneal nerve is rare but should be suspected if there is preservation of function in the EDB muscle (i.e., extension of lateral toes) in a patient with severe deep peroneal neuropathy ( ).
In most people, the brachial plexus arises from the C5 to T1 cervical roots. In some, the plexus origin shifts one level up (prefixed), arising from C4 to C8; in others, it shifts one level down (postfixed), originating from C6 to T2. These anomalies result in error in the precise localization of cervical root lesions based on myotomal and dermatomal representation. In a prefixed plexus, the location of the cervical lesion is one level higher than concluded from findings on the clinical examination and EDX studies. In contrast, with a postfixed plexus, the cervical root lesion is one level lower.
Riche-Cannieu anastomosis is a communication in the palm between the recurrent motor branch of the median nerve and the deep branch of the ulnar nerve. The result is dual innervation of some intrinsic hand muscles such as the first dorsal interosseous, adductor pollicis, and abductor pollicis brevis. Riche-Cannieu anastomosis is rather common but is often not clinically or electrophysiologically apparent. When this anomaly is prominent, denervation in ulnar muscles may follow a median nerve lesion, and vice versa. In addition, a complete median or ulnar nerve lesion may be associated with relative sparing of some median innervated muscles or ulnar innervated muscles in the hand.
The CMAP, evoked by supramaximal stimulation, represents the summation of all individual MUAPs directed to the muscle through the stimulated nerve. Typically, as the stimulus site moves proximally, the CMAP slightly drops in amplitude and area and increases in duration. This is caused by temporal dispersion in which the velocity of impulses in slow-conducting fibers lags increasingly behind those of fast-conducting fibers