Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Chronic lower limb ischaemia often referred to as peripheral arterial disease (PAD) is characterised by impaired circulation in the lower limb extremities. The worldwide prevalence of PAD is over 200 million and this disease burden is expected to increase with the aging global demographics and rise in diabetes. The early assessment and recognition of PAD is crucial as these patients are at 2–4 times the risk for future cardiovascular and cerebrovascular events. Prompt treatment reduces longer-term risks of ischaemic events and major limb amputation.
PAD is a consequence of systemic atherosclerosis, which involves the arterial tree throughout the body and commonly affects the coronary, carotid, iliac, femoral and infra-inguinal arteries. For the purpose of this chapter, PAD will refer to its manifestations in the lower limb. PAD is a common disease and its prevalence can be as high as 20% over the age of 60 and two thirds of these patients remain asymptomatic and undiscovered without specific screening tests. As the degree of arterial disease progresses, symptoms of PAD appear; the severity of which coincides with the extent of arterial bed involvement. These symptoms can range from exertional leg pain, which can subsequently progress to (or present as) severe limb threatening ischaemia.
Intermittent claudication (IC) is often the first symptom of PAD, most commonly located in calf muscles, and usually associated with atherosclerotic occlusion or stenosis involving the iliac or femoro-popliteal (FP) segments. More severe limb ischaemia is usually associated with PAD affecting the lower limb vasculature at two or three anatomical levels. This results in significantly impaired tissue perfusion with rest pain, ulceration or gangrene involving the toes or forefoot. This is referred to as chronic limb-threatening ischaemia ( CLTI ). This chapter deals with the assessment of patients with chronic lower limb ischaemia, including the principles of vascular imaging.
Recent data from the Swedevasc Registry found that the risk of amputation in patients with claudication who had undergone revascularisation was 1.2% at 3 years, compared to a 12% risk of amputation in the first 6 months following revascularisation for those with CLTI and then 2% annually. After 3 years, the cumulative mortality rate for people with claudication was 12% and 41.4% in CLTI.
Severity of PAD is commonly reported by two classification systems. The Fontaine classification is based on clinical symptoms alone ( Table 2.1 ) and in contrast, the Rutherford classification is more detailed and takes into account clinical findings, including Doppler assessment and ankle pressure assessment ( Table 2.2 ). These two classifications are inclusive of the asymptomatic and milder forms of PAD and useful for ensuring consistency in reporting standards.
| Fontaine stage | Description | |
|---|---|---|
| I | Asymptomatic | PAD present but no symptoms |
| II | Intermittent claudication | Cramping pain in leg muscles precipitated by walking and rapidly relieved by rest |
| III | Rest pain | Constant pain in feet (often worse at night) |
| IV | Tissue loss | Ischaemic ulceration or gangrene |
| Grade | Category | Description |
|---|---|---|
| 0 | 0 | Asymptomatic |
| I | 1 | Mild claudication |
| I | 2 | Moderate claudication |
| I | 3 | Severe claudication |
| II | 4 | Ischaemic rest pain (ankle pressure [AP] <40 mmHg of flat waveforms |
| II | 5 | Minor tissue loss (AP <40 mmHg, flat waveforms or toe pressure [TP] <40 mmHg) |
| III | 6 | Major tissue loss (AP <40 mmHg, flat waveforms or TP <40 mmHg) |
IC occurs because of inadequate augmentation of blood flow to the skeletal muscles, which fails to meet the increased metabolic requirements of the exercising muscle groups. IC is present in only 10–20% of patients with PAD with the majority of patients remaining asymptomatic. In addition to the reduced arterial perfusion from fixed atherosclerotic lesions, endothelial dysfunction with diminished vasodilator effect, inadequate angiogenesis combined with failure to develop collaterals, impaired skeletal muscle metabolism because of mitochondrial dysfunction and local inflammatory changes are also proposed as mechanisms for the pathogenesis of IC. The classic feature of IC is of pain developing on walking in the muscle groups distal to the arterial obstruction, with relief of symptoms within 10 minutes of cessation of exercise (usually more quickly). This pain reappears after walking a similar distance and is not felt at rest or within the first few steps taken. The symptoms have a chronic history and are described as an ache, cramp or tightening in the muscle that usually forces the patient to stop. Exercise tolerance is dependent on the balance between tissue perfusion and energy expenditure. In mild PAD, claudication may be felt only while walking uphill or walking quickly. Conversely, in individuals with restricted mobility from other comorbidities, PAD may not manifest with claudication symptoms. Claudication pain most commonly affects the calf muscles, which are extensively used whilst walking. However, in the presence of aorto-iliac disease, pain may be felt more proximally in the buttocks or thigh, as well as in the calf.
