Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The cardiovascular system consists of a complex interaction between the heart and lungs, evolving along a continuum from fetal circulation to adolescence in order to meet the changing needs of an individual’s body as he or she grows and matures. The subcellular makeup of cardiac myocytes produces the heart’s unique structure, which allows for both mechanical and electrical function. In utero, the fetal circulation allows for oxygen delivery to developing organs while removing deoxygenated blood via the placenta. With birth, cardiovascular physiology transitions to allow for gas exchange via the lungs. As the infant grows, the cardiovascular system continues to evolve to meet the body’s needs, which is typified with the change in hemodynamics from the neonatal period through adolescence.
This chapter will explain both the cellular makeup and physiology of the cardiovascular system as it develops from its in utero state through childhood. Assessment of the cardiovascular system will be discussed, including the most advanced tests currently available.
This section will highlight the differentiation and development of subcellular structures. It is important to bear in mind that a single genetic code is responsible for formation of the complex cardiac structures with precise functional roles. The recent advances in this field have come from novel genetic studies on mice and chickens.
Cardiomyocytes are differentiated mesodermal cells in which function is characterized by automaticity, contractility, and impulse propagation. Approximately 25% to 35% of all cardiac cells are cardiomyocytes. The remainder include vascular endothelial cells, fibroblasts, and leukocytes in decreasing prevalence ( ).
Timing of embryologic differentiation and structure development of the human embryo is expressed as Carnegie stages. There are 23 stages that occur over 8 weeks. Beginning in the embryologic development Carnegie stage 8 (CS8, week 3), cardiac crescent cells differentiate into cardiomyocytes via endodermal bone morphogenetic protein and fibroblast growth factors ( ; ). As looping and chamber ballooning begin during CS9 (week 3), cardiomyocytes rapidly increase in number to form the larger cardiac structure. Meanwhile, newly differentiated cardiomyocytes form additional structures based on their position. Further molecular characterization of cardiomyocytes is mediated by T-box transcription factors that promote different degrees of automaticity, contractility, and propagation to atria, ventricles, and conduction pathways. Once differentiated, these cells retain their instructions for further development. An example of this is TBX2 and TBX3 transcription factors that force cardiomyocytes to express predominant conduction and AV-nodal cell types ( ). Genetic studies on transcription factors Hand1 and Hand2 , which regulate genetic expression of ventricular development, have been implicated in structural cardiac diseases, most notably hypoplastic ventricular diseases ( ).
Cardiomyocytes are predominantly formed of unidirectional contractile elements named sarcomeres, which are considered the workhorse of the heart. The structural relationships of the major proteins are grouped by the overlap and relationship to one another, termed bands, discs, and zones. Polarized light defines the isotropic I-band and anisotropic A-band. The interacting filaments of the sarcomere are made of highly complex protein chains with supporting and regulatory proteins ( Fig. 5.1 ). Many genetic mutations have been implicated in sarcomere diseases, such as MYBPC3 and MYH7 in hypertrophic cardiomyopathy.

Actin is a polarized globular molecule that binds itself and projects as a polymerized chain of repeating subunits, creating the thin filament. At terminal ends of the sarcomere, Z-discs, the two thin filaments of overlapping actin, bind to opposing ends of the neighboring sarcomere’s actin ( ). The terminal ends of the sarcomere are also integrated with a complex network of proteins (alpha-ACTININ, FILAMIN-C, MYOTILIN, muscle LIM proteins) that structurally support actin. These supporting proteins not only bind opposing actin thin filaments of neighboring sarcomeres but directly signal sarcomere performance (e.g., muscle LIMP proteins as stretch sensor) ( ). Actin projects along the sarcomere, supported by titin as the I-band. Successively, actin overlaps myosin chains in the A-band to create the contractile sliding motion.
Titin is the largest protein in the human genome and serves to maintain and adjust structure throughout the sarcomere. Anchoring at the Z-disc and terminating at the M-band, titin binds numerous proteins along its chain for signaling tension and promoting remodeling ( ; ). Along the I-band, both thin actin filament and titin proteins create a supportive structure void of contractile myosin elements. The production of three different titin isoforms and later alterations to protein structure within the I-band regulate passive tension and diastolic cardiac function ( ).
The thick filament is composed of chains of myosin proteins and is the largest contribution of cardiac muscle protein by mass ( ). The myosin isoform component consists of two heavy chains and four light chains. Each heavy chain projects a head that binds actin filaments, a neck that transmits movement, and a tail that coalesces myosin filaments ( ). The two isoforms of myosin heavy chains, alpha and beta, have 93% similarity but are significantly different in function. Alpha-myosin is predominantly established in the atria, whereas beta-myosin is in the ventricles ( ). Two alpha-myosin heavy chains (aa-MHC) are considered homodimer V1 and are coded by gene MYH6. Two beta-myosin heavy chains (bb-MHC) combine to form homodimer V3. Animal models of myosin heavy chain activity show V1 to have higher power and speed of contraction compared with V3, which prevails in isometric force ( ; ). Four different myosin light chains are described as essential or regulatory and either atrial or ventricular ( ). Through phosphorylation, myosin light chains initiate movement, modulate force, and adjust the speed of contraction ( ). The combination of different myosin chains produces variable amounts of force and velocity.
