Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Acute aortic dissection is the most common catastrophic event affecting the aorta, with an incidence exceeding that of ruptured abdominal aortic aneurysm. Without treatment, the majority of patients with aortic dissections will die within 3 months of presentation, and few survive the chronic phase more than 5 years due to aneurysmal degeneration and rupture of the outer wall of the false lumen. , Despite improvements in both medical and surgical therapeutic options, overall mortality associated with acute dissection remains significant. In a population-based epidemiologic study by Clouse et al., 38% of aortic dissections were diagnosed at autopsy. This substantial mortality rate in undiagnosed patients underscores the importance of early diagnosis and initiation of appropriate therapy. Indeed, Khan et al. predicted that the mortality of acute dissection (type A and B) left untreated will exceed 22.7% within 6 hours, 50% within 24 hours, and 68% within the first week. Death due to an acute dissection of the ascending thoracic aorta is usually secondary to the central cardio-aortic complications of aortic rupture into the pericardium, acute aortic regurgitation, and coronary ostia compromise, , while patients with descending thoracic aortic dissections are more likely to die from end organ compromise due to obstruction of visceral or extremity vessels in the acute phase of the disease. ,
The management of acute type B aortic dissections has shifted over the last decade. Contributions from multicenter collaborations such as The International Registry of Acute Aortic Dissection (IRAD), the Investigation of Stent Grafts in Aortic Dissection (INSTEAD) trial, and large single institution series have cast doubt on the ability of medical therapy alone to effectively treat acute type B aortic dissections, and it is clear that there is a subset of uncomplicated patients who will benefit from early intervention with a thoracic endovascular aortic repair (TEVAR).
This chapter reviews the classification, pathologic anatomy, clinical presentation, diagnostic, and treatment modalities for acute aortic dissection, with emphasis on the role of the vascular/endovascular surgeon. While a thorough understanding of all components of acute dissection seems requisite for those who propose to treat aortic dissection (AD), the technical principles of graft replacement of the ascending aorta, typically performed by cardiac surgeons, is not detailed. However, it is anticipated that the vascular/endovascular surgeon will become the primary operator in the care of patients with acute dissection of the descending aorta and the complications of peripheral vascular compromise.
Aortic dissections are classified according to the anatomic location of the entry tear and the time between onset of symptoms and patient presentation.
A dissection has traditionally been considered acute within the first 2 weeks after the initial onset of symptoms. While such designation appears arbitrary, it is based on autopsy studies showing that 74% of patients who die from aortic dissections do so in the first 14 days. The period from 2 weeks to 90 days after initial presentation is now considered the subacute phase. During this period the risk of death decreases, but the dissection flap remains pliable such that this is the best time to consider performing elective TEVAR coverage of the proximal entry tear. Finally, after 90 days, the dissection is considered chronic as the flap has become stiff and fibrotic and the aorta will begin to positively or negatively remodel. Indeed, such temporal classification combined with the anatomic location of the entry tear should be used to determine intervention.
Anatomically, aortic dissection is classified based on the location of the intimal tear and the extent of aorta involved in the dissection. There are two classification schemes used to describe aortic dissections. The original, proposed by DeBakey in 1965, delineates both the origin of the entry tear and the extent of the descending aortic dissection. This scheme ( Fig. 83.1 ) uses the following classification:
Type I: Dissection originates in the ascending aorta, extends through the aortic arch, and continues into the descending aorta and/or abdominal aorta for a varying distance.
Type II: Dissection originates in and is confined to the ascending aorta.
Type IIIa: Dissection originates in the descending aorta and is limited to same.
Type IIIb: Dissection involves descending and variable extents of the abdominal aorta.
