Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The bladder is a hollow viscus with the shape of a four-sided inverted pyramid when empty and of a rounded structure when distended. It is divided into the following portions: superior surface (also known as dome , and covered by the pelvic parietal peritoneum), posterior surface (also known as base ), and the two inferolateral surfaces . The trigone is located at the base of the bladder and is continuous with the bladder neck , in which the posterior and inferolateral walls converge to open into the urethra. The lymphatic drainage of the bladder is primarily through the external and internal iliac nodes; portions of the bladder neck region may drain to the sacral or common iliac nodes.
The layers of the bladder are the mucosa, muscularis propria (MP), and adventitia. The latter is covered by serosa at the dome. The mucosa is formed by the epithelium and lamina propria, which often contains (rarely) a continuous or discontinuous muscularis mucosae (MM). The epithelium of the bladder has been traditionally referred to as transitional , but the preferred term urothelium is more informative and accurate. It is generally six to seven cells thick in the contracted bladder, but only two or three cells thick in the distended bladder. It has three layers: superficial, intermediate, and basal. The superficial layer is made up of a single row of large cells having abundant eosinophilic cytoplasm and referred to as umbrella cells. The umbrella cells may also have large multilobated nuclei and prominent cytoplasmic vacuolization. The intermediate cells have a cuboidal to low columnar shape, oval nuclei with fine chromatin and frequent nuclear grooves, moderately abundant cytoplasm, and well-defined margins. The basal layer is made up of a row of cuboidal cells that lie on a thin continuous basal lamina.
The lamina propria is composed of loose connective tissue containing a rich vascular network, lymph vessels, and a few elastic fibers. A rather prominent collection of arteries and veins is present in the midportion of the lamina propria and divides this layer into an inner and an outer zone. Smooth muscle cells are also present in the lamina propria, usually as isolated bundles, sometimes as a discontinuous thin layer, and rarely as a continuous layer. This layer of smooth muscle is designated as MM. These can be confused with the muscle bundles of the MP when evaluating the depth of invasion of a bladder neoplasm (particularly in a biopsy specimen), a serious problem since tumor staging and treatment are largely based on the presence or absence of MP invasion. Isolated bundles of muscle immediately adjacent to urothelium, with loose haphazard fiber orientation and irregular outlines, favor MM. Topographic and morphologic variations exist that complicate this evaluation: (1) the more superficial location of the MP and the inconspicuousness of the MM in the trigone; (2) the occurrence of hyperplastic MM , defined as MM composed of more than three layers of muscle fibers appearing as fascicles parallel to the surface epithelium or as rounded bundles, this being particularly prominent in the dome ; (3) the presence of superficially located MM at the insertion of the ureter into the bladder; and (4) an indistinct border between the MP and the lamina propria. In general, if individual muscle bundles, either as individual small fascicles or separate distinct rounded bundles, are separated by intervening stroma, then MM should be carefully considered. Immunostaining for smoothelin (a novel smooth muscle-specific marker expressed only in fully differentiated smooth muscle cells) has been touted as having utility in this distinction, in the sense that a strong and diffuse reaction is characteristic of MP, in contrast with MM, in which the staining is absent or weak and focal. Not surprisingly, some degree of overlap exists in the degree of reactivity for smoothelin (not to mention significant variability between titers), which calls for extreme caution in its interpretation. Because of these issues, we do not use smoothelin for staging.
The MP is vaguely divided into inner, central circular, and outer longitudinal layers; these are best individualized in the bladder neck region.
Islands of mature adipose tissue are commonly present throughout the wall of the urinary bladder, particularly in the deep portion of the lamina propria; therefore, the presence of carcinoma in adipose tissue is not sufficient for designation as extravesical invasion.
Small paraganglia may be found in the adjacent connective tissue, usually in association with neural structures.
The urachus is a 5–6-cm vestigial structure located between the dome of the bladder and the umbilicus that results from the involution of the allantoic duct and the cloaca. During development, it connects the bladder with the allantois. At birth the urachus retracts from the bladder, and when regressed into a fibrous cord, it is called the median umbilical ligament. Its lumen may persist within the bladder wall and be continuous with the bladder cavity. The lining may be of transitional or columnar type. Schubert et al. found tubular urachal remnants in 32% of 122 bladders studied at autopsy.