as conduction distance increases. With dispersion, a slight positive and negative phase overlap occurs, and phase cancellation of MUAP waveforms is seen ( Fig. 36.5 ). The result of temporal dispersion and phase cancellation is a prolongation of CMAP duration, reduction of CMAP amplitude, and a less obvious decrease in CMAP area.
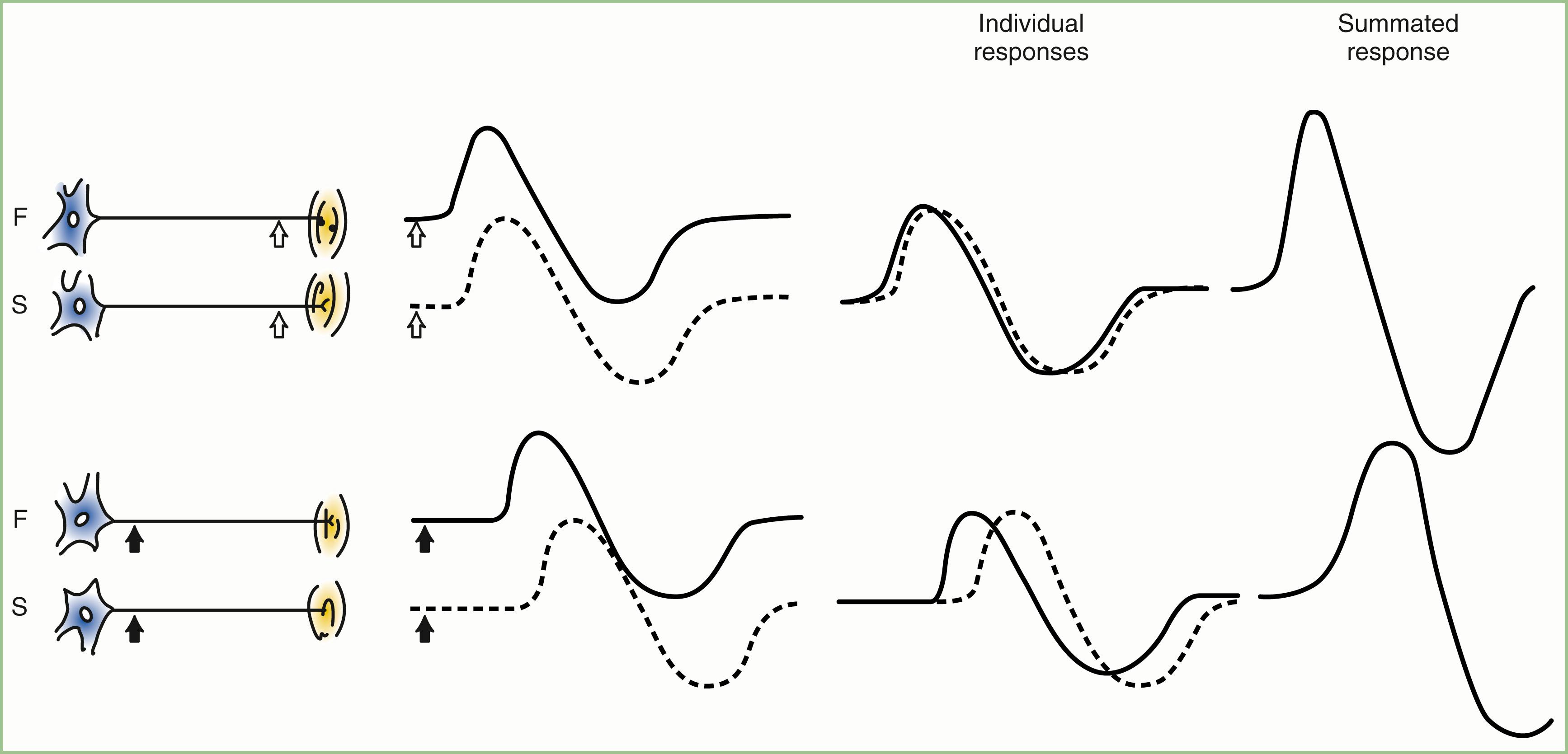
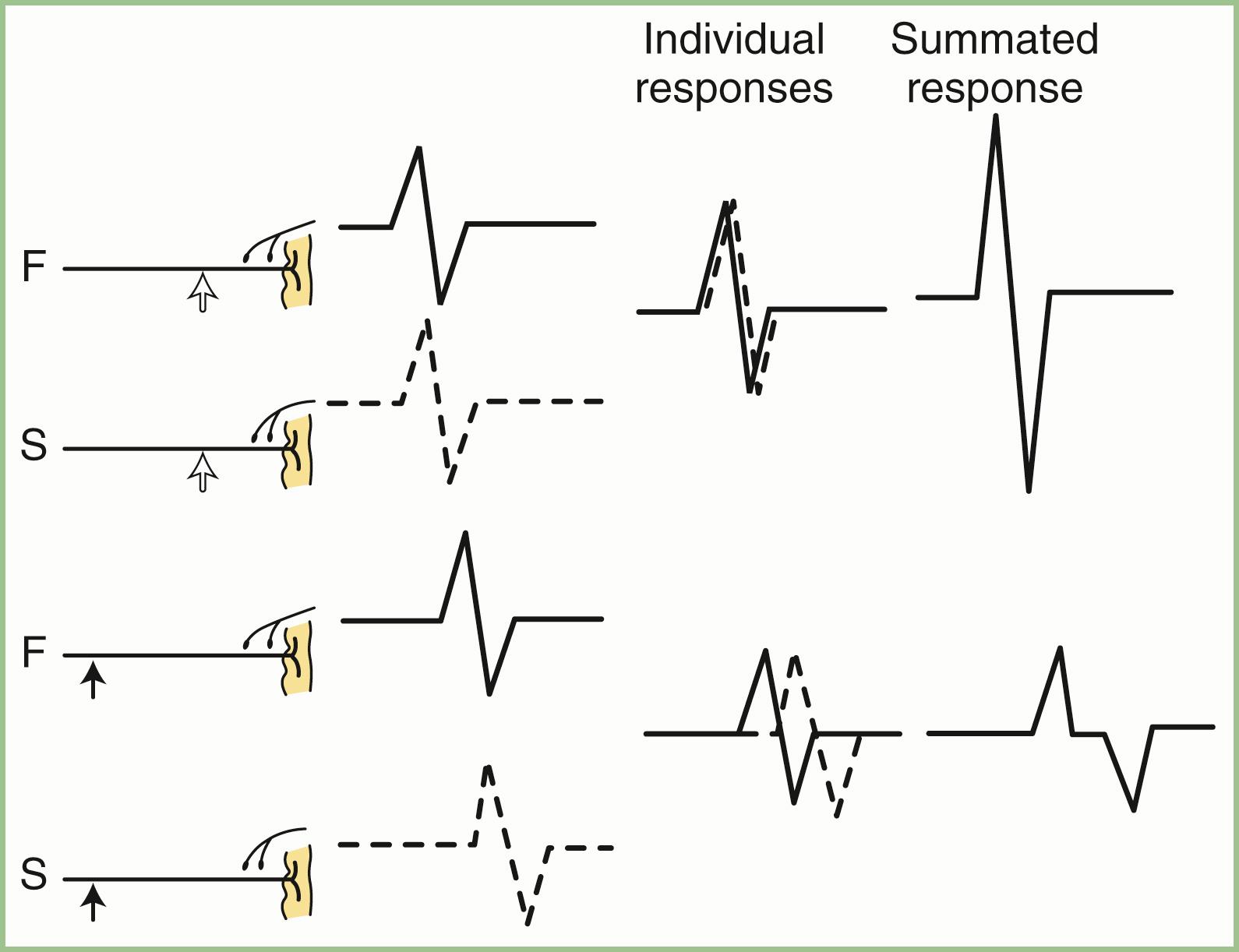
Physiological temporal dispersion affects the SNAP more than the CMAP ( Fig. 36.6 ). This difference relates to two factors. The first relates to the disparity between sensory fiber and motor fiber conduction velocities. The range of conduction velocities between the fastest and slowest individual human myelinated sensory axons is almost twice that for the motor axons (25 m/sec and 12 m/sec, respectively). The second factor is the difference in duration of individual unit discharges between nerve and muscle. With short-duration biphasic or triphasic SNAPs, a slight latency difference could line up the positive peaks of the fast fibers with the negative peaks of the slow fibers and cancel both ( Fig. 36.7 ). In longer-duration biphasic CMAPs, the same latency shift would only partially superimpose peaks of opposite polarity and phase cancellation would be less of a factor.
Principal factors contributing to an intertrial variability include errors in determining surface distance and measuring latencies and amplitudes of the recorded response. Amplitudes vary most, probably reflecting a shift in the recording site. NCSs are more reproducible when they are administered by the same examiner because there is a significant degree of interexaminer difference ( ).
Although both NCSs and needle EMGs are required in most patients to confirm a neuromuscular diagnosis, certain peripheral nerve disorders are evident on NCSs alone.
