Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Despite a progressive decrease in cardiac surgical mortality, the incidence of postoperative neurologic complications has remained relatively unchanged over the decades. During this same interval, the age and the extent of comorbidities in cardiac surgical patients have increased.
Neurologic complications range from coma, stroke, and visual field deficits to impairments of cognitive processes (e.g., delirium, impaired memory and attention, mood alterations). These adverse events are associated with prolonged intensive care unit stay, longer hospital stay, decreased quality of life, and decreased long-term survival in the cardiac surgical population.
Mechanisms for neurologic injury in cardiac surgery include some combination of cerebral embolism, hypoperfusion, inflammation, associated vascular disease, and altered cerebral autoregulation rendering the brain more susceptible to injury.
Perioperative risk factors for neurologic complications include renal dysfunction, hyperthermia, hypoxemia, hyperglycemia, diabetes mellitus, hypertension, prior cerebrovascular disease, aortic atheromatosis, manipulation of ascending aorta, complex surgical procedures, cardiopulmonary bypass (CPB) time longer than 2 hours, hypothermic circulatory arrest, hemodynamic instability during and after CPB, and perioperative atrial fibrillation.
Age-associated increased risk of stroke and other adverse central nervous system (CNS) outcomes appear to be powered primarily by comorbidities, particularly ascending and aortic arch atherosclerosis.
Cerebrovascular disease renders patients who experience wide hemodynamic perturbations during CPB at greater risk for perioperative stroke.
While occlusive carotid artery disease is associated with an increased risk of perioperative stroke, such stroke is not infrequently contralateral, and concomitant perioperative carotid endarterectomy may increase the risk of stroke and other major adverse events.
Routine epiaortic scanning (EAS) before instrumentation of the ascending aorta is a sensitive and specific technique used to detect nonpalpable aortic atheromatosis.
In patients with significant ascending aorta atheromatosis, avoidance of aortic manipulation (“no-touch technique”) is associated with decreased perioperative stroke.
Strategies to decrease the impact of CPB on embolization, inflammation, and coagulation will decrease neurologic complications.
Modular minimally invasive extracorporeal circulation (MiECC) is an evolving approach to physiologically integrated CPB, and it is associated with a variety of improved outcomes.
Pharmacologic management should be directed primarily toward early recovery with intraoperative usage of volatile anesthetics, continuance of perioperative aspirin and statin medications, minimization of hyperglycemia, and vigilant therapy for perioperative atrial arrhythmias.
Multichannel functional cerebral near-infrared spectroscopy (NIRS) allows for real-time monitoring of frontotemporal brain oxygenation, enabling enhanced detection and intervention of ischemic brain region(s) in real time, thus potentially improving neurologic outcomes after cardiac surgery.
Regardless of surgical risk or comorbidity burden, there has been a downward trend in the incidence and severity of neurologic complications in patients undergoing transcatheter aortic valve implantation (TAVI). As a result, the US Food and Drug Administration has broadened the indications for several transcatheter heart valves to include patients with low and intermediate surgical risk.
MitraClip appears to be associated with a significantly lower incidence of mortality and stroke in high-risk surgical patients, and it may even enhance cognitive function in patients with end-stage heart failure.
Any patient suspected of experiencing an acute stroke after cardiac surgery should have appropriate neuroimaging performed as soon as possible. If they meet the criteria for endovascular intervention, an individualized approach should be adopted to weigh the benefits and risks of the procedure.
Central nervous system (CNS) dysfunction after surgery can be devastating, posing a profound humanistic, clinical, social, and economic burden. Despite significant advancements in surgical safety, postcardiac surgery neurologic dysfunction remains a compelling problem that continues to generate debate on the optimal strategy for its prevention and management.
The incidence of clinical stroke after closed-chamber cardiac procedures is generally considered to be approximately 1%. For isolated single valve surgical repair or replacement, the Society of Thoracic Surgeons (STS) database encompassing 109,759 procedures reported a stroke rate of 1.6%, increasing to 2.9% for combined coronary artery bypass graft (CABG) and valve surgery based on reports of 101,661 patients. It is interesting that, whereas the STS database is predicated upon self-reporting of outcome incidences, a meta-analysis of 40 observational studies reporting on 8975 patients aged 80 years or older undergoing combined conventional aortic valve replacement (AVR) and CABG reported a postoperative stroke rate of 3.7% (95% confidence interval [CI], 2.8–4.8; 12 studies, 2770 patients) not inconsistent with the STS database results. As discussed later, however, more sensitive assessments have detected significantly higher stroke rates in both open and closed-chamber procedures.
In a retrospective review of 86,244 CABG and 103,549 percutaneous coronary intervention (PCI) patients who were identified from national registries and were undergoing revascularization for stable multivessel coronary artery disease from 2004 through 2008, the 4-year adjusted cumulative incidence for the composite of death, myocardial infarction, and stroke, favored CABG, whereas the risk of 30-day stroke alone (0.37% vs 1.55%) favored PCI. Although a meta-analysis and a large prospective study appeared to demonstrate that the early excess risk of stroke in CABG was compensated for by a slow but progressive catch-up phenomenon in patients undergoing PCI, it has been argued that these analyses were all underpowered for stroke. , In contrast, Athappan and Head each found a significantly lower cumulative incidence of stroke following PCI compared with CABG up to year five in two independent meta-analyses of 80,314 and 11,518 patient records, respectively. , It is also worth noting that in both meta-analyses the incidence of stroke within 30 days of surgery was lower after PCI, whereas the incidence of late stroke (onset > 30 days post-surgery) was comparable between the two groups. ,
In essence, these and similar studies strongly affirm that perioperative factors play a key role in the reported increased risk of stroke following cardiac surgery. Accordingly, the risk factors, causes, and potential for mitigation of perioperative stroke associated with cardiac surgery and cardiopulmonary bypass (CPB), as well as the management of perioperative acute ischemic stroke are the topic of this chapter.
In a seminal study, Roach and colleagues classified CNS injury into two broad categories: type I (focal injury, stupor, or coma at discharge) and type II (deterioration in intellectual function, memory deficit, or seizures). Cerebral injury after cardiac surgery may also be broadly classified as stroke or encephalopathy (including delirium, neurocognitive dysfunction, seizure, and coma). The neuropsychiatric manifestations of encephalopathy, rather than being discrete entities, reflect a continuum of cerebral dysfunction with overlaps.
Stroke is defined clinically as any new focalized sensorimotor deficit persisting longer than 24 hours, identified either on clinical grounds only or, ideally, as confirmed by magnetic resonance imaging (MRI), computed tomography (CT), or other forms of brain imaging.
Transient ischemic attack (TIA) is defined as brief neurologic dysfunction persisting for less than 24 hours. Neurologic dysfunction lasting longer than 24 hours, but less than 72 hours, is termed a reversible ischemic neurologic deficit.