Anomalies related to urachal remnants most often occur in children, but they can also first present in adults. They include the following: patent urachus through which urine may pass, blind sinuses in the anterior abdominal wall, abscesses , granulomatous omphalitis , and intravesical polypoid multicystic masses ( “hamartomas” ).
Tumors may also develop from this structure, of which adenocarcinoma is the most common; the majority of these cases are well-differentiated, mucin-producing tumors, but enteric type and signet ring carcinomas also occur ( Fig. 25.1 ). Other tumor types include villous adenoma , transitional cell (urothelial) carcinoma , small cell carcinoma , and squamous cell carcinoma . Most of these tumors arise from the intramural portion of the urachus and grow into the wall of the bladder, sometimes in the absence of mucosal involvement. Others occur beneath the peritoneum of the anterior abdominal wall between the umbilicus and the bladder dome. The overall prognosis is poor, the main reason being that the location of the tumor does not result in early symptoms and precludes a timely diagnosis. Depending on the extent of the disease, the treatment can be either umbilectomy with partial cystectomy or en bloc radical cystoprostatectomy/umbilectomy. Because of the unique anatomic considerations, a specific urachal staging system is often utilized.
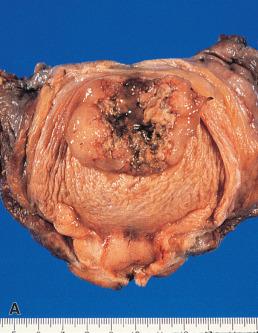
Bladder (cloacal) exstrophy is a congenital abnormality characterized by absence of the anterior vesicle and lower abdominal wall, with eversion of the posterior bladder wall. These changes may be partial or complete and are often associated with other anomalies of the urogenital tract. Malignant change was found in 3 (7.5%) of 42 patients with exstrophic bladder reported by Engel and Wilkinson. It has been calculated that the risk of neoplasia in adults born with exstrophy is 17.5% and that the highest risk (38%) is for those patients who have been exposed to mixing of urine and feces in a colorectal reservoir. The tumor type in most series has usually been adenocarcinoma, occasionally admixed with squamous elements. In addition, the occurrence of a unique benign “bladder exstrophy polyp” has been proposed.
The treatment of bladder exstrophy depends on the specific features of the malformation and may consist of reapproximating or closing the defect, in addition to repairing the other abnormalities that often accompany this condition.
Most diverticula of the bladder are acquired and develop because of partial urinary obstruction in the urethra or bladder neck, usually as a result of prostatic nodular hyperplasia. The longstanding increased muscular contractions required to empty the bladder cause thickening of the wall and mucosal herniation in areas of weakness. Other diverticula are thought to be congenital.
Bladder diverticula are most commonly located in the posterior wall above the trigone, in the region of the ureteral orifices, or in the dome at the site of an obliterated urachus. The communication into the bladder is usually large but may be pinpoint in size. The wall of the diverticulum usually consists of fibrous tissue with little or no muscle. Squamous metaplasia of the lining epithelium often occurs if there is associated inflammation, and associated nephrogenic adenomas are not uncommon.
Complications of bladder diverticula include lithiasis, free perforation into the peritoneal cavity, and tumor development. The tumors are usually urothelial carcinomas, but there seems to be a higher incidence of other tumor types. They may grow to a large size before detection because of their hidden location. The development of neoplasia may be related to the obstruction, chronic inflammation, epithelial hyperplasia, and squamous metaplasia to which these diverticula are prone. Staging is difficult with invasive carcinomas in diverticula because of the absence of MP, but most large tumors are regarded as pT3.
Bladder calculi occur much more often in male than in female individuals. Most of the patients are elderly, but children are at high risk in endemic areas, such as Turkey and the Far East. They represent a frequent complication in paraplegics and quadriplegics. The majority of these stones are solitary and composed of phosphate salts; others are made up of urate and oxalate salts. The most common associated abnormality is nodular hyperplasia of the prostate gland, likely secondary to urinary stasis.
Treatment modalities include removal by cystostomy, mechanical (endoscopic) cystolithotripsy, and extracorporeal shock wave therapy. The latter has been found to be a simple, effective, and safe modality for the treatment of most cases and is becoming the therapy of choice, particularly in adult patients. Recurrence develops in about 10% of the patients.
