Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Bacterial infections of the central nervous system (CNS) in the newborn are common and are of major clinical importance. By far the most frequent of these infections is neonatal bacterial meningitis, and this chapter deals with this disorder in detail. Other bacterial processes include primary intracranial infections (e.g., epidural and subdural empyema and brain abscess) and disorders in which involvement of the CNS is secondary to extraneural infection (e.g., subacute bacterial endocarditis with focal embolic encephalitis, neonatal botulism, and neonatal tetanus). Because most of these bacterial diseases are exceedingly rare, and one (botulism) is discussed in Chapter 36 , only brain abscess and neonatal tetanus will be discussed in this chapter. Systemic candidiasis, a disseminated fungal infection, also is discussed briefly (rather than in Chapter 38 ) because the manifestations thereof, which include meningitis and brain abscess, are related more closely to the subject matter of this chapter.
Bacterial meningitis is the most common and serious variety of neonatal intracranial bacterial infection. In most cases, bacterial meningitis is associated with recognizable bacteremia (i.e., sepsis). The disorder is usually fulminating in evolution but is amenable to therapeutic intervention. Indeed, prompt recognition and appropriate therapy of bacterial meningitis are major challenges in the care of the neonatal patient.
Certain basic and common themes recur in any discussion of neonatal bacterial sepsis and meningitis. Perhaps most prominent of these themes is the dual pattern of illness observed with the major etiological agents for neonatal bacterial meningitis (see following discussion). Thus early-onset and late-onset disease often can be readily distinguished ( Table 39.1 ). In early-onset disease (i.e., onset within 72 hours of life), infection appears to be derived near the time of delivery from an infected birth canal. Understandably, obstetrical complications are common, and infants are often of low birth weight. Multisystemic manifestations are prominent. In late-onset disease (i.e., onset usually after the first few days of life), the infection may still have been acquired from the mother but frequently is acquired from contacts with infected medical personnel, other infants, contaminated equipment, and vascular access devices or other materials. A distinction is made between hospital- and community-acquired late-onset sepsis. Preterm infants are especially at risk to develop late-onset disease in the hospital, whereas community-acquired late-onset disease is mainly reported in late preterm infants and term infants. Meningitis is often the dominant manifestation of late-onset disease.
| MAJOR FEATURE | EARLY-ONSET DISEASE | LATE-ONSET DISEASE |
|---|---|---|
| Usual age of onset | First 72 hours | >7 days |
| Obstetrical complications | Common | Uncommon |
| Dominant clinical signs referable to sepsis and respiratory disease | Common | Uncommon |
| Dominant clinical signs referable to meningitis | Uncommon | Common |
| Mode of transmission | Mother to infant | Mother to infant |
| Human contacts, equipment, vascular access devices, and so forth | ||
| Specific serotype | Uncommon | Common |
The major bacterial causes of early- versus late-onset sepsis are shown in Table 39.2 . Group B Streptococcus (GBS) is the most prominent early-onset pathogen, whereas coagulase-negative Staphylococcus (CONS) (e.g., Staphylococcus epidermidis ) becomes very prominent in late-onset sepsis, especially sepsis after very long hospital stays (>30 days). In a recent surveillance study of infants with early-onset neonatal sepsis and born from April 2015 to March 2017, Escherichia coli (36.6%) and group B Streptococcus (30.2%) were the two most common organisms, with E. coli especially common in the preterm infant with an increasing incidence over the study period. Concerning CONS and late-onset sepsis, these organisms are part of the normal skin and mucosal flora and induce infection in the presence of prolonged indwelling vascular catheters and related features of neonatal intensive care, especially of very premature infants. Pseudomonas and related gram-negative organisms that contaminate moist ventilatory equipment also become prominent in late-onset sepsis. E. coli and Klebsiella species account for 20% to 25% of both early-onset and late-onset cases. Although the causes of neonatal meningitis reflect the causes of neonatal sepsis, the relative proportions for each organism differ somewhat because of the varying propensity to invade the CNS (see later).
| PERCENTAGE OF TOTAL | ||
|---|---|---|
| ORGANISM | EARLY-ONSET SEPSIS | LATE-ONSET SEPSIS |
| Group B beta-hemolytic streptococci | 50 | 5 |
| Group D streptococci | 5 | 10 |
| Coagulase-negative staphylococci | 5 | 40 b |
| Staphylococcus aureus | 5 | 10 |
| Escherichia coli, Klebsiella species | 25 | 20 |
| Pseudomonas, Serratia | 0 | 10 |
| Miscellaneous | 10 | 5 |
a See text for references. Most data are from references , , and .
b More common in cases with onset after 30 days, especially in low-birth weight infants.
In the ensuing discussion, the etiology, pathogenesis, neuropathology, clinical features, diagnostic aspects, prognosis, and management of neonatal bacterial meningitis are reviewed. Particular emphasis is placed on aspects most relevant to neonatal neurology. Where relevant, the discussion centers on specific aspects related to a particular organism, but, in general, the features of neonatal bacterial meningitis caused by most organisms exhibit more similarities than differences.
The major organisms associated with neonatal meningitis and their relative frequency as current etiological agents are shown in Table 39.3 . The gestational age should be taken into account because E. coli disease primarily occurs in preterm infants, and group B Streptococcus disease in full-term infants. Although relative frequencies vary somewhat from one medical center to another and over time, the data are at least representative of cases largely in medical centers in North America, Australia, New Zealand, and Switzerland. Group B Streptococcus is the single most commonly encountered organism, with E. coli second. Listeria monocytogenes is the third most common organism and accounts for approximately 2% to 3% of cases in most series in North America. The remaining cases are accounted for principally by other streptococcal and staphylococcal species, by other gram-negative enteric bacilli, and by a variety of unusual organisms. Particular note should be made of the role of Staphylococcus aureus and S. epidermidis (CONS) in the presence of indwelling catheters, particularly in association with neurosurgical procedures, and the role of Proteus species, Pseudomonas aeruginosa, Serratia marcescens, Bacillus cereus , and Flavobacterium meningosepticum in the presence of respiratory devices using moist inhalation. An occasional newborn infant develops meningitis secondary to Haemophilus influenzae, Salmonella, Pasteurella multocida, Vibrio cholerae, Mycoplasma hominis , or several other less commonly known organisms.
| BACTERIAL ETIOLOGY | PERCENTAGE OF TOTAL |
|---|---|
| Group B Streptococcus | 30–38 |
| Other streptococci and staphylococci (including especially group D streptococci, Streptococcus pneumoniae, Staphylococcus epidermidis , b and Staphylococcus aureus ) | 19–23 |
| Escherichia coli | 23–35 |
| Other gram-negative enteric bacteria (including Pseudomonas aeruginosa, Klebsiella and Enterobacter species, Proteus species, Citrobacter species, and Serratia marcescens ) | 10–27 |
| Listeria monocytogenes | 2–3 |
| Other (including Haemophilus influenzae, Salmonella species, and Flavobacterium meningosepticum ) | 9–14 |
a Data principally from Giannoni E, Agyeman PKA, Stocker M, et al. Neonatal sepsis of early onset, and hospital-acquired and community-acquired late onset: a prospective population-based cohort study. J Pediatr 2018;201:106–114.e4; Stoll BJ, Puopolo KM, Hansen NI, et al. Early-onset neonatal sepsis 2015 to 2017, the rise of Escherichia coli , and the need for novel prevention strategies. JAMA Pediatr 2020;174(7):e200593; Singh T, Barnes EH, Isaacs D. Early-onset neonatal infections in Australia and New Zealand, 2002–2012. Arch Dis Child Fetal Neonatal Ed 2019;104(3):F248–f252.
b Coagulase-negative staphylococci are particularly common etiologies in very-low-birth weight infants with late-onset meningitis.
Meningitis is more common in premature infants than in full-term infants and in the first months of life than in succeeding months. Because the final common pathway in the pathogenetic sequence is almost always bacteremia leading to meningitis, a close association exists between the causes and rates of neonatal sepsis and neonatal meningitis. The association is not perfect, however, because certain organisms are particularly efficient at invading the CNS (see later). Early-onset sepsis occurs at a rate of approximately 0.5 to 1 per 1000 live births, and meningitis occurs at a rate of approximately 0.3 per 1000 live births. Thus, neonatal meningitis is an enormously important problem, and understanding its pathogenesis is critical for attempts to reduce the enormity of the problem in the future.
Most cases of neonatal sepsis and meningitis, especially those of early onset, are related to acquisition of bacteria during labor or delivery, after rupture of membranes. Acquisition is principally by parturitional exposure or, less commonly, ascending infection (e.g., after prolonged rupture of membranes). Thus it is not surprising that important factors predisposing to sepsis and meningitis include a variety of complications of labor and delivery (especially fetal distress, obstetrical trauma, and placental abnormalities), maternal peripartum infection (especially of the genital or urinary tracts), prolonged rupture of membranes (>24 hours), and chorioamnionitis ( Box 39.1 ).
Complications of labor and delivery
Maternal peripartum infection, especially of the genital or urinary tract
Prolonged rupture of membranes
Chorioamnionitis
Maternal urinary tract infection associated with neonatal sepsis and meningitis is usually gram negative, and maternal genital infection associated with these neonatal disorders is usually with group B Streptococcus . Indeed, genital infection secondary to group B Streptococcus (usually asymptomatic) is identifiable at delivery in approximately 15% to 35% of women. Approximately 40% to 70% of infants delivered to such women become colonized at one or more mucocutaneous sites (e.g., ear, throat, rectum, and umbilicus). Approximately 2% to 4% of such colonized infants exhibit invasive, early-onset group B streptococcal disease. The incidence is particularly high (8%) in those infants heavily colonized (i.e., found to have three to four positive mucocutaneous sites). Following the introduction of prevention guidelines in New Zealand, a decrease in the incidence of invasive early-onset GBS disease from 0.5 in the late 1990s to 0.23 per 1000 live births in 2009 to 2011 was shown. However, in a more recent study from Australia and New Zealand, group B Streptococcus was the most prevalent organism for early-onset sepsis with an incidence rate of 0.43/1000 live births, with no evidence of change over time ( P = .3) between 2002 and 2012. A decrease by more than 80% from 1.8 cases/1000 live births in the early 1990s to 0.26 cases/1000 live births in 2010 was shown in the United States. However, there was no effect of the guidelines on the incidence of late-onset disease. A recent study from the United States evaluated data between 2006 and 2015 and reported that the incidence of early-onset disease declined significantly from 0.37 to 0.23 per 1000 live births ( P < .001), whereas late-onset disease rates remained stable (mean, 0.31 per 1000 live births). Interestingly late-onset disease had become more common than early-onset disease. A higher incidence of invasive group B streptococcal infection (0.62 per 1000 live births) was recently reported in Norway. The incidence of early-onset group B Streptococcus disease was 0.41 (0.37–0.45), and the incidence of late-onset disease was 0.20 (0.17–0.23). The annual incidence of late-onset disease increased slightly from 1996 to 2012.
