Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Carcinoma of unknown primary is a diverse group of heterogeneous cancers that accounts for about 2% to 4% of all cancers.
There is not universal agreement on the extent of the appropriate initial evaluation to find a primary cancer; a focused evaluation is recommended.
Early biopsy of a metastatic site is recommended to establish the diagnosis and help direct further workup.
Comprehensive history and physical examination (including breast and pelvic examinations in women and testis and prostate examinations in men) are essential.
Routine laboratory tests, chest–abdominal–pelvic computed tomography scan, and mammography in women should be performed when indicated.
Directed additional invasive tests should be based on symptomatology and pathologic evaluation of the tumor tissue.
Judicious pathologic assessment of the metastatic tumor sample is required, including directed immunohistochemical markers. Based on clinicopathologic evaluation, molecular markers that have therapeutic implications (e.g., KRAS mutational status, Her2 [ERBB2] expression, epidermal growth factor receptor mutations, ALK-mutation studies) should be considered.
The diagnostic utility of positron emission tomography is poorly defined; it is beneficial in selected patients.
The role of tissue of origin molecular profiling and next-generation mutational sequencing assays continues to evolve; these tests are beneficial in selected patients, but prospective outcome data are not yet available for these studies.
There has been a paradigm shift in the choice of treatment away from empiric combination cytotoxic therapy to a personalized approach based on clinic-pathologic evaluation.
When pathologic evaluation falls short, empiric platinum-based combination therapies are usually selected.
In unselected patients, taxanes, gemcitabine, or a fluoropyrimidine + a platinum doublet are common first-line regimens used in patients with good performance status.
Additional lines of therapy depend on performance status, extent of disease, pathologic “profile,” and response to first-line therapy.
Carcinoma of unknown primary (CUP) is a diverse group of cancers, most with a poor prognosis. The reported incidence of CUP varies with the practice setting and the definition used but averages 2% to 4% of all patients who are diagnosed with cancer. Because identification of the primary lesion forms the basis for predicting the expected behavior and assigning appropriate therapy for malignant disease, the absence of a defined primary carcinoma poses a challenge. The inability to identify a primary also generates anxiety for the patient, who may believe that the evaluation has been inadequate or that the prognosis would be improved if a primary site could be established. The management of CUP is evolving, and recently developed sophisticated diagnostic modalities and novel therapies present both an opportunity and a challenge.
We define patients with CUP as having a biopsy-proven malignancy for which the anatomic origin remains unidentified after history and physical examination, laboratory studies including liver and renal function tests, hemogram, computed tomography (CT) of the chest, abdomen, and pelvis; and mammography in women and measurement of prostate-specific antigen (PSA) in men based on presentation. Depending on the clinical situation, additional studies might include directed invasive studies, including upper endoscopy, colonoscopy, or bronchoscopy. Most investigators have excluded from analysis metastatic sarcoma or melanoma without a definite primary site because such presentations have histology- and stage-specific therapy regardless of the primary. CUP research efforts concentrate on the majority of patients with common epithelial histologies such as adenocarcinoma, carcinoma, squamous carcinoma, and neuroendocrine carcinoma.
With the emergence of robust immunohistochemical (IHC) analysis, molecular profiling including tissue-of-origin (ToO) assays, and mutational profiling, there is an increasing need for a tailored approach to CUP management. Empiric trials for CUP were indicated in the era before the development of these tools, and if the results of these reported single-arm and randomized phase II trials are reviewed objectively, no one combination empiric therapy is categorically superior to another. Over the past decade, there has been a shift of focus from empiric therapeutic trials to site-specific profiles that may help define and select CUP patient subtypes that could benefit from individualized treatment.
Although a history of cigarette smoking often can be elicited, the heterogeneity of CUP makes it unlikely that specific etiologic agents will be associated with this disease. The fact that numerous occult anatomic sites can give rise to carcinomas that present with only metastatic disease supports the possibility that specific interactions of genetic and environmental insults could produce genomic and biochemical changes that lead to the early development of a metastatic phenotype without the associated changes supporting local growth in the organ of origin. It is likely that as the genomic and proteomic characterization of malignancies is refined fewer malignancies may be designated as CUP. For example, the incidence of intrahepatic cholangiocarcinoma (ICC) in the United States continues to rise, whereas the incidence of CUP has declined during this same time period. Because the liver is a common site for presentation with CUP, ICC can be misdiagnosed as CUP. Improvements in diagnostic technologies and awareness among clinicians to differentiate the two have contributed to increased recognition of ICC. Although family history often identifies additional cancers with established origins in other family members, no clear familial instances of CUP have been identified or reported.
