Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
In the United States it is estimated that there will be 51,540 new cases of head and neck cancer in 2018, accounting for 3% of all cancer cases and 1.5% of all cancer deaths.
The three major risk factors for head and neck cancer are tobacco use, human papillomavirus (HPV) infection, and alcohol use.
Management of head and neck cancers requires a multidisciplinary approach with effective integration of multiple specialties to achieve the desired goals of cure and functional organ preservation.
Staging of head and neck cancer should be comprehensive and include a complete physical examination, fiberoptic laryngoscopy, computed tomography (CT) and/or magnetic resonance imaging (MRI) of the head and neck, and a positron emission tomography (PET) scan for advanced-stage disease to assess nodal involvement and distant metastases.
The treatment of head and neck cancer is dictated by the primary site.
Management of paranasal sinus malignancies is primarily surgical, with adjuvant radiation and possibly chemotherapy for advanced lesions. In unresectable and nonoperative cases, definitive radiation therapy (RT) should be offered.
In general, surgery is considered the preferred initial treatment modality for oral cavity lesions. Adjuvant RT with or without chemotherapy is indicated for patients with high-risk features at surgical pathologic assessment. Definitive RT is reserved for unresectable tumors, nonoperable tumors, and tumors in which surgical resection would result in significant functional impairment.
Most oropharyngeal cancers in the United States are now due to HPV and therefore have an improved prognosis. Early-stage oropharyngeal cancers can effectively be treated with surgery or RT. The standard of care for locally advanced disease is chemoradiation therapy (CRT); however, incorporation of transoral robotic surgery (TORS) is under active investigation.
In patients with laryngeal cancer, the goal of first-line therapy should be to preserve the function of the larynx without sacrificing tumor control. Early-stage laryngeal cancer can effectively be treated with either surgery or RT. In early-stage disease, the anatomic location and extent of disease will dictate whether surgery is feasible. The treatment of choice in most locally advanced laryngeal cancers is concurrent chemotherapy and radiation. The VA larynx study demonstrated poor outcomes with T4a disease, so total laryngectomy is also a consideration in these patients.
Surgical resection is the mainstay of management in salivary gland cancer, whether arising from the parotid, submandibular, sublingual, or minor salivary glands. Combined therapy is recommended for high-grade malignancies of the salivary gland because postoperative irradiation has been shown to improve locoregional control in patients with positive surgical margins; high-risk features such as advanced stage, high grade, and skin and nerve invasion; and adenoid cystic carcinoma.
The management of locally recurrent head and neck cancer is technically challenging and should be performed at centers that are experienced with these patients. Surgery is typically preferred and offered for resectable lesions in the absence of unacceptable functional sequelae. The roles of adjuvant RT (or reirradiation) and chemotherapy must be determined on a case-by-case basis.
Metastatic head and neck squamous cell carcinoma (HNSCC) carries a poor prognosis, with a median survival of months. Systemic therapies are indicated for widespread disease.
In the United States it is estimated that there will be 51,540 new cases of head and neck cancer (HNC) in 2018, accounting for 3% of all cancer cases and 1.5% of all cancer deaths. Because these tumors are relatively uncommon, misdiagnosis along with patient neglect contributes to an advanced stage at presentation and limited survival. Despite the relatively low numbers of HNCs in clinical practice, research and management of HNCs continue to receive significant emphasis because of the rich anatomic and functional complexity of this body site, which is critical to issues of self-esteem, communication, and social integration. Although squamous cell carcinomas (SCCs) constitute the majority of adult histopathologic patterns in HNC, the variation in histopathologic types and the differing features of the possible anatomic subsites of involvement result in tremendous variability in the natural course of the disease for this small anatomic region.
The head and neck encompasses structures from the base of the skull to the clavicles and includes the temporal bone, paranasal sinuses, nasopharynx, oropharynx (soft palate, tonsil structures, base of the tongue, and oropharyngeal wall), oral cavity (lips, buccal mucosa, alveolar ridges, floor of the mouth, oral tongue, retromolar trigone), major and minor salivary glands, skin, and neck.
Tobacco use, alcohol use, and human papillomavirus (HPV) infection are the three major risk factors for head and neck squamous cell carcinoma (HNSCC) in developed countries. Current smokers have an approximately 10-fold higher risk of HNC than do lifelong nonsmokers. Historically, approximately 80% to 90% of all head and neck carcinomas were attributable to tobacco consumption, particularly cigarette smoking, although currently the attribution is thought to be less, given an increase in HPV-associated HNC. HPV-positive HNC incidence has been steadily increasing and is expected to surpass the incidence of HPV-negative HNC, which was traditionally associated with tobacco and alcohol. It remains to be seen if current HPV vaccination recommendations will result in a decrease in HPV-associated HNC.
The management of HNC requires a multidisciplinary approach with effective integration of multiple specialties to achieve the desired goals of cure and functional organ preservation. The desire for more effective yet organ-preserving therapies with acceptable toxicity is being addressed with technologic advances in surgical and radiation therapy (RT) techniques and with the development of novel biologic agents. These advances, coupled with tremendous clinical research activity, have resulted in a number of therapeutic options for patients with HNC. Although efforts centering on treatment regimens remains active, greater efforts are needed in primary preventative measures such as tobacco and alcohol cessation programs and HPV vaccination campaigns.
Patients with HNC have various symptoms depending on the location of tumor. Some signs, symptoms, and patient characteristics are common regardless of the site of tumor origin. Patients with HNC are mostly male and in their sixth decade of life or older. Most have a history of tobacco and/or alcohol use, and an increasing number of younger nonsmokers and non–alcohol users have evidence of prior HPV infection. Many tumors manifest with symptoms related to the primary site of cancer origin, or with cervical lymph node metastasis as the first sign of disease. Some patients may have dysphagia, odynophagia, hoarseness, pain, swelling, mass lesion, weight loss, and malnutrition. Specific signs and symptoms associated with each subtype are discussed in the individual sections.
An initial examination of the head and neck should be performed with the patient sitting upright in a chair. A standard and complete head and neck examination should then be conducted including inspection of the head and neck skin and the scalp. Cranial nerves II through XII should be assessed. Ear examination with otoscopy should be performed. Nasal examination may be performed with anterior rhinoscopy or nasal endoscopy as indicated. Examination of the oral cavity is performed with a headlight to allow bimanual examination of the lips, buccal mucosa, gingiva, floor of the mouth, and oral tongue. Palpation of the oral tongue and the base of the tongue is crucial for a thorough evaluation. The tongue base and larynx are examined with use of a mirror or, preferably, flexible fiberoptic laryngoscopy. While viewing the larynx fiberoptically, the physician should ask the patient to perform several maneuvers: vocalization to allow assessment of vocal cord motion; tongue protrusion to aid in viewing the vallecula; and filling the cheeks with air to help visualize the pyriform sinuses. After the subglottis has been evaluated, the laryngoscope can be removed and the examination completed with palpation of the parotid glands, the thyroid, and the cervical nodes. The size, mobility, and consistency of any mass should be carefully noted.
Radiography is a necessary component of the evaluation. It is standard practice to order a computed tomography (CT) scan of the head and neck, preferably with contrast material, for any patient with suspected cancer. This examination helps not only with evaluation of the size and location of the primary tumor, but also with assessment for any possible lymph node metastasis. A magnetic resonance imaging (MRI) scan can provide useful additional information in certain cases, such as nasopharyngeal lesions, skull base lesions, or soft tissue tumors, and for the assessment of parapharyngeal space lesions. Routine preoperative laboratory studies may be obtained if surgery is being considered, in addition to medical consultations as indicated.