Factors related to pregnancy and delivery are also particularly important in the pathogenesis of early-onset neonatal listeriosis. Thus various obstetrical complications are present in the history of approximately half of such patients, and in most cases maternal isolates of serotypes identical to those infecting the infants have been found. In the majority of cases of early-onset neonatal listeriosis, a maternal history of flulike syndrome, unexplained fever, or urinary tract symptoms in the several days to weeks before delivery can be elicited. In keeping with probable fecal-oral transmission of this organism, epidemics of maternal infection during pregnancy and subsequent neonatal listeriosis have been associated with consumption of contaminated foods. Indeed, unlike the other varieties of neonatal meningitis, listeriosis probably occurs commonly by transplacental passage, as evidenced by the finding of well-developed placentitis with recognizable organisms in carefully studied cases.
Significant host factors operative in the newborn period relate particularly to defense mechanisms versus bacterial infection. In early life, immunity is characterized by age-dependent adaptations of both innate and adaptive immune responses. The first-line host defense is formed by innate immune cells recognizing pathogens by unique molecular patterns with pathogen recognition receptors. Innate immune responses include opsonization, complement activation, secretion of inflammatory mediators, and induction of apoptosis. The adaptive immune system depends on antigen recognition and comprises particularly the function of T cells and B cells.
Age-dependent adaptations of innate immunity include the following: impaired leukocyte chemotaxis, phagocytosis, and bactericidal activity; defective neutrophilic metabolic responses after phagocytosis (e.g., activation of hexose monophosphate shunt and, especially, oxidative metabolism, with diminished generation of the critical bactericidal hydroxyl radical); impaired costimulatory capacity of antigen presenting cells (dendritic cell populations and monocytes) and decreased production of proinflammatory cytokines upon Toll-like receptor (TLR) stimulation; lower cytotoxic capacity of neonatal NK cells and γδ–T cells than in adults; and diminished concentrations of several complement components. Moreover, lower fibronectin concentrations in the newborn appear to contribute to diminished opsonization and phagocytosis. These adaptations are accentuated and especially affect immune function in premature infants and in infants subjected to illnesses of diverse types.
Studies of T cells in cord blood demonstrate qualitative and quantitative differences in immune responses compared with adult T cells, and these differences are diminished only gradually during infancy. T cells generally express a naïve phenotype but differentiate easily into regulatory T cells. Additionally, T-cell lineage differentiation begins in utero and is highly controlled by epigenetic regulation, which is, interestingly, influenced by environmental factors.
Neonatal B cells have a naïve phenotype and have only a partially developed surface immunoglobulin repertoire. Deficiencies of specific humoral immunity have involved the immunoglobulin M (IgM) and immunoglobulin G (IgG) classes of antibodies. The latter are transferred passively across the placenta, particularly in the third trimester. Thus some premature infants may have decreased levels of IgG antibodies. IgM and immunoglobulin A (IgA) are not transferred passively across the placenta and are in very low concentrations in the normal newborn. IgM antibodies include several antibody types important in the defense against gram-negative bacteria, and deficiency of these antibodies in the newborn infant may play a role in the susceptibility to gram-negative bacteria. IgA antibodies are abundant at later ages at mucosal surfaces (e.g., the respiratory and gastrointestinal tracts), and their deficiency in the newborn may explain in part the relative ease with which mucosal barriers are colonized and penetrated. Newborn infants have diminished production of these antibodies because of both immaturity of the antibody-producing B cells and plasma cells and diminished T-cell help for antibody production. The deficiency in immune globulins led to clinical trials of the use of intravenous immune globulin in prophylaxis and treatment of bacterial infection in newborns, especially premature newborns. This approach has not proven useful for general practice.
Neonates are heavily dependent on innate immune responses for protection against infections. The brain microglial cell, which is derived from monocytes (see Chapter 16 ), is an important part of this defense. Activation of innate immune cells occurs by way of specific cell surface receptors (i.e., Toll-like receptors [TLRs]), which respond to specific molecular motifs shared by the products of large classes of microorganisms. For example, TLR4 is the receptor that recognizes gram-negative bacterial lipopolysaccharide, and TLR2 is the receptor for gram-positive peptidoglycan and lipopeptides. Activation of these receptors triggers an inflammatory response by a mechanism that operates through nuclear factor kappa B and mitogen-activated protein kinase. A study of newborns showed that the basal expression of TLR2 (but not TLR4) is slightly lower in neonatal than in adult phagocytes. In infants with sepsis, TLR2 is sharply upregulated, unlike TLR4. A similar disturbance of TLR4 responsiveness was shown in neonatal monocytes as a function of gestational age. Thus the deficits in TLR2 and TLR4 may be relevant to the importance of such gram-positive organisms as group B Streptococcus and CONS and of such gram-negative organisms as E. coli in neonatal sepsis and meningitis. Furthermore, downstream signaling through the MyD88 and p38 pathways is impaired in neonates following TLR2 and TLR4 stimulation. High levels of adenosine in neonatal blood may increase cyclic adenosine monophosphate levels and protein kinase A–dependent or –independent inhibition of TLR-stimulated tumor necrosis factor-alpha secretion.
Immunological studies of perinatal group B streptococcal infection illustrated the potential roles of both maternal and host defense factors in pathogenesis. Thus using a sensitive, radioactive antigen-binding assay, Baker and Kasper showed that the prevalence of antibody to group B Streptococcus (capsular polysaccharide of the type III strain) in mothers with vaginal colonization was 76% for those whose infants did not develop group B streptococcal disease and only 5% for those whose infants did develop group B streptococcal sepsis or meningitis. These data suggest that this IgG antibody is important in protection of the infant and that passive transfer across the placenta did not occur because of maternal failure to synthesize the antibody. In addition, the infants with sepsis or meningitis had low levels of antibody after recovery, a finding suggesting that, in addition, these infants failed to synthesize this IgG component. Similarly, infants who develop group B streptococcal sepsis have been shown to exhibit defective humoral (opsonic activity) and neutrophilic responses to their infecting strain.
Certain factors related to the neonatal environment increase the risk of sepsis and meningitis, including the following: use of inhalation therapy equipment; use of aerosols; use of vascular, umbilical, and intraventricular indwelling catheters; and exposure to nursery personnel, parents, or other infants harboring pathogenic organisms. Operation of some of these factors may be suggested by the organism causing the sepsis or meningitis, as discussed earlier. Horizontal (or nosocomial) transmission of an organism from human contacts has been studied, particularly for group B Streptococcus . Thus nosocomial infection rates of up to 40% have been reported, particularly in medical centers in which high rates of newborns are already colonized at birth (by vertical transmission from mother) and in which high daily census rates are reported, thereby favoring cross-contamination of infants by nursery personnel. The particular importance of factors related to neonatal intensive care is illustrated by the marked increase in CONS infections with increasing time in neonatal intensive care (see Table 39.2 ). This issue is particularly important in very premature infants, in whom later-onset sepsis is related primarily to CONS and of whom nearly 80% have a central vascular catheter in place at the time of infection. In the subset of very-low-birth weight infants, CONS are important etiological agents. Indeed, in a series of 134 very-low-birth weight (<1500 g) infants with meningitis, 76% of whom weighed less than 1000 g at birth, 29% had positive cerebrospinal fluid (CSF) and blood cultures for CONS.
The propensity for specific strains of group B Streptococcus, E. coli , and L. monocytogenes to be most commonly responsible for neonatal meningitis suggests important pathogenetic roles for the microorganism itself. (Recall the earlier discussion concerning innate immunity and the risk of infection by gram-negative and gram-positive organisms.) An evaluation of serotypes for group B Streptococcus infection among 71 infants, 16 (22.5%) with early-onset disease and 55 (77.5%) with late-onset disease, revealed serotype III ( n = 42, 71.2%) to be the most common serotype among the 59 isolates available for serotyping. A genomic analysis recently performed of 1345 infants with invasive group B Streptococcus infection to understand the increasing incidence in the Netherlands showed an expansion of historical clones from two major lineages: CC17 and CC23. Serotype III of group B Streptococcus , K1 strains of E. coli , and serotype IVb of L. monocytogenes are the predominant specific types of these three bacteria that cause neonatal meningitis ( Table 39.4 ). Approximately 70% of all cases of neonatal meningitis are caused by these three bacterial types.
| CLINICAL DISEASE a | |||
|---|---|---|---|
| ORGANISM | ASYMPTOMATIC | SEPSIS | MENINGITIS |
| Group B Streptococcus serotype III | 36% | 32% | 85% |
| Escherichia coli K1 strain | 12% | 39% | 84% |
| Listeria monocytogenes serotype IVb | ? | 42% (early-onset disease) | 78% (late-onset disease) |
a Data for each clinical disorder expressed as a percentage of total cases (caused by the indicated bacterium) resulting from the specific serotype.
The likelihood that capsular polysaccharides reflect, to a considerable degree, an intrinsic virulence of these organisms is suggested by in vivo and in vitro observations. Studies with immature rats demonstrated, for K1 strains of E. coli relative to other E. coli strains, a high virulence, particularly regarding bacteremia and meningitis, that was age related. The younger the animal, the greater was the likelihood of serious disease. Moreover, investigators showed that group B Streptococcus type III and K1 strains of E. coli have distinctive capsules, which contain polysaccharide with sialic acid in high concentration (≥25% of total carbohydrate). (This distinctive polysaccharide for E. coli is termed K1; hence the name K1 strain .) A relation of the capsular polysaccharide antigen to virulence is suggested by the different outcomes of E. coli meningitis secondary to K1 and non-K1 strains ( Table 39.5 ). In addition to a marked preponderance of cases secondary to K1 strains, whereas 60% of reported infants with meningitis secondary to K1 strains died or exhibited neurological sequelae, only 11% of infants with meningitis secondary to non-K1 strains exhibited the poor outcome (see Table 39.5 ). A protein component of the outer membrane of the K1 strain of E. coli , OmpA, was also shown to be critical for the capacity of this strain to penetrate brain endothelial cells and thus to cause intracranial infection. The relation of the capsular polysaccharide antigen to virulence is supported further by the studies reviewed previously, relating the occurrence of group B streptococcal type III neonatal disease to the deficiency in the mother and in the newborn of antibody against the specific capsular polysaccharide of that organism.
| OUTCOME | |||
|---|---|---|---|
| TYPE OF ESCHERICHIA COLI | NORMAL | NEUROLOGICAL SEQUELAE | DEAD |
| Non-K1 strains | 8 (89%) | 1 (11%) | 0 |
| K1 strains | 19 (40%) | 14 (29%) | 15 (31%) |
The neuropathology of bacterial meningitis may be considered in terms of acute and chronic changes ( Box 39.2 ). Moreover, certain additional histological features are particularly characteristic of infection with specific organisms, and these features are discussed separately.
Arachnoiditis
Ventriculitis: choroid plexitis
Vasculitis
Cerebral edema
Infarction
Associated encephalopathy (cortical neuronal necrosis, periventricular leukomalacia)
Hydrocephalus
Multicystic encephalomalacia: porencephaly
Cerebral cortical and white matter atrophy
Cerebral cortical developmental (organizational) defects (?)
The acute changes of bacterial meningitis are dramatic and include arachnoiditis, ventriculitis, vasculitis, cerebral edema, infarction, and associated encephalopathy (see Box 39.2 ). Because the hallmark of the disease is arachnoiditis, this aspect is discussed first; however, the neuropathological progression of neonatal bacterial meningitis probably begins with choroid plexitis and ventriculitis ( Fig. 39.1 ). Arachnoiditis is discussed first, because it is the dominant feature of bacterial meningitis.

