Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Metastases are responsible for more than 90% of all cancer-related deaths.
Gene dysregulation, the tumor microenvironment, and host cells drive the metastatic spread of tumor cells.
Metastasis can be subdivided into invasion and migration from the primary tumor; intravasation into the vasculature; dissemination and survival in the circulation; extravasation from the vasculature; survival and metabolic adaptation in the distant tissue; dormancy; and reactivation and proliferation in the new tissue microenvironment.
Colonization of metastatic tumor cells requires the ability to metabolically adapt, develop angiogenesis, overcome dormancy, and proliferate in a foreign tissue.
The formation of a premetastatic niche is essential for the growth of extravasating metastatic tumor cells.
Organ specificity of tumor metastases is determined both by blood flow and by tissue-specific factors.
Primary tumors possess stem cells that can recapitulate the tumor from a single cell, and a subset of these cancer stem cells may inherently possess altered gene expression changes with increased metastatic potential.
Antimetastatic therapy will likely require the targeted inhibition of many pathways that control proliferation, invasion, angiogenesis, and immune evasion.
One of the most important challenges in clinical oncology is the prevention and treatment of metastatic disease. With advances in surgical techniques and conventional and targeted therapies, localized disease is effectively managed in the clinic. However, metastatic disease is the primary cause of cancer-related deaths. Tumor metastasis involves tumor cell invasion and migration from the primary tumor, intravasation into the vasculature, dissemination and survival in the circulation, extravasation into distant tissues, survival and metabolic adaptation in the distant tissue, dormancy, reactivation, and overt colonization to form a new macroscopic tumor at a distant site. This process is highly inefficient; it has been estimated that less than 0.01% of tumor cells that enter the circulation develop into metastases. Despite this inefficiency, metastases are responsible for more than 90% of all cancer-related deaths. Understanding the biology and vulnerabilities of metastatic tumor cells is of critical importance to improve overall survival rates in cancer patients. With little evidence to support mutations in “metastasis genes” as drivers of metastasis, current data suggest that microenvironmental factors as well as epigenetic changes may play key roles in metastasis. This chapter describes the cellular and molecular traits driving tumor metastasis and addresses how the tumor microenvironment influences this process. Most important, it discusses how this knowledge can be translated into current and future cancer therapies.
Although cellular intrinsic traits acquired by tumor cells are required for successful metastatic colonization, cellular and molecular factors within the tumor microenvironment significantly contribute to metastatic progression. The tumor microenvironment contains a number of cell types that promote tumor progression, including cancer stem cells, angiogenic vascular cells, infiltrating immune cells, and cancer-associated fibroblasts (CAFs). In addition, extracellular factors within the tumor microenvironment such as hypoxia and extracellular vesicles promote metastatic phenotypes within these cell types.
The traditional view of tumors with a relatively homogeneous population of tumorigenic cells has been significantly revised with the isolation and characterization of cancer stem cells. Stem cells are primal cells that retain the ability to renew themselves through cell division and can differentiate into a wide range of specialized cell types. Cells with stem cell properties have been identified in a variety of solid tumors including colon, breast, head and neck, and pancreatic tumors, glioblastomas, medulloblastomas, and melanoma. This rare subpopulation of tumor cells exhibits enhanced tumor-initiating potential: the ability to self-renew and differentiate into multiple cell types.
The origin of cancer stem cells remains unclear and may differ among tumor types. It has been hypothesized that cancer stem cells arise from either transformed resident stem and/or progenitor cells, or may represent a dedifferentiated epithelial tumor cell. In intestinal cancer, mouse modeling studies have identified intestinal stem cells as the cells of origin. For example, intestinal stem cell–specific deletion of adenomatous polyposis coli (APC) leads to rapid cellular transformation with uncontrolled cellular growth and solid tumor formation. In contrast, deletion of APC in short-lived transit-amplifying cells was not sufficient to induce long-term tumor growth. Stem cell properties can also be acquired by tumors cells through epithelial-mesenchymal transition (EMT). Induction of EMT in immortalized human mammary epithelial cells was sufficient to induce the expression of stem cell markers, enhance self-renewal, and increase the number of tumor-initiating cells. Although the molecular mechanisms that drive the cancer stem cell phenotype in tumor cells remains largely unknown, a study showed that the transcription factors Slug and Sox9 drive EMT and the cancer stem cell phenotype in breast cancer cells. Future studies are eagerly awaited to further delineate the intracellular signaling pathways driving the cancer stem cell phenotype in vivo.
The role of cancer stem cells in multistage tumor progression, particularly with respect to metastasis, is poorly defined. Given that metastasis is an infrequent event that is achieved by only a small portion of cancer cells that reach distant sites, it has been hypothesized that cancer stem cells may be responsible for metastatic disease. Evidence to support this hypothesis has been generated in models of breast and pancreatic cancer. In the MMTV-PyMT model of breast cancer, Malanchi and colleagues observed that only the CD90+ CD24+ population of cancer stem cells isolated from primary breast tumors contained cells with the ability to form metastases in the lung when introduced into the tail vein. Furthermore, this study identified the extracellular matrix protein, periostin, produced by fibroblasts and stromal tumor cells, as a critical factor to maintain the cancer stem cell phenotype and metastatic potential in these cells. In human breast cancers, single-cell analysis of early-stage metastatic cells demonstrated that these samples express distinct stemlike, EMT, prosurvival, and dormancy-associated gene expression signatures compared with metastatic cells from high-burden tissues. Moreover, transplanted stemlike metastatic cells from low-burden tissues exhibited tumor-initiating capacity and were able to differentiate into luminal-like cancer cells. Similarly in pancreatic cancer, lineage-tracing studies showed that circulating pancreatic tumor cells exhibit EMT and cancer stem cell properties, and initiate tumor formation. A critical role for inflammation to maintain the ability of cancer stem cells to metastasize was observed in this study. In addition, the extracellular protein tenascin C (TNC), expressed by stem cell niches and cancer cells, was also found to promote stem cell signaling and lung metastases in breast cancer cells. Collectively, these data strongly implicate the tumor microenvironment in promoting the cancer stem cell phenotype and the initiation of tumor metastasis.