Delirium is described as a transient global impairment of cognitive function, reduced level of consciousness, profound changes in sleep pattern, and attention abnormalities.
Postoperative neurocognitive dysfunction is defined as a decline in cognitive performance, below a predefined threshold, which can be primarily diagnosed when a patient undergoes formal neuropsychological testing before and after surgery.
Seizure is categorized as either convulsive or nonconvulsive. It may be related to overt CNS injury or may reflect transient biochemical or pharmacologically mediated neuroexcitation.
In order to better ascertain causative factors and potential risk reduction strategies, it is important to distinguish perioperative stroke as early (i.e., neurologic deficit apparent on emergence from anesthesia), delayed (i.e., neurologic deficit developing more than 24 h postoperatively), or late (i.e., stroke developing more than 30 days postoperatively). Such an analysis facilitates identification of potentially causal intraoperative events (e.g., hypotension, atherosclerotic aorta) from perioperative events (e.g., atrial fibrillation) and later progression of underlying disease (e.g., cerebrovascular atherosclerosis).
In an analysis of 2516 consecutive patients who underwent either on-pump CABG ( n = 1399) or off-pump coronary artery bypass (OPCAB; n = 1117), more than half of strokes (29 of 46; 63%) were delayed strokes. Patients undergoing OPCAB had a significantly lower risk of early stroke (0.1% vs 1.1%; P = .0009), whereas the incidence of delayed strokes was not significantly different between patients undergoing on-pump CABG and OPCAB (0.9% vs 1.4%; P = .3484). A review of 7839 isolated on-pump CABG operations, 297 OPCAB procedures, and 986 combined CABG and valve surgeries reported an overall early stroke incidence of 1.6% and delayed stroke incidence of 1.1%. Univariate analysis indicated higher early stroke risk for on-pump CABG versus OPCAB (1.4% vs 0.3%; P = .011). Multivariate analysis indicated that risk factors for early stroke were advanced age, high preoperative creatinine level, the extent of aortic atherosclerosis, and duration of CPB. Factors associated with delayed stroke were female gender, unstable angina, previous cerebrovascular disease, inotropic support requirement, and postoperative atrial fibrillation (POAF). Early stroke was associated with increased mortality, mainly in the acute perioperative period, whereas delayed stroke was associated with increased long-term mortality.
These and other studies strongly indicate that intraoperative factors, whether associated with on-pump CABG, OPCAB, or valve surgery, fundamentally impact the incidence of early stroke, whereas delayed stroke reflects the progression of comorbid disease and/or atrial arrhythmias.
In a study by Bucerius and coworkers assessing CNS outcomes from 16,184 patients undergoing cardiac operations with CPB ( n = 14342) or without CPB ( n = 1847), the overall prevalence of postoperative delirium was 8.4%. Stepwise logistic regression revealed that a history of cerebrovascular disease, peripheral vascular disease, atrial fibrillation, diabetes mellitus (DM), left ventricular ejection fraction of 30% or less, urgent operation, and a high perioperative transfusion requirement were independent predictors of delirium; beating-heart surgery and younger patient age were identified as having a significant protective effect. In more recent research, including systematic reviews and meta-analyses, these independent predictors of postoperative delirium in patients undergoing cardiac surgery have been reaffirmed. ,
In a prospective observational study, Hori and associates detected a higher incidence of delirium of 9.2% (42/491 patients) that was associated with mean arterial pressures (MAPs) above the near-infrared spectroscopy (NIRS)–detected upper limit of cerebral autoregulation during CPB (odds ratio [OR], 1.09; 95% CI, 1.03–1.15). A nested randomized clinical trial (RCT) of 199 patients, who underwent nonemergency cardiac surgery at a single academic center, reported that the odds of delirium was reduced by 45% in the group of patients whose MAPs were above their lower limit of autoregulation on CPB (OR, 0.55; 95% CI, 0.31–0.97).
In a prospective study of 221 patients, 31% developed delirium; higher Charlson’s comorbidity index, lower Mini-Mental State Examination (MMSE) score, length of CPB, and systemic inflammatory response syndrome in the intensive care unit (ICU) were independently associated with delirium. Other studies have identified biomarkers such as interleukin-2 (IL-2), IL-6, 26 tumor necrosis factor-α (TNF-α), neopterin, neuroserpin, and C-reactive protein as associated with postoperative delirium.
Compared with stroke, neurocognitive dysfunction is a considerably more frequent sequela of cardiac surgery, and it has been demonstrated in up to 80% of patients early after surgery. , The pathogenesis of neurocognitive dysfunction after cardiac surgery is still uncertain. Advanced age, concomitant cerebrovascular disease, the severity of cardiovascular disease, as well as the progression of underlying comorbid disease, have all been postulated as factors in the development of postoperative neurocognitive decline following cardiac surgery. , Several intraoperative variables have also been implicated, including cerebral emboli, hypoperfusion, activation of inflammatory processes, aortic cross-clamp or CPB time, low MAP, and cerebral venous hypertension. , In addition to the above, some studies have shown correlations between early postoperative neurocognitive dysfunction and intraoperative cerebral oxygen desaturation, as well as new ischemic lesions on postoperative MRI. , Whether early postoperative neurocognitive dysfunction represents permanent neurologic damage, or not, remains controversial.
In many instances, subtle signs of neuropsychological dysfunction are detectable only with sophisticated cognitive testing strategies; depression and personality changes may be noted by family members, however. Although cognitive testing can be used to distinguish between and optimize among various perioperative treatment modalities (e.g., the efficacy of putative cerebroprotectants, equipment modifications, pH management strategies), the incidence of neurocognitive dysfunction in different clinical studies is potentially unreliable as an index of the absolute incidence of neurocognitive dysfunction. Because of the high variability in methodologies among clinical studies (i.e., differing definitions of neurocognitive dysfunction, choice of tests, employment of relevant comparator groups) associated with a given procedure (e.g., CABG surgery), as shown by Murkin et al., cognitive testing is thus best used as a comparator tool to determine appropriate treatment modalities.
In a prospective study of 101 postcardiac surgical patients, the incidence of electroencephalographically detected seizures was 3% (two focal and convulsive, one generalized and electrographic) in which all three patients with seizures were 65 years or older, had open-chamber procedures, and underwent CPB for a duration longer than 120 minutes. Risk factors associated with seizures after cardiac surgery include open-chamber cardiac surgery, deep hypothermic circulatory arrest (HCA), aortic calcification or atheroma, critical preoperative state or high APACHE II score, tranexamic acid (TXA) exposure, , preoperative cardiac arrest, long CPB time, previous cardiac surgery, poor renal function, , older age, and peripheral vascular disease.