If a suspicious mucosal lesion is anatomically accessible, a biopsy may be undertaken in the clinic after administration of a local anesthetic. In general, biopsies of oral cavity lesions and selected oropharyngeal lesions are performed in an outpatient setting. Biopsies of selected lesions of the tongue base, larynx, and hypopharynx may also be performed in the clinic with flexible laryngoscopes with biopsy ports, although most lesions require direct laryngoscopy and biopsy with the patient anesthetized. During this procedure, flexible esophagoscopy can be considered to rule out tumor involvement of the cervical esophagus. If lymphadenopathy is identified at palpation, the abnormal node may be assessed by means of fine-needle aspiration (FNA). Radiographically suspicious nodes that are not easily palpable can be assessed with ultrasound-guided or CT-guided FNA. An open biopsy of a neck node should be considered only if the findings of FNA are inconclusive.
Currently the American Joint Committee on Cancer (AJCC) staging system, which uses unidimensional tumor size and local anatomic invasion (T category), nodal involvement (N category), and presence of metastatic disease (M category), is the most widely accepted and applied prognostic system relating to cancer. In general, T1, T2, and T3 represent increasing tumor size, whereas T4 is defined by invasion of a surrounding structure (i.e., skin, nerve, vessel, and cartilage). The node (N) category is classified largely by size and location (ipsilateral versus contralateral) of involved lymph nodes. The absence or presence of distant metastases is defined as M0 if absent or M1 if present. The T, N, and M categories are combined into overall AJCC stages that are presented in Table 65.1 . Because the natural history of HNC varies somewhat according to specific anatomic location of the primary disease and because stages III and IV include a large number of individual T, N, and M stages, it is customary to refer to specific HNCs by their individual T, N, and M stage and the primary site. Recent work has shown that for HPV-positive oropharyngeal carcinoma, the current AJCC staging system may be suboptimally predictive; this is currently under review.
| Stage | Grouping | ||
|---|---|---|---|
| 0 | Tis | N0 | M0 |
| I | T1 | N0 | M0 |
| II | T2 | N0 | M0 |
| III | T3 | N0 | M0 |
| T1 | N1 | M0 | |
| T2 | N1 | M0 | |
| T3 | N1 | M0 | |
| IVA | T4a | N0 | M0 |
| T4a | N1 | M0 | |
| T1 | N2 | M0 | |
| T2 | N2 | M0 | |
| T3 | N2 | M0 | |
| T4a | N2 | M0 | |
| IVB | T4b | Any N | M0 |
| Any T | N3 | M0 | |
| IVC | Any T | Any N | M1 |
After completion of initial treatment, patients are carefully assessed for response to treatment. For advanced-stage cancers treated with chemoradiation therapy (CRT), CT, positron emission tomography (PET), and/or MRI is performed in order to assess radiographic disease response approximately 12 weeks after completion of treatment. Thereafter, patients are seen at gradually increasing intervals for surveillance examinations. Chest radiographs may be obtained intermittently as indicated for surveillance for metastatic disease or secondary primary malignancies, particularly for patients with a smoking history. Thyroid-stimulating hormone (TSH) levels should be checked every 12 months because 20% to 25% of patients who have had neck irradiation experience hypothyroidism necessitating thyroid hormone supplementation. After 5 years, patients may be examined yearly.
The understanding of HNSCC biology has increased significantly and deepened in recent years. Molecular studies have begun to lend new insight into the pathogenesis and diversity of HNCs. Gene expression profiling has revealed that HNSCC can be categorized into four distinct subtypes: atypical, basal, classical, and mesenchymal. More recently, The Cancer Genome Atlas (TCGA) has performed a comprehensive profiling of 279 HNSCC tumors and identified unique biologic subgroups. In particular, it was found that HPV-positive tumors are enriched for PIK3CA mutations, a druggable target that is being explored in current clinical trials. Other commonly mutated genes in HNSCC include p53, CDKN2A, and NOTCH1. TP53 mutations constitute rare events in the setting of HPV-positive disease because the suppression of TP53 by the HPV E6 oncoprotein likely eliminates selective pressure to acquire such mutations. However, molecular profiling of recurrent and metastatic HPV-positive tumors revealed mutational and genomic signatures more consistent with HPV-negative disease, including an increased frequency of TP53 mutation, whole-genome duplication, and 3p deletion. In the era of precision oncology, it is hoped that clinical application of next-generation sequencing will ultimately yield molecular insight into individual patient's tumors that can be used to identify targetable mutations and signatures on a case-by-case basis and predict response to targeted therapy because the TCGA genomic analysis revealed therapeutic targets in the majority of HNSCC tumors.
Effective management of HNCs requires comprehensive consideration of often competing treatment goals, which requires the integration of various treatment modalities and supportive services. Accordingly, representation from disciplines including head and neck surgery, plastic and reconstructive surgery, radiation oncology, medical oncology, pathology, neuroradiology, dentistry, oral and maxillofacial surgery, nutrition, rehabilitation medicine, and social work is necessary. Before the start of any therapy, it is important not only to discuss the diagnosis, prognosis, and treatment recommendations, but also to review issues that may affect treatment compliance, such as social support. Similarly, effective attention to nutritional support and pain management can significantly improve patient compliance with any subsequent therapeutic plan. Depending on the treatment modalities required, pretreatment evaluation by oral and maxillofacial services practitioners may be indicated.
Determining a prognosis can be complex and imprecise and is influenced not only by the anticipated clinical outcome but also by various patient factors, including treatment tolerance and toxicity. The imprecision of prognostication results in large part from the clinical heterogeneity of established prognostic and treatment predictive factors. In treating patients with curative intent, a higher threshold for treatment- related toxicities is implicitly accepted and should be discussed with the patient.
For HNSCC, therapeutic principles traditionally have considered competing treatment goals of locoregional control and the level of risk and spectrum of toxicity in defining the concept of a therapeutic ratio. Both quantitative and qualitative issues in toxicity must be considered, because a severe but low-probability toxicity may be important in defining what an acceptable therapeutic ratio is for the patient. This concept of a therapeutic ratio is important, because a dose-response relationship exists for both disease control and toxicity from several treatment modalities, including chemotherapy and RT.
The single-modality principle for treatment of early-stage disease attempts to minimize toxicity and emphasizes the importance of patient and tumor selection. When postoperative radiation therapy (PORT) appears likely, definitive RT may be more appropriate, particularly for small-volume disease, because it is unclear that combined-modality therapy is superior to RT alone. This consideration is particularly relevant when initial definitive en bloc resection may, in combination with PORT, increase treatment toxicity and organ dysfunction. Not uncommonly, these issues arise with more extensive early-stage HNSCC or in cases in which the anatomic location limits achievement of adequate surgical margins. For unresectable locally advanced disease, conventionally fractionated RT has been traditionally recommended. Suboptimal locoregional disease control in the absence of surgical options has forced investigation into various modalities of treatment intensification including neoadjuvant chemotherapy, concurrent CRT, and altered fractionated RT.
Although the AJCC staging system is the most widely accepted and applied prognostic system in persons with cancer, much attention has been given to its inability to enable identification of patients with HNC who are at high risk of treatment failure. Accordingly, various radiographic biomarkers have been studied to help improve our ability to prognosticate outcomes for individual patients.
During the past decade, interest has expanded in the use of PET-CT, specifically given its ability to be an accurate and sensitive imaging modality for the pretreatment and posttreatment evaluation of patients with HNC compared with clinical examination and CT alone. Controversy has plagued studies evaluating the prognostic and predictive usefulness of the standardized uptake value (SUV), essentially the metabolic tumor activity. The success of metabolic-based radiographic biomarkers rests on the ability to standardize protocols to enable interinstitutional comparisons.
Similarly, some volumetric indices have been explored as potential prognostic and predictive indices. Multiple studies have demonstrated that the gross tumor volume correlates with locoregional control and survival in patients with HNSCC undergoing surgery, radiation, or CRT treatments in various HNC sites. Further incorporation of metabolic volumetric parameters in defining tumor volume, such as the metabolic tumor volume, has defined yet another prognostic radiologic biomarker. Unfortunately, no volumetric cut points have been prospectively validated.
