Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Cell adhesion is essential for the development and maintenance of multicellular organisms. Cell-to-cell and cell-to-matrix adhesion provide a mechanism for intercellular communication and to define the three-dimensional architecture of organs. The regulated nature of cell adhesion is particularly evident in the hematopoietic system, where blood cells routinely make transitions between nonadherent and adherent phenotypes during differentiation, and in response to stimuli in the circulation or extravascular space.
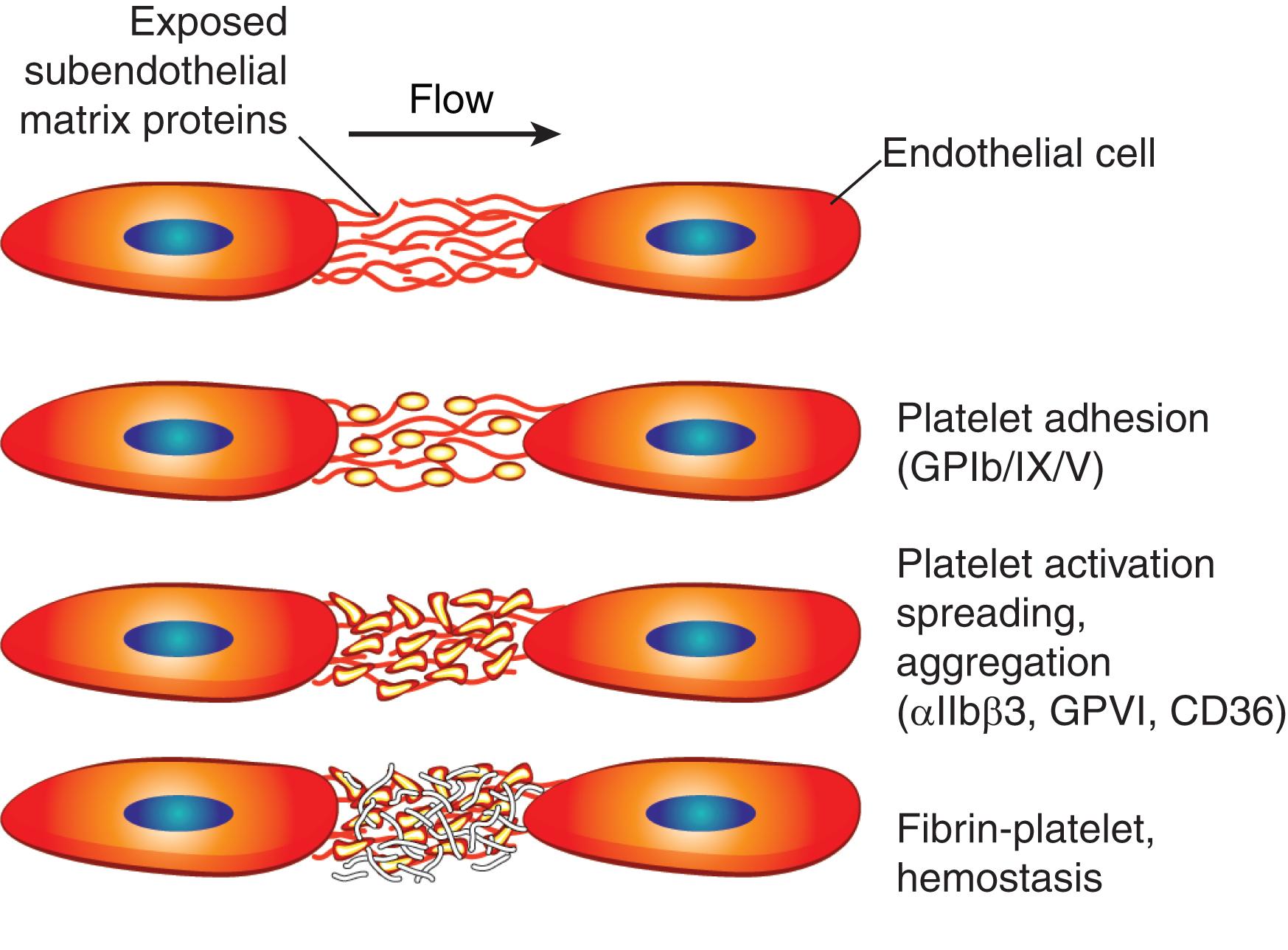
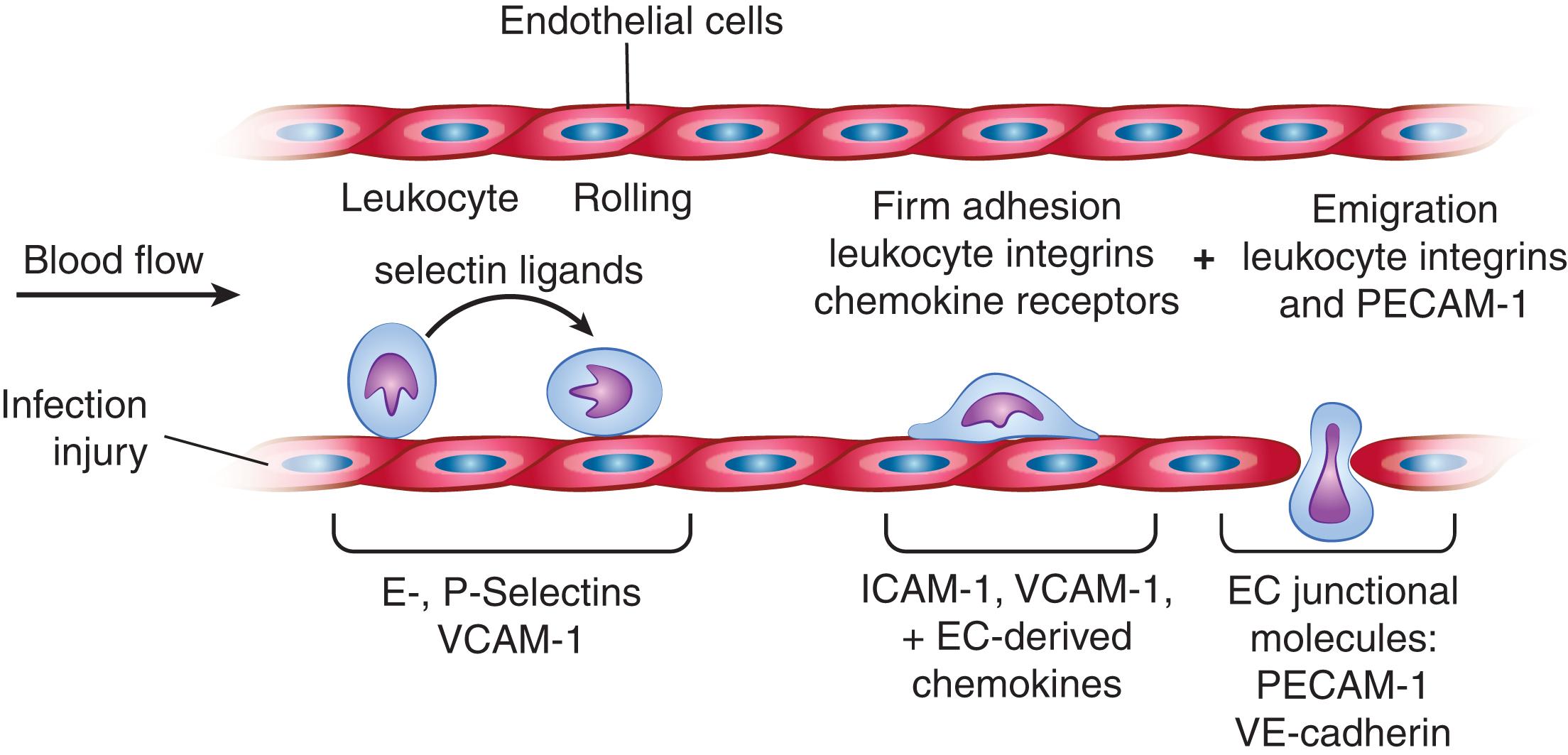
In the bone marrow (BM), hematopoietic stem cells reside in a specialized microenvironment called the stem cell niche , and their proliferation and differentiation are controlled not only by soluble growth factors but also by adhesion to stromal cells and matrix molecules (see Chapter 14, Chapter 16 AU: Please check and confirm cross reference to Chapters 14 and 16. Chapter 14 for more detailed discussion). Weakening of these adhesive interactions is required for mature blood cells to enter the circulation. Circulating erythrocytes normally remain nonadhesive until they become senescent and are finally cleared by the reticuloendothelial system (see Chapter 34 for more discussion). Other circulating blood cells often participate in regulated adhesive events during their lifespan. For example, prothymocytes adhere to thymic stromal cells where they undergo guided movement from the cortex to the medulla during maturation before reentering the circulation (see Chapter 24 for details of T cell development). T