Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The maintenance of bone health is important throughout the entire prostate cancer disease spectrum. The majority of prostate cancer patients are already at increased risk for fragility fractures secondary to age-related bone loss. This is further compounded by the threat of treatment-associated osteoporosis and fractures. In more advanced stages, additional risk arises from disease-associated skeleton-related events (SREs) secondary to bone metastasis. Further complicating the prevention of skeletal morbidity is the fact that a substantial portion of prostate cancer patients have abnormal baseline levels of bone mineral density (BMD) prior to the initiation of therapies.
Most studies looking at bone health use the term “skeletal-related events” as a clinical endpoint, which include pathologic fractures, spinal cord compression, need for surgery, or need for radiation therapy. SREs are associated with a significant decline in quality of life (QOL). Once a single SRE occurs, the risk of subsequent SREs increases. SRE also imposes a substantial economic burden, with a mean annualized SRE cost of $12,469. Without interventions to prevent them, the rate of SREs is estimated to be up to 44% at 15 months in patients with castrate-resistant prostate cancer (CRPC).
The integrity of the skeletal system is a complex and dynamic coupled process that results in bone remodeling ( Figure 53.1 ). Bone remodeling is a complex interaction of various regulators within a rich milieu of growth factors. This process is initiated by bone resorption from osteoclasts, which secrete acid and collagenase. Osteoclasts are large multinucleated cells that are induced from macrophages after exposure to certain cytokines. The bone resorption effects of osteoclasts are balanced by bone formation mediated by osteoblasts. Osteoblasts are single nucleated cells, which are the product of the differentiation and maturation of mesenchymal stem cells. Osteoblasts organize themselves into a functional group of connected cells that produce the extracellular bone matrix. The bone matrix includes an organic component of dense cross-linked collagen with other proteins. The organic component is then mineralized by deposition of a calcium-phosphate-hydroxide salt called hydroxyapatite. Modeling occurs in localized area of cortical and trabecular bone known as bone metabolic units.
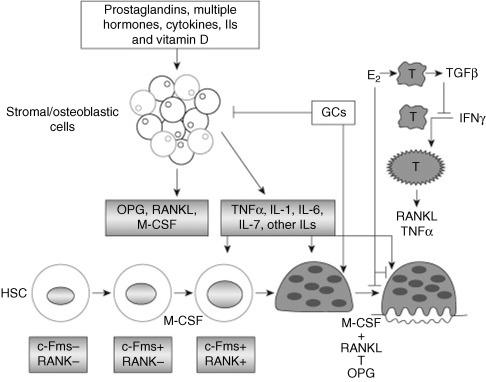
Imbalances within the interactions between osteoclast and osteoblast could lead to a reduction in BMD. If BMD loss is substantial, it increases the risk of bone fractures in situations that would be unexpected in healthy individuals – so-called fragility fractures. Increased bone turnover can be observed with certain laboratory evaluations, such as bone-specific alkaline phosphatase for osteoblast activity and urinary N-telopeptide for osteoclasts.
Androgen deprivation therapy (ADT) is common in many stages of prostate cancer therapy. Unfortunately, ADT is associated with a substantial decrease in BMD, which increases with the duration of therapy. It is not uncommon for patients on ADT to have BMD losses of 3–5% per year, up to even 14% in some studies. This represents a five- to sixfold increased rate compared to healthy men. BMD loss occurs throughout the entire skeletal system in multiple sites. BMD loss is most profound in the first year of initiating ADT but continues annually.
Unfortunately, many men starting on ADT have baseline osteoporosis. The prevalence of osteoporosis in hormone-naive prostate cancer patients is estimated to be approximately 35%. Risk factors for an abnormal baseline BMD include age over 70 years, a low body mass index, and a higher PSA level. The prevalence of osteoporosis increases with ADT duration to nearly 50% within 4 years of therapy and over 80% after 10 years of ADT. Short-term ADT does not appear to increase fracture risk, which suggests that bone loss from hypogonadism may be reversible.