Traditionally, the management of the primary tumor site dictated the treatment modality for the neck as a strategy to facilitate efficient management of HNSCC. Treatment decision making for the neck has adopted a similar therapeutic paradigm of risk stratification to optimize the therapeutic ratio, but the algorithm becomes more complex as a result of the primary treatment considerations. Conceptually, the issue of functional organ preservation also may be extended to the neck, because neck dissection may result in cosmetic changes and compromised neck and shoulder function. Similarly, RT-induced edema and fibrosis may compromise the goal of functional neck preservation and may be exacerbated by a neck dissection.
Typically, combined-modality strategies are used for the treatment of neck disease. In a neck with negative clinical findings, when surgical resection has been elected for the primary site management, elective neck dissection may be omitted for most subsites if preoperative evaluation determines a high risk of requiring PORT and if the risk of occult nodal metastasis is sufficiently high to warrant elective management. When RT has been selected for management of the primary site, neck dissection in a neck with negative clinical findings is not indicated. In fact, pre-RT neck dissection may alter the lymphatic flow of the neck, necessitating that larger volumes of the neck be irradiated. Pre-RT neck dissection also may contribute to delays in the delivery of RT, which have been reported in a retrospective analysis to contribute to an adverse overall survival when compared with post-RT neck dissection.
When conventionally fractionated RT alone has been used for locally advanced HNSCC, suboptimal regional control rates coupled with the morbidity and the limited success of subsequent salvage neck dissection have prompted the incorporation and general acceptance of a planned neck dissection. This approach, in the presence of residual adenopathy after RT, also has been selectively applied to patients with adverse risk factors such as large nodal size (typically ≥3 cm). This risk stratification is based on RT series demonstrating an inverse relationship between nodal size and control rate. In a study of RT alone for treatment of HNSCC in 1251 patients, Dubray and colleagues noted 3-year neck control rates, by maximum nodal size, of 77% for 0.5 cm, 67% for 2 cm; 60% for 4 cm, 52% for 6 cm, 37% for 8 cm, and 7% for 10 cm. Multivariate analysis revealed that regional relapses independently increased with increased nodal size ( P = .0001), decreased radiation dose ( P = .0001), T4 primary disease ( P = .0001), node fixation ( P = .02), bilateral neck disease ( P = .03), and geographic miss ( P = .0001).
Controversy continues, however, regarding the benefit of a planned neck dissection in the setting of a complete clinical response in the neck, particularly in large pretreatment lymph nodes, after completion of RT. Treatment with conventionally fractionated RT has demonstrated that the prognosis for patients with large neck nodes with a complete response (CR) is a comparable with that of patients with smaller nodal metastases, and that the risk of relapse is low. This finding suggests that perhaps the subgroup of patients with a CR in the neck may have more radiosensitive disease and may not require a neck dissection. For patients with oropharyngeal carcinoma, Level I evidence has demonstrated that follow-up with PET-CT surveillance is noninferior to planned neck dissection and results in fewer operations and cost savings. A neck dissection continues to be sometimes favored for persons with advanced neck disease (especially stage N3), because the likelihood of achieving a CR with RT alone is limited. Assessment of disease response by PET-CT allows a more objective evaluation of patients who would likely benefit most from a neck dissection.
Management of patients with HNSCC is further complicated by the risk for the development of second primary carcinomas and relapses within the aerodigestive tract that may have been extensively exposed to prior treatment. An irradiated aerodigestive tract not only limits reirradiation but also may preclude effective surgical salvage, because concerns of residual microscopic disease often exist in cases that would otherwise warrant use of PORT. When a second primary or relapse occurs within a previously irradiated field, surgical resection should be the primary treatment option. Experiences with various repeat external beam radiotherapy (EBRT) strategies, including the integration of chemotherapy, have unfortunately demonstrated limited success, although advances in proton beam therapy may mitigate reirradiation toxicity to some extent. The exception appears to be nasopharyngeal carcinomas (NPCs) that are more radiosensitive. Even then, significant late complications may arise but may need to be considered in view of the limited surgical options.
The decision to treat HNSCC with surgical therapy must be undertaken carefully. To assess whether a patient is a surgical candidate, the input of a multidisciplinary team is invaluable. Of the many issues to be considered is the medical condition of the patient. Severe cardiac or pulmonary disease, profound malnutrition, and generalized debilitation are relative contraindications to immediate surgical intervention, and therefore a detailed assessment by an experienced internist or cardiologist is mandatory when a major resection is planned.
The next important consideration is whether the lesion can be removed safely with adequate margins. The resectability of a tumor is assessed via physical examination and radiographic studies. In general, tumor involvement of certain anatomic landmarks, including skull base involvement, intracranial extension, invasion of the prevertebral fascia, or carotid encasement, renders the lesion unresectable. This is due to the inability to achieve adequate normal tissue margins in these areas. In addition to tumor location, massive tumor size also can preclude total extirpation because tumor-free margins may be impossible to obtain. In some cases, adequate reconstruction may be difficult, and other forms of therapy should be considered.
Postoperative function is a primary concern in considering surgical therapy. Speech articulation, voice quality, swallowing, airway stability and protection, cosmetic appearance, facial motion, shoulder and neck function, facial and neck sensation, and many other functional outcomes may be affected by surgery in the head and neck region. Although complete resection of the malignancy is of paramount importance, reconstruction of the surgical defect must be designed so that the patient has an opportunity to regain as much speech and swallowing function as possible. Site-specific considerations are discussed in the following sections.
The ability of the patient to participate in postoperative rehabilitation should also be reviewed. Patients who are emotionally or socially unable to participate should be identified early in the decision-making process. They may be better served with different forms of therapy.
In general, primary surgical therapy is selected for patients with tumors of the oral cavity, skin, salivary gland, paranasal sinus, thyroid, selected early-stage or advanced-stage larynx cancer, and carefully selected lesions of the oropharynx. Patients with very advanced tumors invading bone, destroying cartilage, or extending into the soft tissues of the neck are considered for primary surgical therapy. When patients have severe organ dysfunction as a consequence of cancer infiltration, surgery should be considered because functional restoration with nonsurgical therapy is unlikely.
A variety of different types of neck dissection may be performed ( Fig. 65.1 ). The radical neck dissection is a procedure wherein the lymph nodes from all five levels of the neck, the sternocleidomastoid muscle, the internal jugular vein, and the spinal accessory nerve are all removed en bloc. This procedure is indicated in the setting of high-volume neck metastases involving these nonlymphatic structures.
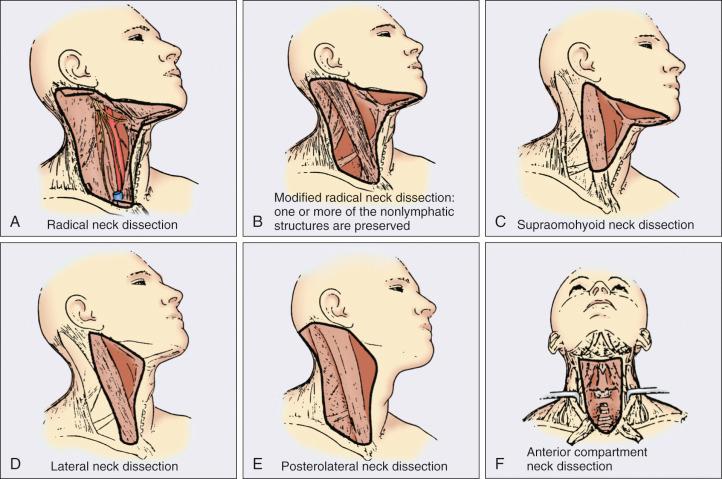
A modified radical neck dissection removes lymph nodes from all five levels of the neck but spares one or more of the nonlymphatic structures. Three types of modified radical neck dissection are type I (which spares one structure, most commonly the spinal accessory nerve), type II (which spares two structures, most commonly the spinal accessory nerve and the internal jugular vein), and type III (spares all three structures—the spinal accessory nerve, internal jugular vein, and sternocleidomastoid muscle). A type III dissection also is termed a functional or Bocca neck dissection. Of these three structures, removal of the accessory nerve is generally considered to result in the most clinically significant functional deficit from shoulder dysfunction. Selective neck dissections involve the resection of fewer than all five levels of lymph nodes, usually involving three or four levels according to the site of the primary cancer.