Independent of CNS injury, current evidence has focused on seizures associated with a high dosage of TXA (>30 mg/kg), an antifibrinolytic administered to decrease blood loss and transfusion requirements. , Studies have generally implicated open-chamber procedures associated with prolonged duration of CPB and renal failure as contributory to TXA-induced seizures.
The mechanism of seizures is thought to be due to increased TXA concentration in the cerebrospinal fluid, whether due to the impact of microgaseous emboli on vascular permeability or excess plasma concentration due to high dosages or impaired renal clearance, with impaired inhibitory GABAergic neurotransmission as a consequence of direct antagonism of cortical γ-aminobutyric acid A (GABA A ) postsynaptic receptors resulting in enhanced neuronal excitation. ,
In a series reported by Malone and colleagues, a correlation was made between neuropathologic lesions found at necropsy and the presence of intraoperative electroencephalographic (EEG) abnormalities (virtual or complete electrical silence) usually seen in conjunction with sustained hypotensive episodes. In all nine patients with clinical evidence of brain damage, cortical boundary zone (watershed) lesions were observed in the parieto-occipital areas. Malone and colleagues suggest that the parieto-occipital region is the most sensitive location for placement of recording EEG electrodes because it was where minimal boundary zone ischemic lesions occurred in the absence of other lesions. It was also where ischemic lesions were found in their maximal severity and extent.
A profound reduction in systemic blood pressure is the most frequent cause of boundary zone (watershed) infarcts. These areas are thought to be more susceptible to ischemia resulting from hypotension because they are situated at the border zone between the territories of two major cerebral arteries (e.g., the middle and posterior, or the anterior and middle cerebral arteries) where terminal and most distant arteriolar anastomoses exist ( Fig. 31.1 ). , Wityk and coworkers studied the pattern of ischemic changes on diffusion- and perfusion-weighted MRI in a case series of 14 patients and four patients, respectively, with neurologic complications after cardiac surgery. Acute ischemic lesions were classified as having a territorial, watershed, or lacunar pattern of infarction. Diffusion-weighted MRI revealed acute infarcts in 10 of the 14 patients, of whom four had combined watershed patterns of ischemia. Findings of perfusion-weighted MRI were abnormal, indicating cerebral hypoperfusion, in two of the four patients. Both patients had either fluctuating neurologic deficits or TIAs and their conditions improved with blood pressure increase.
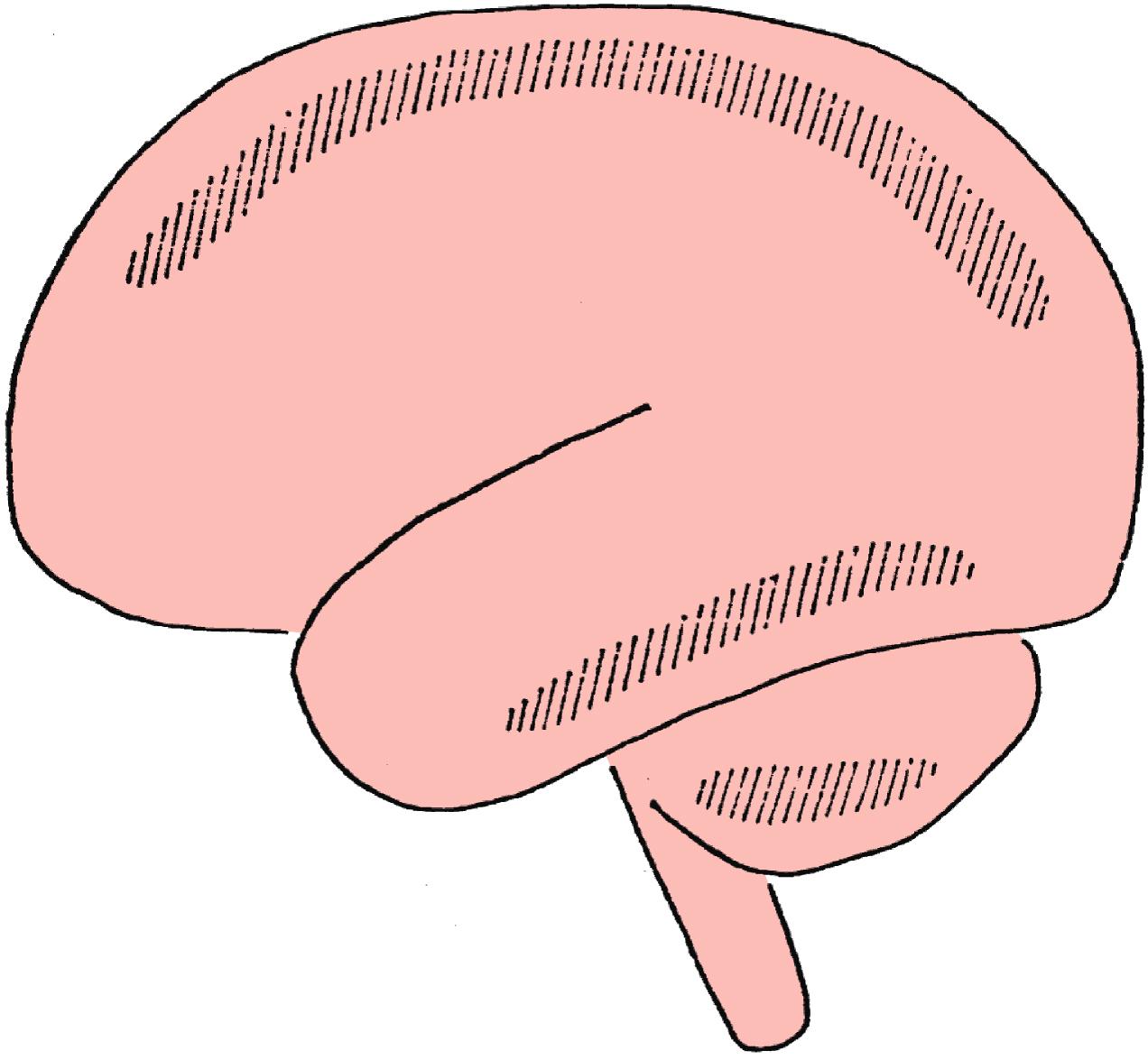
By the same rationale, however, these watershed areas are also highly susceptible to ischemia because of end-artery embolization. It is recognized that although severe hypotension is the most common cause of watershed infarcts, showers of microemboli may lodge preferentially in these areas and cause infarcts in the underlying brain. , As such, although they commonly arise from profoundly hypotensive episodes, watershed lesions are not pathognomonic of a hypotensive episode and may also be the result of cerebral emboli or a combination of both.