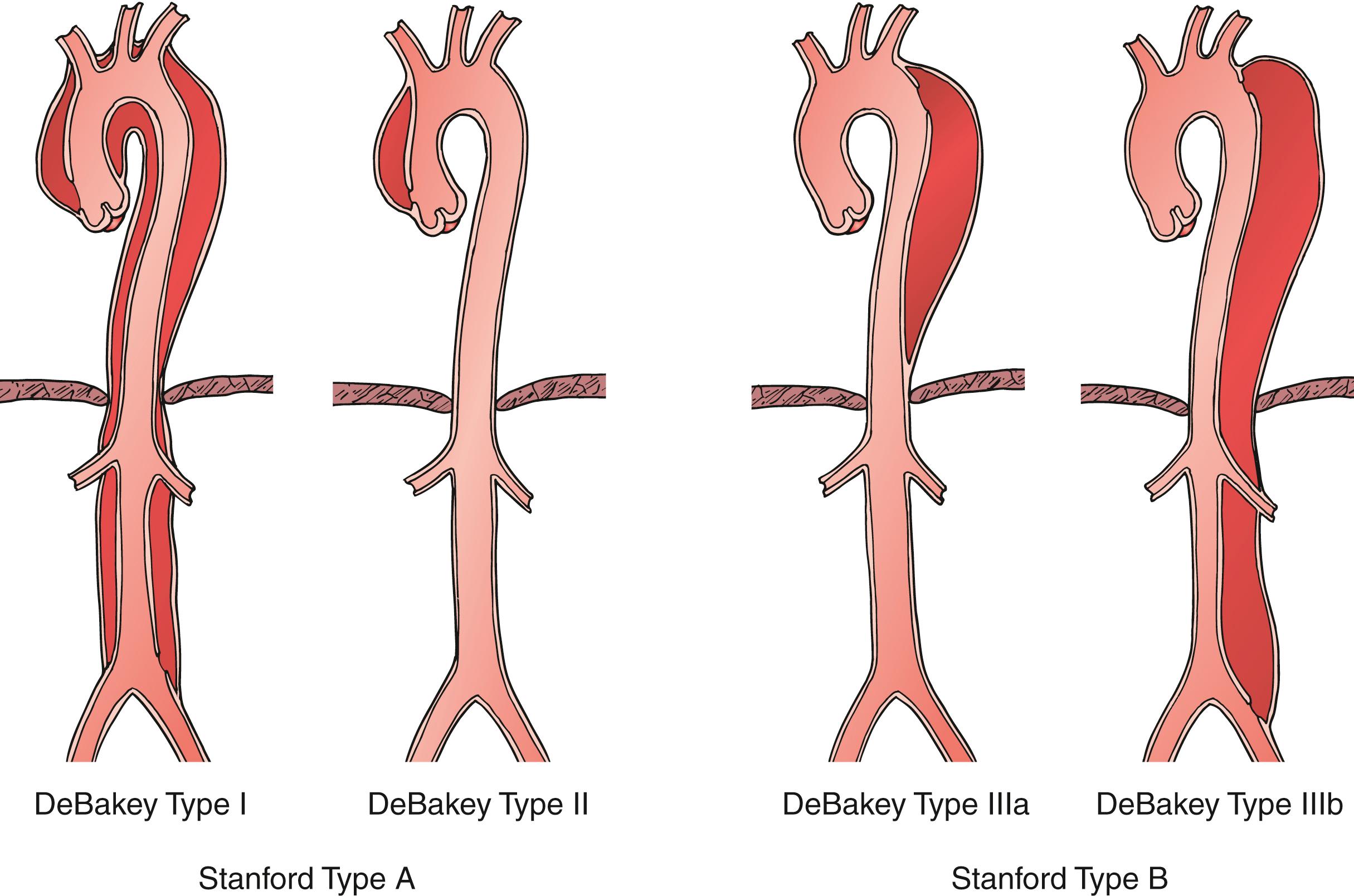
The Stanford Classification that was described by Daily in 1970 simplified the anatomic classification according to the origin of the entry tear alone. A Stanford type A dissection originates in the ascending aorta, and therefore encompasses DeBakey type I and II dissections, while in a Stanford type B dissection, the entry tear originates in the descending aorta distal to the origin of the left subclavian artery (DeBakey type IIIa, IIIb) (see Fig. 83.1 ).
Because the origin of the entry tear is the key predictor of early outcomes, most patients are now stratified into Stanford type A or B at the time of presentation in order to direct initial therapy. However, neither the Stanford nor the DeBakey classification systems address patients whose entry tear originates in the aortic arch between the innominate and left subclavian arteries. The classic Stanford type A dissection is confined to the ascending thoracic aorta, and most cardiac surgeons consider any dissection that originates distal to the innominate artery to be Stanford type B. This is driven by the fact that prompt graft replacement of the ascending aorta is the appropriate treatment for most type A dissections because of the high risk of lethal cardio-aortic complications (principally aortic rupture or myocardial ischemia from extension into the coronary arteries). Alternatively, the classic Stanford type B dissection is confined to the descending thoracic aorta, and vascular surgeons consider any dissection that originates proximal to the left subclavian artery to be type A and the purview of cardiac surgery, because open intervention would require circulatory arrest and placing a stent graft proximal to the left common carotid artery requires surgical debranching or access to investigational branched and fenestrated devices. This distinction underscores the importance of engaging in a multi-specialty approach to patients who present with an acute AD.