Endometriosis may involve the bladder as an isolated focus or in association with similar lesions in other sites ( Fig. 25.2 ). In most instances, there is a history of a previous operation in the area or of symptoms related to the female genital tract. The disease is also rarely reported in men following estrogen therapy for prostatic carcinoma. In the most florid cases, a mass can be palpated at the base of the bladder. Serosal foci are the most common and are usually asymptomatic. Foci occurring in the bladder wall, beneath an intact mucosa, result in a bluish cast on cystoscopic examination and may be accompanied by marked hyperplasia of the vesical muscle in a fashion analogous to that seen in uterine adenomyosis ; these can present with persistent hematuria during menstruation. Radiographically, the lesion may be indistinguishable from an intravesical neoplasm.
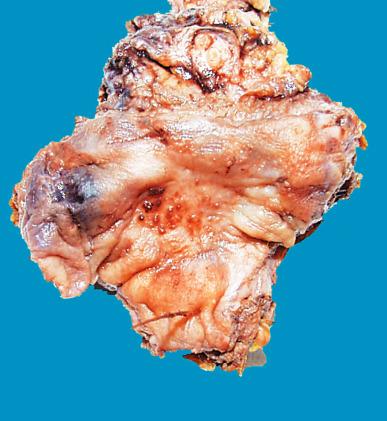
In addition to classic endometriosis composed of endometrial glands and stroma, one can encounter tubal-type epithelium (including ciliated cells, peg cells, and intercalated cells) and endocervical-type epithelium (columnar mucinous cells). The former change is referred to as endosalpingiosis and the latter as endocervicosis . When they occur in combination, the picturesque term müllerianosis is sometimes used. Florid cases of endocervicosis may simulate adenocarcinoma. True malignant tumors can develop in this setting, including endometrioid adenocarcinoma, clear cell carcinoma, endometrioid adenosarcoma, and endometrial stromal sarcoma.
The treatment of endometriosis of the bladder includes hormonal manipulation and resection; depending on the location, the latter may be carried out laparoscopically.
Amyloidosis of the bladder may be the expression of a generalized process or (more commonly) present as a nodular localized mass (“amyloid tumor”). The latter are often misinterpreted as neoplasms, both clinically and cystoscopically. The histologic appearance is diagnostic. The amyloid material is made up in most instances of AL protein (immunoglobulin [Ig] light chain), but some cases have been found to be transthyretin related. Most reported patients with adequate follow-up have remained free of disease following local excision of the mass; therefore, the presence of an amyloid tumor should not be necessarily regarded as a manifestation of myeloma or plasmacytoma.
The classic clinical description of interstitial (Hunner) cystitis (or painful bladder syndrome) is that of an adult or elderly female with ulceration and marked submucosal edema of the bladder resulting in prominent lower abdominal, suprapubic, or perineal pain and urinary frequency, unresponsive to medical therapy. Because of the consistent presence of ulceration, the disease is also known as Hunner ulcer .
The lesion can be located anywhere in the bladder. Microscopically, there is mucosal ulceration covered by fibrin and necrotic material. The underlying lamina propria and muscularis show edema, hemorrhage, granulation tissue, and a mononuclear inflammatory infiltrate that sometimes exhibits a predominantly perineural location. Mast cells are usually present and sometimes numerous; they are found beneath the ulcer, within the detrusor muscle bundles, and between the epithelial cells in the adjacent mucosa, but CD117 immunostains have not proven helpful in establishing a more definitive diagnosis. None of these microscopic features is pathognomonic. Cases lacking ulceration are considered by some authors to represent a different condition affecting a younger patient population.
The etiology of this disorder remains obscure. The search for infectious organisms has been unrewarding. Hyperexpression of human leukocyte antigen (HLA) class I molecules, strong urothelial staining for IgA, and increased sympathetic outflow into the bladder suggest the participation of autoimmune mechanisms, whereas the finding of a reduction in the expression of heat shock protein 60 suggests a defect in the proliferative response of the urothelium. The treatment is usually medical, but surgical intervention (including supratrigonal or total cystectomy) may be necessary in severe cases.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here