The hallmark of bacterial meningitis is infiltration of the arachnoid with inflammatory cells. The exudate is predominant over the base of the brain in approximately one-half of the cases and is distributed more evenly in most of the remainder ( Fig. 39.2 ). The evolution of this inflammatory response was well described by Berman and Banker.
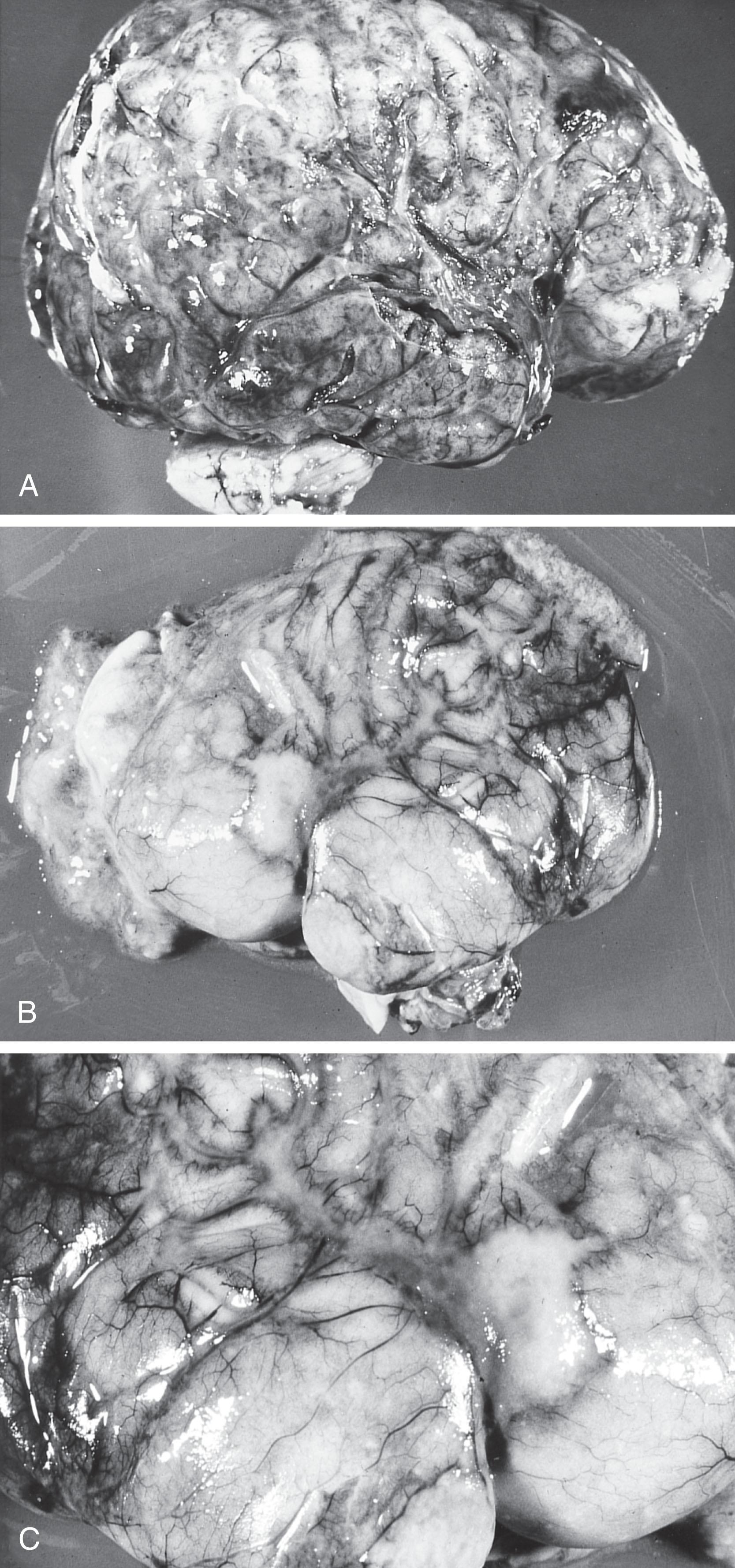
In the acute state (i.e., approximately the first week ), the predominant cells in the arachnoidal (and ventricular) exudate are polymorphonuclear leukocytes. Bacteria are visible, free, and within polymorphonuclear leukocytes and macrophages. The inflammatory exudate is particularly prominent around blood vessels and extends into the brain parenchyma along the Virchow-Robin space. In the second and third weeks of the disease, the proportion of polymorphonuclear leukocytes decreases gradually and constitutes approximately 25% of the cell population. The predominant cells are now mononuclear, mainly histiocytes and macrophages. Lymphocytes and plasma cells are present in relatively small numbers, and this paucity is a characteristic feature of neonatal meningitis. Whether this apparent deficiency of cells involved in the immunological response plays a role in the relative tenacity of neonatal bacterial meningitis remains to be determined but seems plausible. A prominent feature of this stage of the disease is infiltration of cranial nerve roots by the exudate; particular involvement occurs in the subarachnoid space of the posterior fossa and especially affects cranial nerves III through VIII. This involvement is clinically relevant (see subsequent discussion). After approximately 3 weeks , the exudate decreases in amount and consists of mononuclear cells. Thick strands of collagen become apparent as arachnoidal fibrosis begins to develop. This process is probably important in the genesis of the obstructions to CSF flow that result in hydrocephalus.
The characteristics of the arachnoiditis caused by different bacteria vary little. However, early-onset group B streptococcal meningitis is accompanied by much less arachnoidal inflammation than is late-onset meningitis. Indeed, approximately 75% of cases of early-onset meningitis (i.e., positive culture results) are said to exhibit little or no evidence of “leptomeningeal inflammation” at autopsy. It is not clear whether the relative lack of arachnoidal inflammatory response relates to rapidity of death after onset of symptoms or to immaturity of host responses to infection within the CNS of premature infants (who represent the largest proportion of fatal cases of early-onset group B streptococcal meningitis).
Ventriculitis (i.e., inflammatory exudate and bacteria in the ventricular fluid and lining) is a particularly common feature of neonatal meningitis. In the 50 brains studied by Berman and Banker and by Gilles and coworkers, overt ventriculitis was present in 44 (88%). In a collaborative study of 70 infants with meningitis caused by gram-negative bacteria in whom ventricular tap was performed at the time of diagnosis, 51 had ventriculitis (73%), as manifested by positive cultures with pleocytosis in the ventricular fluid (comparable data are not available for group B streptococcal meningitis). Although precise, controlled data are not available, ventriculitis appears to be more common in neonatal meningitis than in meningitis at later ages.
The ventriculitis is initially characterized by exudate most prominent in the choroid plexus stroma and just external to the plexus . Exudative excrescences from the ventricular surface can be visualized in vivo by cranial ultrasonography (see later discussion). In the second and third weeks of the disease course, the ventricular exudate is associated with active ependymitis, characterized by disruption of the ependymal lining and projections of glial tufts into the ventricular lumen. Later, glial bridges may develop and cause obstruction, particularly at the aqueduct of Sylvius ( Fig. 39.3 ). Less commonly, septations in the lateral ventricle may produce a multiloculated state that is similar to abscess formation. The multiple ventricular obstructions, in fact, may “isolate” portions of the lateral ventricles or the fourth ventricle, cause disproportionate and severe dilation of the affected ventricle, and present a difficult therapeutic problem. Ventriculitis with obstructive hydrocephalus may manifest subacutely in newborns (with no history of acute meningitis) after several weeks, especially ventriculitis caused by group B streptococcal and E. coli meningitis.
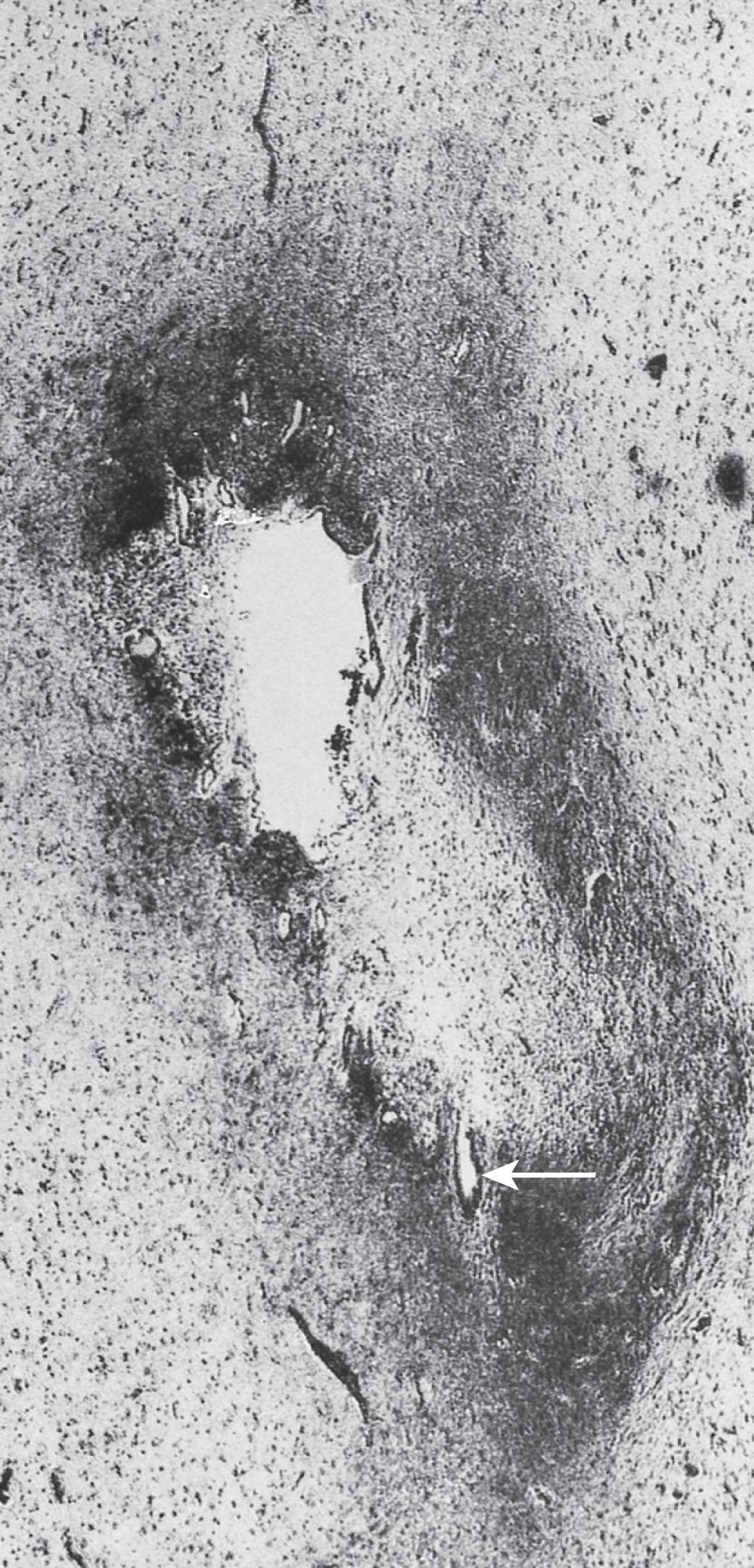
The nearly uniform occurrence of ventriculitis in acute bacterial meningitis in the newborn has pathogenetic and therapeutic implications . Regarding pathogenesis , it appears reasonable to suggest that the initial sequence of events in bacterial invasion of the CNS is for hematogenously borne bacteria to localize first in the choroid plexus and to cause choroid plexitis, with subsequent entrance of bacteria into the ventricular system and later movement to the arachnoid through normal CSF flow (see Fig. 39.1 ). Gilles and coworkers presented considerable data to support this view, including the high glycogen content of the neonatal choroid plexus, which provides an excellent medium for bacteria. Moreover, an age-related effect is suggested by the postnatal decrease in glycogen content of plexus epithelial cells.