cells regularly adhere to the specialized high endothelial venules of lymph nodes in tissues, migrate into these organs for the sampling of processed antigens, and then exit via the lymphatics to recirculate in the blood (see Chapter 12 for more discussion). During inflammation, specific classes of leukocytes roll at very low velocity on acutely or chronically stimulated endothelium that line blood vessels, then adhere more tightly, and finally emigrate between endothelial cells into the tissues. There, neutrophils and monocytes phagocytose invading pathogens, and lymphocytes adhere to antigen-presenting cells, such as dendritic cells, B cells, and macrophages (see Chapter 123 for more discussion). During hemorrhage, platelets adhere to exposed subendothelial matrix components, spread, and recruit additional platelets into large aggregates that serve as an efficient surface for thrombin and fibrin generation. This is discussed in more detail later in Chapter 121 . Leukocytes also adhere to activated platelets and to other leukocytes, and platelets also roll on the endothelium. When activated, endothelial cells increase the expression of molecules that affect the adhesiveness of platelets or leukocytes. Tight contacts between adjacent endothelial cells also regulate access of blood cells to the underlying tissues and passage of plasma proteins and fluid (edema).
Cells adhere through noncovalent bond formation between macromolecules on cell surfaces with macromolecules on other cell surfaces or in the extracellular matrix (ECM). These interactions involve either protein–protein or protein–carbohydrate recognition. Although some adhesion molecules are expressed only by blood or endothelial cells, most also are synthesized by other cells. Many adhesion molecules can be grouped into families according to related structural and functional features.
The ECM provides structural and mechanical support for many tissues and spatial cues that enable cell–cell communication and signaling. The principal constituents of the ECM are adhesive proteins and proteoglycans. The major proteins are collagens, von Willebrand factor (vWF), thrombospondins, elastin, fibronectin, laminin, and vitronectin. These proteins are large and often highly extended and consist of multiple domains with different binding functions. In some proteins such as fibronectin, alternative splicing can increase diversity by producing molecules with variable numbers of domains. In addition, stretching of fibronectin can expose cryptic binding sites. The many binding domains allow adhesive proteins to interact with each other as well as with cell-surface receptors, resulting in multipoint contacts that stabilize matrix structure. One adhesive protein, fibrinogen, is found predominantly in plasma but also may be deposited in the exposed subendothelial matrix after vascular injury. Fibronectin, vitronectin, thrombospondins, and vWF are located predominantly in the ECM but also are found in plasma in lower amounts. Several adhesive proteins also are stored in intracellular α-granules of platelets, where they are secreted after platelet activation at sites of vascular injury. Similarly, the endothelium stores adhesive proteins in cytoplasmic storage granules, called Weibel–Palade bodies , that are released upon injury or activation.