RT has a long and successful history in the treatment of primary head and neck malignancies. Current RT practices have evolved to include a fractionated RT prescription delivered with modern linear accelerators that can produce a spectrum of beam energies. With fractionation, issues of patient immobilization and treatment setup reproducibility become important considerations. For the head and neck, critical normal tissue structures such as the spinal cord, brainstem, and optic chiasm often are in close proximity to the irradiated target. For these reasons, a prerequisite for treatment simulation is that patients be immobilized with various devices such as a custom made facemask and frame, with the setup referenced to a laser light coordinate system in the treatment rooms ( Figs. 65.2 and 65.3 ).
Immobilization addresses the issue of the precision of the treatment delivery as a strategy to optimize the therapeutic ratio. Historically, patients were treated with two-dimensional (2D) RT or three-dimensional (3D) conformal RT, which did not permit significant sparing of normal tissues and resulted in significant toxicity. In these cases, the oncologist would outline the areas to irradiate on a radiograph of the head and neck. Modern treatment with intensity-modulated radiation therapy (IMRT) involves obtaining axial CT images and delineating the tumor and normal tissue on each CT slice. IMRT uses multiple beam angles and allows the beam modulation to achieve exquisite dose conformality, which means that the tumor receives the full dose of RT while the dose delivered to critical normal structure is minimized. Multiple randomized trials have now demonstrated significantly reduced toxicity with IMRT compared with 2D RT and 3D conformal RT.
Given the improved conformality, the success of IMRT is dependent on the ability to identify anatomic sites that harbor subclinical disease. Currently, the basis for this determination is derived from surgical and clinical documentation of disease extension that is often unique to each head and neck subsite. Over the past decade, interest has been expanding in PET-CT, specifically given its ability to be an accurate and sensitive imaging modality for the pretreatment and posttreatment evaluation of patients with HNC compared with clinical examination and CT alone. Currently, the radiation oncologist must incorporate data from the clinical examination, CT, PET-CT, and often MRI when designing the radiation plan.
The identification of nodal groups typically at risk for harboring subclinical nodal metastases has proven more difficult. To aid in this identification, several reports have defined axial anatomic structures that may be used to delineate the various nodal groups ( Table 65.2 ). The incidence of nodal metastases has been summarized by site and can aid the radiation oncologist ( Table 65.3 ). Of note, however, when prior treatment to the neck has occurred, altered flow of lymphatics is a concern. As with head and neck surgery, judgment must be exercised with these precise treatment techniques.
| Level | A natomic B oundary | |||||
|---|---|---|---|---|---|---|
| Cranial | Caudal | Anterior | Posterior | Lateral | Medial | |
| Ia | Geniohyoid muscle | Platysma muscle | Symphysis menti; platysma muscle | Body of hyoid bone | Medial edge of anterior belly of digastric muscle | Not applicable a |
| Ib | Mylohyoid muscle, cranial edge of submandibular gland or caudal edge of medial pterygoid muscle | Platysma muscle | Symphysis menti | Body of hyoid bone; posterior edge of submandibular gland | Basilar edge of mandible; platysma muscle | Lateral edge of anterior belly of digastric muscle |
| II | Bottom edge of the body of C1 | Bottom edge of the body of hyoid bone | Posterior edge of submandibular gland; posterior edge of posterior belly of digastric muscle | Posterior border of sternocleidomastoid muscle | Medial edge of sternocleidomastoid muscle | Internal edge of internal carotid artery, paraspinal (levator scapulae) muscle |
| III | Bottom edge of the body of hyoid bone | Bottom edge of cricoid cartilage | Posterolateral edge of sternohyoid muscle | Posterior edge of sternocleidomastoid muscle | Medial edge of sternocleidomastoid muscle | Internal edge of carotid artery, paraspinal (scalenus) muscle |
| IV | Bottom edge of cricoid cartilage | Cranial border of clavicle | Posterolateral edge of sternohyoid muscle | Posterior edge of sternocleidomastoid muscle | Medial edge of sternocleidomastoid muscle | Internal edge of internal carotid artery, paraspinal (scalenus) muscle |
| V | Skull base | Cranial border of clavicle | Posterior edge of sternocleidomastoid muscle | Anterior border of trapezius muscle; scalenus muscle | Platysma muscle; skin | Paraspinal (levator scapulae, splenius capitis) muscle |
| VI | Bottom edge of the body of hyoid bone | Sternal manubrium | Skin; platysma muscle | Posterolateral edge of sternohyoid muscle | Medial edge of common carotid artery, skin and anterior-medial edge of sternocleidomastoid muscle | n.a. |
| Retropharyngeal | Base of skull | Cranial edge of the body of hyoid bone | Levator veli palatini muscle | Prevertebral muscle (longus colli, longus capitis) | Medial edge of internal carotid artery | Midline |
a Midline structure lying between the medial borders of the anterior belly of the digastric muscle.
| Tumor Size | Patients With Node-Positive Disease (%) | D istribution of M etastatic L ymph N odes per L evel ( % of N ode -P ositive P atients ) | |||||
|---|---|---|---|---|---|---|---|
| I | II | III | IV | V | Other a | ||
| Oral cavity ( N = 787) | 36 | 42/3.5 b | 79/8 | 18/3 | 5/1 | 1/0 | 1.4/0.3 |
| Oropharynx ( N = 1479) | 64 | 13/2 | 81/24 | 23/5 | 9/2.5 | 13/3 | 2/1 |
| Hypopharynx ( N = 847) | 70 | 2/0 | 80/13 | 51/4 | 20/3 | 24/2 | 3/1 |
| Supraglottic larynx ( N = 428) | 55 | 2/0 | 71/21 | 48/10 | 18/7 | 15/4 | 2/0 |
| Nasopharynx ( N = 440) | 80 | 9/5 | 71/56 | 36/32 | 22/15 | 32/26 | 15/10 |
Conventional or daily RT fractionation (typically, daily 1.8- to 2-Gy fractions to a total dose of 70 Gy) has permitted the delivery of high RT doses, which is currently limited by normal tissue radiation dose tolerance levels, such as in the mandible, parotid glands, brainstem, and lens. Conceptually, RT failures may result from insufficient doses of RT relative to the number of tumor clonogens or may be due to intrinsic tumor radioresistance. The former has proven to be more amenable to therapeutic manipulation with the use of altered fractionation schedules, which may be generalized into two groups: hyperfractionation and accelerated fractionation.
A hyperfractionation schedule, or the use of lower doses per treatment fraction, has been hypothesized to reduce the risk of late RT-induced complications associated with an increase in the total RT dose. Typically, the dose per fraction is reduced to 1.2 to 1.5 Gy and exploits a differential radiosensitivity between normal late-responding tissues and most HNSCC. To ensure that the overall treatment time is not adversely protracted, fractions often are delivered twice a day, with an interfraction period of 6 hours. European Organisation for Research and Treatment of Cancer (EORTC) trial 22791 compared hyperfractionation (1.5 Gy twice daily to a total dose of 80.5 Gy) with conventional fractionation (2 Gy once daily to a total dose of 70 Gy) in patients with T2–3N0–1 oropharyngeal cancer (excluding the base of the tongue). This trial demonstrated a statistically significant improvement in local control of the hyperfractionation arm (38% versus 56%; P = .01) at 5 years with no increase in late complications.