Determining which factor or, more likely, which combination of factors is responsible for postoperative neurologic dysfunction in patients undergoing cardiac surgery using CPB is problematic ( Box 31.1 ). How much of this dysfunction is a direct result of exposure to cardiac surgery or CPB, or due to the underlying comorbid disease, is an area of active ongoing investigation. From the few studies in which a surgical control group has been used, it appears that elements inherent to CPB are causative, particularly in dysfunction occurring in the immediate postoperative period. Underlying comorbid diseases, including aortic and cerebrovascular atherosclerosis, hypertension, DM, and atrial fibrillation, which predispose such patients to CNS dysfunction as a result of nonspecific stress associated with major surgery independent of CABG, clearly play a role as well.
Embolization
Hypoperfusion
Inflammation
Influencing factors
Aortic atheroma plaque
Cerebrovascular disease
Altered cerebral autoregulation
Hypotension
Intracardiac debris
Air
Cerebral venous obstruction on bypass
Cardiopulmonary bypass circuit surface
Reinfusion of unprocessed shed blood
Cerebral hyperthermia
Hypoxia
Based on the postmortem studies already highlighted, as well as correlative analyses of intraoperative events with neurologic outcomes, two primary mechanisms appear to be responsible for brain injury in otherwise uncomplicated cardiac operations: cerebral emboli and cerebral hypoperfusion. Whatever the mechanism of the cerebral insult, it appears that coexisting inflammatory processes can exacerbate the magnitude of injury. As such, the effect of inflammatory processes triggered during exposure to cardiac surgery and CPB is receiving more attention. ,
Cerebral emboli during cardiac surgery and CPB can be arbitrarily differentiated into macroemboli (e.g., calcific or atherosclerotic debris) and microemboli (e.g., microgaseous bubbles, microparticulate matter). Overt and focal neurologic damage likely reflects the occurrence of cerebral macroemboli (e.g., calcific and atheromatous debris generated during valve tissue removal or instrumentation of an atheromatous aorta), whereas seizures and less focal neurologic dysfunction have been ascribed to cerebral microemboli. Microemboli are primarily detected based on characteristic transcranial Doppler ultrasound (TCD) signature being transient (<300 ms), high intensity (>3 decibels above spectrum), high frequency (“chirpy”), and unidirectional.
In a study to assess the impact of surgical manipulation of the aorta and correlations with postoperative stroke, Ura and colleagues performed epiaortic scanning (EAS) before aortic cannulation and after decannulation in 472 patients undergoing cardiac surgery with CPB and followed them for new type I neurologic complications. Prior to aortic cannulation, grade 2 aortic atherosclerosis was present in 73 patients (15%) and grade 3 in 28 patients (6%). After aortic decannulation, EAS detected a new lesion in the ascending aorta intima in 16 patients (3.4%), of which 10 were severe with mobility or disruption of the intima ( Fig. 31.2 ). Twelve of the new lesions (some minor, some severe) were attributable to aortic clamping and four (all severe) to aortic cannulation. A total of 10 patients sustained neurologic complications, of whom three were diagnosed with postoperative stroke. The incidence rate of new lesions was directly related to the extent of aortic atheroma: 11.8% if the atheroma was approximately 3 to 4 mm thick, as high as 33.3% if the atheroma was greater than 4 mm, but only 0.8% when it was less than 3 mm.
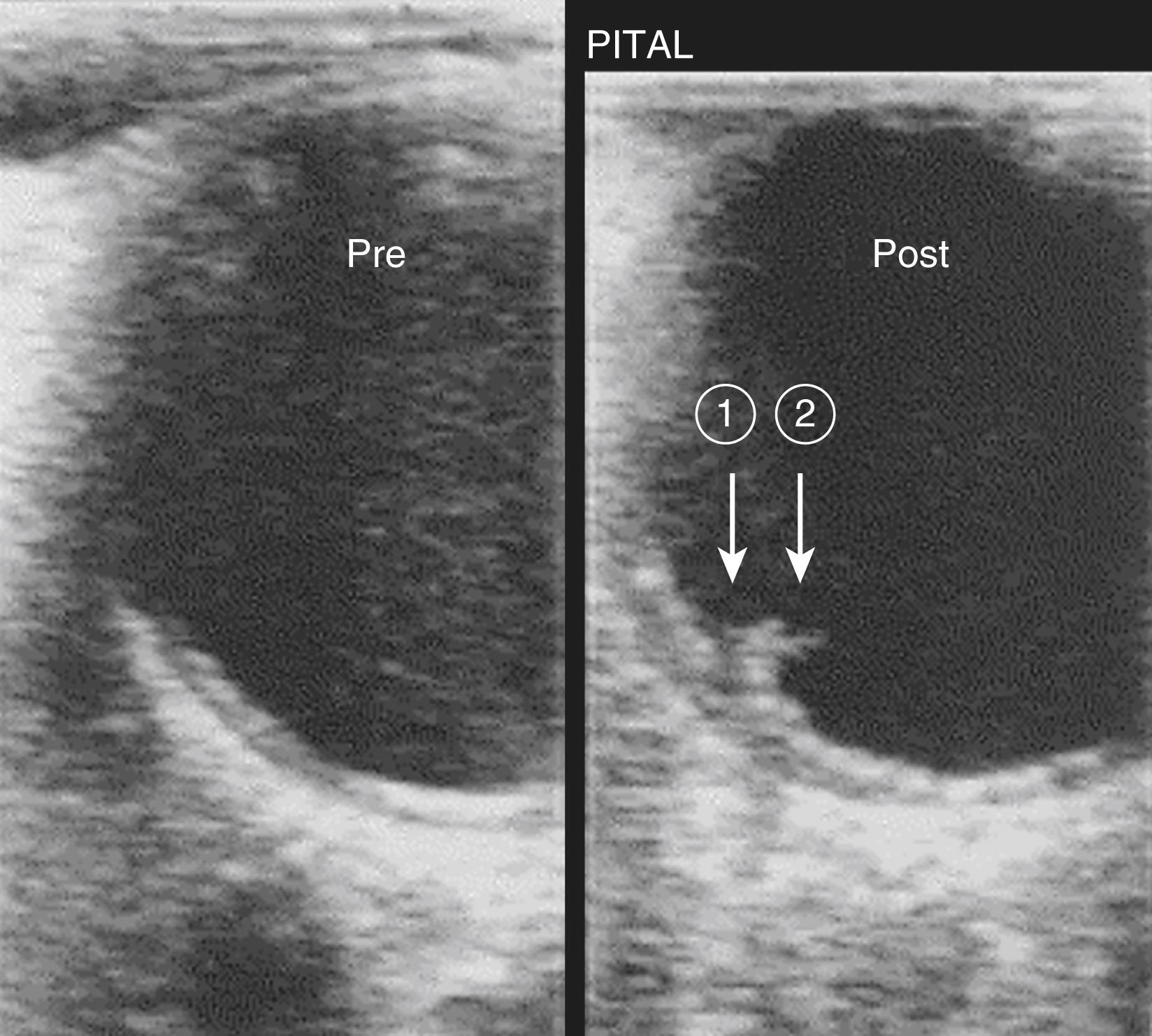
Recent data from the prospective European multicenter study on CABG, which involved 16 European centers, also show that avoiding or reducing aortic manipulation reduces the incidence of postoperative neurologic complications. This study included 7352 patients undergoing OPCAB and on-pump CABG with or without aortic cross-clamping; 684 had an elevated neurologic risk owing to a history of severe carotid stenosis or occlusion, prior carotid artery intervention, or previous stroke or TIA. A higher incidence of neurologic events was observed in the cross-clamp cohort compared with the cohort without an aortic cross-clamp, while the complete aortic “no-touch” cohort had the lowest incidence. These studies underscore the need to reliably detect and ultimately avoid disruption of aortic atherosclerotic plaque.