Associated with the BMD loss is the significantly increased risk of fragility fractures on ADT. One of the first studies to clearly demonstrate that fracture risk was increased with ADT used Surveillance, Epidemiology, and End Results (SEER)-Medicare linked data to show that 19.4% of patients receiving ADT sustained a fracture, as compared to 12.6% not receiving ADT ( p < 0.001). Furthermore, individuals on ADT are more likely to experience a fracture requiring hospitalization (5.2% vs. 2.4% p < 0.001). There is over a twofold increase in the risk of femoral neck and intertrochanter fractures after orchiectomy compared to healthy controls. This increased risk can be seen as early as 6 months after androgen deprivation, and remains elevated for at least 15 years while in a hypogonadal state. Caucasian males are at a greater risk of fractures on ADT.
In addition to the decreased quality of life and economic burden from fractures, approximately 40% of patients will be unable to walk independently 1 year after a hip fracture. After a hip fracture, between 60% and 80% of patients will be unable to perform independent activities of daily living. Most importantly, ADT-related fractures are also associated with a reduced survival by over 3 years (121 vs. 160 months p = 0.04).
The mechanism of bone loss from ADT is thought to be from decreases in serum testosterone, and therefore decreased aromatized estrogens, resulting in increased osteoclast activity. In population studies of elderly men, low estradiol is more strongly associated with low BMD and greater fracture risk than even low testosterone. Even with the addition of antiandrogens, bone loss will continue to occur.
As cancer progresses to the castrate-resistant state, the current treatment paradigm is for these patients to remain on continuous ADT as they receive additional disease-modifying agents. As more novel and effective therapies emerge, patients with CRPC are living longer, which results in a lengthening of the duration of ADT and the associated risk of ADT-related fractures increases.
Early detection of osteoporosis may result in the potential avoidance of the morbidity of fractures by prompt therapeutic interventions and lifestyle changes. Bone mineral testing can be performed by several noninvasive modalities including ultrasound and quantitative computed tomography (CT); however, dual-energy X-ray absorptiometry (DEXA) is the favored gold standard as it provides an assessment of BMD at multiskeletal sites (spine, hip, proximal femur, and total body) with minimal radiation while still being inexpensive, accessible, precise, and validated in clinical trials ( Figure 53.2 ).
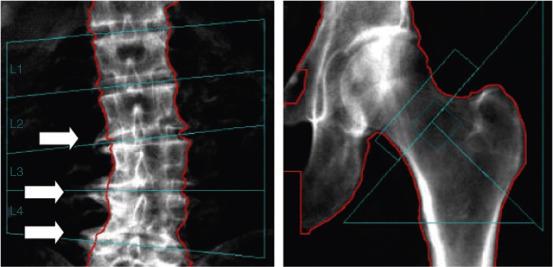
BMD can be expressed in absolute terms, such as grams per centimeter squared, or more commonly as a relative score. The T -score is the difference in standard deviation that an individual’s BMD is from a “young normal” adult of the same gender. A BMD within one standard deviation is considered a normal T -score. A T -score of −1.0 to −2.5 is considered osteopenia. Osteoporosis is defined as the occurrence of either an osteoporotic fracture or a T -score ≤−2.5. DEXA also provides what is known as a Z score, which is the difference in number of standard deviations between the individual and the mean value for a population of the same age and gender. T -score and Z -score have been well established in perimenopausal women, but their validity in men has been questioned by some.
An additional assessment tool to predict fracture risk is the World Health Organization’s FRAX (fracture risk algorithm) score, which considers age, body mass index, and other clinical risk factors to calculate a 10-year probability of hip or major osteoporosis-related fracture.
Patients on ADT at particularly high risk for the development of osteoporosis include those who have been on ADT for over 6 months, previous fractures, a family history of osteoporosis, low body weight, tobacco and alcohol abuse, corticosteroid use, medical comorbidities, and vitamin D deficiency. Although low BMD is a risk factor for bone fractures, it is important to recognize that the majority of men on ADT who experience fractures have BMD that do not meet osteoporosis criteria.
The National Comprehensive Cancer Network (NCCN) guidelines do recommend that all patients receiving ADT receive a baseline assessment of BMD. Routine follow-up BMD scans every 6–12 months while on ADT should also be considered, especially in high-risk patients. Unfortunately, despite these recommendations, few patients on ADT actually receive DEXA screening.