The basic movement of striated muscle involves the formation of a cross-bridge between myosin and actin and rebinding at neighboring sites to promote movement. A strong cross-bridge link is present when no chemical energy is bound to the actin-myosin complex. When the actin-myosin complex first binds chemical energy in the form of adenine triphosphate (ATP), the cross-bridge link becomes weakly associated. Myosin bound to ATP then releases the myosin head from actin. Subsequently, myosin ATPase hydrolyzes the terminal phosphate group, which activates the myosin neck to displace the head 5 nm and rebind actin ( ). The original strong cross-bridge link is reestablished once ADP and phosphate are released. Correspondingly, the myosin neck moves the head to its original position, sliding along the actin filament ( ; ). This process is repeated in the sarcomere and amplified among numerous cardiomyocytes, creating a mechanical contraction from chemical energy.
The troponin complex controls contraction and relaxation of fast, slow, and cardiac striated muscle. Genetic studies have shown that cellular development into adult cardiac tissue is dependent on regulation of genetic expression and posttranslation modification. The troponin complex is composed of three cardiac-specific isoforms: inhibitory troponin (TnI), calcium-binding troponin (TnC), and tropomyosin-binding troponin (TnT). TnC has three isoforms and TnT has two isoforms. Three isoforms of TnI are programmed in striated muscle. As the fetal heart matures, only one isoform, coded by gene TNNI3, is dominant in adult cardiac tissue. With maturation, the dominant isoform switches from the slow skeletal muscle to cardiac-specific muscle. Interestingly, fetal expression of TnI has a higher Ca 2+ sensitivity and pH modulation ( ).
Calcium from the sarcoplasmic reticulum initiates the process by which troponin activates contraction. Cytosolic Ca 2+ binds TnC, activates TnI’s release from actin, and activates myosin ATPase ( ). At higher concentrations of Ca 2+ , troponin T preferentially displaces the tropomyosin, thereby promoting actin-myosin interaction. Low Ca 2+ concentration causes tropomyosin to integrally coil around actin in a closed state and blocks the attachment and sliding of the myosin heads ( ; ). The force of contraction is also Ca 2+ dependent ( ). Recruitment of more activated myosin heads and further overlap between myosin and actin generates a stronger contractile force ( ) ( Fig. 5.2 ).
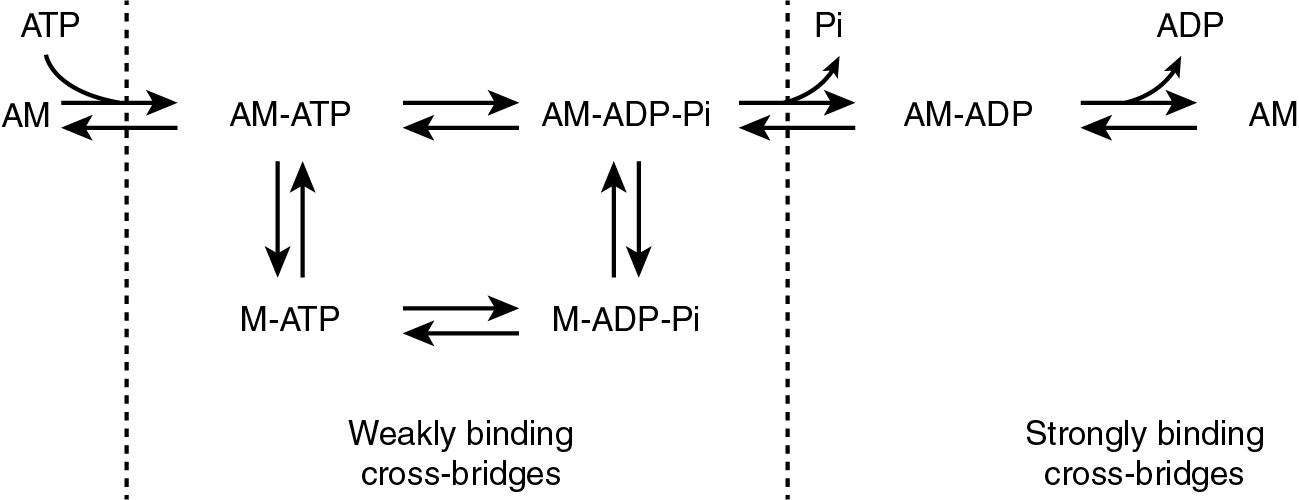
Cardiac myosin-binding protein-C (cardiac MyBP-C) is an important regulatory protein for cardiac contraction. Cardiac MyBP-C promotes cross-link binding of myosin and actin through ATPase signaling. This major protein itself is activated by phosphorylation from β-adrenergic and calcium signals and inhibited by dephosphorylation from cholinergic receptors ( ). Additionally, mutations in the encoding gene for cardiac MyBP-C are closely associated with hypertrophic cardiomyopathy ( ). Advances in genetic studies provide a window into the recovery and regeneration of cardiomyocytes through complex mediated proliferation ( ).