Conditions such as osteoarthritis of the hip or knee, spinal canal stenosis and venous outflow obstruction may be mistaken for IC and should be considered in the differential diagnosis. Other relevant conditions to consider in the differential diagnosis of IC are described in Table 2.3 .
| Condition | Location of pain or discomfort | Characteristic discomfort | Onset relative to exercise | Effect of rest | Effect of body position | Other characteristics |
|---|---|---|---|---|---|---|
| Intermittent claudication | Buttock, thigh or calf muscles and rarely the foot | Cramping, aching, fatigue, weakness or frank pain | After same degree of exercise | Rapid relief with rest | None | Reproducible |
| Nerve root compression (e.g., herniated disk) | Radiates down leg, usually posteriorly | Sharp lancinating pain | Soon, if not immediately, after onset | Not quickly relieved (also often present at rest) | Relief may be aided by adjusting back position | History of back problems |
| Spinal canal stenosis | Hip, thigh, buttocks (follows dermatome) | Motor weakness more prominent than pain | After walking or standing for variable lengths of time | Relieved by stopping only if position changed | Relief by lumbar spine flexion (sitting, using walking frame or stooping forward) | Frequent history of back problems, provoked by intra-abdominal pressure |
| Arthritic, inflammatory processes | Foot, arch | Aching pain | After variable degree of exercise | Not quickly relieved (and may be present at rest) | May be relieved by not bearing weight | Variable, may relate to activity level |
| Hip arthritis | Hip, thigh, buttocks | Aching discomfort, usually localised to hip and gluteal region | After variable degree of exercise | Not quickly relieved (and may be present at rest) | More comfortable sitting, weight taken off legs | Variable, may relate to activity level, weather changes |
| Symptomatic Baker’s cyst | Behind knee, down calf | Swelling, soreness, tenderness | With exercise | Present at rest | None | Not intermittent |
| Venous claudication | Entire leg, but usually worse in thigh and groin | Tight, bursting pain | After walking | Subsides slowly | Relief speeded by elevation | Often history of iliofemoral deep vein thrombosis, signs of venous congestion, oedema |
| Chronic compartment syndrome | Calf muscles | Tight, bursting pain | After much exercise (e.g., jogging) | Subsides very slowly | Relief speeded by elevation | Typically occurs in heavy muscled athletes |
In osteoarthritis of the hip, pain may be referred down the leg to mimic claudication. However, these patients may also give a history of pain in the buttock or groin when turning their body and even in a sitting or supine position. Unlike the pain in IC, this is not relieved by standing still and requires easing the load off the arthritic joint. In addition, pain from osteoarthritis typically begins when walking and gets better after some exercise. Although the diagnosis can usually be established by history and examination alone, non-invasive investigations including an exercise ankle–brachial index (ABI) test to exclude arterial disease will be reassuring.
Patients with spinal canal stenosis may also have symptoms that are very similar to IC although the history of pain may be inconsistent. Postures such as sitting and leaning forward to straighten the lumbar lordosis relieves cord pressure although the pain symptoms may take 60 minutes or even longer to subside. A history of pain on standing as well as walking should raise the suspicion of neurogenic pain because of spinal stenosis.
Lumbar nerve route irritation may also cause aching in the calf or down the back of the leg from buttock to ankle. The sensation appears to be very similar to that of claudication, particularly when confined to the calf. Direct enquiry for these symptoms is helpful, but the key feature is again the need to sit or lie to obtain relief. Spinal flexion may release the involved nerve roots, whereas a straight leg raise will often precipitate the pain. When both sciatic nerve irritation or spinal canal stenosis and PAD coexist, it can be extremely difficult to identify which is contributing most to the patient’s symptoms.
In venous outflow obstruction, exercise-induced arterial flow augmentation is not matched by the venous outflow, resulting in high venous pressures within the lower limbs. Tense dilatation of the veins can mimic claudication-like symptoms. However, this is often described as a severe bursting pain and is relieved slowly with rest or leg elevation. A previous history of iliofemoral deep vein thrombosis and inspection findings of lower limb oedema and other stigmata of chronic venous insufficiency will often help with the diagnosis.
The cornerstones in the assessment of chronic lower limb ischaemia are a careful history, palpation of pulses and ABI measurement. The important point is to verify that the clinical findings correlate well with the patient’s symptoms. As the patients get older so does the arterial tree and the mere presence of PAD (i.e., reduced ABI, atherosclerosis visible on ultrasound examination) does not necessarily mean that it is the cause of the symptoms. The history should include the duration of symptoms and the mode of onset. Most patients gradually become aware of pain on walking, which is typical in progressive PAD.
The blood supply required by resting muscles is relatively small (130–150 mL/min) and may be increased five- to 10-fold during exercise .
Arterial occlusions can be well tolerated when collaterals have developed, which may provide the same volume of blood flow at rest compared to normal individuals. Examples include the thigh muscles, which are perfused by collaterals from the profunda femoris artery in superficial femoral artery occlusion and also occlusions of the iliac arteries, where collaterals develop through the pelvis and buttock and may even result in normal resting ABI and a palpable foot pulse. Significant PAD is usually associated with an ABI of <0.9 (see later), but in such cases, resting ankle pressures may be deceptively normal. However, an exercise challenge will result in a fall in ankle pressures and disappearance of the distal pulses. It is important to bear in mind that these patients may continue to remain asymptomatic if they undertake only limited physical activity below the threshold limit to induce claudication.
CLTI represents the end stage of PAD. Ischaemic rest pain, ischaemic ulceration or gangrene of the foot are the hallmarks of CLTI and this requires urgent investigation and revascularisation to avoid limb loss from progressive tissue necrosis and/or infection. Unlike IC, in CLTI the arterial perfusion is severely compromised to the degree that it is unable to meet the basal metabolic requirements of the ischaemic tissues or adequate perfusion to heal a foot wound. Severely compromised tissue perfusion causes pain even at rest and the inability for cellular repair and regeneration results in ulceration and gangrene commonly seen over areas of minor trauma. Untreated, the prognosis is poor with a higher risk of mortality, limb loss and associated pain and reduced quality of life. Recognition of CLTI is therefore very important in the assessment of chronic limb ischaemia.