Peripheral nerve is composed of unmyelinated and myelinated axons surrounded by Schwann cells and a supporting tissue. Surrounding the unmyelinated axons are only the plasma membranes of Schwann cells. By contrast, wrapped around myelinated axons are multiple myelin layers that have a low capacitance and large resistance. Surrounding the myelinated axon is myelin, along with Schwann cells, except at certain gaps called the nodes of Ranvier , where sodium channels are highly concentrated and saltatory conduction occurs. Three supportive layers—the endoneurium, perineurium, and epineurium—surround nerves; they are highly elastic and protect the myelin and axon from external pressure and tension. Nerve fibers may be injured by a variety of mechanisms, including compression, ischemia, traction, and laceration.
The classification of peripheral nerve lesions is based on the extent of injury to the elements of peripheral nerve, including axon, myelin, and supportive layers. In neurapraxia (first-degree injury) , distortion of myelin occurs near the nodes of Ranvier, producing segmental conduction block without wallerian degeneration. In axonotmesis (second-degree injury) , the axon is interrupted but all the supporting nerve structures remain intact. In neurotmesis , the nerve injury is severe, resulting in complete disruption of the nerve with all the supporting structures (see Chapter 63 ). Often, the neurotmesis group is divisible into three degrees, as follows: third-degree injury , with disruption of the endoneurium and with intact perineurium and epineurium; fourth-degree injury , with disruption of all neural elements except the epineurium; and fifth-degree nerve injury , with complete nerve transection resulting in complete discontinuity of the nerve. EDX studies alone cannot accurately distinguish between the five degrees of nerve injuries, but they can separate the first (neurapraxia) from the other axon-loss types ( ).
