Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Mycobacterium ulcerans infection, commonly called Buruli ulcer (BU), is an indolent, necrotizing disease largely limited to skin and subcutaneous tissue, but it may disseminate to lymph nodes and bone. Molecular studies suggest that M. ulcerans diverged from Mycobacterium marinum in geologic antiquity. Cook wrote the first description of BU, in Uganda in 1897. MacCallum and coworkers (1948) were first to isolate M. ulcerans from patients, and Portaels and colleagues (2008) isolated and characterized the first strain from the environment. After tuberculosis and leprosy, BU is the third most common human mycobacterial infection. Because of its frequent disabling and stigmatizing sequelae, BU is an important public health concern.
BU prevails in rural tropical wetlands, especially in areas with seasonal flooding. The reemergence of this potentially serious disease may be related to environmental factors such as deforestation, topographic alterations (dams, irrigation systems), increased agricultural activities, and global climate changes. West Africa appears to be most highly endemic area (Benin, Burkina Faso, Côte d’Ivoire, Ghana, Guinea, Liberia, Nigeria, Togo, and Sierra Leone) with the highest incidence rates observed in Benin, Ghana, and Côte d’Ivoire. Other endemic African countries include Angola, Cameroon, Democratic Republic of Congo, Equatorial Guinea, Gabon, Kenya, Republic of Congo, Sudan, and Uganda. Fewer cases are reported in Asia (China, Indonesia, Japan, and Malaysia) and the Americas (French Guiana, Mexico, Peru, and Suriname). Australian scientists have contributed enormously to the understanding of BU, although BU is endemic only in Queensland and Victoria. Finally, nonendemic countries that have reported and treated imported cases of BU include Canada, France, Germany, the United Kingdom, and the United States.
The annual global estimation of BU is reported as 9000 cases; however, this estimation undoubtedly represents underreporting of the number of BU patients. Children 15 years old or younger account for 75% to 80% of cases, and lesions develop most frequently on the limbs. Risk factors include tropical climate, exposure to stagnant water, unprotected water sources, trauma to skin, and HIV infection.
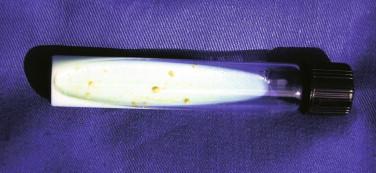
M. ulcerans is strongly acid fast and grows optimally at 30° C to 32° C on routine mycobacterial media (e.g., Löwenstein-Jensen; Figure 22-1 ). It is microaerophilic, slowly growing, and exquisitely sensitive to ultraviolet radiation. Nearly all strains are nonviable after exposure to temperatures of 37° C or higher. Unlike other mycobacterial pathogens, M. ulcerans elaborates a diffusible, necrotizing, and immunosuppressive toxin: a polyketide called mycolactone. Genes in a plasmid of M. ulcerans encode the synthases and modifying enzymes that generate this toxin. Variations in the 3ʹ end of the 16S rRNA gene are used to broadly divide M. ulcerans into four strains: African, American, Asian, and Australian. Definition of single nucleotide polymorphisms (SNPs) now permits the development of SNP typing techniques that may promote a better understanding of the microepidemiology of BU. Proteomic analysis of M. ulcerans is also under study. IS 2404 and IS 2606 are the identifying insertion sequences typically employed in polymerase chain reaction (PCR) analyses and will be discussed later in the section on laboratory diagnosis.
Modes of transmission are not established. The most favored concept is that the etiologic agent enters the skin at sites of trauma or on contaminated fomites such as splinters. Reported antecedent trauma at lesion sites includes gunshot, land mine wound, thrown stone injury, hypodermic injection, penetrating wood splinters, human bite, and snakebite. Insect bite as a mode of transmission is also under study. Interestingly, the first pure culture of M. ulcerans from nature was from a water strider. To date, person-to-person transmission has not been reported; however, animal-to-animal transmission has been postulated in Australia, where koalas and possums acquire BU naturally.
After inoculation of M. ulcerans into skin, development of disease is probably most directly related to temperature (with the optimal growth requirement being lower than body temperature at 30° C to 32° C) and production of the necrotizing toxin mycolactone. Many authorities now believe that M. ulcerans is fundamentally an intracellular pathogen that is engulfed initially and throughout the active disease by variable infiltrations of inflammatory cells. This is evidenced by the fact that all appropriate tissue sections from active BU cases contain inflammatory cells, some of which contain acid-fast bacilli (AFB). This intracellular location is common for pathogenic mycobacteria and plays an essential role in the pathogenesis and immunity in the development of disease. However, to satisfy this conceptualization, the intracellular growth of M. ulcerans must be reconciled with the remarkable cytotoxicity of mycolactone. One proposed mechanism is that intracellular growth of M. ulcerans is controlled by the regulation of mycolactone genes. There is no doubt, however, that the majority of AFB in BU are extracellular and that they proliferate extracellularly.