The Global Vascular Guidelines for CLTI, which is a collaborative effort of the major vascular surgical societies worldwide, has proposed a revised model of care for CLTI. These guidelines aim to redefine the nomenclature and propose a standardised disease staging and revascularisation approach (evidence-based revascularisation [EBR]). Adoption of the term CLTI addresses the shortcomings of the previous terms “ critical limb ischaemia ”, which was based on threshold values of ABI and toe pressures (TPs). CLTI aims to include a diverse group of patients with varying degrees of ischaemia, which can result in delayed wound healing or amputation. More importantly, this definition acknowledges the current global trend of PAD and its close association with diabetes and recognises this as an important risk factor that can result in limb loss.
CLTI is defined as objectively documented PAD and any of the following clinical symptoms or signs:
Ischaemic rest pain present for longer that 2 weeks and associated with ABI <0.4, ankle pressure (AP) <50 mmHg, TP, <30 mmHg, transcutaneous partial pressure of oxygen (TcPO 2 ) <30 mmHg or flat waveforms on pulse volume recordings.
Diabetic foot ulcer (DFU) or any lower limb ulceration present for at least 2 weeks
Gangrene involving any portion of the lower limb or foot.
Pure venous ulcers, pure traumatic wounds, acute limb ischaemia (symptoms less than 2 weeks) embolic disease and non-atherosclerotic chronic vascular conditions of the lower limb extremity (e.g., vasculitis, Buerger’s disease, radiation arteritis) are excluded in this definition.
CLTI encompasses a wide range of clinical disease burden and anatomical complexity. Staging of CLTI therefore becomes paramount not only to estimate risk of amputation and effectiveness of treatment strategies but also in making objective comparisons in research trials and for a uniform and reliable reporting mechanism of disease severity. The Society for Vascular Surgery Lower Extremity Guideline Committee published the Threatened Limb Ischaemia Classification System in 2014 and this staging system takes into account the three major risk factors that determine the probability of lower limb amputation – w ound, i schaemia and f oot i nfection ( WIfI ). Each of the three risk factors are graded from 0–3 based on severity, where 0 represents none, 1 mild, 2 moderate and 3 severe ( Tables 2.4–2.6 ). The scores derived from each component are combined and analysed using two tables: one which estimates the risk of amputation at 1 year and the second table, which estimates the benefit from revascularisation ( Table 2.7 A&B). Accordingly, the limb is stratified into clinical stages 1, 2 , 3 or 4 as very low, low, medium and high risk of amputation and also the likely benefit from revascularisation. Stage 5 is clinically assessed as a non-salvageable limb. Several studies have now validated WIfI as a reliable staging system in predicting lower limb outcome in CLTI. It is recommended that all patients with CLTI are staged with WIfI initially and this should be repeated after vascular intervention or foot surgery, treatment of infection or when there is a clinical deterioration.
| Grade (W) | Ulcer | Gangrene |
|---|---|---|
| 0 | No ulcer | No gangrene |
| Clinical description: ischaemic rest pain (requires typical symptoms + ischaemia grade 3); no wound. | ||
| 1 | Small, shallow ulcer on distal leg or foot; no exposed bone, unless limited to distal phalanx | No gangrene |
| Clinical description: minor tissue loss. Salvageable with simple digital amputation (1 or 2 digits) or skin coverage. | ||
| 2 | Deeper ulcer with exposed bone, joint or tendon; generally not involving the heel; shallow heel ulcer, without calcaneal involvement | Gangrenous changes limited to digits |
| Clinical description: major tissue loss salvageable with multiple (>3) digital amputations or standard TMA ± skin coverage. | ||
| 3 | Extensive, deep ulcer involving forefoot and/or midfoot; deep, full-thickness heel ulcer ± calcaneal involvement | Extensive gangrene involving forefoot and/or midfoot; full-thickness heel necrosis ± calcaneal involvement |
| Clinical description: extensive tissue loss salvageable only with a complex foot reconstruction or non-traditional TMA (Chopart, or Lisfranc amputation); flap coverage or complex wound management needed for large soft tissue defect. | ||
| Grade (I) | ABI | Ankle systolic pressure | TP, TcPO 2 |
|---|---|---|---|
| 0 | ≥0.80 | >100 mmHg | ≥60 mmHg |
| 1 | 0.6–0.79 | 70–100 mmHg | 40–59 mmHg |
| 2 | 0.4–0.59 | 50–70 mmHg | 30–39 mmHg |
| 3 | ≤0.39 | <50 mmHg | <30 mmHg |
| Clinical manifestation of infection | SVS | IDSA/PEDIS infection severity |
|---|---|---|
| No symptoms or signs of infection | 0 | Uninfected |
| Local infection involving only the skin and the subcutaneous tissue (without involvement of deeper tissues and without systemic signs as described later). Infection present, as defined by the presence of at least 2 of the following items:
Exclude other causes of an inflammatory response of the skin (e.g., trauma, gout, acute Charcot neuro-osteoarthropathy, fracture, thrombosis, venous stasis) |