Population-based studies have estimated the incidence of acute aortic dissection to range from 2.9 to 3.5 per 100,000 person-years. , Factors that predispose one to develop aortic dissection include age, hypertension, and structural abnormalities of the aortic wall. , Men are more frequently affected, with a male/female ratio of 3 or 4:1. , In the author’s cumulative experience, type A dissections account for 60% of cases, and this is consistent with the IRAD data, wherein 62.5% of 1417 patients presented with a type A dissection. The incidence of type A dissection peaks between 50 and 60 years, while type B dissections occur more frequently between 60 and 70 years of age. Hypertension is prevalent and was present in 72% of patients in our longitudinal database ( Table 83.1 ).
| Variable | MGH (%) | IRAD (%) | Total (%) |
|---|---|---|---|
| Patients | 512 | 1078 | 1590 |
| Age (mean) | 62 years | 62.4 years | |
| Male | 347 (67.8%) | 732 (67.9%) | 1079 (67.9%) |
| Type A | 278 (54%) | 674 (62.6%) | 952 (60%) |
| Hypertension | 361 (70.5%) | 755 (71.8%) | 1116 (70.2%) |
| VC/pulse deficit | 159 (31%) | 259 (27.7%) | 418 (26.3%) |
Acute aortic dissections tend to demonstrate chronobiologic patterns of occurrence. The onset of dissection occurs most frequently in the morning hours between 6 a.m. and 12 p.m., and more often in the winter (28%) than the summer (20%), independent of endogenous climate. , Aortic wall structural abnormalities and the presence of a bicuspid aortic valve with or without its accompanying aortic root dilation are well-established risk factors for ascending aortic dissections. Indeed, the presence of a bicuspid aortic valve has been documented in 7% to 14% of all patients with aortic dissections. , Other aortic diseases, such as coarctation of the aorta, annulo-aortic ectasia, chromosomal abnormalities (Turner syndrome and Noonan syndrome), aortic arch hypoplasia, and hereditary conditions (Marfan syndrome and Ehlers–Danlos syndrome), are also risk factors for the development of acute aortic dissection. Marfan syndrome accounts for 50% of cases of acute aortic dissection in patients less than 40 years of age.
Pregnancy is associated with a fourfold increased risk of aortic dissection and rupture when compared with nonpregnant women of child-bearing age. However, it still remains rare, with an incidence of aortic complications of 5.5 per million pregnant women. Preeclampsia with resultant hypertension is the most common etiology of peripartum aortic dissection, but pregnant women with Marfan syndrome are also at high risk, and the presence of a dilated aortic root (>4 cm) is the best predictor of dissection in the pregnant patient with Marfan syndrome. The most common site of aortic dissection in young (<40 years) patients is the ascending aorta involving the sinuses of Valsalva or the sinotubular junction, while type A dissections in older patients are more likely to originate higher in the ascending aorta.