Sylivris and investigators studied 41 consecutive patients undergoing CABG with TCD monitoring and preoperative and postoperative MRI brain scans. A subgroup of 32 patients underwent neuropsychological testing the day before and 5 to 6 days after surgery, of whom 27 had reliable TCD data. Among the subgroup of patients with reliable TCD data, 17 (63%) of the 27 patients had early neuropsychological deficit after CABG. On univariate analysis, the duration of CPB and total microembolic load during bypass were significantly greater in the group with neuropsychological decline alone. Five patients had neuropsychological decline and radiologic evidence of cerebral infarction, of which four had a significant decline in neuropsychological functioning. Unlike the association between neuropsychological decline alone and higher total microembolic load during bypass, there was no relationship between these microembolic parameters and radiologic evidence of cerebral infarction. Not inconsistent with the findings of Ura and other studies, , there was a significantly greater microembolic load during aortic instrumentation in patients with cerebral infarction, temporally suggestive of particulate emboli, which was not apparent in comparison with patients with neuropsychological deficits alone.
A study with a newer generation TCD, which uses two different frequencies of insonation and purportedly distinguishes between gaseous and particulate emboli, compared the number and nature of intraoperative microemboli in 45 patients undergoing cardiac surgical procedures (15 OPCAB, 15 on-pump CABG, and 15 open cardiac procedures). The investigators demonstrated significantly fewer emboli in the OPCAB versus on-pump CABG and open procedure groups, averaging 40 (range, 28–80), 275 (range, 199–472), and 860 (range, 393–1321) emboli, respectively ( P < .01). Twelve percent of microemboli in the OPCAB group were defined as solid compared with 28% and 22% in the on-pump CABG and open procedure groups, respectively. In the on-pump surgery groups, 24% of microemboli occurred during CPB, and 56% occurred during aortic manipulation, that is, cannulation, decannulation, application, and removal of cross-clamp or side clamp, again underscoring the importance of minimizing aortic instrumentation.
Relatively little information regarding cerebral circulation in human beings during CPB appeared until 1983, when Henriksen and colleagues reported evidence of cerebral hyperemia occurring during CPB. This report was followed in 1984 by a seminal paper from Govier and coworkers; they incited controversy with their observations of ischemic threshold levels of cerebral blood flow (CBF) during CPB, in direct contrast with the hyperperfusion reported by Henriksen, and made preliminary observations on many of the other critical variables thought to influence CBF during CPB.