Whether the biology of CUP is fundamentally different from known primary carcinomas with systemic metastases remains controversial ( Fig. 91.1 ). Nystrom and associates have argued that the distribution of metastatic sites in patients with CUP in whom the primary cancer is subsequently found is sufficiently different from known primary carcinomas to support the hypothesis that CUP is biologically unique. However, analysis of a series from the MD Anderson Cancer Center demonstrates few significant differences in the pattern of metastases or in overall survival for true CUP versus patients in whom the primary lesion was found. Whether CUP metastases are genetically and phenotypically unique remains to be determined.
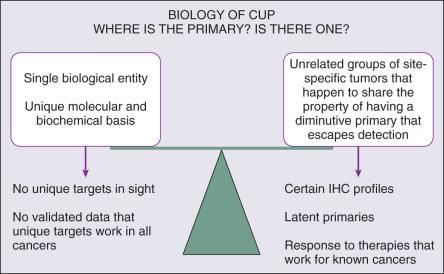
The underlying cause of “occult” primary tumors is unknown. Previous investigators have speculated that the primary tumor may remain below the limits of clinical or radiographic detection or that it spontaneously regressed. It is possible that CUP falls within a continuum of cancer presentation in which the primary has been contained or eliminated by the immune system. Alternately, CUP may represent a specific malignant event that results in an increase in metastatic spread or survival relative to the primary, with immunogenic or antiangiogenic factors that impede local growth.
Aneuploidy has been described in 70% of patients with CUP with metastatic adenocarcinoma or undifferentiated carcinoma. The overexpression of various genes, including RAS , bcl-2 (40%), HER-2 (11%), and p53 (26–53%), has been studied in CUP samples, but expression of these genes has no effect on response to therapy or survival. The extent of angiogenesis in CUP relative to that in metastases from known primary tumors has also been evaluated, but no consistent findings have emerged. The current focus is on comprehensive genomic profiling that may identify targeted therapeutic approaches to improve outcomes for this disease.
An effective diagnostic strategy should take into account the projected natural history and duration of survival based on extent of disease and performance status without compromising quality of life with difficult and time-consuming diagnostic studies. The overall goal is to rapidly identify treatable patient subsets or occult primary lesions through a rational, focused approach.
A thorough history and physical examination, including a detailed review of systems, is mandatory because it may elicit symptoms or signs that were not immediately brought to the physician's attention. Attention should also be paid to a history of previous biopsies, as well as spontaneously regressing lesions. The family history can be helpful, especially if the patient belongs to a specific genetically predisposed ethnic group that is known to be at high risk for malignancies at specific sites (e.g., Ashkenazi Jewish ancestry and risk for BRCA1 and BRCA2 mutations causing breast, ovarian, and pancreatic cancer).
No tumor marker is diagnostic in determining the primary site in CUP. β-Human chorionic gonadotropin (β-hCG) and α-fetoprotein (AFP) are indicated in patients with CUP with a midline tumor and a concern for extragonadal germ cell cancer. AFP is also indicated in patients with liver-predominant presentations and a risk for hepatocellular carcinoma. Measurement of PSA is useful in men with adenocarcinoma and predominantly skeletal metastases. Elevation of PSA can provide a confirmation of metastatic prostate cancer, but the physician should be wary of the occasional coexistence of early prostate cancer with a more aggressive synchronous second neoplasm. Serum measurements of PSA commonly should be coupled with IHC for PSA in tumor tissue because rare patients have been reported with metastatic cancer and clinical features atypical for metastatic prostate cancer.
Most tumor markers, including carcinoembryonic antigen (CEA), CA-125, CA 19-9, and CA 15-3, are not specific and are thus not helpful in determining the site of the primary tumor. Similarly, with the wide availability of IHC, cytogenetic analysis is rarely helpful.
An accurate pathologic assessment of biopsy material is essential in the initial evaluation of the patient with suspected CUP; core biopsy is preferred over a fine-needle aspirate specimen. The pathologist typically begins by examining the tissue specimen using light microscopy and several commonly used IHC stains. Electron microscopy and chromosomal studies, including cytogenetics, are rarely needed. With increased availability and interest in mutational analysis, testing for specific-molecular alterations based on IHC is becoming increasingly common.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here