Several medications have been shown to increase BMD in nonmetastatic patients receiving ADT. These include the bisphosphonates pamidronate (60 mg every 12 weeks), alendronate (70 mg once per week), and the more potent zoledronic acid 5 mg once per year, in addition to the receptor activator of nuclear factor-kappa B ligand (RANKL) monoclonal antibody denosumab (60 mg every 6 months). However, to date, only denosumab has been shown to reduce the actual risk of fracture for patients on ADT.
The recommendations by the National Osteoporosis Foundation and NCCN are for consideration of pharmacologic therapy with a T -score between −1.5 and −2.0. The guidelines recommend strong consideration for a T -score <−2.0, FRAX fracture risk probability ≥3% for their hip, or FRAX fracture risk probability ≥20% at other sites.
Bone is the most common site of metastasis in prostate cancer, with osseous metastasis occurring in nearly 80% of patients with metastatic prostate cancer. The most common site of osseous metastasis corresponds with the amount of bone marrow present, such as the vertebral column, pelvis, ribs, and long bones. In contrast to other solid malignancies, bone metastases from prostate cancer usually appear as osteoblastic lesions. Despite the radiographic appearance of osteoblastic lesions, the complex interaction between the metastasis with bone stroma result in abnormal bone remodeling leading to biomechanical instability and pathological fractures. Expansion of tumor within bone and progressive destruction of bony matrix may lead to pain, compression, or pathologic fractures.
Replacement of bone marrow with metastasis also impairs hematologic production, which is further complicated by myelosuppression from long-term ADT, radiation, and cytotoxic therapies. Hypercalcemia is uncommon from prostate metastasis, if present; it is more suggestive of parathyroid hormone-related protein expression from differentiation.
The exact mechanism for the development of bony metastasis has not been conclusively defined but there exists robust support for the “seed” and “soil” hypothesis, originated by Stephen Paget in 1889. This hypothesis postulates that metastasis development results from a complex series of interactions between metastatic cells (the “seed”) and the bone microenvironment (the “soil”). Primary tumor sites are a heterogeneous group of cells with various mutations. For metastasis to occur a tumor cell must be able to separate itself from the extracellular matrix. Detachment of cells from the primary tumor is believed to be through an epithelial–mesenchymal transition with alteration in integrin-mediated attachments. The detached cancer cells must then invade through the tissue stroma and into the vasculature. Circulating tumor cells must then survive immune surveillance and attach to cortical or medullary bone spaces via specific receptors, including the integrin family. The establishment of metastatic sites results from the reciprocal interactions between cancer cells and bone microenvironment. Central to this complex interaction is the rich milieu of growth factors within the bone microenvironment. Many of these interactions are mediated by androgen responsive elements and other bone-derived proteins ( Figure 53.3 ).
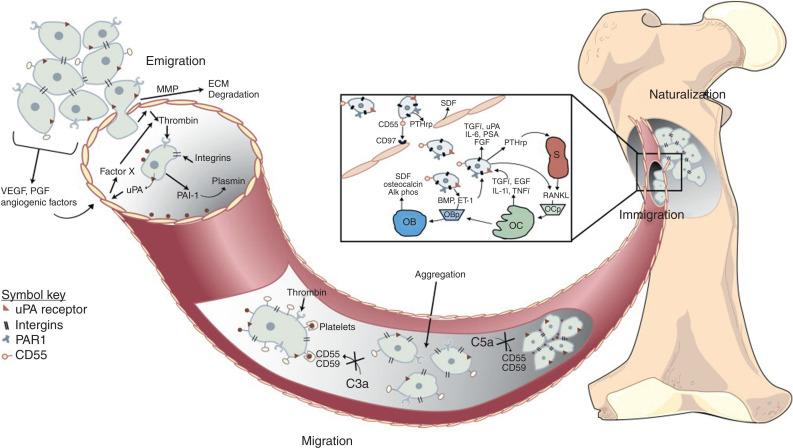
The time to the development of bony metastasis is extremely variable for castrate-sensitive and castrate-resistant prostate cancer. This is because they are both very heterogeneous groups of patients. In CRPC, the median time from a rising PSA despite ADT to the development of a bony metastasis is approximately 2 years, but ranges as widely as 9 months to 30 months. It is estimated that without the addition of bone-targeted therapy, the rate of SREs for patients with CRPC would be about 44% at 15 months.