Derived from prokaryotic organisms, mitochondria are maternally inherited and possess their own DNA and proteins encoded by nuclear DNA. Oxidative phosphorylation along the mitochondrial membrane provides an enormous source of ATP for persistent and adjustable cardiomyocyte contraction. Mitochondria make up one-third of cell volume and are structurally integrated to the sarcomere’s mobile filaments ( ). Although mitochondria can use all forms of stored energy, the main cytosolic energy sources for cardiac mitochondria come from glucose in the form of pyruvate and fatty acids converted to acyl CoAs (Acetyl Coenzyme A). Storage of these substrates is in the form of glycogen and triacylglycerol, respectively. The fetal myocardium is dependent on energy from glycolysis, whereas adult myocardium is dependent on fatty acid oxidation ( ). The fetal heart consumes a large amount of ATP (6 kg/day) with insufficient reserve.
Mitochondrial diseases directly affect the performance and structure of cardiac myocytes. Mutation in mitochondrial DNA have been implicated in noncompaction, ventricular preexcitation, accessory pathways, and nodal arrhythmias ( ).
The outer cell membrane of cardiac cells, the sarcolemma, communicates with intracellular structures via multiple calcium regulatory protein channels (e.g., NCX, PMCA, Orai) and transverse tubules (t-tubules). T-tubules are deep operational cell membrane invaginations along the Z-discs of the sarcomere. It is here that the coupling of electrical excitation and contraction of the sarcomere is promoted. The L-type Ca 2+ channel is the voltage-gated protein at the t-tubule membrane that undergoes conformational change to allow an influx of calcium ( ). The L-type Ca 2+ channel family of proteins has four isoforms in various genetic expression and activation throughout the heart, attributing to differences in nodal, atrial, and ventricular activation. Genetic studies have linked mutations in L-type Ca 2+ channels with disease processes of heart failure, cardiomyopathy, and inherited arrhythmias ( ). L-type Ca 2+ channels linked with cardiac ryanodine receptor type-2 (RyR2) in a macromolecule produce a larger release of calcium from the sarcoplasmic reticulum (SR) ( ). Mutations in RYR2 have been implicated in inherited cardiac arrhythmias such as catecholaminergic polymorphic ventricular tachycardia (Meissner 2017).
As important as cytosolic Ca 2+ is for the activation of troponin complex C, so too is the removal of cytosolic Ca 2+ for termination of coupling and cardiac relaxation. As mentioned, multiple regulatory proteins in the cellular membrane and SR membrane account for the flux of cytosolic calcium. Once the ryanodine receptor protein is closed, the sarco/endoplasmic reticulum Ca-ATPase, or SERCA protein, transports Ca 2+ into the SR for cardiac relaxation. Cytosolic Ca 2+ is also transported out by the sodium calcium cell membrane exchange (NCX) and plasma membrane Ca-ATPase (PMCA) ( ).
Heart failure has been linked to various stages of the coupling of electrical stimulus and mechanical contraction, including remodeling of t-tubules, calcium leak from SR by phosphorylation of RYR2, and decreased function of SERCA proteins ( ).
Transmission of cardiac impulse along the longitudinal axis of the heart is directed by gap junctions on intercalated disks. Here, cellular communication occurs by ion and small molecule exchange. Vertebrate gap junction proteins, or connexins, have multiple different isoforms that propagate electrical current between modified cardiomyocytes. Isoform Cx40 is predominant in conduction pathways, whereas major contractile elements contain predominant isoform Cx43. Furthermore, alterations in protein isoforms and expression directly affect structural development and conduction performance ( ; ). As an example, in Brugada syndrome, a mutation in the sodium channel protein Nav1.5 is potentially linked to abnormal expression of gap junction protein Cx43 and responsible for life-threatening ventricular arrhythmias ( ; ).
Cardiac fibroblasts develop and maintain structure, provide cell communication, and repair damaged tissue. They are derived from several different cell lineages—namely, the epicardium and endocardium. The majority of fibroblasts from the epicardial line transition to mesenchymal cells to form and maintain cardiac structure and integrity. Valvular interstitial cells are derived from mesenchymal transformation of endocardial cells ( ). Fibroblasts are spindle-shaped cells without a basement membrane and extend between cardiomyocytes. Fibroblasts manufacture growth factors, cytokines, collagen, and fibronectin from a sizable Golgi apparatus and rough endoplasmic reticulum. During injury, fibroblasts transform into myofibroblasts and provide contractile collagen links and limit wound progression ( ). Rodent experiments show that the prevalence of fibroblasts increases in the postnatal period to adulthood ( ). Current research is focused on fibroblast proliferation and wound healing to mitigate pathologic cardiac remodeling.
At Carnegie stage 16 of development (week 6), the primitive vascular plexus matures into the coronary vasculature through endothelial cell angiogenesis. Endothelial cells arranged in a simple squamous layer form the internal tubular shape of cardiac vasculature ( ). In addition to functional transport, these blood-barrier cells promote signaling and wound healing and mediate the immune response. The luminal side of endothelial tissue promotes anticoagulants and facilitates blood transport. Endothelial injury sets into motion a cascade of reactions, including binding of platelets, in order to limit pathologic processes. Von Willebrand factor and tissue factor in the subendothelium promote coagulation and healing.