Proteoglycans contain protein cores to which are covalently attached many glycosaminoglycans—long linear polymers of repeating disaccharides. Most proteoglycans are in the ECM, but some are anchored on cell surfaces through a core protein that contains a membrane-spanning domain. Hyaluronan is a unique glycosaminoglycan that forms polymers with molecular masses up to several million daltons that are not covalently attached to a protein. Hyaluronan forms noncovalent interactions with globular domains on the protein core of proteoglycans and with a small molecule called link protein . The resultant hyaluronan–proteoglycan complexes can become very large, contributing to the structural stability of the matrix and function as space fillers during embryonic development. Hyaluronan can also bind to cell-surface receptors and is also abundantly produced during wound healing.
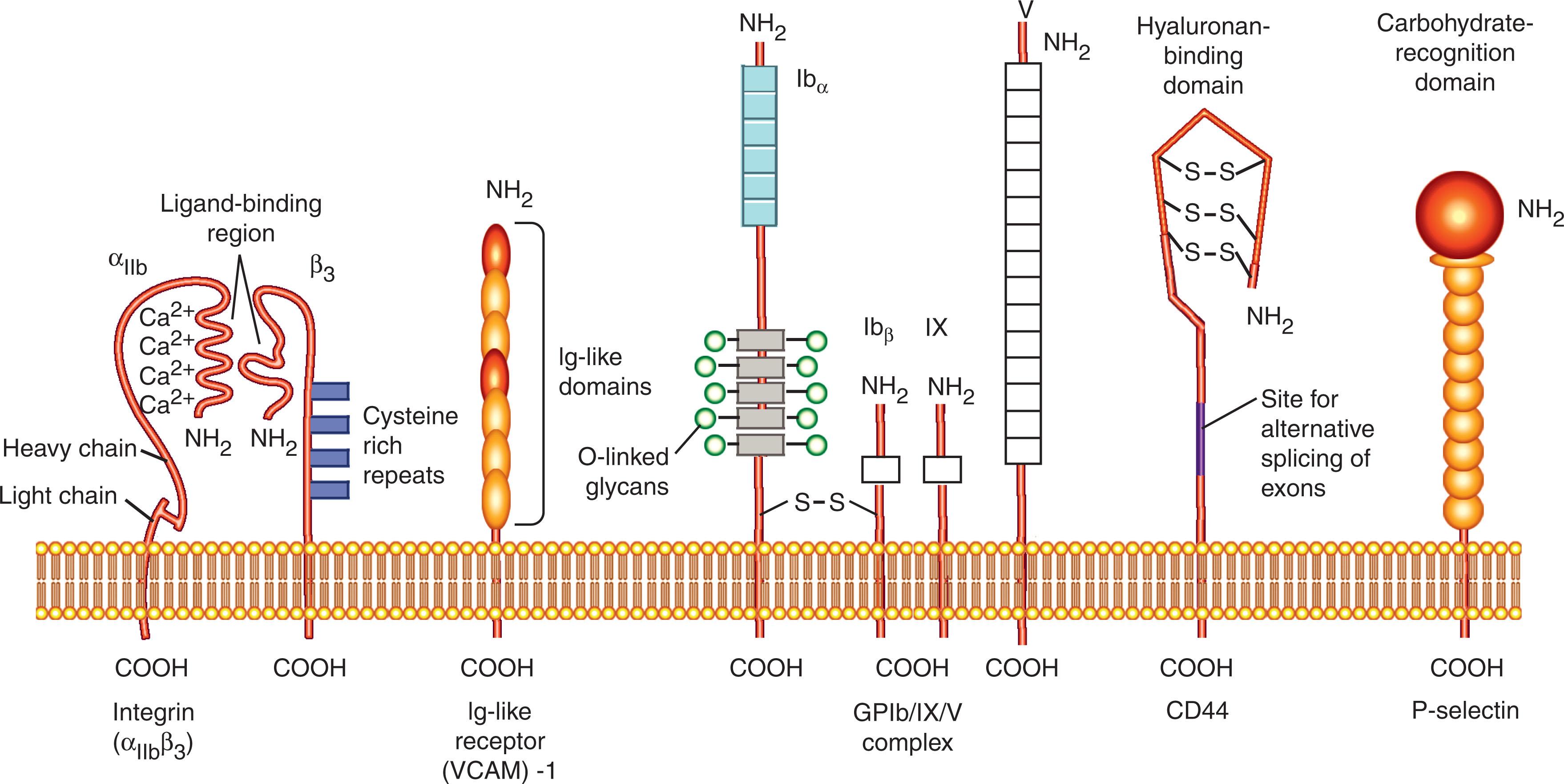
Integrins are a broadly distributed group of cell-surface adhesion receptors that consist of noncovalently associated α- and β-subunits ( Fig. 15.1 and Table 15.1 ). There are 18 α-chains and eight β-chains that pair in many, but not all, of the possible combinations. All blood cells have multiple different integrins. The four β2 integrins, each paired with a unique α-subunit, are expressed only by leukocytes, and the αIIbβ3 integrin (glycoprotein IIb–IIIa [GPIIb–IIIa]) is expressed only by megakaryocytes and platelets. Multidomain adhesive proteins of the ECM are ligands for many integrins. Integrins are unusual adhesion molecules because they usually reside in an inactive state on the cell surface until they receive an activating signal. Some integrins bind to specific domains of several different proteins, and some adhesive proteins bind to several different integrins. These interactions generally mediate cell–matrix and cell–cell adhesion. A unique feature of integrins is the transmission of signals in both directions across the cell plasma membrane. Integrin binding to matrix informs the interior of the cell (outside-in), and intracellular signals or conditions inside cells transmit signals outward (inside-out) that regulate binding to matrix or to adhesion