Pretreatment PSA, PSA nadir on ADT, and PSA velocity has been associated with the time to development of bony metastasis. After development of metastasis despite radical localized therapy, the median time till death is approximately 5 years. The extent of bony metastasis does have prognostic significance, and elevated serum alkaline phosphatase levels are independently associated with shorter survival.
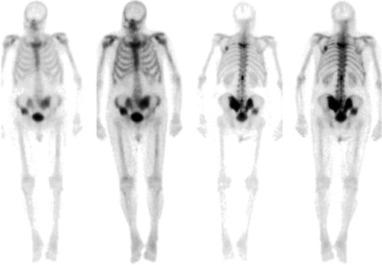
The most commonly used imaging modality for the detection of bony metastases is a radionucleotide bone scintigraphy using Technetium 99m-diphosphonate, commonly referred to as a bone scan. While it is estimated that nearly 30–50% of bone marrow needs to be replaced by tumor cells to be detected on plain film X-ray, only 10% of the marrow needs to be replaced for the area to appear abnormal on bone scan. Bone scintigraphy is limited in early stage lesion as the osteoblastic response may not be substantial enough for detection on bone scan. Furthermore, healing fractures and degenerative changes can be difficult to differentiate from metastasis. MRI can also be used and has a high sensitivity (82–100%) and specificity (73–100%) in detecting tumor within marrow, even if an osteoblastic response is not yet present.
Bone scintigraphy scans are an established and important component of most clinical trials in advanced prostate cancer. As the disease progresses after ADT initiation, many patients may be relatively asymptomatic despite the presence of occult bony metastasis. The prevalence of a positive bone scan correlates with serum PSA. Depending on the patient population characteristics the rate of bone scan positivity is estimated to be <1% if serum PSA ≤10, approximately 10% if PSA is between 10 and 50, and over 50% for a PSA ≥50. These rates vary depending on the study design and population; for example, others have estimated the rate of a positive scan for a patient with CRPC and PSA ≤10 to be between 12% and 26%.
PSA kinetics also correlates with bone scintigraphy scan positivity as a shorter PSA doubling time (PSADT) is also associated with an increased risk of bony metastasis. In nonmetastatic CRPC, a PSADT of ≤10, ≤6, and ≤4 months is associated with a median time to first bone metastasis of 26, 22.1, and 18.5 months, respectively.
Guideline recommendations for obtaining bone scans from the NCCN are for patients who are either symptomatic, have clinical T3/T4 disease, clinical T2 with PSA >10, clinical T1 with PSA >20, or any Gleason Score ≥8 as long as life expectancy is over 5 years. The AUA guidelines recommend a bone scan for patients with a PSA >10 or have poorly differentiated/high-grade tumors. The EUA guidelines suggest that a bone scan is usually not indicated for asymptomatic patients with a PSA <20 ng/mL “in the presence of well-differentiated or moderately differentiated tumors.”
Despite these recommendations approximately 45% of low-risk patients receive unnecessary imaging (39% bone scans), while only 66% of high-risk patients receive appropriate imaging. The overutilization of unnecessary routine bone scans in low-risk prostate cancer patients has been identified by the AUA and ASCO in their “choosing wisely campaign” as a practice that needs to be stopped. A Swedish effort to disseminate utilization data and guideline recommendations to urologists led to a decrease in inappropriate imaging of low risk (from 45% to 3% p < 0.001), but also had the unintended consequence of decreased appropriate imaging of high risk (from 63% to 47% p < 0.001) over a 10-year time period. Although it is unclear if a national level initiate in the United States would be successful, regional collaboration that provides feedback on imaging utilization and guideline recommendation have been shown to reduce inappropriate imaging and variation among a group of urologists in Michigan, Ohio, and Indiana.
For patients who receive localized treatment, the NCCN guidelines suggest imaging should be based on individual risk, age, PSADT, Gleason score, and overall health, but they acknowledge that bone scans are rarely positive in asymptomatic men with PSA <10 ng/mL ( Figure 53.4 ).