Experimental studies of bacterial meningitis produced in infant rhesus monkeys indicated that infection of the lateral ventricle is a uniform feature. Moreover, when discordance between the amount of bacteria in ventricular and subarachnoid CSF samples was observed in these studies of rhesus monkeys, ventricular bacterial densities were greater. These observations further support the notion that the initial events in the genesis of bacterial meningitis are bacteremia and infection of the choroid plexus and lateral ventricles.
The therapeutic implications of these data concerning ventriculitis are important and are discussed later in “Management.” Suffice it to say here that the ventricle may be a major reservoir of bacterial infection, inaccessible either to systemic antibiotics, which are unable to penetrate the purulent covering over the ventricular lining, or to intraventricular antibiotics, which are unable to reach the choroid plexus epithelium (and particularly inaccessible to intrathecal antibiotics, which are unable to reach the ventricles because of normal CSF flow). Moreover, if obstruction of the ventricular system is added (e.g., at the aqueduct), a closed infection, approximating an extraparenchymal abscess, will result.
Vasculitis is an almost invariable feature of neonatal bacterial meningitis. The involvement of both arteries and veins can be considered an extension of the inflammatory reaction in the arachnoid and within the ventricles. The arteritis is manifested particularly by inflammatory cells in the adventitia, although involvement of the intima is not uncommon. Although the arterial lumen may be narrowed, only rarely is complete occlusion observed. Involvement of veins is severe and includes arachnoidal, cortical, and subependymal veins. In contrast to arterial involvement, phlebitis is frequently complicated by thrombosis and complete occlusion. Multiple fibrin thrombi of adjacent veins are often observed in association with areas of hemorrhagic infarction (see “Infarction,” later). The vasculitic changes are apparent in the first days of meningitis and become particularly prominent by the second and third weeks.
More similarities than differences exist among various bacteria with regard to the nature and severity of the vascular changes with meningitis. However, vasculitis with thrombosis, sometimes resulting in stroke in the distribution of perforating arteries, or even hemorrhage is relatively frequent in early-onset group B streptococcal disease, despite the relatively modest arachnoidal inflammation.
Cerebral edema is a characteristic of the acute stage of neonatal bacterial meningitis ( Fig. 39.4 ). Indeed, the swelling of brain parenchyma is often so severe that the ventricles are reduced to small slits. The difficulty and hazard of performing ventricular puncture in the acute stage are significant because of this phenomenon. The cause of the edema is related primarily to the vasculitis and increased permeability of blood vessels (i.e., vasogenic edema; see Fig. 39.1 ). This vasogenic component may be complicated by a cytotoxic component, when parenchymal injury occurs (e.g., through infarction), or when impairment of CSF flow results in development of interstitial edema (see later discussion).
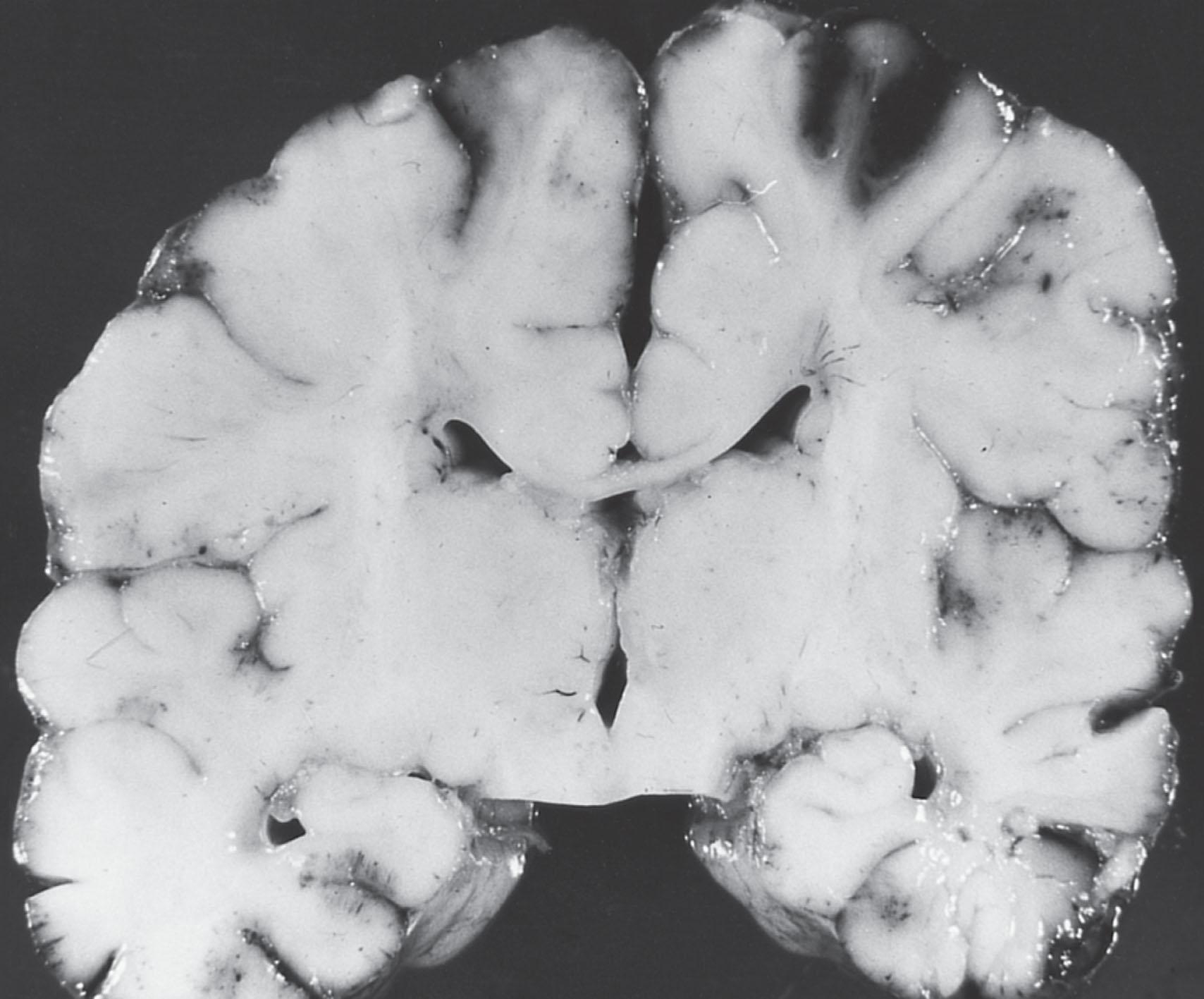
Despite the edema, another feature of neonatal bacterial meningitis that is distinctive relative to disease at later ages is the rarity of evidence, clinical or pathological, for herniation of supratentorial structures through the tentorial notch or of cerebellar tonsils into the foramen magnum. This feature may relate to the distensibility of the neonatal cranium, especially because of the separable sutures. This factor also may prevent marked increases in intracranial pressure and therefore impaired cerebral perfusion; studies of intracranial pressure and cerebral blood flow velocity in neonatal bacterial meningitis supported this suggestion (see later discussion). Nevertheless, lethal uncal and cerebellar tonsillar herniation has been documented in neonatal bacterial meningitis. Moreover, the rare phenomenon of anterior fontanelle herniation has been reported to complicate neonatal meningitis.
Infarction may be a prominent and serious feature of neonatal bacterial meningitis (see Fig. 39.5 ). Studies of autopsy cases indicate an incidence of 30% to 50% ( Box 39.3 ). More than one-half of the cases in Friede’s neuropathological series sustained their lesions in the first week after the diagnosis of meningitis. Thus although the vascular lesions become particularly prominent in the second and third weeks, infarction often may be an early event. The lesions most frequently are related to venous occlusions and are often hemorrhagic (characteristic of venous infarcts). Occlusion of multiple adjacent veins appears to be necessary to result in infarction, as evidenced by the usual demonstration of several thrombosed vessels contiguous to the infarct. The loci of the infarcts, most often, are cerebral cortex and underlying white matter (see Fig. 39.6 ), although subependymal and deep white matter lesions are not uncommon. Involvement of major cerebral arteries may be more common than previously expected because brain imaging in living infants not uncommonly demonstrates lesions in an arterial distribution (see later discussion; see Box 39.3 ). Additionally, stroke patterns within the distribution of perforating arteries were shown to be especially common in neonatal group B Streptococcus meningitis. The origin of the infarctions is related particularly to the vasculitis, in combination with concomitant thrombotic effects of arachidonic acid metabolites (e.g., thromboxanes, platelet-activating factor), impaired cerebrovascular autoregulation, and decreased cerebral perfusion pressure (caused by systemic hypotension or increased intracranial pressure, or both; see later “Mechanisms of Brain Injury” section). Such a combination of factors presumably could lead to infarction in the presence of vascular narrowing and not necessarily complete thrombotic occlusion.