1 | Mild |
| Local infection (as described earlier) with erythema >2 cm, or involving 2 structures deeper than skin and subcutaneous tissues (e.g., abscess, osteomyelitis, septic arthritis, fasciitis), and no systemic inflammatory response signs (as described later) | 2 | Moderate |
Local infection (as described earlier) with the signs of SIRS, as manifested by two or more of the following:
|
3 | Severe |
| Ischaemia - 0 | Ischaemia - 1 | Ischaemia -2 | Ischaemia - 3 | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| W-0 | VL | VL | L | M | VL | L | M | H | L | L | M | H | L | M | M | H | |
| W-1 | VL | VL | L | M | VL | L | M | H | L | M | H | H | M | M | H | H | |
| W-2 | L | L | M | H | M | M | H | H | M | H | H | H | H | H | H | H | |
| W-3 | M | M | H | H | H | H | H | H | H | H | H | H | H | H | H | H | |
| fI-0 | fI-1 | fI-2 | fI-3 | fI-0 | fI-1 | fI-2 | fI-3 | fI-0 | fI-1 | fI-2 | fI-3 | fI-0 | fI-1 | fI-2 | fI-3 | ||
| B. Estimate benefit of/requirement for revascularisation (after control of infection first) | |||||||||||||||||
| Ischaemia - 0 | Ischaemia - 1 | Ischaemia -2 | Ischaemia - 3 | ||||||||||||||
| W-0 | VL | VL | VL | VL | VL | L | L | M | L | L | M | M | M | H | H | H | |
| W-1 | VL | VL | VL | VL | L | M | M | M | M | H | H | H | H | H | H | H | |
| W-2 | VL | VL | VL | VL | M | M | H | H | H | H | H | H | H | H | H | H | |
| W-3 | VL | VL | VL | VL | M | M | H | H | H | H | H | H | H | H | H | H | |
| fI-0 | fI-1 | fI-2 | fI-3 | fI-0 | fI-1 | fI-2 | fI-3 | fI-0 | fI-1 | fI-2 | fI-3 | fI-0 | fI-1 | fI-2 | fI-3 | ||
Rest pain often presents with pain in the forefoot at night sufficient to disturb the patient’s sleep when the patient is supine. When the patient hangs the leg out of bed, sits in a chair or if they stand up and walk (thereby increasing the blood flow to the foot), the pain is often relieved. If the patient constantly hangs their feet out of bed at night, or even sleeps sitting in a chair, the limb tends to swell because of dependent oedema. This oedema in turn increases the hydrostatic pressure of the peripheral tissue and compresses the already compromised capillaries and further interferes with tissue perfusion and nutrition. These patients may often require inpatient treatment with analgesia (usually including opiates) so that the limb can be kept in the supine position overnight to relieve the oedema before arterial reconstruction.
To the experienced eye, the diagnosis of CLTI seems obvious, but it is often easy to miss a small ischaemic lesion on the heel or between the toes. When established necrosis or gangrene is present with absent limb pulses, there is no doubt about the diagnosis. The stage of CLTI without necrosis or gangrene is characterised by pallor when the leg is elevated above the heart and changing to a deep red colour when hanging down (Buerger’s test-positive). The red colour is caused by the dilated capillaries of the foot. In CLTI, the natural compensatory mechanisms for ischaemia including angiogenesis (new capillary formation) and arteriogenesis (enlargement of pre-existing capillaries) are exhausted and the capillaries are maximally vasodilated and hence unresponsive to pro-vasodilatory stimuli. , Therefore it may take a while for pallor on elevation to occur but capillary refill will be abolished immediately.
Although the vast majority of cases of chronic lower limb ischaemia are caused by atherosclerotic PAD, other rare conditions exist and these are more frequently seen in the younger age group of patients. History of IC in a patient who is young and also in older patients without risk factors for atherosclerosis should alert one to the possibility of these rare diagnoses. Thorough history and meticulous examination including peripheral pulses and resting ABIs should be recorded. In patients giving a convincing history of IC with palpable foot pulses or normal ABI, onset of symptoms with exercise testing and post-exercise ABI’s may help clinch the diagnosis.
This condition should be suspected particularly when a young patient, especially an athletic individual complains of claudication symptoms or CLTI. There is a male preponderance for popliteal artery entrapment syndrome (PAES) with 85% being male and over 60% of cases occur in athletes. , The presentation is bilateral in 30% of cases. During embryonic development, the medial head of the gastrocnemius muscle (MHG) migrates medially from the posterior aspect of the fibula and lateral tibia across the popliteal fossa to its attachment to the posterior aspect of the medial femoral condyle. The popliteal artery simultaneously develops superficial to the popliteus muscle during this stage. These complex and dynamic changes associated with the foetal limb bud rotation and knee extension raises the chance for various anatomical variations to develop in the relationship between the popliteal artery and the MHG. There are six variants of popliteal entrapment described ( Table 2.8 ). Fig. 2.1 demonstrates the anatomical variants of PAES.