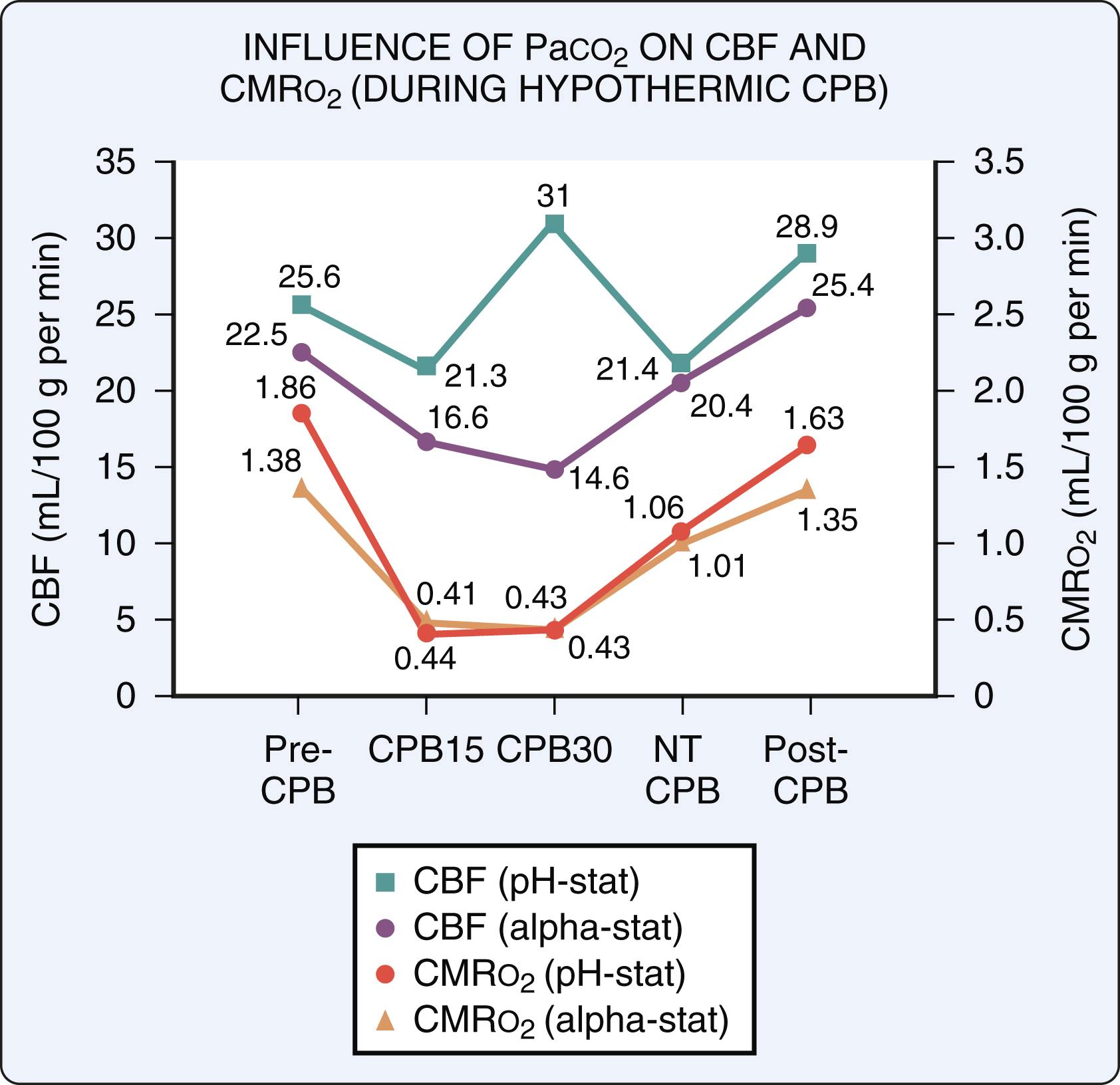
Without the measurement of concomitant cerebral metabolism (CMR o 2 ), these apparently discordant observations of CBF could not be reconciled. Murkin and coworkers subsequently reported their observations of both CBF and CMR o 2 during hypothermic CPB in humans, using a xenon-133 clearance technique for measurement of CBF, similar to techniques used by Govier and coworkers, and Henriksen and colleagues, but with the addition of a jugular bulb catheter for sampling effluent cerebral venous blood for measurement of cerebral metabolic activity. It was hypothesized that differences in pH management and thus arterial partial pressure of carbon dioxide (Pa co 2 ) accounted for the divergent values previously reported for CBF during hypothermic CPB. Accordingly, patients were managed with either alpha-stat or pH-stat pH management during hypothermic CPB. A similar and pronounced reduction in CMR o 2 was observed in both groups during hypothermia ( Fig. 31.3 ), and in the alpha-stat group, global cerebral flow/metabolism coupling was preserved in comparison with the group managed with pH-stat ( Fig. 31.4 ). Decreases in CBF and CMR o 2 , significantly lower than similar measures before and after CPB, were still evident after rewarming during normothermic nonpulsatile CPB. These low values for CBF and CMR o 2 were restored to control levels shortly after separation from CPB. Alpha-stat management preserved cerebral autoregulation and the relation between CBF and metabolism, and it has become the standard of care for adult patients undergoing CPB with mild and moderate hypothermia. ,
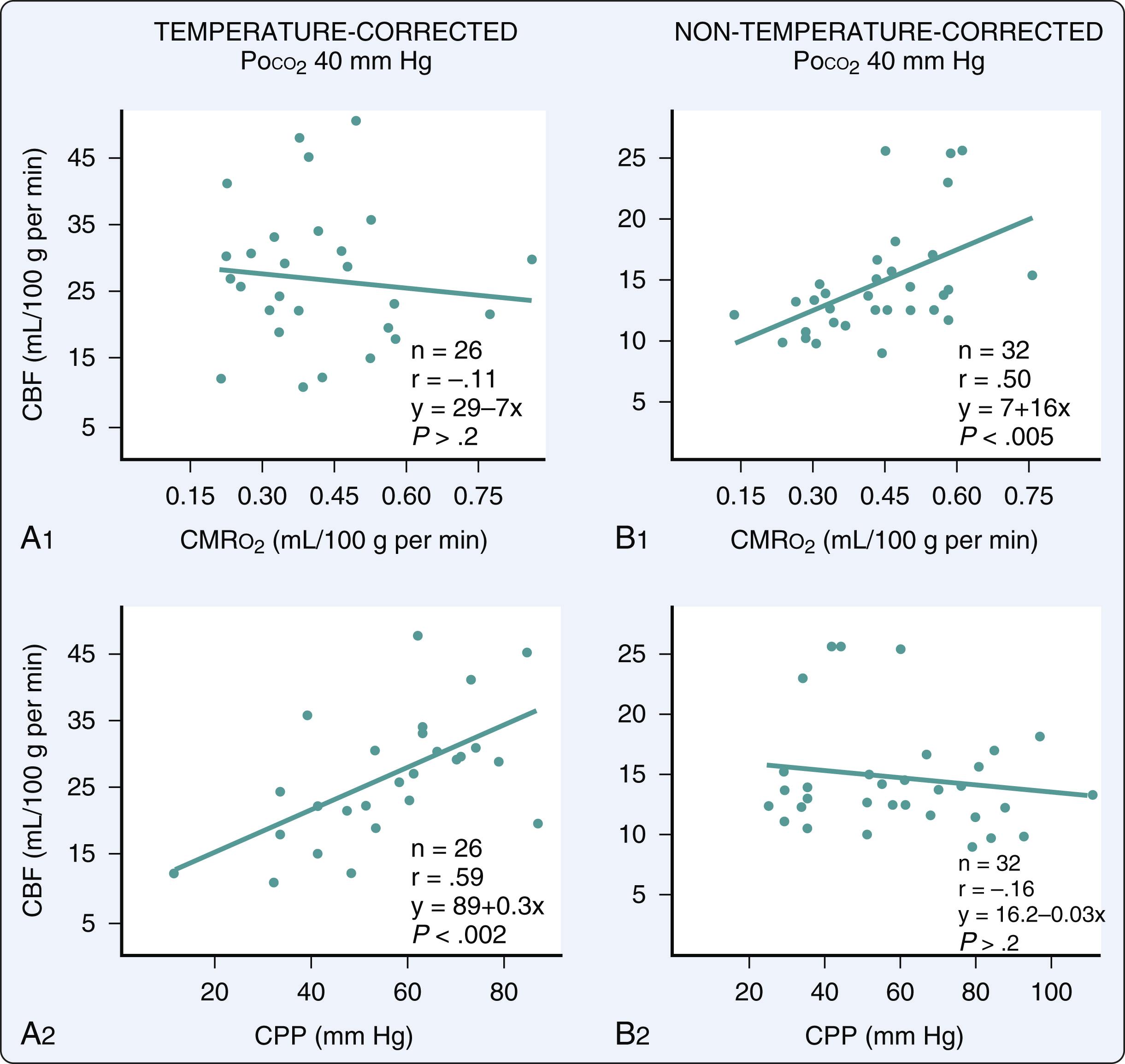
During the transition to CPB, the brain is particularly vulnerable to ischemia. The brain is initially perfused with an asanguinous prime, and even after equilibration during established CPB, hematocrit is generally maintained at a range between 20% and 30%. As a result, any further decreases in cerebral perfusion, in the absence of concomitant decreases in CMR o 2 , are poorly tolerated. As discussed later, it cannot be overemphasized that cerebral perfusion pressure (CPP) is not simply MAP, but rather represents the difference between MAP inflow and jugular venous or superior vena cava (SVC) outflow pressures; venous hypertension such as may result from cannula malplacement or dislocation of the heart thus has an impact to decrease CPP proportionately.