Alternatively, an accelerated fractionation RT schedule attempts to deliver the prescribed total dose over a shorter treatment duration. This strategy was founded on observations of adverse locoregional control rates with protracted treatment durations (with conventional fractionated schedules) such that higher total doses are required to maintain the same probability of tumor control. These results have been interpreted to be consistent with a model whereby tumor clonogens surviving each daily RT fraction undergo an accelerated rate of repopulation. As a consequence, a larger tumor burden would be expected with increasing duration of treatment interruptions. It has been rationalized that when the overall treatment time is reduced, the opportunity and impact of accelerated tumor repopulation would be minimized. As the severity of acute toxicities is increased, some accelerated schedule studies have attempted to modify the risk of unacceptable acute toxicities by modifying either the dose per fraction or the total dose as a strategy to achieve an acceptable therapeutic ratio. EORTC 22851 compared accelerated fractionation (1.6 Gy three times daily to a total dose of 72 Gy) with conventional fractionation (1.8 to 2 Gy once daily to a total dose of 70 Gy) in patients with intermediate to advanced HNCs, excluding the hypopharynx. A significantly improved 5-year locoregional control rate ( P = .02) was noted, but acute and late toxicities were increased in the accelerated fractionation arm.
To date no single fractionation schedule has proven optimal. Radiation Therapy Oncology Group (RTOG) 90-03, a phase III randomized study comparing hyperfractionation, two variants of accelerated fractionation, and standard fractionation, found that both accelerated fractionation with a concomitant boost and hyperfractionation were associated with improved locoregional control and disease-free survival as compared with standard fractionation, but acute toxicity was increased.
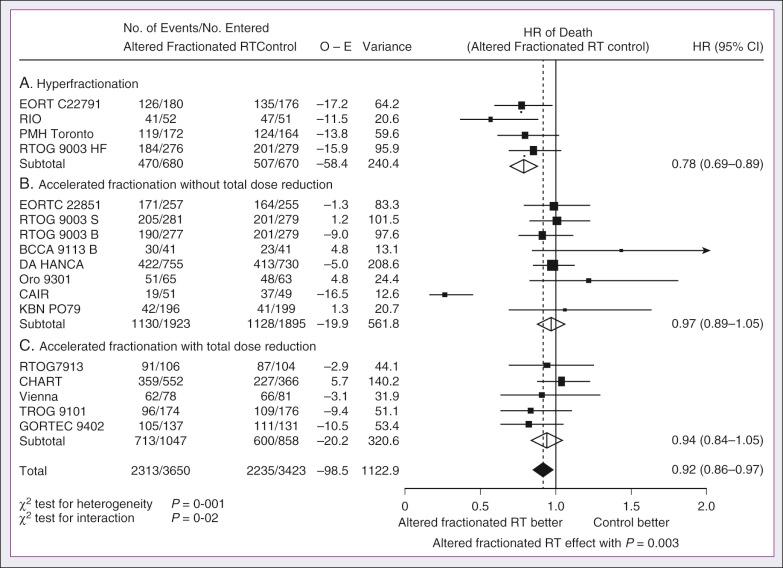
The Meta-Analysis of Radiotherapy in Carcinomas of the Head and Neck (MARCH) Collaborative Group pooled 15 HNSCC randomized trials for a total of 6515 patients and reported that altered fraction schedules, hyperfractionated or accelerated RT, had a 5-year absolute survival benefit of 3.4% (hazard ratio [HR], 0.92; 95% confidence interval [CI], 0.86–0.97; P = .003) and an absolute local control benefit of 6.4% (P < .0001) when compared with standard fractionation ( Fig. 65.4 ). It is important to note that the benefit was significantly higher for younger patients (aged <50 years). Because RTOG 01-29 and GORTEC 99-02 both demonstrated no significant improvement in concurrent conventionally fractionated CRT versus altered fractionated CRT, altered fractionated therapy is typically reserved for patients who are not candidates for systemic therapy.
For more advanced primary lesions (typically defined as stage T3 and T4 disease), increased treatment-related toxicities generally have been accepted because of their inferior local control rates. Accordingly, a combined-modality approach typically has been used. Traditionally, this approach has included surgery and RT with either preoperative or postoperative conventionally fractionated RT. In general, a postoperative protocol has been favored because the RT can be delayed until more accurate delineation of the tumor and histopathologic stratification of patients requiring PORT can be achieved.
PORT has been demonstrated to provide superior outcomes in a subset of patients with HNSCC compared with preoperative RT. The indications for PORT are based on factors associated with an increased risk of locoregional recurrence after surgery, including advanced tumor stage (T3 or T4), presence of a positive margin, extracapsular spread outside lymph nodes, lymphovascular invasion, perineural invasion, presence of a lymph node greater than 3 cm, and multiple positive lymph nodes. When evaluated in a randomized trial in 277 patients with supraglottic larynx and hypopharynx carcinomas (RTOG 73–03), PORT demonstrated superior locoregional control rates compared with preoperative RT, although the results were confounded by the use of a higher dose delivered in the postoperative setting.
Poor risk factors such as the presence of extracapsular extension or positive margins require the addition of chemotherapy to PORT. In a study of 334 patients with tumors possessing high-risk pathologic features and who underwent surgery with curative intent (EORTC 22931), patients were randomly assigned to PORT alone (66 Gy) versus PORT with concurrent cis- diamminedichloroplatinum (CDDP; cisplatin) (with a dose of 100 mg/m 2 on days 1, 22, and 43 of the RT regimen). Postoperative concurrent chemoradiation therapy (POCRT) significantly improved progression-free survival (HR, 0.75 [95% CI, 0.56 to 0.99]) and overall survival (HR, 0.70 [95% CI, 0.52 to 0.95]) and reduced the cumulative incidence of locoregional recurrence in patients receiving PORT as compared with RT alone. Similarly, RTOG 95-01, in which 459 patients with high-risk pathologic features were randomized to receive PORT alone (60 to 66 Gy in 30 to 33 fractions) or RT plus concurrent cisplatin (100 mg/m 2 on days 1, 22, and 43), found that POCRT yielded significantly improved rates of locoregional control (HR, 0.61 [95% CI, 0.41 to 0.91]) and disease-free survival (HR, 0.78 [95% CI, 0.61 to 0.99]). Not surprisingly, CRT resulted in greater grade 3 or greater acute toxicity as compared with RT alone (77% versus 34%; P < .001). Pooled analysis of these two trials suggested that PORT with cisplatin should be considered for patients with either a positive margin or extracapsular extension.
Randomized trials have permitted the identification of prognostic factors that adversely influence the outcome, particularly in high-risk patients. Of greater significance, it is now clear that the time from surgery to the start of PORT and the overall treatment time of PORT are important determinants of locoregional control.
Proton beam therapy allows for more conformal radiation dose deposition owing to differences in physical interactions of protons with tissue compared with photons. Specifically, when the proton beam enters the body, most of the radiation dose is delivered at the specified tumor depth, a phenomenon known as the Bragg peak. The Bragg peak allows for elimination of exit dose, or irradiation of normal structures beyond the tumor, and therefore may allow for improved toxicity and dose escalation in certain settings when compared with the use of IMRT. The accessibility of proton beam technology is limited because of the cost associated with development of facilities, although recent advances have made proton beam therapy more affordable and it is expected to become increasingly available in the coming years.