| Type 1 | The popliteal artery takes an aberrant medial course around the normally placed medial head of the gastrocnemius muscle (MHG) |
| Type II | The MHG has a more lateral insertion on the femoral condyle and hence the popliteal artery runs medially and inferiorly |
| Type III | Abnormal muscle slips or fibrous bands tether the artery to the medial or lateral femoral condyles |
| Type IV | Popliteal artery is entrapped by the popliteus muscle and here a persisting axial artery replaces the popliteal artery |
| Type V | The popliteal artery and vein are involved in any of the aforementioned subtypes |
| Type VI | (Functional variant): Muscle hypertrophy causes functional compression of the popliteal artery and vein. No anatomical abnormality is noted |
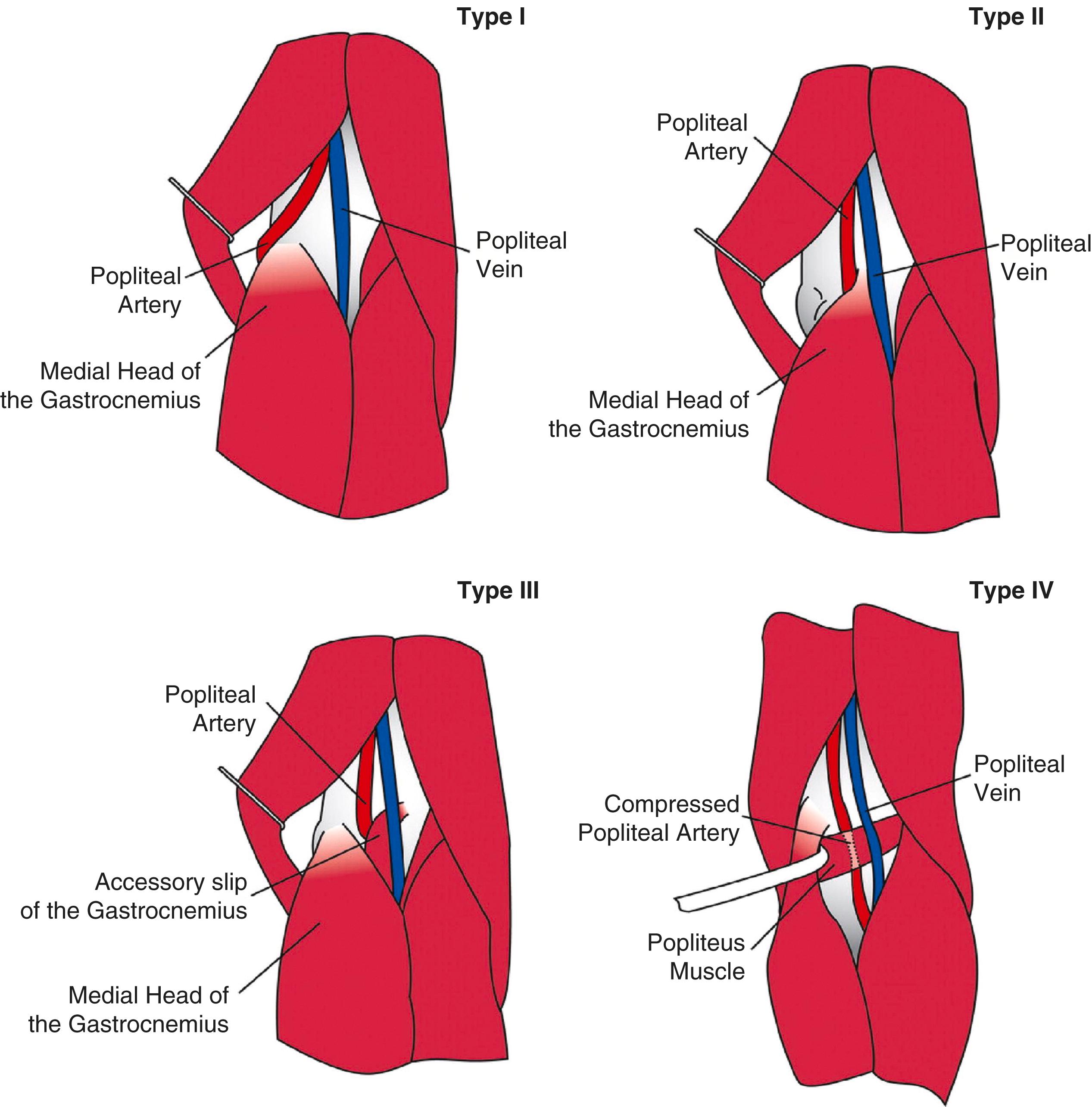
Continuous compression of the artery can result in fibrotic change, which progresses from the outer adventitial layer of the artery to the intima. As a consequence, aneurysmal degeneration and/or thrombosis may develop. Examination may reveal reduction or obliteration of pedal pulses during active plantar flexion. Duplex scanning or arteriography using this manoeuvre may also demonstrate kinking or compression of the popliteal artery. Computed tomography (CT) or magnetic resonance (MR) scanning can also demonstrate the anatomical abnormality. Symptomatic patients should be treated by the division of the medial head of gastrocnemius and/or reconstruction of the popliteal artery. Surgery may be indicated for an asymptomatic contralateral limb whenever anatomical entrapment is detected. The functional variant of the condition, unlike the anatomical variant, should only be treated when symptomatic.