In addition to healing and remodeling, endothelial cells have direct effects on vascular smooth muscle cells and cardiomyocyte contractility. Endothelin-1 is an endothelial secretory protein that increases vascular tone by binding ETA or ETB receptors on smooth muscle cells. Production and secretion of endothelin-1 is promoted by stress, thrombin, angiotensin II, and inflammatory mediators. Conversely, atrial natriuretic peptide, prostacyclin, and nitric oxide relax vascular tone ( ).
Vascular smooth muscle cells and pericytes are mural cells that create a structure and contractile element in the cardiac endothelial vasculature. Proepicardial cells form the epicardial cell layer and transition to mesodermal cells that generate the prenatal vasculature. Rodent studies have shown that postnatal coronary vasculogenesis is likely new rather than from further vascular growth ( ). Multiple protein isoforms of smooth muscle actin and myosin create the contractile mechanism of the cardiac vasculature ( ).
The cardiac conduction system develops early in the embryonic stage, during the proliferation of myocardial cells in the primitive tube. Here, cells of the inflow tract, or venous pole, acquire automaticity in a unidirectional sinusoidal electrocardiogram. Then, as the cardiac structure forms and further divides into chambers, the conduction system forms a complex pathway of both slow and fast electrical conduction. The final form of the conduction system propagates an impulse from the sinoatrial node to the atrioventricular node and finally through the fast-ventricular conducting system. Nodal and conduction myocardial cells possess automaticity but lack developed contraction elements and densely packed sarcomeres ( ). Regulation and amplification of gene expression occur in a myriad of protein interactions, beginning at the earliest stages of cardiac development. Each of these conduction cells has an intrinsic automaticity apparent only when earlier depolarizations are not connected or present.
Genetic studies in the beginning of the 21st century have assigned numerous transcriptional factors (e.g., TBX3 and TBX5) to programming sinus venosus cells to sinoatrial nodal cells ( ). The sinus node’s final orientation is broad and complex, spreading from the superior vena cava (SVC)/RA (right atrium) junction to the inferior vena cava (IVC) ( ). These primary nodal cells are responsible for faster automaticity, which stimulates an electrical impulse through the atria and toward the atrioventricular node. Subsequent depolarization of bilateral atria, the atrioventricular node, and the atrioventricular bundle is depicted as the p-wave on an electrocardiogram.
The atrioventricular node is located posteriorly and between the atria and ventricles. It is formed from early designation and differentiation of cellular function rather than recruitment of cells ( ). Transcriptional factors Tbx2, Tbx3, GATA, and Bmp2 control the differentiation and proliferation of these slow conduction nodal cells ( ). The AV node maintains the separation of atrial and ventricular conduction systems, which allows for ventricular diastole and insulates the heart from rapid atrial rhythms ( ).
As the bulbus cordis forms the rudimentary ventricular chambers, the atrioventricular bundle (bundle of His) forms at the crest of the intraventricular septum and becomes the bridge from slow atrial to fast ventricular conducting cells. Transgenic mouse models have revealed that transcriptional factors NKX2.5 and T-box protein are among many factors that stimulate atrioventricular bundle (AVB) and bundle branch (BB) development ( ). Amplification of genes Scn5a and Cx40 by T-box protein Tbx5 induces and maintains fast ventricular conduction in these cells ( ; ).
Maturation and remodeling of fast ventricular conducting cells begins during ventricular formation and continues after birth. Ventricle-specific bundle branches and Purkinje fibers are generated from endocardial trabeculations. Differentiation of fast conducting Purkinje fibers is directed by numerous signaling proteins (e.g., endocardial Notch and neuregulin) ( ). Proliferation of Purkinje fibers is propelled by Notch expression of Bmp10 suppressing coding inhibitors ( ). These trabeculations overexpress the junctional protein Cx40, a marker solely present in the ventricular conducting system ( ).
Studies are deciphering genetic variations and their association with cardiac conduction pathology. Brugada syndrome is an example of a conduction disease with ST elevation and high occurrence of ventricular fibrillation and sudden death . A mutation in Scn5a , the sodium channel gene, explains 80% of genotyped patients with Brugada ( ).
The primary function of the fetal circulatory system is to deliver oxygenated blood to metabolizing organs and return deoxygenated blood to the placenta in order to eliminate carbon dioxide and waste products as well as replenish oxygen. The fetal cardiovascular system is anatomically arranged in such a way as to allow blood to bypass the lungs and, as a result, the fetal systemic circulation receives cardiac output from both the left and the right ventricles ( Fig. 5.3 ). This fetal circulation is characterized by increased pulmonary vascular resistance (PVR) with very little pulmonary blood flow, decreased systemic vascular resistance (SVR) with the placenta operating as the major low-resistance vascular bed, and right-to-left blood flow through the ductus arteriosus and foramen ovale. In utero, the right and left ventricles are working in parallel, which is in contrast to the circulatory pattern in postnatal life whereby the left and right circulations are separated and the ventricles work in series. As seen in some forms of congenital heart disease, such as critical aortic stenosis, hypoplastic left heart syndrome, and pulmonary atresia, limited flow to either ventricle seems to preclude the growth of that ventricle ( ). Blood flow into both the right and left ventricles is considered to be important in facilitating their growth.