receptors on the surface of adjacent cells. Force can also regulate integrin adhesive function. The application of tension to integrins can increase ligand binding and a reduction in tension lessens integrin adhesiveness. Cell–cell interactions result from integrin recognition of cell-surface members of the immunoglobulin superfamily. Binding of fibrinogen to αIIbβ3 integrins on adjacent platelets creates a molecular bridge that promotes platelet aggregation ( Fig. 15.2 ).
| Integrin Designation | Other Name(s) | Expressed by | Ligand(s) | Function(s) |
|---|---|---|---|---|
| α 1 β 1 | VLA-1 | Leukocytes, other cells | Collagens, LM | Adhesion to ECM |
| α 2 β 1 | VLA-2 GPla/lla | Leukocytes, platelets, other cells | Collagens, LM | Adhesion to ECM |
| α 3 β 1 | VLA-3 | Leukocytes, other cells | Collagens, LM, FN | Adhesion to ECM |
| α 4 β 1 | VLA-4 | Monocytes, lymphocytes, eosinophils | VCAM-1, FN | Adhesion to cells, ECM |
| α 5 β 1 | VLA-5 GPlc/lla | Leukocytes, platelets, other cells | FN | Adhesion to ECM |
| α 6 β 1 | VLA-6 GPlc/lla | Leukocytes, platelets, other cells | LM | Adhesion to ECM |
| α 9 β 1 | Neutrophils | VCAM-1 | Adhesion to ECs | |
| α L β 2 | LFA-1 CD11a/CD18 | Leukocytes | ICAM-1, -2, -3 | Leukocyte aggregation and adhesion |
| α M β 2 | MAC-1 CR3 CD11b/CD18 | Neutrophils, monocytes | ICAM-1, FIB, CR for iC3b | Neutrophil aggregation and adhesion to ECs |
| α X β 2 | P150,95 CD11c/CD18 | Neutrophils, monocytes | CR for iC3b | Adhesion to ECs |
| α D β 2 | CD11d/CD18 | Eosinophils, monocytes, lymphocytes | VCAM-1, ICAM-3 | Adhesion to leukocytes and to ECs |
| α IIb β 3 | GPllb/llla | Platelets | FIB, FN, vWF, VN, TSP | Platelet adhesion and aggregation |
| α V β 3 | VN receptor | Platelets, ECs | FIB, FN, vWF, VN, TSP, collagens | Platelet adhesion, angiogenesis |
| α 4 β 7 | LPAM-1 | Lymphocytes | VCAM-1, MAdCAM-1, FN | Lymphocyte adhesion to ECs and ECM |