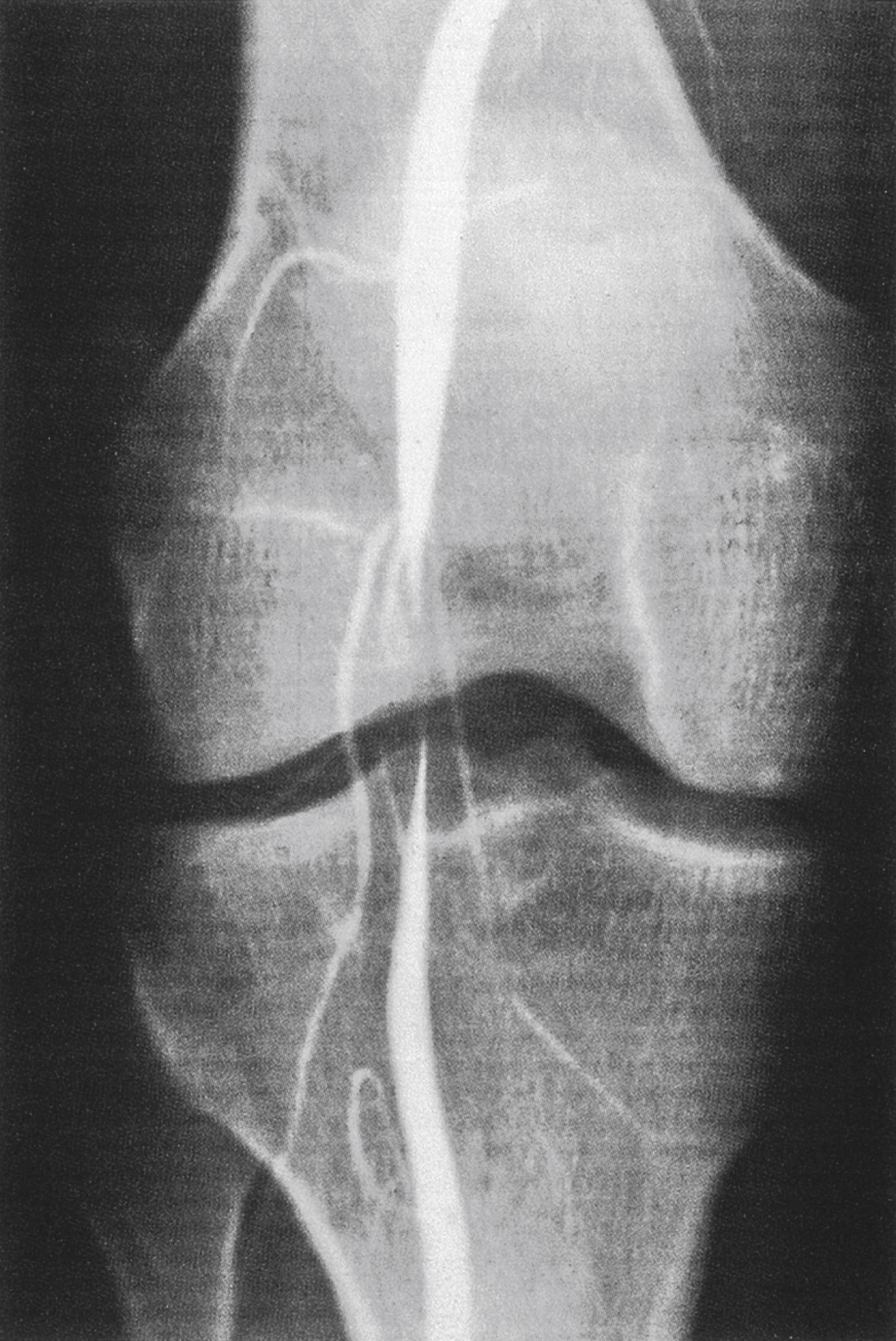
Cystic adventitial disease (CAD) is caused by cyst formation in the adventitia of the artery because of implantation of mucin-secreting mesenchymal cells on the adventitial wall during development. CAD commonly affects the popliteal artery and less often the external iliac and femoral vessels. It predominantly affects males in the mid-30s, is unilateral in presentation and patients commonly present with IC. The contents of the cyst resemble that of a ganglion and the cysts may be connected to the synovium of the knee joint. IC may be severe and of rapid onset. The condition should be particularly suspected in young patients without significant risk factors for PAD. Pedal pulses sometimes disappear on knee flexion (Ishikawa’s sign). Arteriography may show an unusually smooth ‘hourglass’ stenosis referred to as the ‘ scimitar’ sign ( Fig. 2.2 ). Ultrasound scanning will demonstrate the cystic abnormality with absence of flow within it and CT may help delineate CAD from popliteal entrapment syndrome and aneurysm. The appearance of CAD on MR imaging (MRI) is distinctive. CAD demonstrates homogenous low intensity signal on T1-weighted images and high signal intensity on T2-weighted images. On post-contrast T1-weighted images, the cyst does not enhance and can be seen compressing the arterial lumen. The distinctive findings on MRI make this modality the imaging technique of choice when the condition is suspected or diagnosed on ultrasound. Resection of the affected segment of artery and repair with an interposition vein graft via a posterior approach is the most widely practised technique and is mandatory in popliteal artery thrombosis or in extensive arterial involvement. Non-resectional techniques such as cyst excision or evacuation have been described with some success although a high incidence of recurrence has been observed with these techniques.