The clinical and histologic spectrum of features of the various forms of BU suggests that some patients are innately immune to the disease, others acquire immunity during the infection, and still others are incapable of developing resistance. Reactions to the skin test reagent “burulin,” a sonicate of M. ulcerans , reveal that noninfected people, or patients in the early stages of disease are usually nonreactive, whereas later in disease, patients become positive reactors with the development of delayed-type hypersensitivity (DTH) and subsequent healing.
Sections of localized lesions contain large amounts of gamma interferon corresponding to a Th-1 type response, whereas the more progressive forms of BU show abundant IL-10 corresponding to a Th-2 type reaction. Such investigations on minor ulcers are not available, but because these ulcers regularly self-heal, studies focused on this form could provide valuable immunologic information.
Apoptosis, probably mediated by mycolactone, plays an important role in tissue destruction in BU. In addition, mycolactone has been experimentally shown to be immunosuppressive for T and B cells, partly by inhibition of IL-2, tumor necrosis factor (TNF)-α, and other cytokines. Mycolactone also affects circulating leukocytes, dendritic cells, and lymphoid tissues. Whether or not circulating mycolactone affects the function of visceral organs and other tissues in patients with extremely severe edematous disease is uncertain. No clinical laboratory metabolic profiles, for example, have been reported, and there are no published results of autopsies of such patients. One such patient we studied was anuric and died in hospital.
Vaccination with Bacille Calmette-Guérin (BCG) moderates the severity of BU infection but full protection is short-lived. Vaccine research for BU is of high priority.
In summary, the pathogenesis of the natural history of untreated BU infection proceeds through the following stages:
Infection, followed by a latent period. Factors that may determine the pathway of the infection may include depth of inoculation of M. ulcerans , number of organisms inoculated, and virulence of the etiologic agent.
Intracellular proliferation of M. ulcerans followed by lysis of inflammatory cells by mycolactone.
Development of a mycolactone-created necrotic nidus and enhanced cell necrosis.
Increased immunosuppression from extension of necrosis inhibits cell-mediated immunity (CMI).
Eventually there is sufficient stimulation of immunoresponsive cells to raise the level of DTH and develop a granulomatous response to promote healing.
Variations in the duration and types of each of these stages may be related to genetic variance in the etiologic agent, type of host response, and other factors.
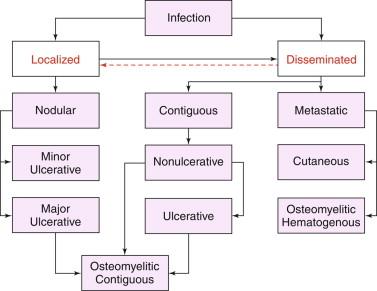
Following infection, the etiologic agent may remain dormant, provoke localized disease, disseminate from an obvious localized lesion, or disseminate rapidly without localized signs. Estimated incubation periods range from 2 weeks to 3 years, or longer, based on antecedent trauma records. The median incubation period is estimated to be 3 months. Figure 22-2 depicts a classification of the spectrum of forms of BU proposed by the authors and shows possible pathways of how untreated disease may advance. Further details of localized and disseminated disease are discussed later.
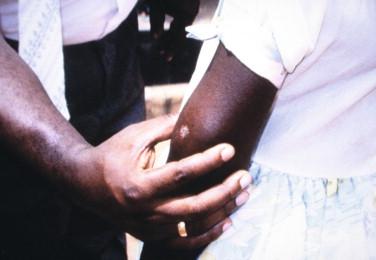
Localized disease includes nodular and ulcerative forms. Nodules are firm, movable masses in the dermis and subcutis that are nonpainful and measure 2 to 4 cm ( Figure 22-3 ). In Australia, the initial lesion may present as a painless pustule (< 1 cm in diameter).
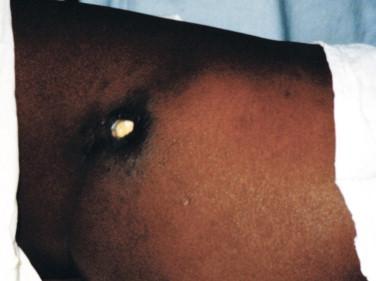
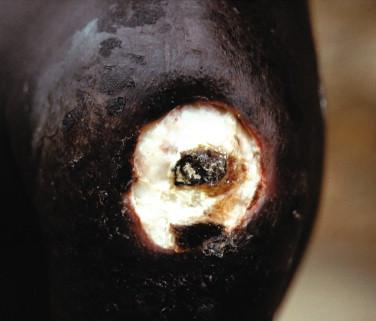
Ulcers can be classified as minor (1 to 2 cm in diameter) or major (> 2 cm). Minor ulcers typically arise at the site of a former nodule and are nonpainful, are sharply demarcated with undermined edges, and usually heal without treatment soon after presentation ( Figure 22-4 ). Major ulcers also arise at sites of former nodules, but they usually present with widely undermined edges, necrotic slough in center of the ulcer, and, initially, edema of surrounding skin ( Figure 22-5 ). Occasionally subjacent bone is eroded and often disabling cicatricial contractures over joints follow. Metastatic disease to nonadjacent bone or distant skin sites also may develop.