Immunoglobulin superfamily members contain a variable number of disulfide-stabilized motifs similar to those in antibodies, which are linked to transmembrane and cytoplasmic domains ( Table 15.2 ; see also Fig. 15.1 ). The immunoglobulin-like motif provides a framework on which specific recognition structures for other proteins can be added. Some of these motifs also recognize glycoconjugates. The immunoglobulin-like molecules, intercellular adhesion molecule 1 and 2 (ICAM-1 and ICAM-2), and vascular cell adhesion molecule 1 (VCAM-1), expressed on endothelial cells, as well as ICAM-3, expressed on leukocytes, mediate cell–cell contact through recognition of specific integrins on leukocytes. ICAM-4, expressed on erythroid precursors, binds to integrins on stromal cells of BM, which may regulate erythropoiesis. ICAM-5 is restricted to neural tissues. The immunoglobulin-like GPVI on platelets promotes cell activation by binding to collagen in the subendothelial matrix exposed on damaged blood vessels. Interactions between immunoglobulin-like molecules help to mediate adhesion between T cells and antigen-presenting cells. Thus, whereas the immunoglobulin-like molecules CD8 and CD4 on T cells bind to the conserved membrane-proximal domains of class I and class II major histocompatibility complex (MHC) proteins, respectively, the α- and β-chains of the T-cell receptor (TCR) bind to the polymorphic antigen-presenting domain. In addition, the immunoglobulin-like proteins CD2 and CD28 on T cells bind to the immunoglobulin-like protein leukocyte function-associated antigen-3 (LFA-3) and B7-1 (CD80) and B7-2 (CD86), respectively, on antigen-presenting cells. The immunoglobulin-like receptor, platelet, and endothelial cell adhesion molecule-1 (PECAM-1; CD31) uses homotypical interactions to promote contacts between adjacent endothelial cells and to mediate transmigration of leukocytes across vascular endothelium ( Fig. 15.3 ). The immunoglobulin-like junctional adhesion molecules (JAMs), expressed on endothelial and epithelial cells and leukocytes, regulate endothelial and epithelial cell junctions, paracellular permeability, and leukocyte trafficking between endothelial and epithelial cells by homotypical interactions or by heterotypical interactions with integrins. JAM-A, the founding member of this family, functions as a homodimer and transmits intracellular signals critical for its function in the regulation of endothelial and epithelial permeability.