Cocaine use is a rare cause of acute aortic dissection in otherwise healthy individuals that has an incidence as high as 37% in an urban setting, but was present in only 1.7% of the IRAD data and less than 5% in the author’s institution. , , The typical patient with a cocaine-induced dissection is a young (average age 47 years), male (86%) smoker (92%) with a history of hypertension (82%) and no particular trend toward dissection type (type A 52% vs. type B 48%). One possible explanation for development of acute dissection after cocaine use is the associated catecholamine release, which causes a triad of profound hypertension, vasoconstriction, and increased cardiac output. This creates a dramatic, acute increase in ventricular contraction (shearing) force (dP/dt) on the aortic wall. ,
The process of aortic dissection is dynamic and can occur anywhere along the course of the aorta, resulting in a wide spectrum of clinical manifestations. The pathognomonic lesion is one of an intimal tear followed by blood surging either antegrade (typically) or retrograde (depending on the hemodynamic gradient between the true and false lumens), and cleaving the intima and media layers of the aortic wall longitudinally for a variable distance. , The typical tear is transverse, not circumferential, and the intimomedial layer can be cleaved both longitudinally and circumferentially. The adventitia-bound blood-filled space created between the dissected layers of the aortic wall becomes the false lumen. Fenestrations (connections between the true and false lumens) occur within the intimal flap downstream, usually at branch vessel ostia, which are cleaved by the dissection process; these serve as sites of reentry of blood flow into the true lumen (fenestrations), thus maintaining false lumen patency. The presence of an “intimal flap,” representing the intimomedial septum between the true and false lumen, is the most characteristic pathology in acute aortic dissection. The origin of the intimal flap/tear is in the ascending aorta in 65%, the descending aorta in 25%, and in the arch and abdominal aorta in 10% of patients. In the descending aorta, the intimal tear typically originates within a few centimeters of the left subclavian artery, because this segment of the aorta is subject to the greatest dP/dt pressure fluctuations. , , In the usual pattern of a Stanford type B dissection, the cleavage plane will progress with the distal false lumen on the left posterolateral aspect of the aorta (80% of patients); the celiac, superior mesenteric artery (SMA), and right renal arteries typically emanate from the true lumen; and the left renal artery arises from the false lumen. It is to be emphasized, however, that variations in this pattern are frequently encountered.