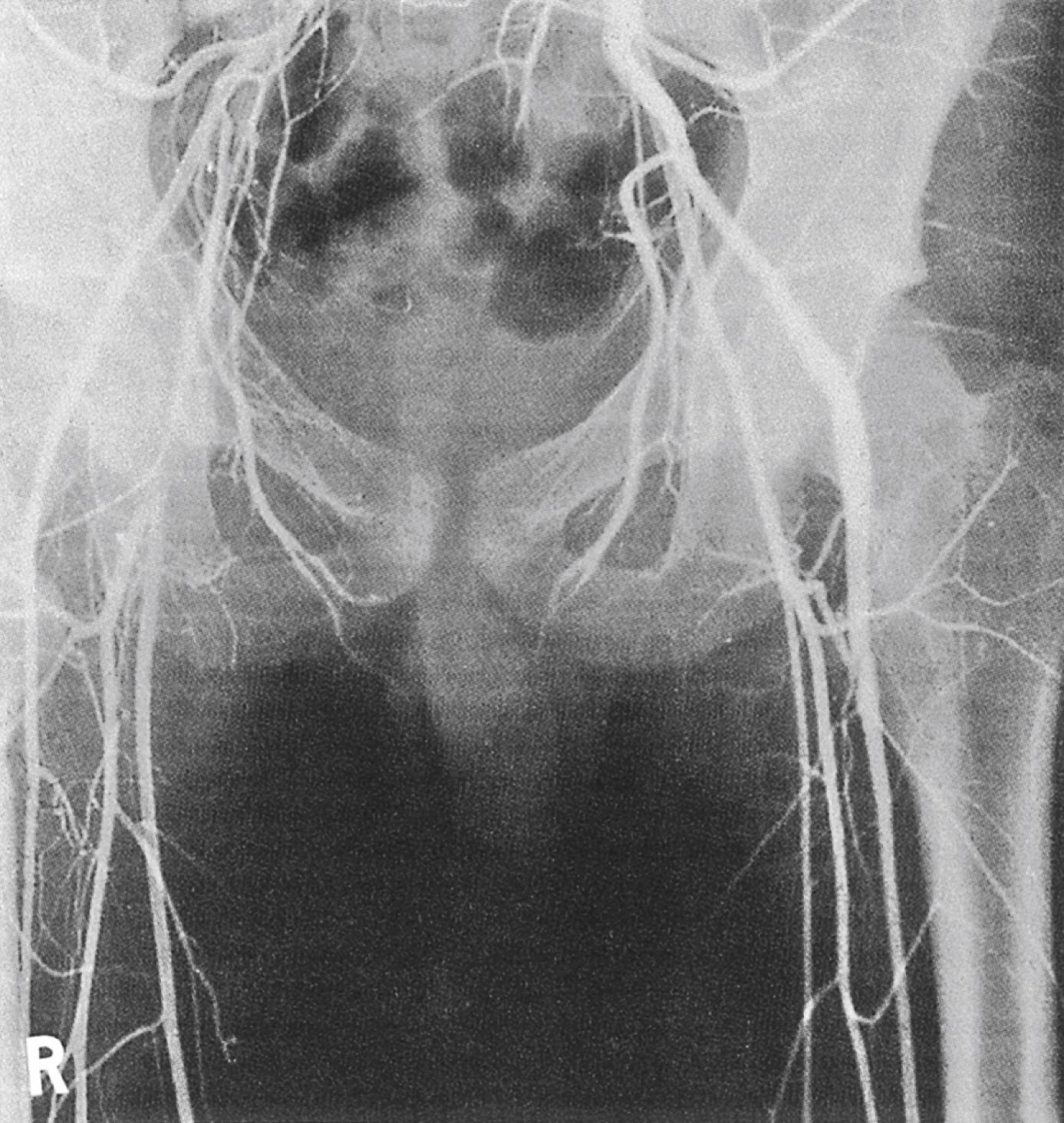
This is a rare congenital anomaly with only a couple of hundred cases reported in the literature. In early embryonic development, the sciatic artery, which is the embryonic limb artery, disappears when the superficial femoral artery has developed. However, in persistent sciatic artery (PSA), the sciatic artery does not obliterate and remains continuous with the popliteal artery, providing the major blood supply to the lower limb. The anomaly is bilateral in approximately 30% of cases and is commonly associated with failure of the iliofemoral vessels to develop normally. The presenting symptoms are variable and include IC, limb ischaemia, a pulsatile mass in the buttock or neurological symptoms from sciatic nerve compression. Aneurysmal degeneration of the artery as it emerges from the sciatic foramen is a common presentation and thrombosis or distal embolism may lead to acute ischaemia. In a review of 159 patients with PSA, the mean patient age at the time of diagnosis was 59 years and 80% were symptomatic. Although pedal pulses may be present, the femoral pulse will be reduced or absent if the iliofemoral vessels are hypoplastic (Cowie’s sign) and IC will result if neither system has developed properly ( Fig. 2.3 ). Symptomatic patients can be treated by combined bypass grafting and endovascular exclusion of the aneurysm. Asymptomatic patients should be monitored for aneurysm development.