Much of what we have observed in the human fetus and neonate with respect to cardiac anatomy and physiology has been confirmed in animal studies. Rudolph and colleagues were pioneers in studies on cardiovascular development and cardiac function ( ; ; ; ). Although animal models were used in many studies, thereby limiting the ability to extrapolate findings to humans, a large body of knowledge has still been gained.
The placenta is a large low-resistance circuit that functions as the prenatal respiratory organ. In addition to the umbilical arteries and umbilical vein, the ductus venosus, foramen ovale, and ductus arteriosus are very important vascular structures that form early in gestation. These three structures serve as shunts that allow the most highly oxygenated blood to perfuse the heart and brain. In utero, blood is transported between the fetus and placenta through the umbilical cord, which contains two umbilical arteries and a single umbilical vein. The umbilical arteries originate from the fetal internal iliac arteries and carry deoxygenated fetal blood toward the placenta. The umbilical arterial flow is in part dependent on the placental resistance. The placenta has the lowest vascular resistance of any structure in the fetal circulation and therefore is the major contributor to umbilical arterial flow. The pulsatility in the umbilical arteries is low and progressively decreases during pregnancy. Reversal of diastolic flow indicates flow toward other vascular regions in the fetus where resistance is low, such as an AVM, or severe elevation in placental resistance.
The umbilical vein carries highly oxygenated, nutrient-rich blood under high pressure from the placenta back to the fetus. Flow in the umbilical vein is generally phasic in the IVC toward the heart. Phasic periods with absent forward flow or reversal of flow are markers of impaired relaxation of the right ventricle or right atrium secondary to decreased compliance. This can be the result of a cardiomyopathy, ductal restriction, and/or severe volume overload. When the umbilical vein approaches the liver, it branches into two veins: the portal vein and the ductus venosus. This allows for delivery of some oxygenated blood to the liver while the majority of blood is shunted around the liver and into the IVC. Approximately 50% of the blood passes directly into the ductus venosus and the other 50% of the blood flows via the portal vein into the sinusoids of the liver and enters the suprahepatic IVC through the hepatic veins ( Fig. 5.4 ). By midgestation, approximately 30% of the umbilical venous blood is shunted through the ductus venosus, and by 30 to 40 weeks’ gestation, this fraction decreases to approximately 20%, although a significant increase in flow through the ductus venosus can occur in response to hypoxia ( ).

The portal vein is the smaller branch from the umbilical vein and carries well-oxygenated blood primarily to the right lobe of the liver. The ductus venosus is the larger branch and essentially connects the umbilical vein with the suprahepatic IVC. It is estimated that 50% to 60% of umbilical venous blood bypasses the liver via the ductus venosus. The remainder of the umbilical venous flow perfuses the left lobe of the liver. The ductus venosus drains primarily oxygenated blood into the suprahepatic IVC. Blood flow through the ductus venosus is regulated by a sphincter mechanism that is located close to the umbilical vein. When the sphincter contracts, more blood is diverted to the portal vein and hepatic sinusoids and less is diverted to the ductus venosus. This prevents overloading of the heart when blood flow through the umbilical vein is high, such as during uterine contractions.
The right and left hepatic veins, along with the ductus venosus, merge into the suprahepatic IVC. Because the umbilical venous blood in the ductus venosus bypasses the high-resistance hepatic microcirculation, it is not only more oxygenated, it also flows at a higher velocity. Within the suprahepatic IVC, there are now two streams of blood. The higher velocity stream of blood is derived from the ductus venosus and drainage from the left hepatic vein while the lower velocity stream of blood consists of drainage from the right hepatic vein mixed with venous return from the abdominal IVC (deoxygenated blood from the lower limbs, abdomen, and pelvis). Upon entering the right atrium, the anatomic orientation of the IVC–right atrial junction favors the streaming of the well-oxygenated, high-velocity blood across the foramen ovale to the left atrium, which is facilitated by the crista dividens and Eustachian valve. The right-to-left shunt is possible because left atrial pressure is low due to minimal pulmonary venous return.
The crista dividens is a structure at the cephalad margin of the foramen ovale. The orientation of the crista dividens is such that it overrides the orifice of the IVC and as a result splits the IVC bloodstream into an anterior and rightward stream that enters the right atrium and a posterior and leftward stream that passes through the foramen ovale into the left atrium. The latter stream allows the higher velocity, more highly oxygenated blood to enter the left atrium where it mixes with the small quantity of poorly oxygenated blood returning from the lungs. In addition to the crista dividens, the Eustachian valve is a flap of tissue at the junction of the IVC and the right atrium, which also functions to help direct the higher velocity stream of blood across the foramen ovale and into the left atrium. Not only are these anatomic structures important but the umbilical venous pressure and the portocaval pressure gradient also play an important role in the preferential shunting of ductus venosus blood through the foramen ovale into the left atrium. It is also important to note that the fetal lungs consume oxygen within fetal circulation instead of replenishing it.
