Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Acute aortic dissection is an uncommon but potentially catastrophic illness that occurs with an incidence of approximately 3.5 per 1 million person-years, with at least 8000 cases occurring annually in the United States.
Aortic stent grafts are primarily used to reconstruct the compressed true lumen cranial to major aortic branches and to increase distal aortic flow. Proximal communications are sealed to direct flow into the true lumen, depressurize the false lumen, and induce thrombosis in the false lumen, with fibrotic transformation and subsequent remodeling of the aortic wall.
Percutaneous stent graft placement in the dissected aorta is safer and produces better results than surgery for type B dissections.
Use of endovascular stent grafts for repair of thoracic aortic aneurysms is a promising and less invasive therapeutic alternative to conventional surgical treatment. Most devices use a metal skeleton made of stainless steel, nitinol, or Elgiloy.
Evaluation for repair of a thoracic aortic aneurysm considers the patient’s overall risk profile, evidence of rapid enlargement of the aneurysm, diameter equal to or greater than 5.5 cm, and symptoms. Suitability for endovascular repair is based on clinical and anatomic considerations.
Endoleaks are the most prevalent complications after stent graft treatment of thoracic aortic aneurysms. Treatment options include transcatheter coil or glue embolization, balloon angioplasty, placement of endovascular graft extensions, and open repair.
Procedural success and long-term outcome of endovascular repair of abdominal aortic aneurysms depend on proximal and distal fixation and sealing.
Acute aortic dissection is an uncommon but potentially catastrophic illness. It occurs with an incidence of approximately 3.5 per 100,000 person-years, and at least 8000 cases occur each year in the United States. About 0.5% of patients with chest or back pain have an aortic dissection. Two-thirds of patients are male, with an average age at presentation of approximately 65 years. A history of systemic hypertension, found in up to 72% of patients, is by far the most common risk factor.
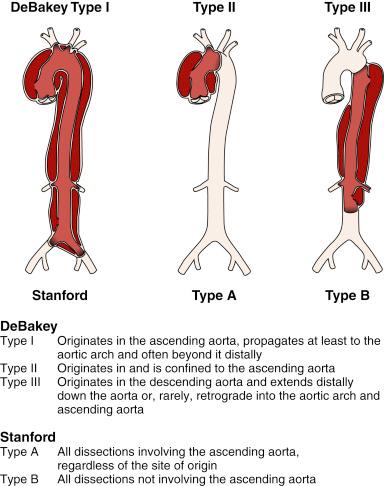
Acute aortic dissection is diagnosed within 2 weeks of onset of symptoms, which is the high-mortality period. Patients surviving 2 weeks are considered to have subacute disease, and chronic aortic dissection is diagnosed after 8 weeks. Aortic dissections are further classified according to their anatomic location using the Stanford and DeBakey classifications. The fundamental distinction is whether the dissection is proximal (i.e., involving the aortic root or ascending aorta) or distal (i.e., beyond the left subclavian artery) ( Fig. 44.1 ).
Important precursors of typical aortic dissection include intramural hematoma (IMH), penetrating aortic ulcers (PAU), and localized intimal tears that are variants of a wall-dissecting process. Chronic hypertension affects arterial wall composition, causing intimal thickening, fibrosis, and calcification and extracellular fatty acid deposition. The extracellular matrix also undergoes accelerated degradation, apoptosis, and elastolysis with hyalinization of collagen. Both mechanisms may eventually lead to intimal disruption. Adventitial fibrosis may obstruct nutrient vessels feeding the arterial wall and the small intramural vasa vasorum. Both result in necrosis of smooth muscle cells and fibrosis of elastic structures, rendering the vessel wall vulnerable to pulsatile forces and creating a substrate for aneurysms and dissections.
In addition to chronic hypertension, smoking, dyslipidemia, and use of crack cocaine are risk factors. Rarely, inflammatory diseases destroy the media layers and cause weakening, expansion, and dissection of the aortic wall. Iatrogenic aortic dissection may be associated with invasive retrograde catheter interventions or occur during or after valve or aortic surgery.