Given the complex and confined anatomy of the head and neck, tumors often lie in close proximity to critical normal structures including brain, nerves, salivary glands, and oropharyngeal mucosa. In certain scenarios, proton beam therapy has offered significant advantages over the most advanced photon techniques. For skull base tumors, proton therapy may allow for safer dose escalation adjacent to the optic nerves and chiasm and the brainstem. For treatment of lateralized tumors, such as primary tumors of the major salivary glands or skin, the elimination of exit dose results in an improved toxicity profile. In the setting of recurrent disease or second primary tumors, proton beam therapy can minimize retreatment of previously irradiated tissue to minimize toxicity. Aside from the benefits in toxicity, a meta-analysis comparing proton therapy and photon therapy in the treatment of nasal cavity and paranasal sinus tumors demonstrated improvements in 5-year locoregional control (relative risk [RR], 1.25; P = .011) and disease-free survival (RR, 1.44; P = .04) with the use of protons compared with photons. The role of proton beam therapy in the multidisciplinary management of HNCs is evolving rapidly and is the subject of multiple ongoing clinical trials.
Although brachytherapy previously had a significant role in the management of HNSCC, its current role in the initial management of HNSCC has decreased. With the advent of IMRT and the routine use of concurrent chemotherapy, brachytherapy has become less used in the up-front setting, although this is not true in the recurrent setting. It is important to note that studies have demonstrated good outcomes with brachytherapy implants in the definitive setting for several tumor sites including the tonsil and soft palate, oral tongue, base of the tongue, and lip.
Induction chemotherapy, which has been studied for more than four decades, has been repeatedly associated with significant tumor shrinkage and possibly with a decrease in the risk of distant metastases. Unfortunately, an improvement in overall survival for induction chemotherapy over standard treatment approaches has not been definitively demonstrated.
The concept of induction chemotherapy arose from several principles. It was postulated that chemotherapy could promote tumor regression, enhance locoregional therapy through sensitization, and identify potential candidates for a more conservative surgical approach as the need for improved quality of life through functional preservation arose. Thus organ preservation, rather than extensive, potentially morbid surgical procedures, came into vogue as a philosophical consideration in the management of advanced-stage disease. An additional attractive feature with this approach was the conceptual ability to treat micrometastatic disease in hopes of reducing distant failure rates.
Nonrandomized phase II trials in the 1970s used single-agent chemotherapy based on strategies used in the recurrent and metastatic setting. In these single-agent trials, 30% to 40% response rates were reported. Induction strategies subsequently evolved to include multiple chemotherapy regimens. The first reported trials by Wittes and colleagues demonstrated a 71% response rate, with CRs noted in 21% of patients with use of cisplatin and continuous infusion bleomycin in 21 patients. Other studies followed using cisplatin-bleomycin with other drugs such as hydroxyurea, and revealed increased toxicity with no improvement in response rates or survival. Investigators from Wayne State University reported the first trial using neoadjuvant cisplatin with infusional 5-fluorouracil (5-FU), with an overall response rate of 88% and a CR rate of 54%. Trials using carboplatin with 5-FU have also produced good rates of response.
In 1985, the increasing interest in laryngeal preservation coupled with disappointing experiences with up-front RT for advanced disease laid the foundation for the Veterans Affairs Cooperative Studies Program to initiate a multiinstitutional randomized trial of neoadjuvant chemotherapy as an organ preservation strategy. Patients with previously untreated, locally advanced but potentially resectable stage III (T2–3N1 or T3N0) or stage IV (T1–3N2–3 or T4N0–1) disease of the supraglottic or glottic larynx were randomized either to receive neoadjuvant chemotherapy or to undergo surgical resection. The chemotherapy regimen was cisplatin (100 m/m 2 ) with continuous infusion 5-FU (1000 mg/m 2 ) for 5 days every 21 days, with response after two cycles used to stratify patients to either continue with an additional cycle of chemotherapy followed by RT or, for nonresponders, salvage surgery followed by PORT. In total, 332 patients were enrolled: 216 patients with T3 disease, 85 with T4 disease, and 240 patients with N0–1 disease. Laryngeal preservation was achieved in 64% of patients enrolled in the chemotherapy arm. The local failure rate was significantly higher in the chemotherapy plus irradiation arm, but the distant failure rate was significantly lower in this arm. The estimated 2-year overall survival rate was 68% in each arm. This trial demonstrated that laryngeal conservation was achievable in a significant proportion of patients with advanced laryngeal carcinoma without sacrificing overall survival. However, the incremental value of neoadjuvant chemotherapy over RT alone was not answered in this trial.
These findings led to the RTOG 91-11 trial, which was conducted in patients with stage III or IV resectable disease of the larynx. Patients were randomized to three treatment groups: chemotherapy (cisplatin plus 5-FU) followed by RT, concurrent CRT with high-dose cisplatin as the radiosensitizer, or standard fractionated EBRT. The rate of laryngeal preservation at a median follow-up of 3.8 years was significantly higher among patients who received RT with concurrent cisplatin (84%) than in those who received induction chemotherapy followed by RT (72%; P = .005) or RT alone (67%, P < .001). Both chemotherapy regimens suppressed distant metastases and resulted in better disease-free survival than did RT alone. Despite these differences, the overall survival rates were similar in all three groups. As expected, the rate of high-grade toxic effects was greater with the chemotherapy-based regimens as compared with RT alone. This study demonstrated that concurrent CRT was the preferred approach for laryngeal preservation.
Efforts to improve the clinical efficacy of neoadjuvant chemotherapy continue, in hopes of achieving significant activity to yield consistent survival benefits. Largely based on evidence from two European studies (TAX 323 and TAX 324) the Food and Drug Administration (FDA) approved induction chemotherapy with docetaxel (Taxotere), cisplatin (Platin), and 5-FU (TPF) in patients with inoperable and operable HNSCC. The TAX 323 trial was a multicenter, randomized, phase III trial of 358 patients with previously untreated, unresectable, locally advanced stage III and IV tumors who were randomized to cisplatin and 5-FU with or without docetaxel (TPF and PF regimens, respectively). Four to 7 weeks after chemotherapy, patients who did not have progressive disease underwent RT. Patients treated with TPF had a reduction in their risk of death (27%; P = .02) and improved progression-free survival (HR, 0.72; P = .007). In the TAX 324 trial, 501 patients with stage III or IV HNSCC were randomly assigned to receive either PF or TPF as induction chemotherapy, followed by CRT with concurrent carboplatin (sequential therapy) in patients who did not have progressive disease. In this trial, patients who received TPF had improved overall survival (HR, 0.70; P = .006) and locoregional control ( P = .004). Benefits with TPF did come at the cost of higher rates of toxicity. Notably, neither of these trials had a control arm with standard CRT alone.
The meta-analysis of chemotherapy for HNC (MACH-NC) from Pignon and colleagues included 31 induction chemotherapy trials for a total of 5311 patients over a median follow-up of 6.1 years. Overall, induction chemotherapy had a nonsignificant absolute survival benefit of 2.4% at 5 years (HR, 0.96 [95% CI, 0.90 to 1.02]; P = .18; Fig. 65.5 ). No significant variation ( P = .23) of the effect according to the type of chemotherapy was found. These investigators concluded that no clear evidence existed of a differential effect of induction chemotherapy on survival according to age, sex, performance status, stage, or tumor site. In the DeCIDE trial, 285 patients from 20 sites in five countries with N2 or N3 disease were randomized to two cycles of induction chemotherapy with TPF followed by concurrent CRT with docetaxel, 5-FU, and hydroxyurea versus CRT alone. No significant difference in overall survival, recurrence-free survival, or distant failure-free survival was observed, and severe adverse events were more common in the induction arm. A modest benefit was seen in cumulative incidence of distant metastasis without prior locoregional recurrence ( P = .043). The study, however, did not meet planned accrual and so was underpowered.