The purpose of the ductus venosus and foramen ovale is to allow the most oxygenated blood from the umbilical vein to reach the left ventricle with the least drop in oxygen saturation possible. Once the well-oxygenated blood enters the left ventricle from the left atrium, it is ejected into the ascending aorta, proximal to the entrance of the ductus arteriosus, and preferentially directed to the fetal coronary and cerebral circulations ( ). This anatomic arrangement allows the higher oxygenated blood from the umbilical vein to bypass the liver and the right side of heart, thereby ensuring the delivery of well-oxygenated blood to the heart and the brain, which are the two organs with the highest oxygen requirements. Oxygen saturation in the umbilical vein is approximately 80%. However, after mixing with more deoxygenated blood from the liver, IVC, and SVC, the left ventricle ejects blood with a saturation of 65% to 70% when the mother is breathing room air ( Fig. 5.5 ).
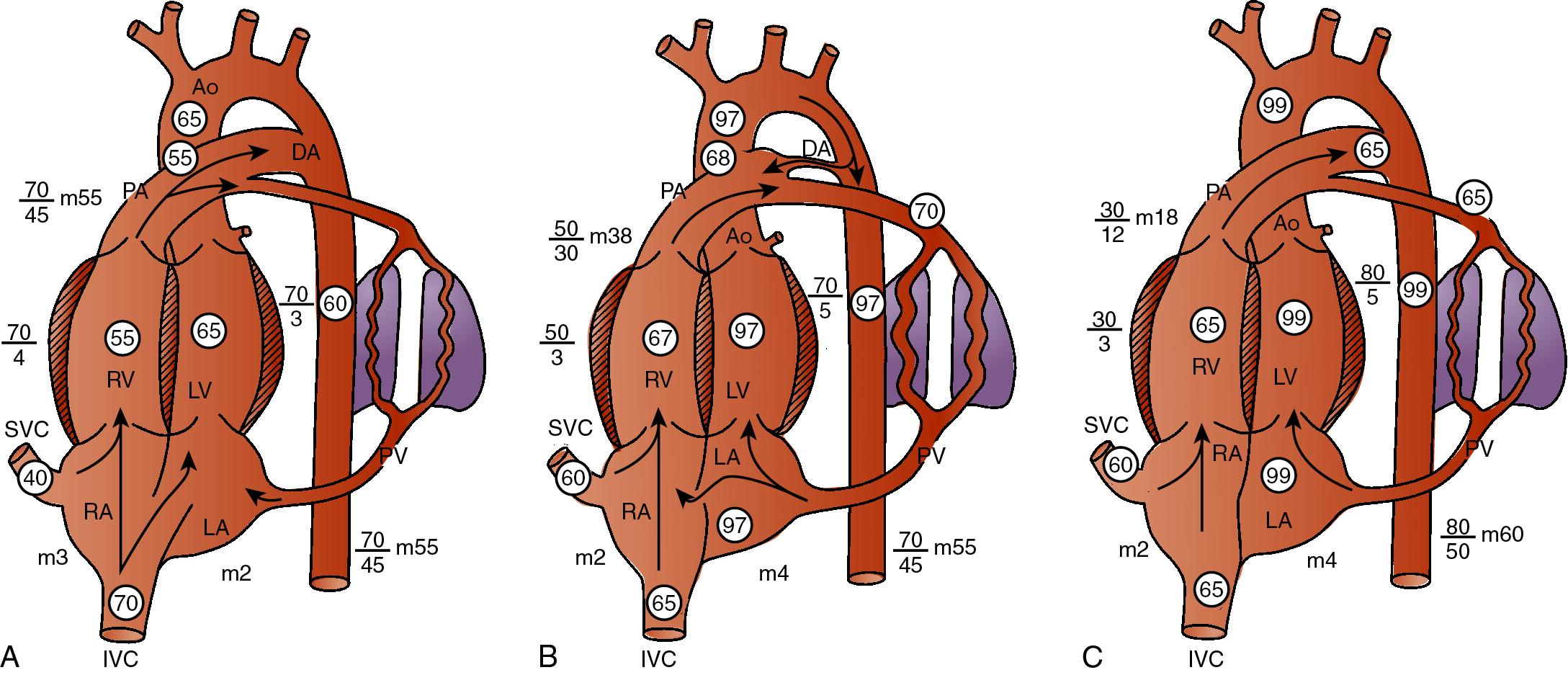
A small fraction of well-oxygenated blood from the suprahepatic IVC remains in the right atrium and does not cross the foramen ovale into the left atrium. This blood instead mixes with poorly oxygenated blood from the low-velocity stream of deoxygenated suprahepatic IVC blood as well as deoxygenated blood from the coronary sinus and SVC. After entering the right atrium, this blood is preferentially directed across the tricuspid valve and is ejected by the right ventricle into the pulmonary artery. In the fetus, PVR is higher than SVR; therefore approximately 10% of the blood exiting the right ventricle makes its way to the lungs and the remaining 90% of the right ventricular output passes through the ductus arteriosus and enters the aorta distal to the branches of the ascending aorta and aortic arch. The ductus arteriosus usually inserts into the aorta at a point immediately distal to the origin of the left subclavian artery or between the origins of the left common carotid artery and the left subclavian artery. The blood in the descending aorta is a mixture of left and right ventricular outputs, with the right ventricle predominating. The ductus arteriosus protects the lungs from circulatory overload and allows the right ventricle to strengthen. Although the majority of this poorly oxygenated blood in the descending aorta passes into the umbilical arteries and is returned to the placenta for gas and nutrient exchange, a fraction of it supplies the viscera and the inferior part of the body, which has a lower oxygen consumption than the heart and brain. As a result, the gut, kidneys, and lower extremities are perfused with blood that has an oxygen saturation of approximately 55%. The patency of the ductus arteriosus in utero is controlled by the lower partial pressure of oxygen in fetal blood and by endogenously produced prostaglandins that act on the smooth muscle in the wall of the ductus arteriosus. The prostaglandins cause the ductus arteriosus to relax.