Men are affected twice as often as women by acute aortic dissection, with 60% of cases classified as proximal (type A) and 40% as distal (type B) according to the Stanford classification. Historical data for untreated aortic dissection of the ascending aorta show a mortality rate of 1% to 2% per hour within the first 24 hours, resulting in a mortality rate of up to 50% to 74% during the acute phase. Uncomplicated acute type B dissection is less frequently lethal, with survival rates for medically treated patients of 89% at 1 month, 84% at 1 year, and up to 80% within 5 years. Survival may be significantly improved by the timely institution of appropriate therapy. Prompt clinical recognition and definitive diagnostic testing are essential in the management of aortic dissection.
Conventional treatment of Stanford type A (DeBakey types I and II) dissection (see Fig. 44.1 ) consists of surgical reconstruction of the ascending aorta with complete or partial resection of the dissected aortic segment. In type A dissections, interventional endovascular strategies have no clinical application except to relieve critical malperfusion before surgery of the ascending aorta. This is done by distal endovascular interventions in cases of thoracoabdominal extension of a proximal dissection (DeBakey type I) with distal ischemic complications. In Stanford type B dissections, stent graft placement aims to remodel the thoracic descending aorta, typically by sealing one or multiple proximal entry tears with a Dacron-covered stent, initiating thrombosis of the false lumen.
Reconstruction of a collapsed true lumen may reestablish side branch flow ( Fig. 44.2 ). Various scenarios of malperfusion syndrome are amenable to endovascular management, including static or dynamic collapse (i.e., intima invagination) of the aortic true lumen (i.e., pseudocoarctation) ( Fig. 44.3 ), static or dynamic occlusion of one or more vital side branches, and an enlarging false aneurysm due to a patent proximal entry tear. Although peripheral pulse deficits often can be reversed with surgical repair of the dissected thoracic aorta, patients with signs of mesenteric or renal ischemia do not fare well. Mortality rates for patients with renal ischemia are between 50% and 70% and may be as high as 87% in cases with mesenteric ischemia.
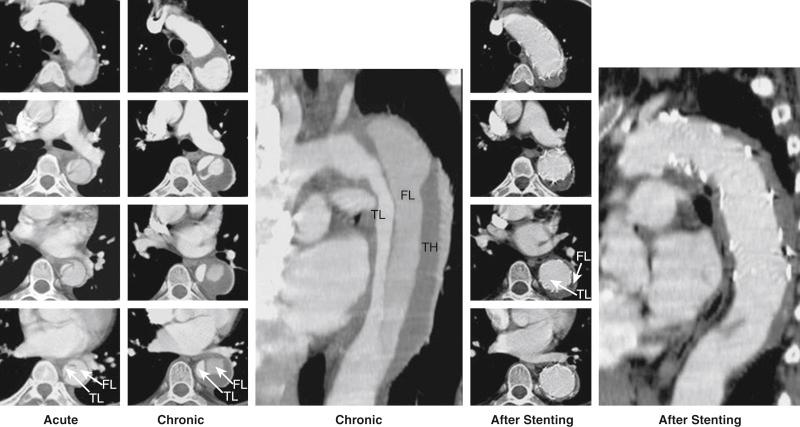
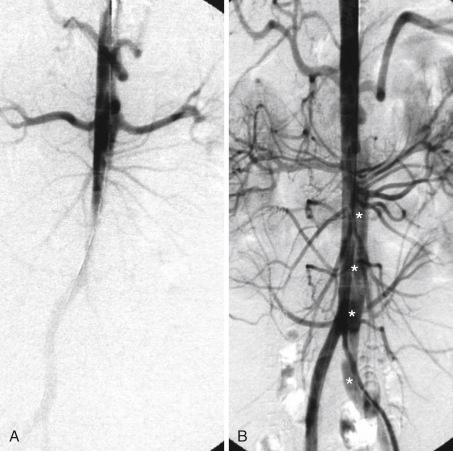
Surgical mortality rates for patients with acute peripheral vascular ischemic complications are similar to those for patients with mesenteric ischemia, and the in-hospital mortality rate may reach 89%. Operative mortality rates for surgical fenestration are between 21% and 61%, which has encouraged percutaneous interventional management by endovascular balloon fenestration of a dissecting aortic membrane to treat mesenteric ischemia, a concept often discussed as a niche indication in complicated cases of malperfusion.