Similarly, the PARADIGM trial enrolled 145 patients across 16 sites for randomization between induction TPF followed by docetaxel- or carboplatin-based CRT versus cisplatin-based CRT alone. No difference was seen in progression-free survival or overall survival, although the trial was terminated early. The lack of observed benefit may partly have been due to unexpectedly good survival in the CRT arm and lack of stratification for HPV-related disease. Nevertheless, without randomized data demonstrating the benefit of induction chemotherapy compared with CRT alone, the use of induction chemotherapy remains an investigational approach and reserved for select cases.
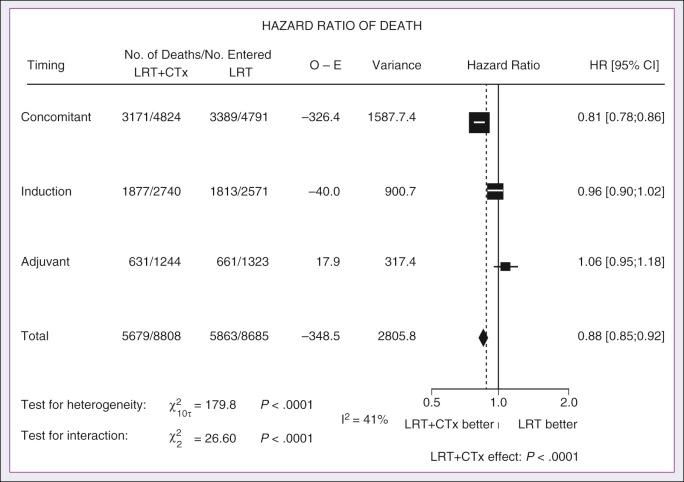
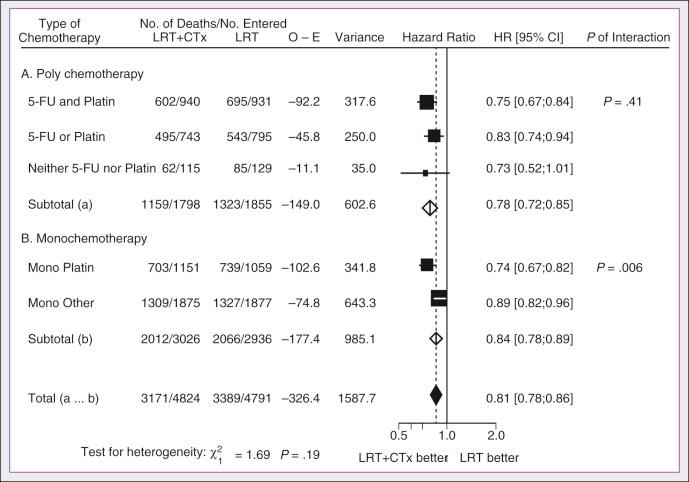
A significant body of literature exists, including numerous randomized trials of concurrent chemotherapy, systematically analyzed by several investigators. These independent reviews have consistently favored the concurrent integration of chemotherapy with RT. Pignon and colleagues from the MACH-NC have reported the largest and most recently updated of these meta-analyses. A patient-based meta-analysis of 9605 patients who underwent concomitant CRT was derived from 50 randomized trials and confirmed an absolute 5-year survival benefit of 6.5% (HR, 0.81 [95% CI, 0.78 to 0.86]; see Fig. 65.5 ). The benefit of chemotherapy was due to its effect on deaths related to HNC (HR, 0.78 [95% CI, 0.73 to 0.84]), because no benefit was seen for noncancer deaths (HR, 0.96 [95% CI, 0.82 to 1.12]). Similarly, an absolute benefit of 6.2% was seen in event-free survival (HR, 0.79 [95% CI, 0.76 to 0.83]). This benefit was largely restricted to patients 70 years of age and younger because a statistically significant decreasing effect of chemotherapy on survival with increasing age was found ( P = .003). Important to note, no significant difference was seen between monochemotherapy and polychemotherapy regimens ( P = .19), and no difference was noted among different polychemotherapy subgroups ( Fig. 65.6 ). In patients receiving monochemotherapy, the effect was significantly higher with platin agents than with other types of monochemotherapies (see Fig. 65.6 ).
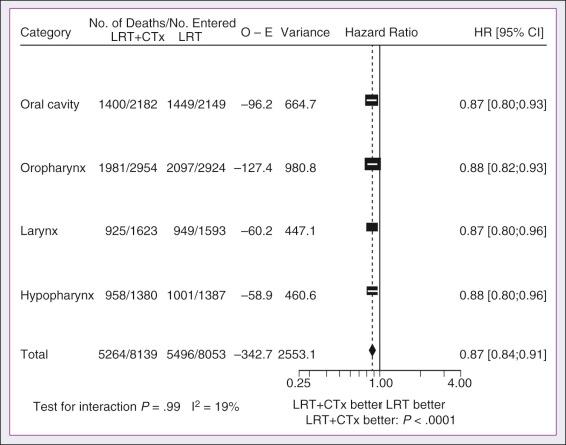
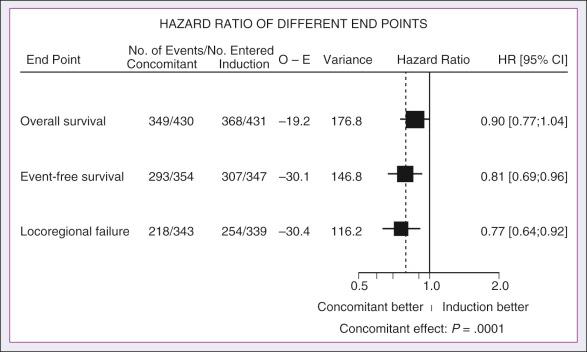
In its most recent update the MACH-NC collaborative group published a comprehensive analysis by tumor site. This analysis included 4331 patients with cancer of the oral cavity, 5878 patients with oropharyngeal cancer, 3216 patients with laryngeal cancer, and 2767 patients with hypopharyngeal cancer. Overall, the addition of chemotherapy resulted in a reduction of the risk of death by 13% ( Fig. 65.7 ), with greater benefit for tumor sites associated with concomitant administration. The 5-year absolute overall survival benefits associated with concomitant chemotherapy were 8.9%, 8.1%, 5.4%, and 4% for oral cavity, oropharynx, larynx, and hypopharynx tumors, respectively ( Table 65.4 ). Similarly, the 5-year absolute event-free survival benefits associated with concomitant chemotherapy were 6.9%, 8.4%, 5.4%, and 3.2% for oral cavity, oropharynx, larynx, and hypopharynx tumors, respectively. In a direct comparison of concomitant versus induction chemotherapy, MACH-NC analyzed six trials with a total of 861 patients over a median follow-up of 10.9 years. Patients who underwent concomitant CRT had a nonsignificant improvement in overall survival with an absolute benefit of 3.5%, significantly decreased locoregional failure, and significantly improved event-free survival compared with patients who were treated with induction chemotherapy patients ( Fig. 65.8 ).