When focal injury to myelin occurs, conduction along the affected nerve fibers may alter. This may result in conduction slowing or block along the nerve fibers. The cause of conduction block is interruption of action potential transmission across the nerve lesion; it is the electrophysiological correlate of neurapraxia and usually results from loss of more than one myelin segment (segmental or internodal demyelination) . Bracketing two stimulation points, one distal and one proximal to the site of injury, best localizes a nerve lesion with conduction block. With such lesions, stimulation distal to the lesion elicits a normal CMAP, whereas proximal stimulation evokes a response with reduced amplitude or fails to evoke any response; these are respectively defined as partial or complete conduction block ( Fig. 36.8 , A ). There are several limitations to the diagnosis of demyelinative conduction block:
Phase cancellation between peaks of opposite polarity may reduce CMAP size because of abnormally increased temporal dispersion. Such excessive desynchronization often develops in acquired demyelinative neuropathies. If the distal and proximal responses have dissimilar waveforms, the discrepancy in amplitude or area between the two may be the result of phase cancellation rather than conduction block. Therefore, for a diagnosis of partial conduction block, findings should include a significantly lower CMAP amplitude as well as CMAP area with stimulation proximal to the injury site than with the CMAP distal to it, and without any significant prolongation of CMAP duration. More than 50% decay of both the CMAP amplitude and area across the lesion is usually the criterion for definite conduction block.
Distal demyelinating lesions causing conduction block of the nerve segment between the most distal stimulating point and the recording site manifest as unelicitable or low CMAP amplitudes at both distal and proximal stimulation sites. This finding mimics the NCSs seen with axonal degeneration. Repeated NCSs often show rapid improvement of CMAP within weeks, consistent with remyelination but not with axonal loss and reinnervation.
Conduction block may also follow axonal loss before the completion of wallerian degeneration. This is referred to as axon-loss conduction block, or axon-discontinuity conduction block. Repeated NCSs will show rapid decline of distal CMAP within a week, resulting in equal CMAPs at all points of stimulation (see “Axon-loss mononeuropathy,” later).
The prominent temporal dispersion normally seen in evaluating SNAPs precludes the use of sensory potentials to diagnose conduction block.
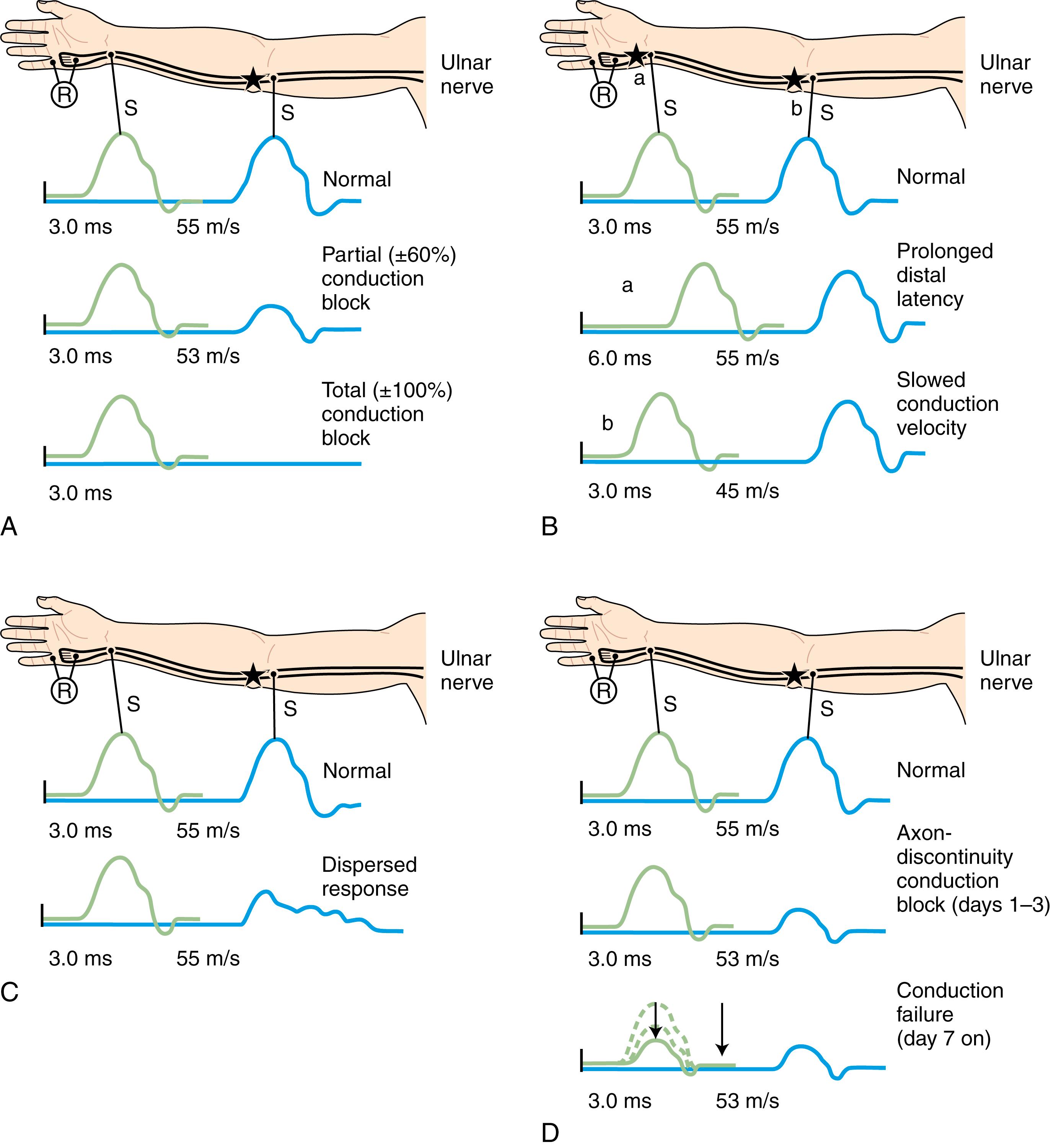