One pathologic process that is associated with an increased risk of aortic dissection is medial degeneration of the aortic wall (cystic medial necrosis) that diminishes the structural integrity of the aortic layers. , The central lesion appears to be deterioration of medial collagen and elastin fibers through elastolysis; this is considered to be a factor in most cases of aortic dissection. In particular, classic cystic medial necrosis appears to be an essential feature of several hereditary conditions, such as Ehlers–Danlos syndrome and Marfan disease. Yet, specific connective tissue diseases account for only 10% to 15% of all acute aortic dissections (see Ch. 141 , Arterial Disease in Patients with Connective Tissue Disorders). Even in “normals,” in which dissections occur without any antecedent diagnosis of a specific underlying contributing disease, the degree of medial degeneration still tends to be greater than expected as part of normal aging. The exact cause of this medial degeneration remains unclear, but advanced age and hypertension appear to be the most important factors. , ,
Malperfusion syndrome occurs when there is end organ ischemia secondary to aortic branch compromise from the dissecting process. This can involve one or more vascular beds simultaneously, with early symptoms often subtle and of varying severity over the hours and days after initial symptom onset. Consistent over several series, aortic branch compromise is present in up to 31% of patients with acute aortic dissection, , with progression to the malperfusion syndrome correlating with early mortality. , , Any aortic branch can be affected, and as intuitively suspected, the morbid clinical events will vary as a function of the vascular territory involved. Mesenteric involvement is associated with intestinal infarction, whereas subclavian and/or lower extremity occlusive events are often well-tolerated.
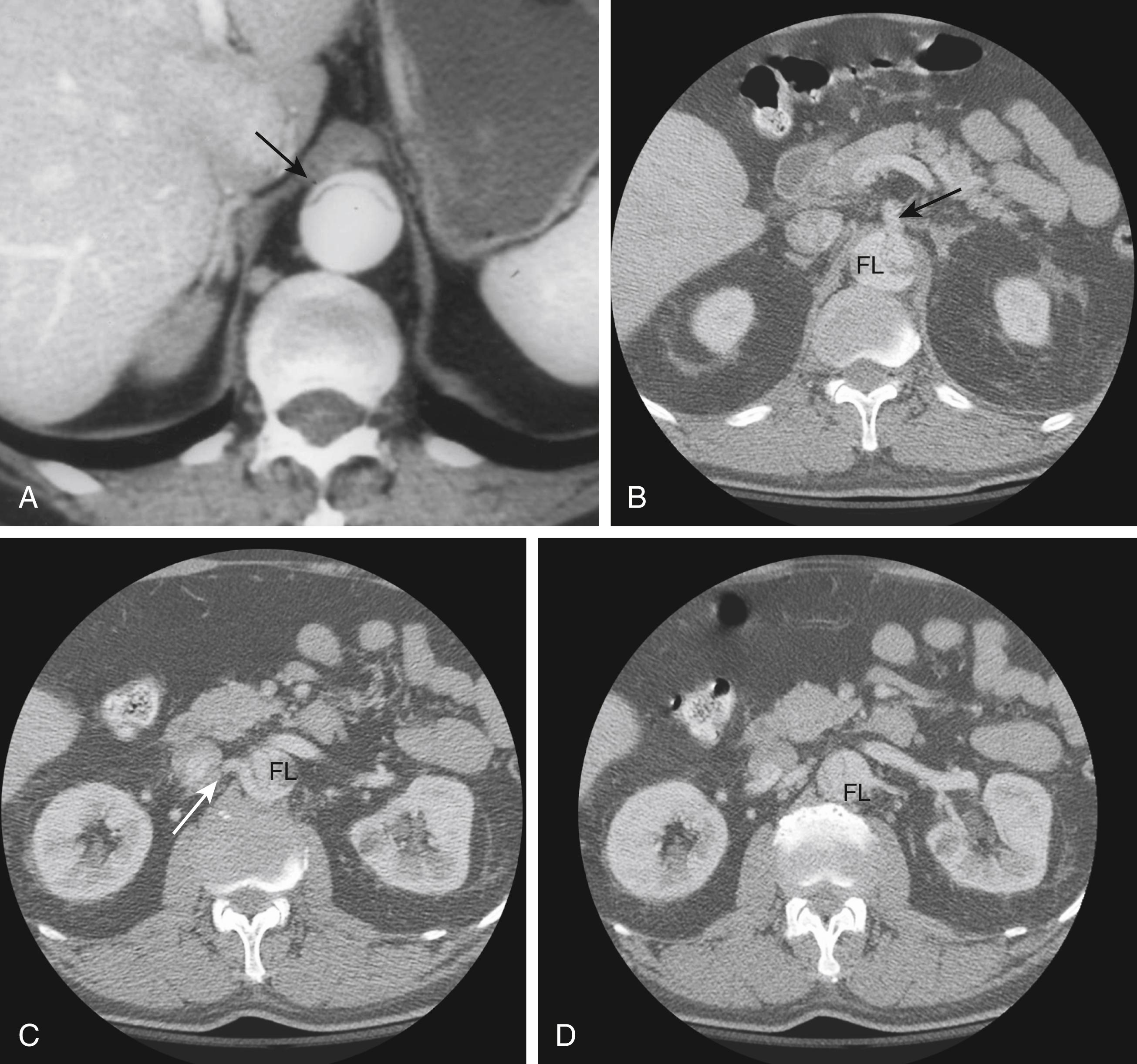
Identifying the mechanisms of branch compromise is a critical step in formulating effective treatment plans. The anatomic and physiologic variables underpinning any compromised vascular bed include (1) the percentage of aortic circumference dissected, (2) the presence of a distal re-entrant focus in the false lumen or true lumen outflow, and (3) the topography of branch ostia to the true versus false lumen. In the minutes after an aortic dissection is initiated, the true lumen (representing the remnant of the original aortic lumen) collapses to a variable degree, and the false lumen expands. The adventitia-bound outer wall of the false lumen must expand to a larger diameter in order to accommodate the same wall tension at a given blood pressure, as governed by the law of LaPlace. In contrast, the true lumen, which contains the majority of the elastic components of the aortic wall, undergoes radial elastic collapse. Therefore, the degree to which the true lumen recoils and the false lumen expands (i.e., their respective cross-sectional area) is dependent on the percentage of the total aortic circumference involved with the dissection. In the presence of a deep proximal tear and the absence of distal fenestrations, the mean false lumen pressure increases leading to compression of the true lumen ( Fig. 83.2 ). A compressed true lumen will lead to impaired perfusion of distal structures and should increase the index of suspicion for visceral and renal ischemia.