| T iming of C hemotherapy | Test of Interaction a | ||||
|---|---|---|---|---|---|
| Adjuvant | Neoadjuvant | Concomitant | |||
| Oral cavity | HR (95% CI) | 0.94 (0.76–1.17) | 0.93 (0.82–1.05) | 0.80 (0.72–0.89) | P = .15 |
| 5-year absolute benefit (CI) | 0.4% (−7.6 to 8.4) | 2.2% (−2.9 to 7.3) | 8.9% (4.4–13.4) | ||
| Oropharynx | HR (95% CI) | 1.15 (0.92–1.44) | 1.00 (0.90–1.1) | 0.78 (0.72–0.85) | P < .0001 |
| 5-year absolute benefit (CI) | −0.4% (−9.6 to 8.8) | +1.4% (−2.9 to 5.7) | +8.1% (4.8–11.4) | ||
| Larynx | HR (95% CI) | 1.05 (0.84–1.33) | 1.00 (0.81–1.23) | 0.80 (0.71–0.90) | P = .05 |
| 5-year absolute benefit (CI) | 0.1 (−8.5 to 8.7) | 3.8% (−4.6 to 12.2) | 5.4% (0.5–10.3) | ||
| Hypopharynx | HR (95% CI) | 1.06 (0.82–1.38) | 0.88 (0.75–1.02) | 0.85 (0.75–0.96) | P = .31 |
| 5-year absolute benefit (CI) | −2.3% (−13.7 to 9.1) | 5.3% (−0.8 to 11.4) | 4% (−1.1 to 9.1) | ||
Cetuximab, a monoclonal immunoglobulin G1 (IgG1) antibody against the ligand binding domain of the epidermal growth factor receptor (EGFR), has been studied as a single agent or in combination with cisplatin in patients with HNC with promising results. Bonner and colleagues randomized 424 patients with stage III or IV HNC to treatment with high-dose RT alone ( n = 213) or high-dose RT plus weekly cetuximab ( n = 211) at an initial dose of 400 mg/m 2 followed by weekly 250 mg/m 2 doses for the duration of RT. RT with concurrent cetuximab resulted in a significant 32% reduction in the risk of locoregional progression, increased overall survival (HR for death, 0.74; P = .03), and prolonged progression-free survival (HR for disease progression or death, 0.70; P = .006). This overall survival advantage was maintained in a subsequent report on the updated 5-year analysis (HR, 0.73; P = .018). Unfortunately, in the MACH-NC meta-analysis, concurrent CRT trials with cetuximab were limited, preventing definitive conclusions.
In patients with locally advanced unresectable HNSCC, interest has been increasing in the use of sequential therapy (induction chemotherapy followed by concurrent CRT). As described previously, the MACH-NC chemotherapy meta-analysis demonstrated the significant superiority of concurrent CRT (absolute benefit 6.5%) and a nonsignificant benefit from induction chemotherapy (absolute benefit 2.4%). Subsequently, the TAX 323 and TAX 324 trials demonstrated additional benefit with the addition of a taxane to cisplatin and 5-FU induction (TPF regimen). Although the TAX 324 trial demonstrated the principle of sequential therapy (induction TPF with subsequent CRT), it did not include a CRT-alone arm to allow formulation of conclusions regarding the comparative efficacy of these two approaches. Randomized trials comparing sequential therapy and CRT are ongoing.
The proximity of HNCs to the oral and esophageal mucosa results in an increased risk of treatment toxicity. Approximately 50% of patients undergoing concurrent CRT develop severe dysphagia, and the majority develop significant mucositis throughout treatment, both of which result in decreased oral intake and resultant malnutrition.
Prophylactic feeding tubes were routinely used in HNC patients undergoing CRT because it was thought that by providing adequate nutritional support one could decrease dehydration, malnutrition, fatigue, and other treatment-related complications and improve overall treatment outcomes by limiting medically indicated radiation treatment breaks, which historically have been associated with worse outcomes. Although multiple single-institution reports have reported decreased weight loss in patients with HNC who underwent percutaneous endoscopic gastrostomy (PEG) placement, none has demonstrated improved clinical outcomes. A multiinstitutional analysis of 2315 oropharynx patients treated with IMRT demonstrated that only 5% to 10% of patients required continued use of a PEG tube at 1 year after CRT, about 48% of which were placed prophylactically.
The routine use of prophylactic feeding tubes was called into question in a report from Memorial Sloan Kettering Cancer Center (MSKCC). Placement of a prophylactic percutaneous gastrostomy (pPEG) in patients with oropharyngeal HNC who were undergoing definitive CRT failed to significantly improve overall survival, increase albumin levels, decrease acute toxicity rates (e.g., dysphagia, mucositis, xerostomia), decrease chronic dysphagia, and affect treatment duration compared with findings in patients who refused pPEG placement. Given these data, pPEG likely has a role in the management of HNC, but it remains to be defined who will benefit most, and we recommend placement on a case-by-case basis. Current recommendations from our institution and from the National Comprehensive Cancer Network (NCCN) are to consider the prophylactic placement of PEG in patients who have severe weight loss before treatment or who are at high risk of weight loss and dehydration that would necessitate interventions that could potentially delay treatment.
The nasopharynx is a cuboidal structure bounded by the sphenoid bone superiorly, the posterior choanae anteriorly, the clivus and the first two cervical vertebrae posteriorly, and the soft palate inferiorly. The eustachian tube enters through the lateral wall, with the posterior portion of the tube being cartilaginous and forming the portion of the lateral nasopharyngeal wall known as the torus tubarius; just posterior to this is the fossa of Rosenmüller. The nasopharynx afferent innervation is dominantly provided by cranial nerve V2 anterior to the eustachian orifice and cranial nerve IX posterior to the tubal orifice. Motor innervation is dominated by pharyngeal branches of cranial nerves IX and X and sympathetic fibers from the superior cervical ganglion. Arterial supply is provided by the ascending pharyngeal artery, sphenopalatine artery, and vidian artery. The pharyngeal plexus provides venous drainage and communicates with the internal jugular veins directly or via communication with the pterygoid plexus.
The vast majority of nasopharyngeal malignancies are epithelial neoplasms that arise from the lateral wall, particularly in the fossa of Rosenmüller. NPC most commonly manifests as a nontender neck mass, but given its location, nasal and aural symptoms (e.g., epistaxis, nasal obstruction and discharge, headache, and cranial neuropathies) are also common presenting complaints.
Local spread may include extension anteriorly through the submucosa including the nasal cavity; superiorly through the foramen lacerum, located superior to the Rosenmüller fossa, into the base of the skull; laterally to the parapharyngeal space; and inferiorly into the oropharynx. In patients with skull base involvement, two cranial neuropathies have commonly been described. The petrosphenoidal syndrome—that is, superior invasion into the base of the skull—involves cranial nerves III, IV, V, and VI, and patients commonly describe facial pain with V2 involvement with or without ocular muscle and efferent papillary reflex deficits. The retroparotidian syndrome—that is, lateral invasion into the parapharyngeal space—involves cranial nerves IX, X, XI, and XII. Metastatic spread to adjacent upper cervical lymph nodes is seen in 80% to 90% of patients at time of presentation, with more than 50% having bilateral neck disease, most commonly involving the retropharyngeal lymph nodes, but also other major routes of lymphatic drainage including jugular chain and spinal accessory chain pathways. Hematogenous dissemination, most commonly to bone thereafter lung and liver, is present in 3% to 6% of patients at time of presentation, most commonly in patients with advanced neck node metastases, especially with low-neck involvement.
Nasopharyngeal cancer exhibits regional bias with age-adjusted incidence rates (per 100,000 people per year) among men: 0.6 in the United States and Japan, 5.4 in Algeria, 11.0 in Singapore, 17.2 among Eskimos, and 26.9 in Hong Kong and Guangdong providence in Southern China. This discriminative geographic distribution is likely of a multifactorial etiology related to geographic virus infectivity rates, genetic associations, and regional environmental causes. The high consumption of salted fish in Southern China has been implicated, largely as a result of high concentrations of dimethylnitrosamine, a suspected human carcinogen. Genome-wide association studies have identified human leukocyte antigen (HLA) A2, B46, and B17 as associated with an increased risk of developing NPC. Cigarette smoke, alcohol consumption, and exposure to formaldehyde have also been associated with increased risk of NPC. Epstein-Barr virus (EBV), one of the seven classified oncoviruses discussed in Chapter 12 , has been associated with NPC, specifically the nonkeratinizing type, irrespectively of ethnic or geographic origin. Increased IgA antibodies to EBV have been reported to appear months to years before the clinical onset of NPC and can be used to define populations at high risk of EBV-associated epithelial cancer. HPV (high-risk types) may play a role in a small subset of NPC. As in the oropharynx, HPV-related NPC occurs in nonsmoking patients younger than 60 years.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here