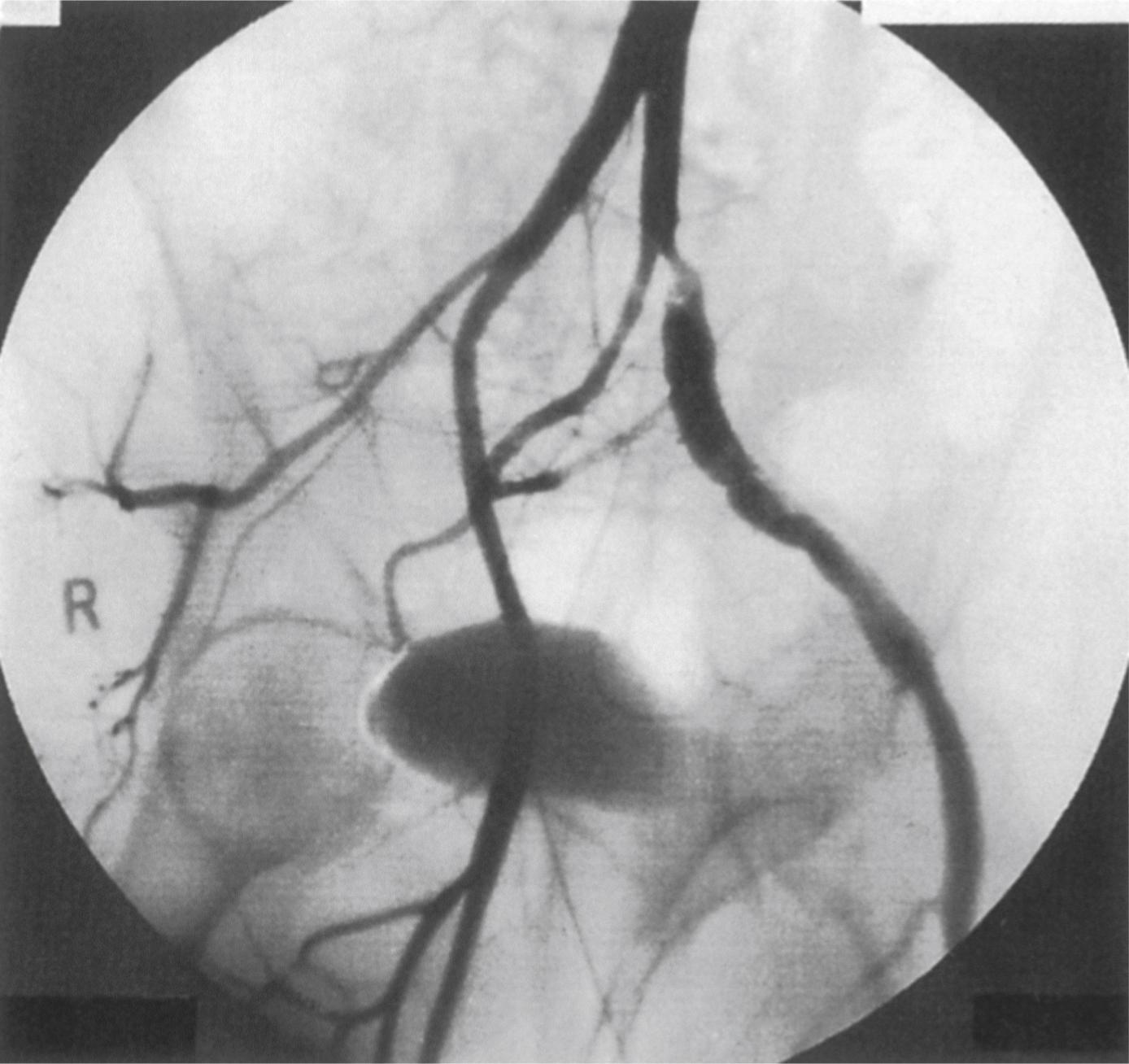
Fibromuscular dysplasia (FMD) is a non-atherosclerotic and non-inflammatory arterial disease, which is more commonly seen in middle-aged women and usually affects the renal, carotid and vertebral arteries. It can also affect the iliac, femoral or popliteal arteries although this is less common. In younger patients, FMD can cause IC or microembolisation and rarely severe limb ischaemia from arterial dissection. Depending on the layer of the arterial wall that is predominantly affected, three varieties of FMD are recognised – medial fibroplasia (most common type), intimal fibroplasia and adventitial fibroplasia. It is characterised by luminal fibrotic webs that give rise to stenoses and post-stenotic dilatations giving the classic beaded appearance. The external iliac artery is the commonest site of involvement in the lower limb vasculature ( Fig. 2.4 ). Patients with iliac FMD should be screened for renal and carotid/vertebral involvement. Symptomatic stenoses usually respond well to angioplasty. Stenting should be reserved for cases with suboptimal angioplasty results or procedural complications following angioplasty.
This is a rare cause of arterial stenosis seen particularly in the external iliac artery among competitive cyclists. It is thought to develop as a result of repetitive trauma to the external iliac artery and the commonest presentation is IC with maximal exertion. Clinical examination may be normal and occasionally, a femoral bruit may be heard. Exercise testing with immediate post-exercise ABIs and duplex ultrasound (DUS) help confirm the lesion. DUS and contrast angiography may demonstrate concentric stenosis but the lesions may be very subtle at rest. Techniques widely reported for repair are endarterectomy and patch angioplasty and interposition graft. ,
Buerger’s disease (thromboangiitis obliterans) is a systemic vasculopathy associated with tobacco use that affects medium-sized arteries and veins in both the upper and lower limbs. It should be considered in any heavy-smoking young male patient with claudication, especially if they are of Middle or Far Eastern origin. Vasospastic symptoms and superficial thrombophlebitis commonly occur and patients may progress rapidly to, or present with, CLTI. The pathophysiology and management of this condition and other causes of vasculitis such as Takayasu’s are covered in Chapter 12 . Patients who smoke large quantities of cannabis may present with symptoms and imaging indistinguishable from those of Buerger’s disease.
The crural vessels are usually severely affected, with patent arteries to the knee joint and typical ‘corkscrew’ collaterals in the calf.
A meticulous approach to the patient presenting with suspected arterial disease is paramount in the diagnosis of PAD. This includes a detailed history to delineate symptoms from other conditions, clinical examination and non-invasive hemodynamic tests to stage severity followed by dedicated vascular imaging for those presenting with CLTI or disabling claudication.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here