Tumor angiogenesis facilitates the hematogenous spread of metastatic tumors. The aberrant production of proangiogenic factors by tumor cells results in malformed and irregular tumor blood vessels that often contain breaks in their lining that facilitate tumor cell intravasation and dissemination. Endothelial cells and pericytes are important structural and functional components of the tumor and distant tissue vasculature, and both have been considered as potential targets in metastatic therapy.
Endothelial cells play an active role in tumor cell intravasation, extravasation, and dormancy ( Fig. 3.1 ). Studies have shown that metabolic reprogramming of tumor endothelial cells toward a glycolytic phenotype contributes to the early stages of metastasis by promoting an abnormal tumor vasculature, tumor intravasation, and dissemination. Cantelmo and colleagues performed mRNA sequencing analysis of tumor-associated endothelial cells and discovered that tumor endothelial cells expressed a hyperglycolytic signature compared with normal endothelial cells. The glycolytic phenotype within tumor-associated endothelial cells could be inhibited through genetic and pharmacologic inhibition of the glycolytic enzyme 6-phosphofructo-2-kinase/fructose-2,6-bisphosphatase-3 (PFKFB3). Moreover, PFKFB3 inhibition in endothelial cells was sufficient to normalize the tumor vasculature and significantly reduce tumor cell intravasation and metastasis. There are multiple mechanisms by which PFKFB3 inhibition in endothelial cells may have impaired metastatic dissemination, including enhancing the integrity of the endothelial cell barrier, improving vessel maturation and pericyte coverage, and inhibiting cancer cell adhesion molecules. These findings suggest that targeting glucose metabolism in tumor endothelial cells may offer therapeutic benefit for anticancer therapy.
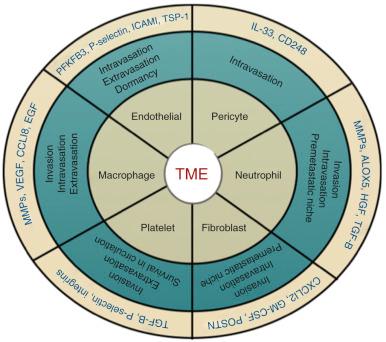
Adhesion to the endothelium is an initial step in extravasation that is followed by transendothelial cell migration. Tumor cell adhesion to the endothelium requires the expression of cognate ligands and receptors by cancer cells and endothelial cells. A variety of ligand-receptor interactions have been shown to contribute to tumor extravasation, including interactions with selectins, integrins, cadherins, CD44, and immunoglobulin (Ig) superfamily receptors (for a review, see Reymond and colleagues ). For example, endothelial cell P- and E-selectins bind to tumor cells through cell-cell adhesion molecules such as integrins and CD44. Studies have suggested a role for the immunosuppressive cytokine interleukin (IL)-35 in facilitating cancer cell adhesion to endothelial cells. IL-35 was found to be highly expressed by human pancreatic cancer cells in which it promoted ICAM1 expression to facilitate endothelial cell adhesion and transendothelial migration via an ICAM1-fibrogen-ICAM1 bridge.
Finally, endothelial cells within in the perivascular niche of the bone marrow have been implicated in promoting tumor cell dormancy. Similar to hematopoietic stem cells, dormant cancer cells have been found to be localized within perivascular niches of the bone marrow microenvironment. Within stable microvasculature niches, endothelial cells promote tumor cell quiescence through the production of thrombospondin-1 (TSP1) . In contrast, endothelial cells isolated from sprouting neovasculature promote tumor proliferation through the production of tumor-promoting factors including transforming growth factor–β1 (TGF-β1) and periostin from sprouting endothelial cell tips.
Similar to endothelial cells, pericytes are thought to play a protumorigenic role by supporting blood vessel maturation and function (see Fig. 3.1 ). Pericytes promote endothelial cell survival and stabilize the tumor vasculature. In addition, pericytes can directly facilitate distant metastasis by promoting tumor cell intravasation through the upregulation of the transmembrane receptor endosialin (CD248). Studies have demonstrated that pericytes can also promote tumor cell intravasation and metastasis through the IL-33–dependent recruitment of macrophages. Therefore it has been proposed that targeting pericytes alone or in combination with endothelial cells may be an effective strategy in the treatment of cancer. However, clinical data indicate that low pericyte coverage is associated with metastasis and poor prognosis in a number of cancers. A recent study demonstrated that depletion of pericytes suppressed tumor growth and enhanced metastasis of murine models of breast cancer. Enhanced metastasis was associated with increased levels of tumor hypoxia, EMT, and Met receptor activation. These findings indicate that pericyte coverage may be important to stabilize the tumor vasculature and prevent the hypoxic selection of aggressive tumor cells (see later discussion of hypoxia and metastasis). These findings raise concerns when considering antipericyte treatments in cancer therapy, especially in the setting of metastasis.
Most solid tumors contain immune infiltrates derived from both the myeloid and lymphoid lineages that can have both positive and negative effects on metastasis. This section focuses on immune cells that stimulate metastatic progression.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here