In the fetus, the systemic circulation receives blood from both the right and left ventricles in parallel, with the exception of a proportion of the right ventricular output that is delivered to the fetal lungs. This is in contrast to the postnatal series circulation seen in children and adults. The descending aorta receives flow from the left ventricle and the right ventricle via the ductus arteriosus. The ascending aorta receives well-oxygenated blood that is ejected from the left ventricle proximal to the entrance of the ductus arteriosus. Since the lower body organs are perfused by both the right and the left ventricles, it is customary to consider fetal cardiac output as being the total output of the heart or the combined ventricular output. At midgestation, the combined ventricular output is approximately 210 mL/min, and it increases to approximately 1900 mL/min (450 mL/kg/min) at 38 weeks’ gestation ( ). In utero blood flow to critical organs is often described as a percentage of combined ventricular output. The demands of a parallel circulation result in increased myocardial work superimposed on the demands of a fetus having to grow and develop in a cyanotic milieu. Nevertheless, the fetal circulation is an efficient arrangement with adequate physiologic reserve.
Given the various sites of intra- and extracardiac shunting in the fetus, the left and right ventricles do not have the same stroke volume. Based on studies in the fetal lamb, the right ventricle ejects approximately two-thirds of total fetal cardiac output (approximately 300 mL/kg/min) and the left ventricle ejects the remainder (approximately 150 mL/kg/min) ( ). Echocardiographic studies in human pregnancy also suggest that the right ventricle dominates the left ( ; ; ; ; ). Of the 65% of the total cardiac output ejected by the right ventricle, only a small amount (8%) flows through the pulmonary arteries to the lungs. The remainder (57%) crosses the ductus arteriosus and enters the descending aorta. With respect to left ventricular output, which accounts for 35% of the total cardiac output, 21% flows to the brain, head, upper limbs, and upper thorax, and approximately 10% joins the blood flowing across the ductus arteriosus to perfuse the descending aorta. The majority of the combined ventricular output is distributed to the placenta (41%) and the bone and skeletal muscle (38%), with the remainder going to the gastrointestinal system (6%), the heart (4%), the brain (3%), and the kidneys (2%).
Because the right ventricle in the fetus handles more volume, its radius is greater, the radius-to-wall thickness is greater, and it hypertrophies to maintain appropriate wall tension. The volume load imposed on the right ventricle combined with the high fetal pulmonary vascular resistance results in significant right ventricular hypertrophy. As a result, the wall thickness of the right ventricle is approximately equal to that of the left ventricle in fetal life. This also leads to impaired passive filling of the right ventricle. Therefore active atrial filling is responsible for emptying the right atrium into the right ventricle. After birth, the right ventricle remodels under the influence of a series circulation and lowered PVR. Postnatally, right ventricular apoptosis is four times that in the left ventricle and decreases with time, resulting in a thinner right ventricular wall because of atrophy from a lighter workload ( ).
It is unclear whether fetal myocardium possesses similar responses to preload and afterload as the adult myocardium ( ; ). The adult heart functions in accordance with the Frank-Starling curve, which indicates that ventricular distention lengthens the diastolic fibers and results in augmented contractility. There are mixed results as to whether the fetal heart has the capacity to increase its stroke volume in response to an increase in preload; however, for the aforementioned reasons the right ventricle tends to be more sensitive to changes in preload. A possible explanation for the inability to augment cardiac output in response to increasing preload is ventricular constraint arising from tissues that surround the heart, such as the chest wall, pericardium, and lungs, which limit fetal ventricular preload and overall cardiac function in utero. Relief of this constraint at birth, as a result of lung aeration and clearance of liquid from the lungs, may then allow for an increase in left ventricular preload and subsequent stroke volume in the newborn ( ).
The fetal myocardium is similar to the neonatal myocardium in that it is composed of a greater proportion of noncontractile elements (60% versus 30%); however, the fetal cardiomyocytes grow primarily through cell division, whereas after delivery, cardiac mass increases as a result of cardiomyocyte hypertrophy ( ). In addition, the removal of calcium from troponin C is slower in the fetus, resulting in slower muscle relaxation.
Abnormal heart rates are possible in the fetus. Fetal tachycardia is generally defined as fetal heart rates greater than 180 beats per minute. The majority of fetal tachycardias are supraventricular in origin, and ventricular tachycardias are very rare. Intermittent tachycardia is generally well tolerated by the fetus, with the risk for hydrops increasing proportionally with increased time spent in the abnormal rhythm. Therapy is generally felt to be indicated for sustained tachycardias that last for greater than 50% of time monitored. When treatment is elected, most of these tachyarrhythmias can be treated through maternal administration of antiarrhythmic medications, with transplacental delivery to the fetus ( ).