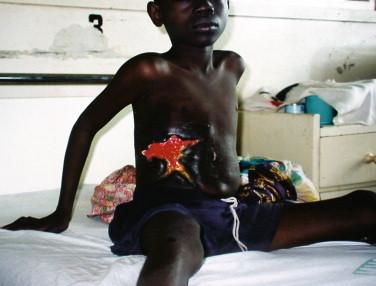
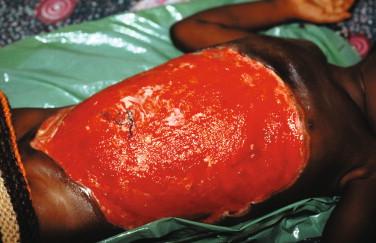
Disseminated disease refers to either wide contiguous spread from a localized lesion, or noncontiguous metastatic spread via the lymphatic or hematogenous routes. Plaques and edematous forms fall under the category of contiguous disseminated disease. Plaques are indurated lesions, > 2 cm in diameter, which may or may not arise from nodules and frequently display late ragged stellate ulceration ( Figure 22-6 ). The edematous form is a diffuse, nonpainful, firm induration of the skin with margins that are difficult to discern. Lesions range from relatively small to massive (involving a whole limb or trunk). There is usually no history of a preceding nodule or plaque in edematous forms. Figure 22-7 shows the postsurgical appearance of a large edematous lesion that underwent debridement to remove necrotic tissue. Ulceration or a nodule was never noted in this case.
The disease may metastasize via lymphatic or hematogenous routes to regional lymph nodes, distant cutaneous sites, or bone. Initial bone lesions are often at the epiphysial/metaphysial area of long bones. Resultant disease often leads to amputation. Osteomyelitis develops in 10% to 15% of BU patients.
In endemic areas, experienced clinicians are usually proficient in diagnosing BU, particularly the ulcerative and plaque forms. Nodules and widely disseminated BU, however, are often difficult to diagnose because of the broad differential diagnostic possibilities.
The four laboratory tests that are used for confirmation of suspected BU are direct smear for AFB, in vitro cultivation, molecular diagnosis, and histopathologic examination.
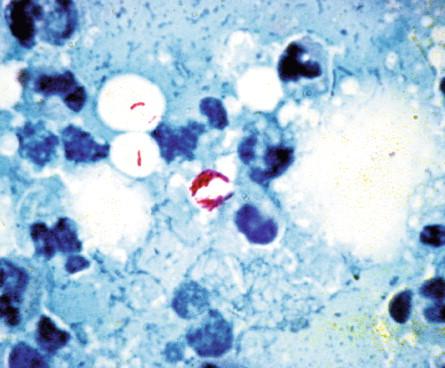
The bacilli of M. ulcerans stain well with Ziehl-Neelsen or auramine O stains ( Figure 22-8 ). Material that may be examined by direct staining includes exudates, fine-needle aspirates (FNA) of nonulcerated lesions, and biopsy specimens. Unfortunately, the sensitivity of AFB smears alone is only ≤ 60%.
In vitro cultivation of M. ulcerans may also be performed from exudates, FNA, or biopsy specimens using Löwenstein-Jensen medium incubated at 30°C to 32 °C, following decontamination with oxalic acid (see Figure 22-1 ). The organism is a slow grower, often requiring many months to appear in primary culture. Pigmentation of colonies ranges from dull white to yellow, and this variation in pigmentation of M. ulcerans colonies seems to be related to the geographic origin of the strain of the organisms. Sensitivity of this test is 70% to 80% if the specimen is plated directly after obtaining it from patient. Transport media are available if cultivation cannot be done on site, but prolonged transportation times may decrease the rate of recovery of the etiologic agent.
Molecular diagnosis may be performed using assays targeting the IS 2404 gene by gel-based conventional PCR or quantitative real-time PCR. Success has been described by extracting DNA from nondecontaminated specimens using the modified Boom method, followed by real-time PCR. Sensitivity of these procedures is > 90%.
Some other mycobacteria may be positive for the IS 2404 gene, but none of them is known to cause lesions mimicking BU. Thus, PCR should only be performed on material obtained from patients with probable clinical evidence of BU. Variable number of tandem repeats (VNTR) analysis also separates M. ulcerans from other mycobacteria with IS 2404 . Histopathologic findings (described later) are > 90% effective in diagnosing BU.
The sensitivities (or efficacies) stated here for each test reflect data from comparative studies and from the authors’ experience. Although a gold standard has not been established, PCR and histopathology seem to be the most dependable procedures for a firm diagnosis.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here