Positron emission tomography (PET) combined with CT (PET/CT) has shown promising results in the detection of bony metastasis. 18 F-Fluorodeoxyglucose (FDG) is a glucose analog that is taken up by metabolically active tumor cells, and allows PET/CT to detect active disease within the marrow before a secondary bone reaction occurs. 18 F-Fluoride is another PET tracer, which is taken up by bone surface cells with the most active turnover. The combination of 18 F-fluoride with FDG is under investigation, along with several other PET tracers. The sensitivity for PET/CT ranges from 62% to 100% with a specificity from 96% to 100%. However, data on PET/CT remains limited in metastatic prostate cancer. At this time, bone scintigraphy is still considered the preferred first-line imaging modality for prostate cancer. Currently, 18 F-FDG-PET /CT is usually reserved for equivocal cases after bone scan to help differentiate active metastases from healing bones (EUA).
Bone matrix is comprised of nearly 90% Type 1 collagen. Serum concentration of the precursor molecule, procollagen, is elevated in the setting of early bone formation and osteoblast activity. Similarly, the degradation products of Type 1 collagen can be used as markers of bone resorption. Elevated levels of these and other bone biomarkers have been shown to correlate with tumor burden within bone, which may provide some prognostic and predictive value. For example, a reduction in bone biomarkers of osteoclast activity, such as urinary N-terminus procollagen, may predict the impact of denosumab in reducing subsequent SREs, although other results have been mixed and prospective validation is needed.
Alkaline phosphatase is another biomarker of bone metabolism with a role in the diagnosis and monitoring of patients with bony metastasis and has been used extensively for some time. Alkaline phosphatase is found in osteoblasts, but can be a rather nonspecific serum marker of metastasis as it is also found in liver and the intestine. The bone isoform accounts for 40–50% and is more specific than total alkaline phosphatase but can still have 15–20% cross-reactivity with liver alkaline phosphatase. Alkaline phosphatase is recommended by the NCCN and EUA for the detection of bony metastasis.
Although bone scintigraphy scans can demonstrate the presence of osseous metastases, bone scans are limited in quantifying disease burden and measuring the degree of treatment response. Bone Scan Index (BSI) is an attempt to improve upon this. BSI measures the tumor burden in bone as a percent of the total skeletal mass based on a reference male skeleton. BSI can be automated to reduce time and interobserver differences. In a preliminary analysis, BSI was associated with overall survival in patients receiving cytotoxic chemotherapy. These findings will need to be validated in the expanding array of systemic therapies for metastatic prostate cancer.
Historically, patients dying from prostate cancer usually presented with bony metastasis and died of their disease after rapidly progressing to CRPC. This presentation is far less common in the PSA screening era. More so, PSA monitoring has led to the creation of “the clinical state of a rising PSA” despite radical localized therapy. Although some argue this is an artificial disease context, these patients are frequently started on ADT and might progress to CRPC without any evidence of metastatic disease. Patients with nonmetastatic CRPC are a very heterogeneous group with a variable length of time till they develop metastatic disease; however, when metastasis does develop, they will most likely occur in the axial skeleton.
Currently, no bone-targeting agent is approved for the prevention of osseous metastasis. Several negative trials have been conducted using bisphosphonates and endothelin antagonists. Only the RANKL inhibitor denosumab in the Denosumab 147 metastasis prevention trial demonstrated a 4.2 month improvement in bone metastasis-free survival in nonmetastatic CRPC patients with either a PSA ≥8 or PSADT ≤10 months. However, no benefit from denosumab was seen in OS in this setting and there are several criticisms in the trial design. To date, denosumab is not approved for the prevention of bony metastasis.
Two on-going bone metastasis prevention trials with zoledronic acid include the RADAR (NCT00193856) and STAMPEDE (NCT00268476). RADAR randomized 1071 “high-risk” patients (clinical stage T2b–4 or clinical stage T2a with Gleason ≥7 and PSA ≥10) without metastasis to definitive radiation with ADT (short term or long term), with or without zoledronic acid. The STAMPEDE trial is a complex seven-arm phase II/III trial, which plans to enroll 4000 men with high-risk localized, metastatic, or relapsed prostate cancer starting on ADT. STAMPEDE examines several combinations of ADT, zoledronic acid, docetaxel, abiraterone, and celecoxib. The primary endpoint for STAMPEDE is OS.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here