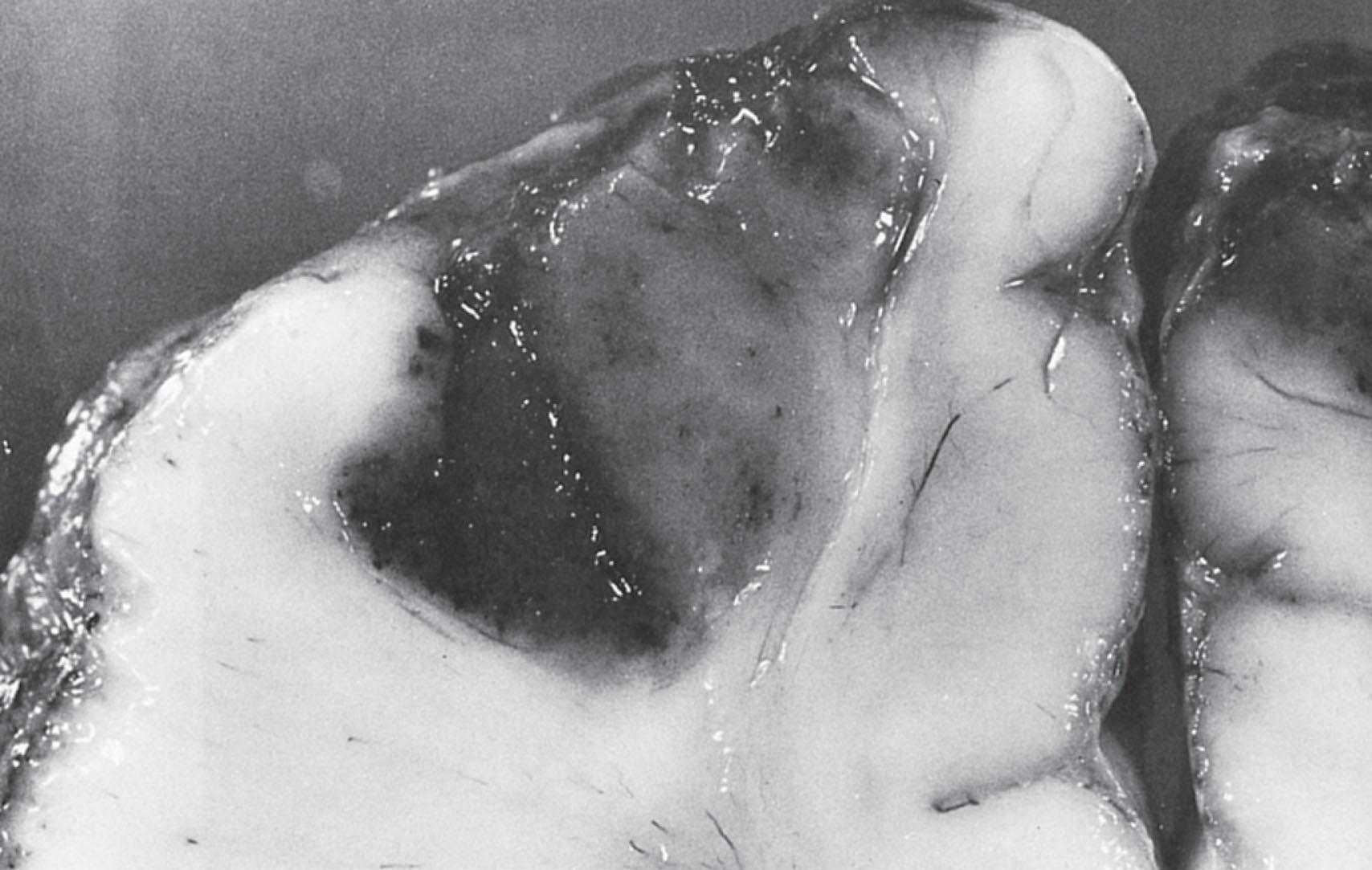
≈30%–50% of autopsy cases
Associated with thrombi in inflamed meningeal, cortical, and/or subependymal veins; venous sinuses; arteries (“arterial distribution” often suggested by brain imaging findings)
Related to a combination of vasculitis, endothelial injury, thrombotic effects of arachidonic acid metabolites, impaired autoregulation, decreased cerebral perfusion pressure
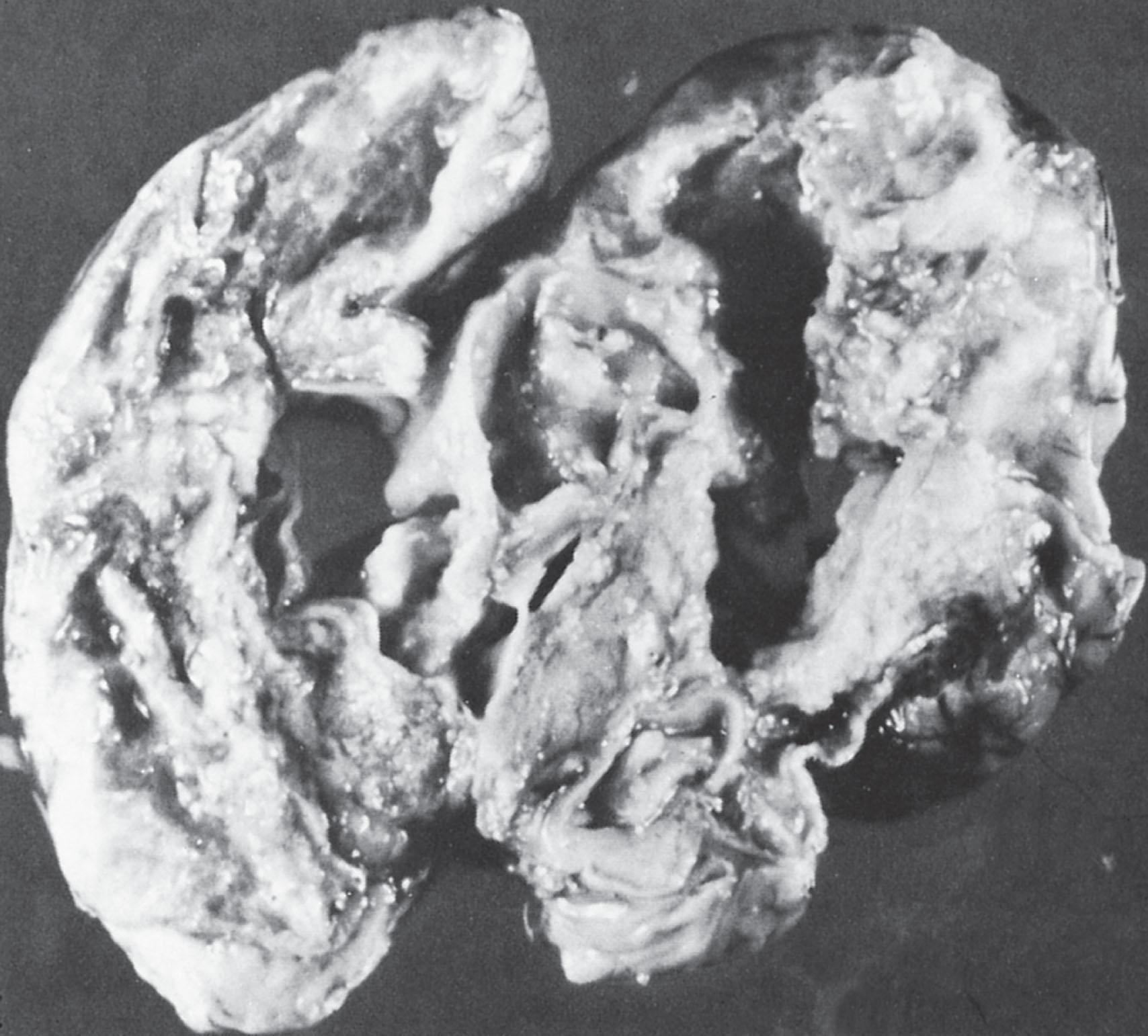
Although venous or arterial infarction of cerebral structures predominates, involvement of spinal cord may occur. Indeed, spinal cord necrosis and the clinical picture of a segmental myelopathy may rarely develop. The onset may be delayed, as shown in two infants who developed severe progressive late-onset myelopathy precipitated by a fall following presumed chronic arachnoditis.
Associated with neonatal bacterial meningitis are parenchymal changes (i.e., “associated encephalopathy”), the causes of which have been elucidated increasingly in recent years (see later discussion in “Mechanisms of Brain Injury”). The major changes are (1) diffuse gliosis of regions subjacent to inflammatory exudate, (2) neuronal loss in cerebral cortex and several other brain regions, and (3) periventricular leukomalacia. The first change, parenchymal gliosis, is observed in cerebral cortex (molecular layer and superficial cortical layers), cerebellar cortex (molecular layer), brainstem and spinal cord (marginal white matter), and subependymal regions. These various regions are immediately subjacent to the inflammatory exudate and presumably are injured by the toxic and metabolic factors associated with the bacterial process (see later discussion). The clinical significance of the gliosis is not entirely clear. The second and third changes, involving cortical neurons and periventricular white matter, are similar in topography, in many ways, to hypoxic-ischemic encephalopathy. The cause of these neuronal and white matter lesions probably relates at least in part to ischemia (see later discussion; Box 39.4 ). Moreover, it is likely that concentrations of endotoxin and related cytokines in brain and ventricular fluid are high in gram-negative bacterial meningitis and that the endotoxin could injure cerebral white matter through activation of innate immunity in brain (see later and Chapter 17 ). A similar conclusion applies to the capsular polysaccharides and proteins of group B Streptococcus , which also have been shown to be neurotoxic (see Chapter 17 ). Further data are needed on these issues, because the clinical significance of the neuronal loss and of the white matter injury is almost certainly considerable.
One neuropathological feature of neonatal bacterial meningitis, conspicuous by its relative lack of prominence, is subdural effusion. The reason for the notable difference in incidence of significant subdural effusion between meningitis in the newborn and in the infant of 2 to 3 months of age and older is not clear.
Subdural empyema is a very rare acute feature of bacterial meningitis.
The major neuropathological sequelae of neonatal bacterial meningitis are hydrocephalus, multicystic encephalomalacia, and cerebral cortical and white matter atrophy (see Box 39.2 ). Experimental observations suggest the possibility of a subsequent impairment in brain development, involving organizational events (see later discussion).
In studies of postmortem material, hydrocephalus is apparent in approximately 50% of cases. Of the 14 autopsy cases of hydrocephalus studied carefully by Berman and Banker, the major obstruction to CSF flow appeared to be at the level of the aqueduct in 4, at the outflow of the fourth ventricle in 2, and in the subarachnoid space (i.e., communicating) in 8. Multiple sites of impairment in CSF flow are probable. Ventricular dilation secondary to obstruction to CSF flow should be distinguished from that secondary to loss of cerebral substance. Because both hydrocephalus and cerebral atrophy are usually present concurrently, this distinction may be difficult.
Multicystic encephalomalacia and porencephaly are the end of the continuum of multifocal parenchymal injury secondary to neonatal bacterial meningitis. The single or multiple cystic areas of destruction in the cerebral hemispheres appear to reflect primarily residua of infarction ( Fig. 39.6 ). In postmortem material, some of the cavities rarely appear to represent abscesses. The implications of multicystic encephalomalacia for neurological outcome are obviously grave.
Most commonly, the major neuropathological sequela of neonatal bacterial meningitis is cerebral atrophy, manifested particularly by loss of cerebral cortical neurons and periventricular white matter. This state appears to be a sequela principally of the associated encephalopathy described previously. Neuronal loss in deep cortical layers and myelin loss in the periventricular region, both areas also infiltrated with glial fibers, are the major histological findings.
Experimental data suggest that more refined techniques may reveal subsequent aberrations of brain development following neonatal bacterial meningitis. In an infant rat model of bacterial meningitis, disturbances of subsequent dendritic arborization and synaptogenesis were observed. Because of the timing of neonatal bacterial meningitis with regard to brain developmental events (see Chapter 5, Chapter 6, Chapter 7, Chapter 8 ), it is reasonable to postulate that application of Golgi, immunocytochemical, and electron microscopic techniques to the study of cerebral cortex and myelin of infants who survive the acute state of neonatal meningitis would reveal impairment of cerebral organizational events and, perhaps, myelination. Further data in this regard would be of particular importance concerning the effects of neonatal bacterial meningitis on subsequent neurological outcome. Similarly, advanced magnetic resonance imaging (MRI) studies of subsequent cerebral gray matter and white matter development would be of considerable interest in this context.
Certain neuropathological changes are particularly characteristic of specific microorganisms ( Box 39.5 ).