Using radioisotope techniques for measurement of CBF, and incorporating a jugular venous catheter for calculation of CMR o 2 , it was determined that there is a profound reduction in CMR o 2, exceeding 50% for a 10°C reduction in temperature, during hypothermic CPB and that CBF decreases proportionately and will autoregulate down to a CPP of 20 mm Hg, in the presence of alpha-stat pH management. This suggests that low CPP during the hypothermic phase of CPB is unlikely to result in cerebral ischemia in the absence of cerebrovascular disease. Unfortunately, the continuous increase in age and associated comorbidities in patients undergoing CPB renders the likelihood of cerebrovascular disease even more likely. This is not inconsistent with data demonstrating a high incidence of cerebrovascular disease in coronary revascularization patients. ,
Using NIRS to detect cerebral autoregulation in 450 patients undergoing CABG and/or valve surgery, the area under the curve of the product of the duration and magnitude of blood pressure, below the limits of cerebral autoregulation, was compared between patients with and without major postoperative morbidity or mortality (e.g., stroke, renal failure, mechanical lung ventilation > 48 h, inotrope use > 24 h, or intraaortic balloon pump insertion). Eighty-three of the 450 patients experienced major morbidity or operative mortality, and the study demonstrated that the area under the curve below the limits of cerebral autoregulation was independently associated with major morbidity or operative mortality after cardiac surgery. This is directionally similar to what Murkin and coworkers initially reported in a prospective study of NIRS that also demonstrated an association between cerebral NIRS desaturation and major morbidity or operative mortality.
Given the uncertainty on the optimum MAP for the entire cardiac surgical population during CPB, , real-time cerebral autoregulation monitoring might be useful in guiding individualization of MAP targets to potentially improve morbidity and mortality outcomes following cardiac surgery. ,
It should also be appreciated that during CPB, cerebral venous hypertension can result from partial obstruction of the SVC ( Fig. 31.5 ), particularly in the presence of a single two-stage venous cannula, and may cause cerebral edema and produce a disproportionate decline in CPP relative to arterial pressure.
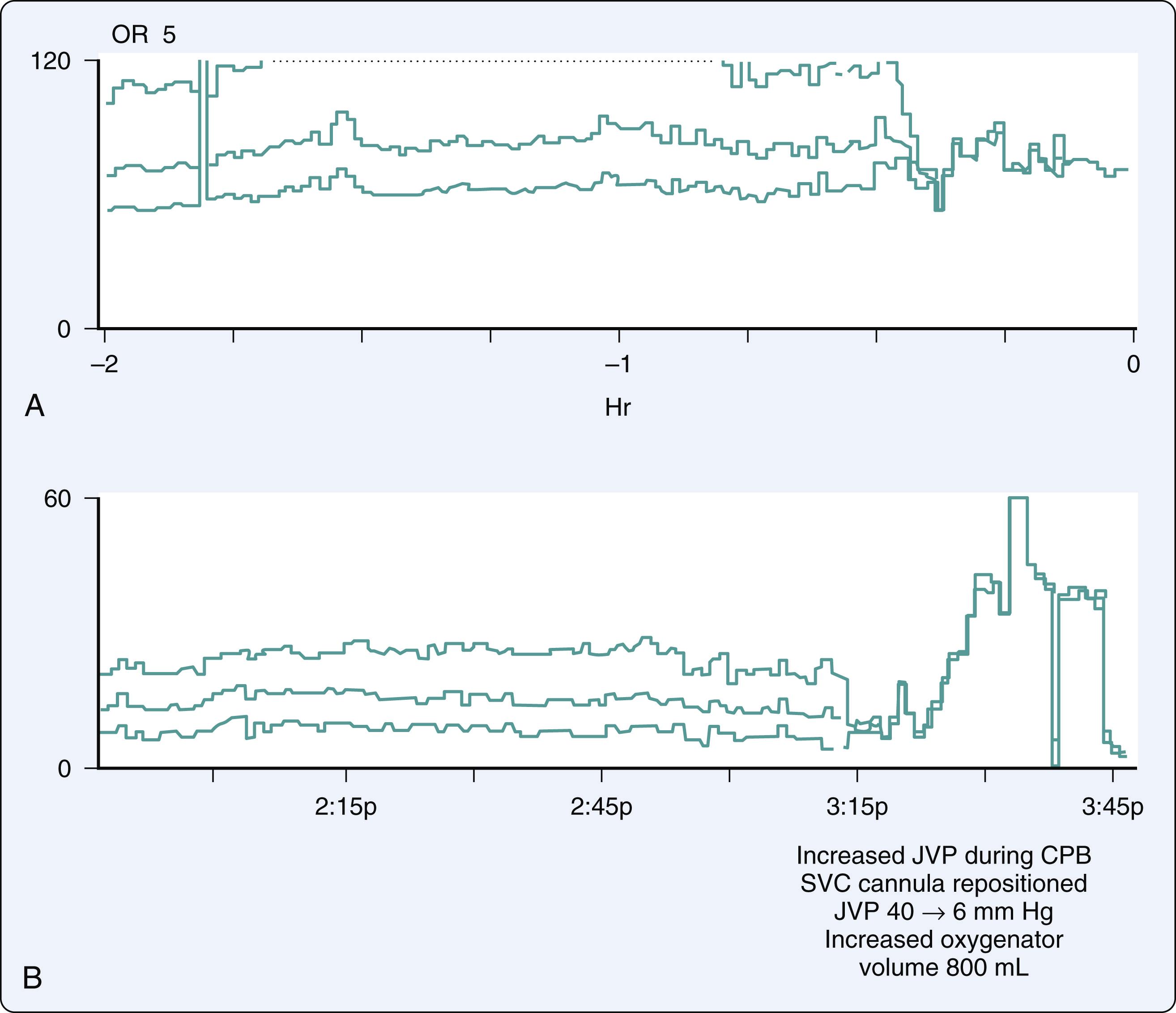
In a study by Avraamides, surgical displacement of the heart during CPB produced increases in proximal SVC pressure and resulted in significant decreases in CBF velocity as measured with TCD, despite stable arterial pressure and pump flow rates. This strongly suggests that cerebral venous hypertension, as can occur during CPB with myocardial dislocation and impaired drainage of the SVC, may result in cerebral ischemia if unrecognized and untreated. It is feasible that such unrecognized cerebral venous hypertension has resulted in some of the postoperative neurologic syndromes that have been reported.
In a study of 92 patients undergoing CABG, the effect of changes in jugular venous bulb pressure (JVBP) on the blood-brain barrier was detected by retrograde cannulation of the jugular bulb to measure concentrations of plasma S-100β protein, matrix metalloproteinase 9, creatine kinase isoenzyme BB (CK-BB), and arteriovenous total and ionized magnesium as biomarkers of blood-brain barrier dysfunction. CPB increased JVBP, and that JVBP, elevated above 12 mm Hg, increased biomarkers of blood-brain barrier disruption.