Focal slowing of conduction is usually the result of widening of the nodes of Ranvier (paranodal demyelination) . Slowing, often synchronized, affects all large myelinated fibers equally. This results in prolongation of distal latency if the focal lesion is distal (see Fig. 36.8 , B, a ), or slowing in conduction velocity if the focal lesion is proximal (see Fig. 36.8 , B, b ). CMAP amplitude, duration, and area, however, are normal and do not change when the nerve is stimulated proximal to the lesion. Desynchronized slowing (differential slowing) occurs when conduction velocity reduces at the lesion site along a variable number of the medium-sized or small nerve fibers (average- or slower-conducting axons). Here the CMAP disperses with prolonged duration on stimulations proximal to the lesion. The speed of conduction along the injury site (latency or conduction velocity) is normal because of sparing of at least some of the fastest-conducting axons (see Fig. 36.8 , C ). When synchronized and desynchronized slowing coexist, slowing of distal latency or conduction velocity accompanies the dispersed CMAP with prolonged duration.
After acute focal axonal damage, the distal nerve segment undergoes wallerian degeneration. Characteristically, unelicitable or low CMAP amplitudes with distal and proximal stimulations are signs of complete or partial lesions, respectively, involving motor axonal loss. The CMAP amplitudes provide a reliable estimate of the amount of axonal loss except in the chronic phase, in which effective reinnervation via collateral sprouting may increase the CMAP and give a misleadingly low estimate of the extent of original axonal loss.
In partial axon-loss lesions, distal latencies and conduction velocities are normal or borderline. Selective loss of fast-conducting fibers associated with more than a 50% reduction in mean CMAP amplitude may slow conduction velocity up to 80% of the normal value because the velocity represents the remaining slow-conducting fibers. Motor conduction velocity may slow to 70% of normal value with a reduction of CMAP amplitude to less than 10% of the lower limit of normal.
Soon after axonal transection (i.e., for the first 48 hours), the distal axon remains excitable. Therefore stimulation distal to the lesion elicits a normal CMAP, whereas proximal stimulation elicits a response with reduced amplitude and area, producing a conduction block pattern (see Fig. 36.8 , D , middle panel ). This pattern is axonal noncontinuity, early axon loss, or axon-discontinuity conduction block. Soon, however, the distal axons undergo wallerian degeneration, and the distal CMAP decreases in size to equal the proximal CMAP (see Fig. 36.8 , D , lower panel ). With wallerian degeneration, the distal CMAP decreases in amplitude and area starting 1 or 2 days after nerve injury and reaches its nadir in 5–6 days. In contrast, the distal SNAP lags slightly behind and reaches its nadir in 10 or 11 days ( Fig. 36.9 ). The difference between the decline of the SNAP and CMAP amplitudes and areas after axon loss probably relates to neuromuscular transmission failure, which affects only the CMAP amplitude and area. Supporting this hypothesis is the fact that MNAPs recorded directly from nerve trunks follow the time course of SNAPs.