Fetal bradycardia is defined as a fetal heart rate less than 110 beats per minute. The most common form of bradycardia in the fetus is sinus bradycardia, usually secondary to compression during ultrasound examinations. Fetal atrioventricular block can present with varying degrees of bradycardia and is rarely an isolated phenomenon but is usually associated with maternal Sjögren antibodies (SSA/SSB) and complex structural heart disease ( ; ). In the setting of immune-mediated heart block, fluorinated glucocorticoids and intravenous immunoglobulin have been used in therapy.
In utero, oxygen transport to the fetus must be achieved in a relatively hypoxic environment ( ). Fetal hemoglobin (HbF) constitutes approximately 80% of the fetus’s hemoglobin and has unique properties that facilitate the transport of oxygen to the fetus despite a low partial pressure of oxygen in the fetal blood (Pao 2 ). In the adult, adult hemoglobin (HbA) dominates (>90%). Although HbF and HbA both have a sigmoidal-shaped oxyhemoglobin dissociation curve, this curve is shifted to the left for fetal hemoglobin, demonstrating the increased affinity of HbF for oxygen. The P 50 represents the Pao 2 at which hemoglobin is 50% saturated, and the P 50 for HbF is 19 mm Hg compared with the HbA, which has a P 50 of 26 mm Hg.
A low level of 2,3-diphosphoglycerate (2,3-DPG) and the decreased affinity of HbF for 2,3-DPG further contributes to the leftward shift of the HbF oxyhemoglobin dissociation curve. The net result of this left shift is that for any given oxygen tension, the oxygen saturation is higher for HbF than for HbA. For example, a Pao 2 of 30 mm Hg falls in the steepest part of the dissociation curve, and at this Pao 2 fetal hemoglobin would be approximately 70% saturated, whereas adult hemoglobin would only be 50% saturated ( Fig. 5.6 ). This also justifies administration of oxygen to a mother in which the fetus demonstrates signs of distress and uteroplacental insufficiency. Since HbF has a different oxygen-hemoglobin dissociation curve than HbA, an increase in Pao 2 within the umbilical vein can result in a large increase in fetal oxygen saturation. However, the transfer of oxygen across the placental bed is inefficient compared with the transfer of oxygen in the lungs. It would not be unusual for the partial pressure of oxygen in the umbilical vein to range from 20 to 35 mm Hg with a maternal arterial oxygen tension of close to 100 mm Hg. The large gradient for oxygen across the placenta means that when the mother is breathing 100% oxygen, maternal partial pressure of oxygen can be as high as 400 to 500 mm Hg, but the partial pressure of oxygen in the umbilical vein is no higher than 40 mm Hg. With such a low fetal oxygen tension, any reductions in maternal oxygenation could severely impact the fetus.

Although fetal hemoglobin’s greater affinity for oxygen improves oxygen uptake at the placenta, it has a drawback for the unloading of oxygen at the tissue level, which poses a problem with respect to oxygen delivery. The fetus copes with this problem with another modification in its pH. Fetal pH (normal values 7.25 to 7.35) is lower than it is in adults, resulting in acidosis and a rightward shift of the oxygen-hemoglobin curve. Also known as the Bohr effect, this rightward shift facilitates oxygen release from HbF. The loading conditions for HbF draw attention to the importance of oxygen content within the blood; however, subsequent delivery of oxygen can only be achieved when an adequate oxygen-carrying capacity is matched with a cardiac output sufficient to meet the metabolic needs.
Oxygen content of blood (Cao 2 ) is defined by the following equation:
Hb is the hemoglobin concentration in grams/deciliter; 1.34 is the binding capacity of hemoglobin, which can range from 1.31 to 1.39 and represents the negative effect on binding of oxygen to hemoglobin that occurs when nonoxygen species are bound to hemoglobin; Sao 2 is the arterial oxygen saturation of hemoglobin expressed as a fraction instead of a percentage; Pao 2 is the partial pressure of oxygen in mm Hg; and 0.003 represents the amount of dissolved oxygen in the blood in mL/dL/mm Hg.
Typically, even when breathing room air, the amount of oxygen dissolved in blood is negligible because it is only 0.003 mL o 2 /dL/mm Hg. Therefore the Cao 2 calculation is usually simplified by using only the amount of oxygen that is carried bound to hemoglobin. For example, an adult with an Sao 2 of 100% and an Hb of 11 g/dL would have the following Cao 2 :
By using the equation for oxygen content of the blood, one can demonstrate how the fetus achieves levels of oxygen transport that are near those of an adult. The fetus maintains Cao 2 through two mechanisms: erythrocytosis and the leftward shift of HbF. Therefore if one calculated the Cao 2 for a fetus with an Hb of 17 g/dL and an oxygen saturation of 65%, it would be as follows:
The addition of more hemoglobin represents a readily available way to increase arterial oxygen capacity and subsequent oxygen delivery to tissues. Other factors such as tissue perfusion, oxygen uptake, and oxygen extraction also play significant roles in supplying oxygen to end organs, but again augmentation of hemoglobin content within blood is the best way to improve oxygen content.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here