Management of Stanford type B (DeBakey type III) dissection with the use of endovascular stent grafts has evolved slowly in anticipation of an unknown risk of paraplegia from spinal artery occlusion, as seen in up to 18% of cases after open surgery. After technical improvements, a large series of patients has been successfully treated in various specialized centers by endovascular stent grafts placed to cover entry tears in the descending aorta and in the aortic arch. Studies have demonstrated that closure of proximal entry tears is essential to reconstruct the aortic wall and reduce the total aortic diameter. Entry tear closure promotes depressurization of the false lumen, thrombus formation in the false lumen ( Fig. 44.4 ), and remodeling of the entire aorta. In the near future, combined surgical and interventional procedures are likely to be developed for proximal dissections.
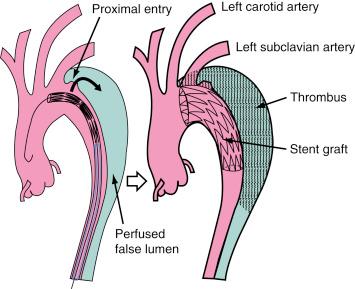
Significant perioperative morbidity and mortality in cases of acute, complicated type B dissection have prompted alternative therapeutic concepts. Thoracic endovascular aortic repair (TEVAR), even though it is an off-label indication for dissection, has been approved by the U.S. Food and Drug Administration (FDA) for aneurysmal disease of the aorta. The natural course of aortic dissection is determined by two elements: early complications and late events. In the acute phase, TEVAR has abrogated impending rupture and relieved static and dynamic malperfusion. A later benefit appears to be false lumen thrombosis, mitigating the risk of aneurysmal dilation and subsequent rupture.
The role of interventional management of static or dynamic obstruction of aortic branch arteries in complicated and complex distal dissection was settled in 2010, when static obstruction of a branch was overcome by placing endovascular stents in the ostium of a compromised side branch. Dynamic obstruction may benefit from stents in the aortic true lumen, sometimes combined with side branch stenting and preferentially without additional balloon fenestration because fenestration does not improve stress and tension on the thin aortic wall.
Sometimes, bare-metal stents deployed from the true lumen into side branches can buttress the flap in a stable position. In rare cases, fenestration helps to create a reentry tear for the dead-end false lumen back into the true lumen, with the aim of preventing thrombosis of the false lumen and compromise of branches fed exclusively from the false lumen; however, this concept lacks clinical proof of benefit. Conversely, fenestration increases the long-term risk of aortic rupture because a large reentry tear promotes flow in the false lumen, which promotes aneurysmal expansion. There is also a risk of peripheral embolism from a patent but partly thrombosed false lumen.
The most effective method to avoid an enlarging false lumen is sealing of proximal entry tears with a customized stent graft ( Fig. 44.5 ). Absence of a distal reentry tear is desirable for optimal results but is not a prerequisite. Compression of the true aortic lumen cranial to the main abdominal branches with distal malperfusion (i.e., pseudocoarctation) is usually corrected by stent grafts that expand the compressed true lumen and improve distal aortic blood flow. Depressurization and shrinking of the false lumen is the most beneficial result to be gained, ideally followed by complete thrombosis of the false lumen and remodeling of the entire dissected aorta (see Fig. 44.2 ).

Like previously accepted indications for open surgical repair in complicated type B dissections, factors such as intractable pain with descending dissection, rapidly expanding false lumen diameter, extraaortic blood collection as a sign of imminent rupture, and distal malperfusion syndrome are accepted indications for emergent stent graft placement. Even late-onset complications such as malperfusion of vital aortic side branches are likely to justify endovascular stent grafting of an occlusive lamella to improve distal true lumen flow as a first option. Surgery is employed only after an unsuccessful attempt because surgical repair has failed to prove superior to interventional treatment even in uncomplicated cases. In complicated cases, endoluminal treatment has replaced open surgery in advanced aortic centers. Even in some cases of retrogradely extended type A dissections, TEVAR is feasible as a primary approach to seal the entry or as a secondary step after open repair of the ascending aorta. Open surgery may include an “elephant trunk” or transposition of arch vessels to provide optimal landing zones for endovascular completion in a hybrid approach. Treatment options are summarized in Table 44.1 .