Citrobacter, Serratia marcescens, Proteus, Pseudomonas, Bacillus cereus , Enterobacter species
Tissue necrosis, often hemorrhagic, and/or brain abscess
Listeria monocytogenes (Transplacental)
Multifocal granulomata with or without microabscesses
These bacteria have a particular propensity to cause severe cerebral necrosis, usually hemorrhagic (see Box 39.5 ). One consequence of this tissue necrosis, in the presence of bacteremia, is the occurrence of brain abscess. Indeed, the brain abscess may dominate the clinical syndrome, which occasionally may evolve in a less acute fashion than uncomplicated meningitis. (The most common organism leading to meningitis complicated by brain abscess is Citrobacter , which is discussed later in the section “Brain Abscess.”)
An even more dramatic and malignant form of neonatal bacterial meningitis is caused by Serratia marcescens . Widespread hemorrhagic necrosis of cerebral cortex and white matter occurs ( Fig. 39.7 ). Striking invasion of brain parenchyma by bacteria, which can be seen streaming from blood vessels (see Fig. 39.8 ), is a prominent feature.
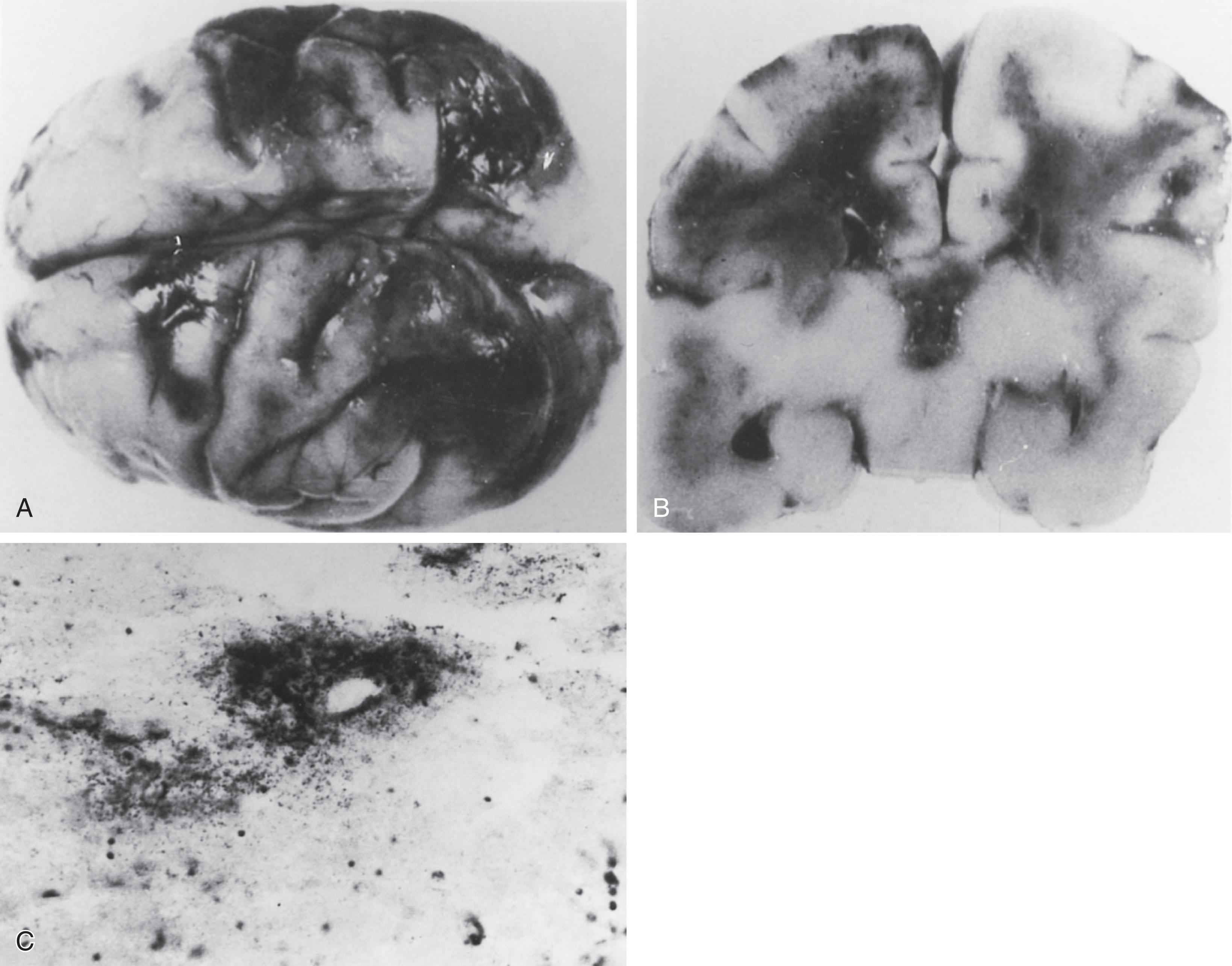
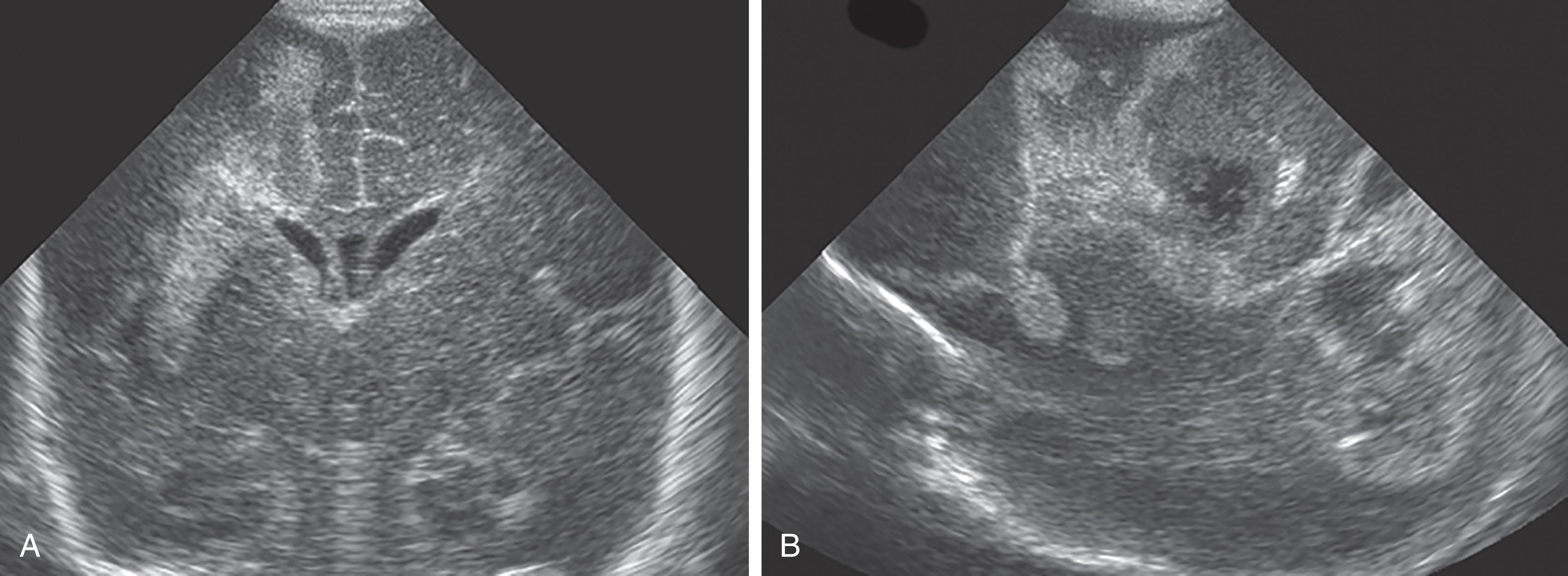
A rapid destruction of brain tissue may also occur with Bacillus cereus meningo-encephalitis, a gram-positive spore forming rod. The organism has often been considered to be a contaminant but is now recognized as a pathogen in patients who are immunocompromised. Invasive infections are related to the use of central or peripheral catheters, contaminated dressings, hospital linens, ventilator equipment, balloons used for manual ventilation, and breast milk. Following an uneventful initial neonatal period, the infant may become unwell, with signs of sepsis. The disease can be rapidly progressive, owing to the production of several toxins (necrotizing enterotoxin, emetic toxin, hemolysin, and phospholipase C), resulting in hemolysis, tissue invasion, and tissue necrosis. A recent review of the literature showed an increase in the number of published cases since 2017. It is possible that earlier infections were overlooked, because Bacillus cereus has long been regarded as a contaminant. A total of 145 preterm infants were reported in a recent review. The majority had a bacteremia (48%) and 36 (25%) had CNS involvement. Twenty-three had a meningitis and 13 had brain abscesses, empyema, or necrosis. Sixteen of those with CNS involvement died (42%). The overall mortality is less than a previous review of 15 newborn infants with neonatal meningitis caused by this organism, with gestational age ranging from 26 to 37 weeks with birth weights from 830 to 2780 g. Twelve of the infants died (75%), the majority within 3 days of onset. Of the four who survived the infection, one developed cerebral palsy, and the other three had no sequelae. Hemorrhagic necrosis and liquefaction of brain tissue have been reported in postmortem studies. Special stains reveal Gram-variable rods in brain tissue. An important aspect of treatment is recognition that this organism produces beta-lactamase that renders it resistant to most penicillins and cephalosporins. Treatment involves a combination of vancomycin and an aminoglycoside. Imaging characteristics may show destructive changes of the white matter, cortex, and basal ganglia, which can develop within 12 to 24 hours. Cranial ultrasound shows a typical irregular cauliflower-like pattern with extensive areas of increased echogenicity, followed by rapid cystic evolution ( Fig. 39.8 ). Many infants are unstable and die before they can be transferred to the MR unit. In one study, three infants assessed with MRI showed extensive hemorrhagic lesions in the cerebral white matter on conventional T1- and T2-weighted sequences, as well as areas of restricted diffusion on diffusion-weighted imaging (DWI).
Although L. monocytogenes may cause typical bacterial meningitis in the newborn, especially in infants with late-onset disease, transplacental infection of the fetus may occur and may produce a particularly fulminating infection. The latter may be manifest in utero and may result in fetal death or in an early-onset septicemic type of syndrome. The pathological features of this multifocal variety of listeriosis are miliary granulomatous lesions in many organs, including the CNS (see Box 39.5 ). The characteristic lesion is a necrotizing granuloma, which occurs particularly in the meninges, ventricular walls, and choroid plexus. Granulomata may form microabscesses, and organisms can be demonstrated in the necrotic portions of the lesions. As may be expected, this variety of infection is not readily accessible to antibiotics, and death occurs in 25% to 50% of cases, often in utero. Notably, however, treatment of the mother early in her infection can prevent infection and sequelae in the fetus and newborn. In a national observational cohort study, 189 infants with neonatal listeriosis were enrolled, 132 of whom developed early-onset listeriosis (70%) and 12 who developed late-onset listeriosis (6%). In 57 of the 132 infants with early-onset listeriosis the CSF culture was positive (43%). All 12 with late-onset listeriosis developed a meningitis within 7 to 17 days after birth. Outcome was better than reported previously, with a much lower mortality. Overall, 17 infants had major adverse outcomes (9%), including 5 deaths (3%), 12 with severe brain injury (16%), and 3 with severe bronchopulmonary dysplasia (2%). All but 2 of these 17 infants had a gestational age <34 weeks. The infant was noted to be less severely affected when the mother received antimicrobial treatment ≥1 day before delivery.