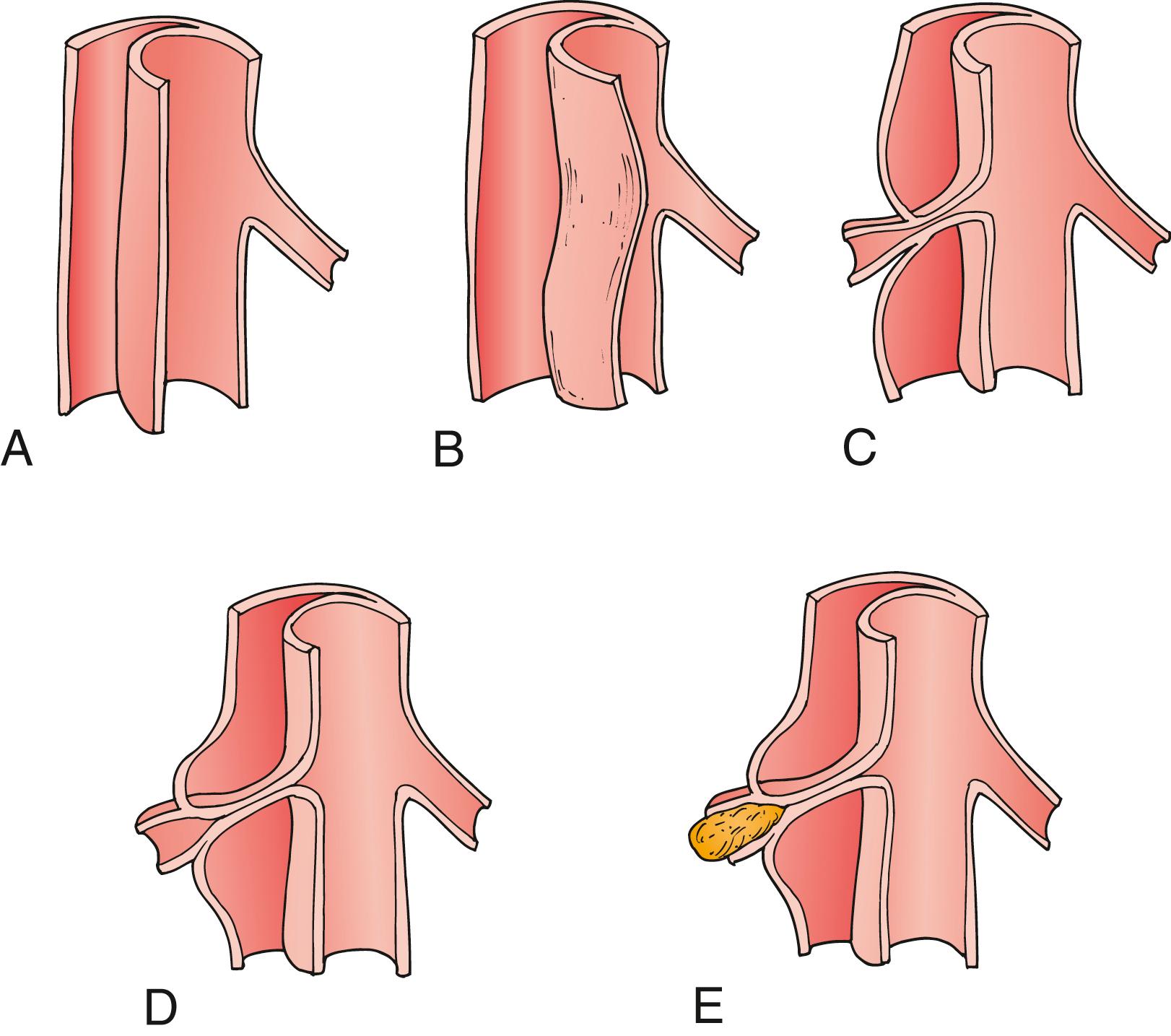
Two mechanisms for aortic branch vessel compromise have been identified, and each has specific treatment implications in the management of malperfusion syndromes, as originally described by Williams et al. In dynamic obstruction , the compressed true lumen is unable to provide adequate volume flow, or the dissection flap may prolapse into the vessel ostium, which remains anatomically intact ( Fig. 83.3 ). This is the more common mechanism of branch compromise and is responsible at least in part for some 80% of malperfusion syndromes. The severity of the true lumen collapse and the degree of aortic-level ostial vessel occlusion is determined by the circumference of the dissected aorta, cardiac output, blood pressure, heart rate, and the peripheral resistance of the outflow vessel. Pulse deficits based on dynamic obstruction may wax and wane over time, due to the variability of the aforementioned factors. , In static obstruction , malperfusion is caused by the dissecting process extending into the branch vessel proper, narrowing it to a variable degree. The false lumen is highly thrombogenic as a result of the exposed adventitial and medial layers, and thrombus formation may occur in the blind end of the dissection. If this enters and constricts the ostia of a branch vessel, organ injury can occur by thrombosis or hypoperfusion of the involved vessel. This obstruction is unlikely to resolve with restoration of aortic true lumen flow alone, and some manipulation of the vessel itself (e.g., stent or bypass graft) will typically be required (see Fig. 83.3 ).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here