| Surgery |
|---|
|
| Medical Therapy |
|
| Interventional Therapy |
|
Patients with suspected acute aortic dissection should be admitted to intensive care and subjected to immediate diagnostic evaluation. Reduction of systolic blood pressure to 100 to 120 mm Hg with an eye on renal function and pain relief is an initial priority and is usually achieved by morphine sulfate and intravenous β-blocking agents with or without vasodilating drugs such as sodium nitroprusside or angiotensin-converting enzyme inhibitors ( Table 44.2 ).
| Drug | Mechanism | Dose | Cautions and Contraindications |
|---|---|---|---|
| Esmolol | Cardioselective β 1 -blocker | Load: 500 μg/kg IV Drip: 50 μg/kg/min IV Increase by increments of 50 μg/min |
Asthma or bronchospasm Bradycardia Second- or third-degree AV block Cocaine or methamphetamine abuse |
| Labetalol | Nonselective β 1,2 -blocker Selective α 1 -blocker |
Load: 20 mg IV Drip: 2 mg/min IV |
Asthma or bronchospasm Bradycardia Second- or third-degree AV block Cocaine or methamphetamine abuse |
| Enalaprilat | ACE inhibitor | 0.625–1.25 mg IV q6h Maximum dose: 5 mg q6h |
Angioedema Pregnancy Renal artery stenosis Severe renal insufficiency |
| Nitroprusside | Direct arterial vasodilator | Begin at 0.3 μg/kg/min IV Maximum dose: 10 μg/kg/min |
May cause reflex tachycardia Cyanide/thiocyanate toxicity, especially in renal or hepatic insufficiency |
| Nitroglycerin | Vascular smooth muscle relaxation | 5–200 μg/min IV | Decreases preload; contraindicated in tamponade or other preload-dependent states Concomitant use of sildenafil or similar agents |
The heart rate should be kept low; a heart rate below 60 beats per minute significantly decreases secondary adverse events (e.g., aortic expansion, recurrent aortic dissection, aortic rupture, need for aortic surgery) in type B aortic dissections compared with a conventional rate of more than 60 beats per minute. After stable blood pressure and symptom relief are obtained, a patient with an uncomplicated type B aortic dissection can be discharged (usually within 14 days) on oral drugs. Clinical and imaging follow-up are advised at 3 and 6 months and annually thereafter.
Of 384 patients with type B dissections from the International Registry of Aortic Dissection (IRAD), 73% were managed medically with an in-hospital mortality rate of 10%. Short-term survival rates were 91% at 1 month and 89% at 1 year. The reported long-term survival rate with medical therapy varies between 60% and 80% at 4 to 5 years and is about 40% to 45% at 10 years. Potential beneficial effects of early stenting is proved in the ADSORB trial (Acute Uncomplicated Aortic Dissection Type B: Evaluation of Stent Graft Placement or Best Medical Treatment Alone). Of 61 patients being randomized in best medical therapy group ( n = 31) and best medical therapy group in addition to aortic stent graft placement ( n = 30), the latter group had a significantly lower rate of primary end point, which was a combination of incomplete/no false lumen thrombosis, aortic dilatation, or aortic rupture at 1 year ( P < .001).
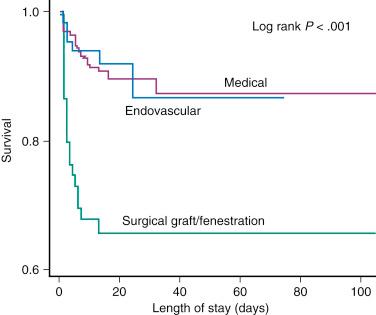
About 30% to 42% of acute type B aortic dissections are complicated, as evidenced by hemodynamic instability or peripheral vascular ischemia, and they have an unpredictable outcome. Acute lower limb and visceral ischemia has been reported in 30% to 50%, and malperfusion syndrome occurs frequently with extended dissections, with mortality between 50% and 85% if untreated. However, operative mortality rates for patients with acute aortic dissection complicated by renal ischemia are about 50% and can be as high as 88% for those with mesenteric malperfusion. Of 571 patients with acute type B aortic dissection in the IRAD, 390 were treated medically. In the 125 complicated cases, 59 patients underwent standard open surgery, and 66 were subjected to TEVAR. The in-hospital mortality rate was significantly lower with TEVAR than after open surgery (10.2% vs. 33.9%, P = .002) ( Fig. 44.6 ).