The neuropathological features of neonatal bacterial meningitis are caused by the action of a complex series of mechanisms summarized very broadly in Fig. 39.1 and in detail in Fig. 39.9 and Fig. 39.10 . The mechanisms shown in Fig. 39.9 and Fig. 39.10 represent a summary of more detailed physiological and biochemical processes, described briefly next and based on a variety of studies, primarily in animal models and cellular systems but also in human infants. The process begins with sepsis, followed by invasion of the CNS. Penetration of the bacteria into the choroid plexus and across the blood-brain barrier is one of the most critical steps in the pathogenesis of bacterial meningitis. The role of specific capsular polysaccharides in this invasion is crucial, as discussed earlier. Once the bacteria invade the blood-brain barrier and the local tissue barrier, most pathogens can activate the transcription factor nuclear factor kappa B, another important feature of bacterial meningitis, resulting in high levels of inflammatory cytokines in the blood and CSF, a result of the stimulation of phagocytic cells. This secretion of proinflammatory cytokines is followed by recruitment of leukocytes into the CNS across the blood-brain barrier. The meningitic process begins with the action of specific bacterial components, especially in gram-positive bacteria, the peptidoglycan layers and the teichoic acid of the cell wall, and in gram-negative bacteria, the lipopolysaccharide molecules of the outer cell membrane. Among the early events induced by these products, of particular importance is activation of the immediate immune response (i.e., the innate immune response; see Fig. 39.10 ). As noted earlier, these molecular products of gram-positive organisms (e.g., group B Streptococcus ) and gram-negative organisms (e.g., E. coli ) activate specific receptors on brain microglia (the immune cell of the CNS). For gram-positive organisms, the receptor is TLR2, and for gram-negative organisms, it is TLR4. The resulting microglial activation results in several effects, the most important of which is generation of free radicals, both reactive oxygen and nitrogen species. These reactive compounds ultimately lead to neuronal and oligodendroglial death (see also Chapter 18 ), the key feature of the associated encephalopathy of neonatal bacterial meningitis. Evidence of neonatal bacterial meningitis leading to generation of free radicals and periventricular leukomalacia has been obtained by direct serial measurements of markers of free radical attack in CSF and correlation of elevations of these markers with the occurrence of MRI-documented cerebral white matter lesions.
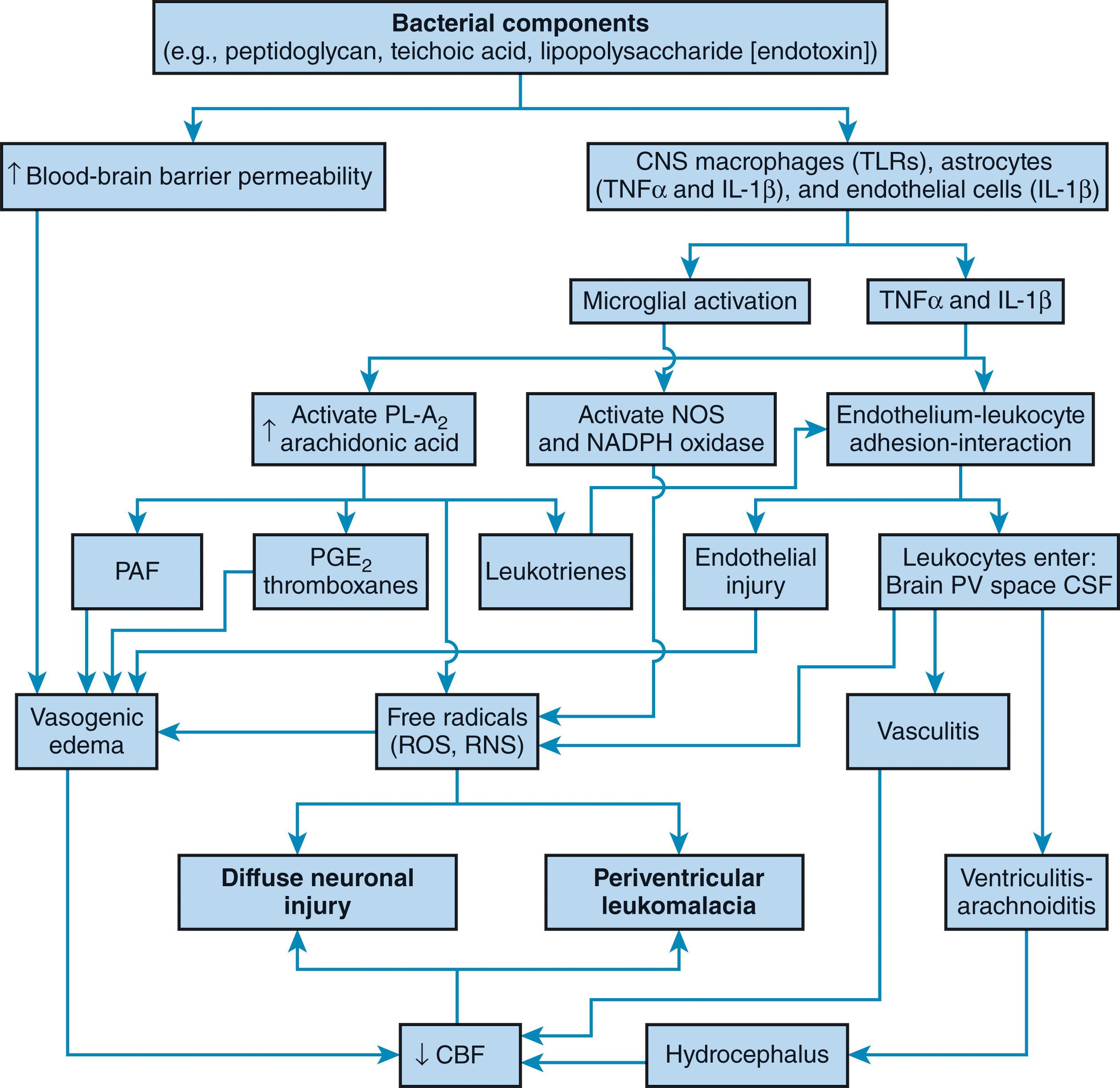
The myriad deleterious biochemical and physiological events initiated by the bacterial products discussed earlier are shown in Fig. 39.10 . Thus these components induce an increase in blood-brain barrier permeability and the early phases of the inflammatory response (i.e., the synthesis and secretion of cytokines, especially tumor necrosis factor from CNS macrophages [microglia] and astrocytes and interleukin-1 beta from astrocytes and endothelial cells). The cytokines lead to the adhesion and interaction of leukocytes with endothelial cells by induction of specific cell surface molecules on both leukocytes and endothelial cells. The result is the ventriculitis, arachnoiditis, and vasculitis described earlier; the generation of free radicals by leukocytes; and the activated microglia just discussed (see Fig. 39.10 ). A second crucial effect of the cytokines is the activation of phospholipase A 2 and thereby arachidonic acid release and subsequent metabolism, the products of which include free radicals, platelet-activating factor, prostaglandins, thromboxanes, and leukotrienes. The interaction of these effects leading to diffuse neuronal injury, periventricular leukomalacia, and thrombotic cerebral infarction is shown in Fig. 39.10 . The importance of impaired cerebral blood flow is substantial, as discussed earlier. Impairment of cerebrovascular autoregulation is suggested by experimental data ( Fig. 39.11 ), and the importance of systemic hypotension is implied by clinical observations. Ischemia as an important final common denominator is suggested further by the demonstrations of elevations of extracellular glutamate and reactive oxygen and nitrogen species in experimental models of bacterial meningitis.
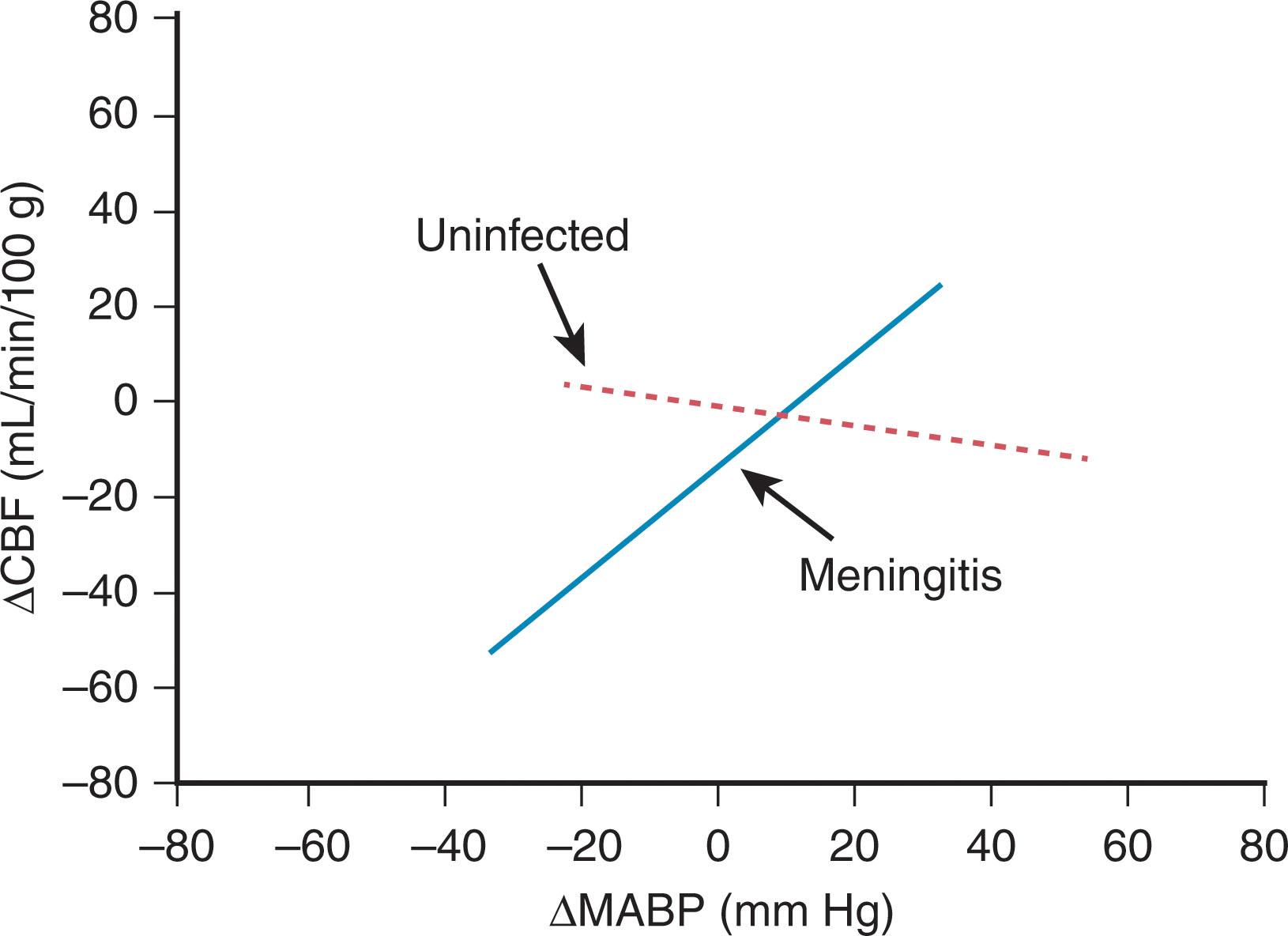
Finally, activation of innate immunity and of many of the mechanisms shown in Fig. 39.9 and Fig. 39.10 may occur without bacterial invasion of the CNS, as discussed in Chapters 16 , 17 , and 19 concerning the roles of systemic infection in white matter and neuronal injury, especially in the premature infant. Indeed, in a large study of more than 6000 premature infants (401 to 1000 g at birth), infants with sepsis alone (without meningitis) had 50% to 100% higher rates of cognitive deficits, cerebral palsy, visual impairment, hearing impairment, and neurodevelopmental disability compared with rates of these outcomes in uninfected infants .