| Name | Other Name | Expressed by | Ligand | Function(s) |
|---|---|---|---|---|
| ICAM-1 | Macrophages, EC, other cells | α M β 2 , α L β 2 , FIB | T-cell responses, leukocyte adhesion to EC | |
| ICAM-2 | EC | α L β 2 | Leukocyte adhesion to EC | |
| ICAM-3 | Leukocytes | α L β 2 | T-cell responses, leukocyte aggregation | |
| ICAM-4 | Erythroid precursors | α 4 β 1 , α V β 3 , α llb β 3 | Regulate erythropoiesis | |
| ICAM-5 | Telencephalin | Telencephalic neurons | ICAM-5, αLβ2, α5β1 | Neuron-neuron and neuron-leukocyte |
| GPVI | Platelets | Collagen | Platelet adhesion and activation | |
| PECAM-1 | CD31 | Leukocytes, platelets, EC | PECAM-1 | EC junctions, leukocyte transmigration, cell signaling |
| VCAM-1 | Activated EC, smooth muscle cells | α 4 β 1 , α 4 β 7 | Mononuclear cell adhesion to EC | |
| MAdCAM-1 | EC of Peyer patches | α 4 β 7 | Lymphocyte homing | |
| Siglecs | Leukocyte subsets | Sialylated glycans | Regulates B-cell activation, innate immunity?, hematopoiesis? | |
| JAMs | EC | JAMs, α L β 2 , α 4 β 1 | EC junctions, leukocyte transmigration | |
| CD2 | T cells | LFA-3 a | T-cell responses | |
| CD4 | T cells | Class II MHC a | T-cell responses | |
| CD8 | T cells | Class I MHC a | T-cell responses | |
| CD3 | T-cell receptor | T cells | Antigen on MHC a | T-cell responses |
| CD28 | Costimulatory molecule | T cells |
|
T-cell responses |
a LFA-3 and classes I and II MHC molecules are also immunoglobulin-like receptors.
Cadherins are cytoskeletal-linked type 1 transmembrane proteins that mediate calcium-dependent, cell-to-cell contact in many organs through homotypical binding to cadherins on adjacent cells ( Table 15.3 ). Cadherins are found on endothelial cells, where they form adherens junctions and regulate permeability and leukocyte migration across endothelial cell-to-cell borders, termed diapedesis or transendothelial migration . Cadherins are also expressed in the epithelium and help form cell-to-cell junctions.
| Name | Other Name | Expressed by | Ligand | Function(s) |
|---|---|---|---|---|
| Cadherins | EC, many other cells | Homotypic binding | Formation of EC junctions | |
| GPlb/IX/V | Platelets | vWF | Platelet adhesion to ECM under shear | |
| CD36 | GPIV | Platelets, many other cells | Collagens, TSP | Platelet adhesion to ECM |
| CD44 | Leukocytes, other cells | Hyaluronan, serglycin | Lymphopoiesis, lymphocyte activation | |
| DC-SIGN | Dendritic cells | Mannosylated glycans, other glycans | Regulate T-cell–dendritic cell interactions, recognize pathogens | |
| NK cell receptors | NK cells | MHC molecules | Recognition of virus-infected or other foreign cells |
The GPIb–IX–V complex on platelets consists of leucine-rich protein subunits (see Figs. 15.1 and 15.2 ). Under conditions of high shear stress such as those found in arterial circulation, this complex promotes the initial platelet adhesion to injured vessels by binding to vWF exposed in the subendothelial matrix. It also assists interactions with other platelets or with endothelial cells by binding to P-selectin, which normally binds to glycoconjugates, and it may assist platelet adhesion to leukocytes by binding to the integrin α m β 2 .
CD36 is a multifunctional receptor with at least two membrane-spanning domains that is expressed on many cell types. On platelets, it has been implicated as a receptor for collagen and for thrombospondin and as a receptor to activate NADPH oxidase and generate reactive oxygen species (ROS); both processes could facilitate adhesion and activation at sites of hemorrhage.
CD44 is an unusual transmembrane GP expressed to variable degrees on many subsets of leukocytes (see Fig. 15.1 ). It has a membrane-distal domain that is structurally related to the link protein of the ECM, and similar to link protein, CD44 can bind to hyaluronan. The hyaluronan-binding function of CD44 may modulate a number of leukocyte responses. The most clearly demonstrated function is in lymphopoiesis, where maturation of lymphocyte precursors requires contact with BM stromal cells bearing surface hyaluronan. CD44–hyaluronate interactions also may promote lymphocyte entry to and transit through organized lymphoid tissues. The membrane-proximal regions of CD44 are structurally diverse because of the insertion of variable numbers of domains through alternative splicing. These insertions may regulate the ability of CD44 to bind hyaluronan and may mediate post-binding events that affect cell signaling, proliferation, differentiation, and motility.