The provisional extension to induce complete attachment (PETTICOAT) technique pushes the idea of endothoracic reconstruction further by extending the stent graft scaffold distally with open-cell bare-metal stents until distal malperfusion is corrected. Using this technique, aortic fenestration maneuvers and branch vessel revascularization with side branch stents usually are not needed and have almost become obsolete.
A meta-analysis of outcomes for 942 patients from 29 studies found an in-hospital mortality rate of 9% and reintervention rate of 10.4%. Emergency surgical conversion or periprocedural stroke was rare (0.6% and 3.1%, respectively), and the survival rate was 88% at mean follow-up of 20 months. A second meta-analysis of outcomes for 1304 patients subjected to TEVAR for complicated acute type B aortic dissection found a technical success rate of 99% and a 30-day mortality rate of 2.6%. At late follow-up, false lumen thrombosis was documented in 92.9% of patients, and surgical conversion was required in 0.8%, with endovascular reintervention performed in 1.6%. Retrograde extension into the ascending aorta and neurologic complications were reported in 0.4% and 0.6%, respectively. Further results are listed in Table 44.3 and elsewhere.
| Study | Year | n | Technical Success (%) | Paraplegia (%) | Mortality (%) | Follow-Up (Months) |
|---|---|---|---|---|---|---|
| Acute Complicated Type B Dissection | ||||||
| Bortone et al. | 2004 | 43 | 100 | 0 | 7 | 21 |
| Xu et al. | 2006 | 63 | 95 | 0 | 10.6 | 48 |
| Verhoye et al. | 2008 | 16 | 100 | 0 | 27 | 36 |
| Fattori et al. | 2008 | 66 | 100 | 3.4 | 10.6 | 1 |
| Szeto et al. | 2008 | 35 | 97.1 | 2.8 | 2.8 | 18 |
| Khoynezhad et al. | 2009 | 28 | 90 | NA | 18 (1 year) 22 (5 years) |
36 |
| Alves et al. | 2009 | 106 | 99 | 1.8 | 18 (acute AD) 7 (chronic AD) |
35.9 |
| Parsa et al. | 2010 | 55 | 100 | 2 | 37 (overall) 6 (aorta related) |
14.4 |
| Shu et al. | 2011 | 45 | 100 | NA | 4.4 (1 year) | 13 |
| Ehrlich et al. | 2013 | 29 | 100 | 10 | 21 (1 year) 39 (5 years) |
53 |
| Wiedemann et al. | 2014 | 110 | 100 | 4.5 | 15 (1 year) 27 (5 years) |
60 |
| Massmann et al. | 2014 | 14 | 93 | 0 | 0 | 30 |
| Bavaria et al. | 2015 | 50 | 100 | NA | 15 (1 year) | 12 |
| Chronic Type B Dissection | ||||||
| Nienaber et al. | 1999 | 12 | 100 | 0 | 0 | 12 |
| Kato et al. | 2001 | 15 | 100 | 0 | 0 | 24 |
| Eggebrecht et al. | 2005 | 28 | 100 | 0 | 13.6 | 12 |
| Jing et al. | 2008 | 35 | 100 | 0 | 7.6 | 48 |
| Nienaber et al. | 2009 | 72 | 95.7 | 2.8 | 11.1 (overall) 5.6 (aorta related) |
24 |
| Kang et al. | 2011 | 76 | 100 | 0 | 15.7 | 34 |
| Nienaber | 2013 | 72 | 100 | 2.8 | 11.3 (overall) 6.9 (aorta) |
60 |
| Andersen | 2014 | 75 | 100 | 0 | 14 (1 year) 35 (5 years) |
34 |
| Lu et al. | 2015 | 51 | 100 | 0 | 1.9 (3.5) | 44 |
| Conway et al. | 2017 | 125 | 98.4 | NA | 2.4 (in-hospital) | 8 |
In patients deemed unsuitable candidates for conventional open surgical repair, 1- and 5-year survival rates were 74% and 31% with TEVAR compared with 93% and 78% ( P < .001) survival rates for patients considered candidates for conventional open repair. A comparison between endovascular treatment of complicated type B aortic dissection and medical therapy of uncomplicated type B dissection in 56 patients with a follow-up of 18.1 ± 16.9 months found similar outcomes in both groups but a better midterm fate of the descending thoracic aorta in the stent graft group and no associated paraplegia. There were no differences in the 5-year survival rates (86.3% for both groups).
