Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Bone and cartilage abnormalities of the facial skeleton have a tremendous impact on facial aesthetics and may cause significant psychosocial burden to the patient. Anomalies of facial form may arise from acquired loss, congenital anomalies present at birth, or developmental anomalies that become evident with growth.
There are various grafting materials that can be used in skeletal facial surgery, including autologous tissues, allogeneic grafts, xenografts, or alloplastic implants. Autologous grafts will be the focus of this chapter. Autologous grafts remain the ideal option due to their limited immunogenicity, disease transmission, and risk of foreign body reaction and, thereby, higher success rates. The main disadvantages are the limited supply of autologous tissue, donor site morbidity, and variable resorption. Autologous grafts are useful tools in maxillofacial surgery. A thorough understanding of the biology of grafts will minimize complications and optimize results.
Although bone and cartilage both originate from a mesenchymal stem cell, their differentiation, proliferation, environmental niche, and reparative function differ significantly and will be discussed separately.
Bone, unlike other connective tissues, is a physiologically mineralized tissue. Bone is composed of three cell types, osteoblasts, osteocytes, and osteoclasts, as well as extracellular matrix (ECM). The ECM is composed of:
Inorganic matrix, hydroxyapatite (50–70%)—the function of which is to strengthen the collagen composite, provide mechanical support, and serve as a source of calcium, phosphate, and magnesium.
Organic matrix (20–40%)
Type I collagen—this predominant form is a triple-helical molecule composed of two α 1 (I) chains and one α 2 (I) chain; there are trace amounts of type III, V, and fibril-associated collagens.
Noncollagen proteins (NCPs)—coordinate cell–matrix or mineral–matrix interactions and regulate the mineralization process via osteoblast and osteoclast metabolism. There are four categories of NCPs: (1) proteoglycans, (2) glycosylated proteins, (3) glycosylated proteins with cell attachment activities, and (4) γ-carboxylated (gla) proteins.
Water (5–10%)
Lipids (<3%)
Cells—osteoblasts, osteoclasts, osteocytes
Inorganic matrix—hydroxyapatite
Organic matrix
Type I collagen
Noncollagen proteins
Water
Lipids
The cell lineages that contribute to skeletal development include osteoblasts and osteoclasts. Terminally differentiated osteocytes also reside in bone, although they are thought to have limited regenerative capacity. Through complex cellular interactions and signaling molecules, these cells are responsible for the formation, growth, remodeling, and resorption of bone. Through the presence of mesenchymal stem cells, bones have the capacity for complete renewal, structurally and functionally. The mesenchymal stem cells allow for pluripotency, or the ability to differentiate into more than one cellular precursor ( Fig. 11.1 ). Those progenitors with upregulated Sox9 and Runx2 can differentiate into chondrocytes and osteoblasts, respectively, for endochondral or intramembranous bone formation.
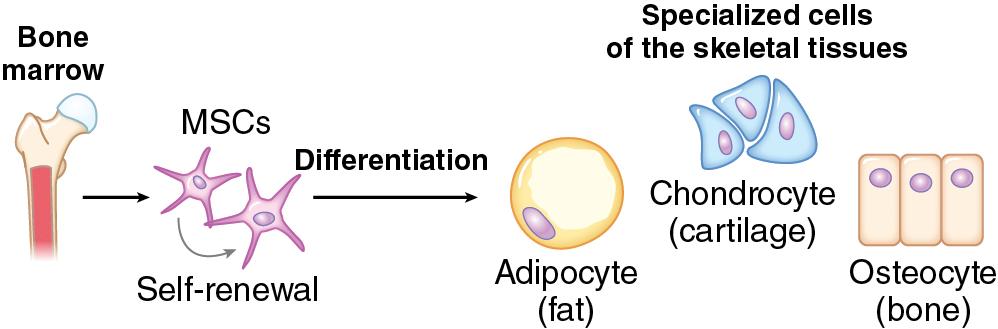
Osteoblasts produce bone and can be further separated into cellular subsets based on their various genetic expressions. In early osteoblast maturation, type I collagen is upregulated, and this is later followed by expression of alkaline phosphatase and matrix gla protein. In the mineralization phase of osteoblasts, there is notable expression of osteopontin, osteocalcin, and collagenase. The bony ECM synthesis is partially regulated by C-type natriuretic peptide and the mitogen-activated protein kinase (MAPK) pathway of fibroblast growth factor signaling, whereas the NCPs (noted previously) are thought to play a central role in coordinating the cell–matrix interface, which can regulate osteoblast function. This complex network of cells and ECM works together to orchestrate bone formation.
Osteoclasts are responsible for resorption of the mineralized matrix, functioning to achieve optimal patterning and creating space for new bone formation during remodeling. Osteoclasts are unique because they are derived from the hematopoietic cell lineages, which are found in bone marrow, spleen, and peripheral blood. Osteoclasts have a common differentiation pathway with macrophages and have thus evolved distinct features, such as their multinucleated phenotype. Transcription factors, such as c-Fos, nuclear factor–kappa B (NF-κB), and microphthalmia-associated transcription factor, are necessary for the maturation of osteoclasts. In their functional state, osteoclasts secrete acids, collagenases, and proteases to resorb the mineralized matrix (osteoid) and associated proteins.
A critical balance between these two cell types is necessary to maintain bone. Receptors that mediate osteoclast formation are present on osteoblastic cells, indicating the importance of mesenchymal cell interaction in the recruitment of osteoclast progenitors. Receptor-activator of the NF-κB ligand (RANKL), a member of the tumor necrosis factor (TNF) superfamily, is located on osteoblasts and mesenchymal cells. Working together with its receptor RANK, found on osteoclast precursors, this interaction is essential for osteoclast activation and bone resorption.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here