The clinical features of neonatal bacterial meningitis occur in the setting of two basic clinical syndromes, separable principally by age of onset: early-onset disease and late-onset disease (see Table 39.1 ). Both syndromes have been described with the three major organisms associated with neonatal bacterial meningitis: group B Streptococcus, E. coli , and L. monocytogenes .
Early-onset disease is associated with clinical phenomena usually in the first 72 hours of life. As noted earlier, a history of obstetrical complications and premature birth is common, and, understandably, the mode of transmission is primarily from mother to infant near the time of delivery. In a recent population-based study a distinction was made between early-onset, late-onset, and community-acquired disease ( Fig. 39.12 ). With early-onset disease , group B streptococcal infection was the most common etiological organism, and, together with E coli , accounted for >60% of cases. Mortality rates in recent years are much lower than reported previously (see “Prognosis,” later).
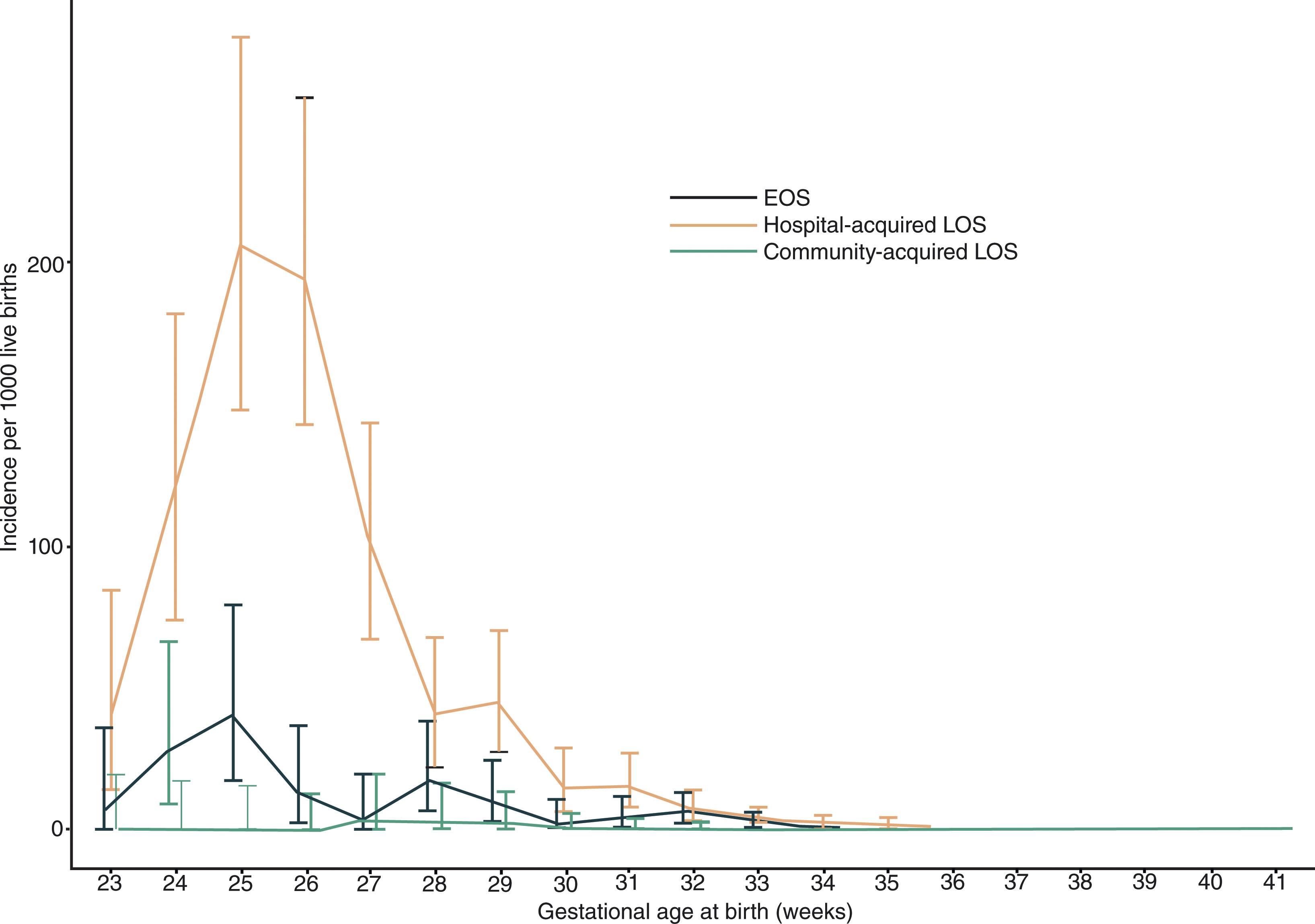
The clinical presentation is dominated by nonneurological signs (i.e., signs related to sepsis and respiratory disease). The most common signs are hyperthermia, apnea, hypotension, disturbances of feeding, jaundice, hepatomegaly, and respiratory distress; less common signs are hypothermia, skin lesions (e.g., petechiae and sclerema), and overt focal infection (e.g., otitis media, omphalitis, arthritis, and osteomyelitis). Neurological signs are usually limited to stupor (usually termed lethargy ) and irritability. Signs suggestive of meningitis (see later discussion) are unusual, in part because overt meningitis occurs in only approximately 30% of the patients with early-onset disease. Indeed, even those infants with culture-proven meningitis often do not exhibit striking CSF pleocytosis.
The respiratory manifestations of early-onset sepsis-meningitis may be particularly prominent with group B streptococcal infection and, indeed, may simultaneously present serious diagnostic problems and important diagnostic clues. Approximately 40% to 60% of infants with early-onset group B streptococcal disease have manifestations of prominent respiratory disease in the first 6 hours of life. Only approximately 35% of these infants exhibit radiographic infiltrates suggestive of congenital pneumonia, whereas approximately 50% exhibit radiographic features of respiratory distress syndrome. Distinction from nonbacterial respiratory disease is suggested by the history of premature rupture of membranes, low Apgar scores (<4 at 1 minute in 85%), rapid progression of pulmonary disease, and low or declining absolute neutrophil counts in the first 24 hours of life.
Late-onset bacterial sepsis-meningitis is much more likely to manifest as a neurological syndrome with overt signs of meningitis. Indeed, approximately 80% to 90% of affected infants have CSF findings clearly indicative of meningitis. As noted earlier, unlike in early-onset disease, obstetrical complications and prematurity are uncommon, and onset is usually after the first week of life. The mode of transmission may be from mother to infant, but often horizontal transmission from human contacts, indwelling catheters, or contaminated equipment can be documented. Specific serotypes are usually involved (serotype III for group B Streptococcus , IVb for L. monocytogenes , and K1 strains for E. coli ); the course is not so fulminating as in early-onset disease, and the mortality rate is slightly lower.
Although neurological signs associated with meningitis are prominent in late-onset disease , some of the signs prominent in early-onset disease are also common, especially fever and feeding disturbances. Most impressive, however, are the neurological signs ( Table 39.6 ). Most patients exhibit impairment of consciousness , manifested usually as varying degrees of stupor, with or without irritability. The disturbances of level of consciousness presumably relate to the cerebral edema and, perhaps, the associated encephalopathy (see “Neuropathology”). Seizures develop at some time in the illness in nearly 50% of cases, although these seizures may be predominantly subtle. (Seizures represent a strong predictor of neurodevelopmental impairment, as discussed later. ) The convulsive phenomena presumably relate to the cortical effects of the arachnoidal inflammatory infiltration. Focal seizures occur in approximately 50% of infants with seizures and may be prominent. These focal episodes may be related to ischemic vascular lesions. Similar bases are probable for the focal cerebral signs (e.g., hemiparesis and horizontal deviation of eyes), which are movable with the doll’s eyes maneuver. These signs are reported only uncommonly in the literature, but they can be elicited with careful examination in nearly one-half of cases. Extensor rigidity occurs in approximately one-third of cases and may be so severe that opisthotonos occurs. These phenomena probably relate to the arachnoidal inflammation, especially in the posterior fossa. A similar basis is likely for the nuchal rigidity, which occurs in fewer than 25% of cases. Apparent in most patients, however, is a distinct increase in irritability, elicitable by flexion of the neck, and, presumably, the neonatal counterpart of more overt nuchal rigidity. Cranial nerve signs involve usually the seventh, third, and sixth nerves, in that order of frequency, and are more common than often suggested in the literature. These signs relate to involvement of cranial nerve roots by the arachnoidal inflammation (see “Neuropathology”). A bulging or full anterior fontanelle is present, often later in the course of the disease, in approximately 35% to 50% of patients. This feature relates to an increase in intracranial pressure (see following section). Rarely, anterior fontanelle herniation of cerebral tissue occurs and imparts a “doughy” consistency to the bulging fontanelle. Moreover, very uncommonly, signs of segmental cord involvement (sensory level, flaccidity below the level of the lesion), secondary to myelopathy, may be present.
| NEUROLOGICAL SIGN | APPROXIMATE FREQUENCY |
|---|---|
| Stupor with or without irritability | 76%–100% |
| Seizures | 26%–50% |
| Bulging or full anterior fontanelle | 26%–50% |
| Extensor rigidity, opisthotonos | 26%–50% |
| Focal cerebral signs | 26%–50% |
| Cranial nerve signs | 26%–50% |
| Nuchal rigidity | 0%–25% |
The clinical course of neonatal bacterial meningitis may be complicated by the following four major and often interrelated events: (1) severe increase in intracranial pressure, (2) ventriculitis with localization of infection, (3) acute hydrocephalus, and (4) an intracerebral mass or extraparenchymal collection (e.g., abscess, hemorrhagic infarct, and subdural effusion) ( Box 39.6 ).
Increased intracranial pressure
Ventriculitis with localized, inaccessible infection
Acute hydrocephalus
Intracerebral mass or extracerebral collection
Intracranial pressure, although only uncommonly monitored, may be markedly increased in neonatal bacterial meningitis. Major causes include cerebral edema, hydrocephalus, and, uncommonly, formation of an intracranial mass or extracerebral collection ( Box 39.7 ). Cerebral edema (vasogenic and cytotoxic) is a frequent feature in the first several days of the disease and may be aggravated by water retention secondary to inappropriate antidiuretic hormone secretion. Increased intracranial pressure may persist or worsen in the ensuing days, with the development of acute hydrocephalus or an intracranial space-occupying lesion (see subsequent discussion).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here