Evolution of an acute dissection to a chronic dissection involves progressive thickening of the intimal flap due to fibrosis. More intimal tears are reported in chronic type B aortic dissection than acute dissection. The growth rate of the chronically dissected distal aorta is estimated to be between 0.10 and 0.74 cm in diameter per year, depending on the initial aortic diameter and state of hypertension.
Unfortunately, the long-term outcome after medical therapy alone is suboptimal, with a reported 50% mortality rate at 5 years and delayed expansion of the false lumen in 20% to 50% of patients at 4 years. Expansion of the false lumen, for which an initial diameter beyond 4 cm and persistent perfusion of the false lumen are predictors, predisposes patients to aortic rupture or retrograde migration of the dissection toward the ascending aorta. TEVAR should be considered when the aortic diameter exceeds 5.5 mm or when there is persistent thoracic pain or uncontrolled hypertension and rapid growth of the dissecting aneurysm (>1 cm/year).
Our group prospectively evaluated elective TEVAR in 12 patients with chronic type B dissection and compared the results with 12 matched surgical controls. Proximal entry closure and complete thrombosis of the false lumen at 3 months were achieved in all patients. Stent graft treatment resulted in no morbidity or mortality, whereas surgical treatment resulted in four deaths (33%, P = .04) and five adverse events (42%, P = .04). These results were confirmed by similar observations (see Table 44.3 ).
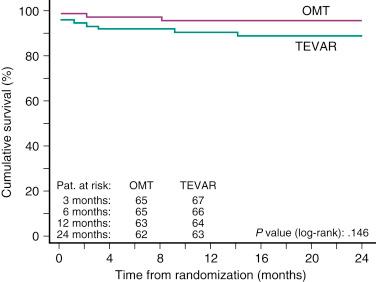
Whether prophylactic use of TEVAR in patients with chronic type B aortic dissections is superior to medical treatment alone was evaluated in the prospective, randomized, controlled Investigation of Stent Grafts in Aortic Dissection (INSTEAD) trial. A total of 140 patients in stable clinical condition at least 2 weeks after the index dissection were randomly subjected to elective stent graft placement in addition to optimal medical therapy ( n = 72) or to optimal medical therapy alone ( n = 68). There was no difference in the all-cause death rate and a 2-year cumulative survival rate of 95.5% ± 2.5% with optimal medical therapy versus 88.9% ± 3.7% with TEVAR ( P = .15) ( Fig. 44.7 ). Moreover, the aorta-related death rate was not different (2.9% vs. 5.6%, P = .68), and the risk for the combined end point of aorta-related death and progression was similar ( P = .65). Aortic remodeling (with true lumen recovery and thoracic false lumen thrombosis) occurred in 91.3% of patients with TEVAR and in 19.4% of those who received medical treatment ( P < .001), which suggests ongoing aortic remodeling.
The 5-year follow up, which was evaluated in the INSTEAD-XL trial, revealed an all-cause mortality rate of 11.1% for TEVAR versus 19.3% for medical therapy ( P = .13), whereas the aorta-specific mortality rate was 6.9% versus 19.3 ( P = .04), with a progression rate of 27.0% versus 46.1% ( P = .04). Landmark analysis suggested a benefit of TEVAR for all-cause mortality (0% vs. 16.9%, P = .0003), aorta-specific mortality (0% vs. 16.9%, P = .0005), and progression (4.1% vs. 28.1%, P = .004) between 2 and 5 years.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here