Before, during, or after surgery, hemodynamic complications have been found to increase cerebral injury in cardiac surgical patients. Ganushchak and coworkers tested the hypothesis that combinations of hemodynamic events from apparently normal CPB procedures are related to the development of postoperative neurologic complications and affect the impact of common clinical risk factors. In a retrospective analysis of 1395 patients, they reported that the frequency of neurologic complications was 3.9% in the group of patients who experienced large fluctuations in hemodynamic parameters while on CPB, whereas in the group of patients with more stable values on CPB, the incidence rate of neurologic complications was 0.3%. This finding suggests an increased susceptibility of the brain to cerebral injury in hemodynamically unstable cardiac surgical patients resulting from increased inflammatory processes.
In the study by Ganushchak and coworkers, a multivariate statistical procedure was applied to a data set of automatically recorded perfusions from 1395 patients who underwent CABG. The following five parameters emerged for analysis: MAP, dispersion of MAP, dispersion of systemic vascular resistance, dispersion of arterial pulse pressure, and the maximum value of mixed venous saturation. Using these parameters, the researchers identified four significantly different clusters; cluster 4 represented procedures with high alterations of hemodynamic parameters during CPB compared with stable procedures in cluster 1. Importantly, the impact of common clinical risk factors on postoperative neurologic complications was affected by the performance of the CPB procedure. For example, the frequency of neurologic complications among patients with cerebrovascular disease in their medical history was 22% in the fourth cluster, whereas it was zero in the second cluster. Hence, patients with cerebrovascular disease who undergo CPB procedures with large fluctuations in hemodynamic parameters may manifest a greater inflammatory response and are thus at particularly increased risk for developing postoperative neurologic complications. , This is of particular importance since it has been estimated that more than 50% of CABG patients have coexisting cerebrovascular disease. ,
Hyperthermia can strongly impact neurologic outcomes by altering cerebral oxygen transfer and glutamate levels, leading to eventual cell death. Okano and associates assessed the effects of normothermia and mild hypothermia (32°C) during CPB on jugular venous oxygen saturation (Sjv o 2 ) in 20 patients scheduled for elective CABG. The Sjv o 2 in the normothermic group was decreased significantly at 20 and 40 minutes after CPB onset compared with pre-CPB, whereas Sjv o 2 in the mild hypothermic group did not change during the study. The investigators concluded that cerebral oxygenation, as assessed by Sjv o 2 , was increased during mild hypothermic CPB compared with normothermic CPB.
Kawahara and colleagues examined the effect of rewarming rates on jugular venous oxygen saturation in 100 patients scheduled for elective CABG and randomly divided them into a control group and a slow rewarming group. Cerebral desaturation (defined as a Sjv o 2 value < 50%) during rewarming was more frequent in the control group than in the slow group. The ratio of the cerebral desaturation time to the total CPB time in the control group differed significantly from those in the slow group (control group: 17 ± 11 min, 12% ± 4%; slow group: 10 ± 8 min, 7% ± 4%, respectively; P < .05). Consistent with this, in a study of the impact of rate of rewarming on cognitive outcomes in 165 CABG patients, randomized to two differing rewarming strategies, Grigore and colleagues demonstrated a significant association between change in cognitive function and rate of rewarming. Hence, rapid rewarming may also exacerbate a preexisting cerebral injury by creating a mismatch between cerebral oxygen consumption and delivery.
Grocott and colleagues recorded hourly postoperative temperatures in 300 patients undergoing CABG on CPB and determined the degree of postoperative hyperthermia using peak temperature within 24 hours and area under the curve for temperatures higher than 37°C. Patients underwent a battery of cognitive testing before surgery and 6 weeks after surgery. The maximum temperature within 24 hours after CABG ranged from 37.2°C to 39.3°C. Findings showed that the maximum postoperative temperature was independently associated with cognitive dysfunction at 6 weeks.
Accordingly, a slow rewarming rate, with lower peak temperatures during CPB, may be an important factor in the prevention of neurocognitive decline after hypothermic CPB, and interventions to avoid postoperative hyperthermia may be warranted to improve cerebral outcomes after cardiac surgery.
In a review of 67,764 cardiac surgical patients who underwent cardiac surgery at 22 centers in the National Cardiovascular Network, of whom 4743 were octogenarians, Alexander and coworkers reported that the incidence of type I cerebral injury (defined by Roach et al.) was 10.2% in patients older than 80 years versus 4.2% in patients younger than 80. Although the crude mortality rate for cardiac surgery in octogenarians was greater than in younger patients, the researchers reported that when octogenarians without significant comorbidities were considered, their mortality rates were similar to those of younger patients.
This observation has been confirmed in a single-center study of 418 consecutive patients older than 80 years who underwent cardiac surgery between 2000 and 2012 and were matched according to gender, surgical procedure, and comorbidities with 426 younger patients. This study identified postoperative stroke in 4.1% of octogenarian patients and 3.5% of matched controls ( P = .65). In younger patients, peripheral vascular disease and cardiac rhythm disturbances were significant risk factors, whereas factors related to intraoperative brain oxygenation (i.e., preoperative anemia) were the most critical determinant of stroke in older patients. In a review from the STS National Adult Cardiac Surgery database of 774,881 patients undergoing isolated CABG between January 2002 and December 2006, the overall incidence rate of stroke was 1.4%, increasing to 2.3% in patients aged 75 years and older. Stroke rate was inversely related to body surface area, which may again reflect a decrease in red cell mass and a greater degree of hemodilution during CPB, and was directly proportional to serum creatinine concentration, as well as the presence of valvular heart disease and other comorbidities.
In this regard, reports from Europe and North America consistently describe DM, hypertension, previous cerebrovascular disease, peripheral vascular disease (including carotid disease), aortic atherosclerosis, renal dysfunction, myocardial infarction or unstable angina within 24 hours before surgery, as well as intraoperative and postoperative complications, as being additional factors increasing the incidence of cerebral injury in cardiac surgical patients ( Box 31.2 ). Determining the impact of age-associated cerebral injury in cardiac surgery is becoming more relevant because of the progressive increase in the average age of the general population and, in particular, of the cardiac surgical population. The presence of preoperative comorbidities is increasingly recognized as the primary determinant of the age-associated risk for CNS complications. As overall survival and quality of life after cardiac surgery continue to improve in older patients, advanced age alone is no longer considered a deterrent when evaluating a patient for cardiac surgery, with some centers reporting similar incidences of stroke in octogenarians independent of whether on-pump CABG or OPCAB was employed. , The presence and extent of comorbidities should be considered as being of equal or greater importance than age itself as a risk factor for cerebral injury in cardiac surgical patients.
Aorta atheromatosis
Carotid disease
Diabetes mellitus
Hypertension
Peripheral vascular disease
Renal dysfunction
Stroke or cerebrovascular disease
Recent unstable angina or acute myocardial infarction
Preoperative low output/low ejection fraction
Combined/complex procedures
Redo surgery
Prolonged cardiopulmonary bypass time
Intraoperative hemodynamic instability
Postoperative atrial fibrillation
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here