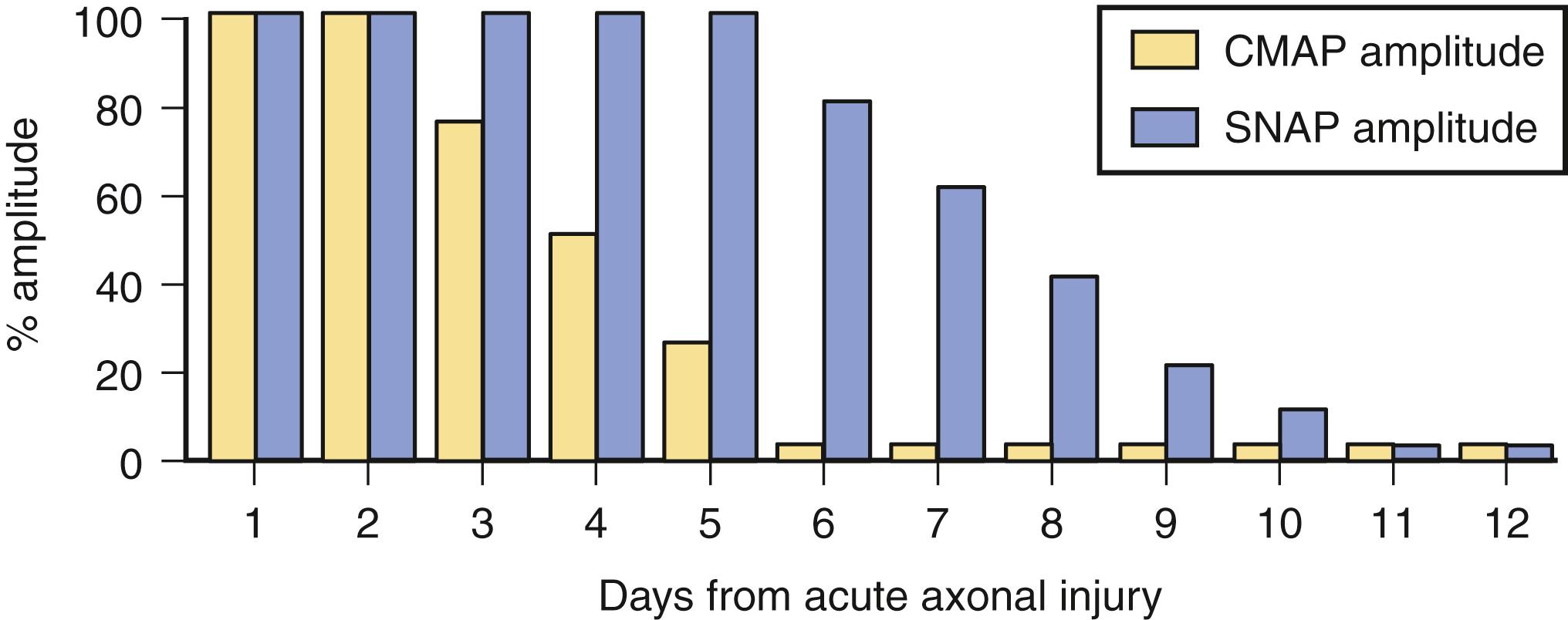
The study is repeated after 10 or 11 days, when degenerating axons have lost excitability, to distinguish between conduction block due to demyelination and that due to axonal loss. A reduction in amplitude and area of the evoked potential from stimulation above and below the lesion indicates axonal loss (see Fig. 36.8 , D ). By contrast, if the distally evoked CMAP still has preserved amplitude and area greater than that of the proximally elicited response, it indicates partial segmental demyelination.
Identification of conduction block in the early days of axonal loss is extremely helpful in localizing a peripheral nerve injury, particularly the closed type in which the exact site of lesion is not apparent. Awaiting the completion of wallerian degeneration leads to diffusely low or unevokable CMAPs (regardless of stimulation site), which does not allow accurate localization of the injury site. Needle EMG study is useful, but localization by this method is suboptimal (see later discussion).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here