The selectins are a group of three receptors that terminate in a membrane-distal carbohydrate-recognition domain related to those in Ca 2+ -dependent (C-type) animal lectins such as the hepatic asialoglycoprotein receptor (see Fig. 15.1 ). L-selectin is expressed on leukocytes, E-selectin on cytokine-activated endothelium, and P-selectin on macrophages, platelets, and endothelial cells exposed to secretagogues such as thrombin or histamine ( Table 15.4 ). The selectins mediate leukocyte adhesion to platelets, endothelium, or other leukocytes through Ca 2+ -dependent interactions of the carbohydrate-recognition domains with cell-surface carbohydrates on opposing cells (see Fig. 15.3 ). High-affinity binding appears to require specific carbohydrate structures displayed on a limited number of membrane GPs. The best-characterized GP ligands for selectins are mucins, which have large numbers of clustered, sialylated O -linked oligosaccharides. Site-specific construction of O -glycans with specific sialylated, fucosylated, and (in some cases) sulfated moieties is required for these mucins to bind optimally to selectins. In the case of one mucin, P-selectin GP ligand-1 (PSGL-1), sulfation of tyrosine residues near a specific O-glycan is required for binding to P- and L-selectin.
| Name | Other Name (s) | Expressed by | Ligand | Ligands Expressed by | Function(s) |
|---|---|---|---|---|---|
| P-selectin | CD62P GMP-140 PADGEM | Thrombin-activated platelets and ECs, cytokine-activated ECs | PSGL-1, GPlbα | Leukocytes, platelets | Leukocyte adhesion to activated ECs and platelets |
| E-selectin | CD62E ELAM-1 | Cytokine-activated ECs | PSGL-1, other sialylated and fucosylated GPs | Leukocytes | Leukocyte adhesion to activated ECs |
| L-selectin | CD62L LECAM-1 LAM-1 | Leukocytes | PSGL-1, also GlyCAM-1, CD34, and other mucins on ECs of lymph nodes | Leukocytes, ECs or lymph nodes | Leukocyte adhesion to other leukocytes; lymphocyte homing to lymph nodes |
Dendritic cells and related macrophages express a novel group of C-type lectins, of which the best characterized is dendritic cell-specific ICAM-3-grabbing nonintegrin (DC-SIGN). DC-SIGN binds to particular oligosaccharides on ICAMs, thereby regulating T-cell and dendritic cell function during antigen presentation. It also binds to glycans on a variety of pathogens, which may have critical roles in innate immunity. Natural killer cells express a different group of proteins, with some containing membrane-distal C-type lectin-like domains (e.g., NKG2D). Although these receptors are important for interactions of natural killer cells with target cells, they may bind to proteins rather than to glycoconjugates.
Siglecs are a subgroup of membrane proteins of the immunoglobulin superfamily that bind to carbohydrates instead of to proteins (see Table 15.2 ). The first two amino-terminal ( N -terminal) domains appear to be necessary and sufficient for carbohydrate recognition. The N -terminal domain is a V-type structure that includes an unusual disulfide bond that is not found in the more common C-type immunoglobulin domains. Siglecs bind well to sialylated glycans on some but not all GPs. Different siglecs preferentially recognize sialic acid that is linked α2,6-, α2,8-, or α2,3- to an underlying galactose residue. Most siglecs have immune receptor tyrosine-based inhibitory motifs and transmit inhibitory signals. Siglecs can form cis interactions with other GPs on the same cell or trans interactions with GPs on another cell. The best-characterized example is CD22, which negatively regulates B-cell activation when it engages sialylated GPs. Sialoadhesin, expressed on BM macrophages, may regulate hematopoietic cell differentiation.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here