Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Platelets and coagulation play a synergistic role in the generation of thrombus. Improved antithrombin approaches reduce the dependence on antiplatelet therapy for achieving suppression of ischemic events.
Although the impact of ischemic events on late mortality is well appreciated, a robust link between bleeding events and late mortality is evident. These factors bear careful consideration when weighing efficacy and safety considerations among antithrombotic therapies.
Unfractionated heparin (UFH), enoxaparin, and bivalirudin are the anticoagulants with established efficacy in contemporary percutaneous coronary intervention (PCI) practice.
A large collection of clinical trial data now exists that compares the different anticoagulants in various clinical settings, from elective PCI through to ST-elevation myocardial infarction (STEMI); however, substantial heterogeneity exists in the trial protocols and inclusion criteria, and these require careful consideration when choosing among agents.
Multiple factors need to be considered when selecting an agent, such as indication for PCI, patient bleeding risk, renal function, concomitant use of the newer antiplatelet P2Y 12 agents and glycoprotein (GP) IIb/IIIa inhibitors, familiarity with the anticoagulant, hospital protocol, and current European Society of Cardiology and American College of Cardiology guidelines.
The last two decades have witnessed the introduction of a number of new pharmacologic agents aimed at improving patient outcomes after percutaneous coronary intervention (PCI). Optimizing ischemic prevention while minimizing bleeding complications remains the cornerstone of contemporary pharmacologic practice. Notably, this period has seen improvements in PCI with the use of radial artery access, more potent antiplatelet P2Y 12 receptor inhibitors, and the availability of bleeding risk calculators (i.e., Can Rapid Risk Stratification of Unstable Angina Patients Suppress Adverse Outcomes with Early Implementation of the American College of Cardiology/American Heart Association Guidelines? [CRUSADE]); these advances, together with improvements in drug-eluting stent technology, have helped minimize the bleeding and ischemic risks during PCI.
Heparin, enoxaparin, and bivalirudin are the most widespread anticoagulant agents used in current PCI practice, and these form the basis of recommendations from major society guidelines. This chapter will discuss the current biologic basis of atherothrombosis and its key effector, thrombin; the pharmacology of the anticoagulant agents in current use; and the targets for future therapy. Also reviewed will be the landmark trials that support anticoagulant use in ST-elevation myocardial infarction (STEMI), non–ST-elevation myocardial infarction (NSTEMI), and elective PCI.
The coagulation cascade reflects the complex interplay among the coagulation proteins, platelets, and cellular phospholipid membranes of not only vascular endothelial cells but of a wide range of inflammatory cells that include monocytes and vascular smooth muscle cells. The subsequent discussion will focus on the coagulation factors that serve as targets, although modern antithrombotic regimens, additional effects on platelet-mediated thrombosis, and vascular tissue function should not be ignored.
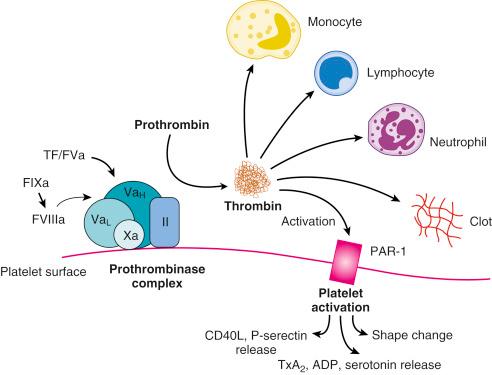
Disruption of endothelial integrity and expression of prothrombotic molecules such as tissue factor (TF) lead to the activation of the soluble coagulation proteins ( Fig. 11.1 ). This amplifying cascade converges on the generation of activated factor X (FXa) and the prothrombinase complex, which leads to the conversion of thrombin from its parent molecule, prothrombin. Thrombin generation leads to multiple effects that influence the formation of thrombosis. Specifically, thrombin catalyzes the conversion of fibrin from fibrinogen; this enables clot formation and also activates factors V, VIII, and X; thus it promotes its own generation. In addition, via its direct effects on protease-activated receptors (PARs) 1 and 4, thrombin promotes platelet activation that leads to the expression of CD40 ligand, P-selectin, and the glycoprotein (GP) IIb/IIIa receptor as well as to the secretion of vasoactive agents that include adenosine diphosphate (ADP), serotonin, and thromboxane A 2 (TxA 2 ). Direct effects of thrombin on endothelial cells and smooth muscle cells result in the expression of adhesion molecules that enables platelet and leukocyte attachment, and its effect on endothelial membrane permeability contributes to the transmigration of the cellular- and cytokine-mediated inflammatory response within the vascular wall. Thrombin promotes vasodilation in the intact endothelium, and it contributes to vasoconstriction where the endothelium is damaged or denuded. Thrombin also appears to promote fibroblast cytokine production and is mitogenic ( Fig. 11.2 ).
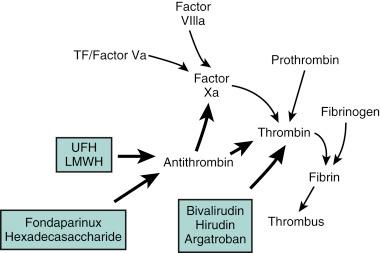
However, thrombin has a short circulating half-life, and in the context of a normal endothelial barrier, the effects of thrombin are tightly controlled by a negative-feedback mechanism. Antithrombin is a single-chain plasma glycoprotein produced by the liver. As an inhibitor of coagulation, this molecule has the ability to bind to thrombin and FXa and activated factor IX (FIXa) in equimolar concentrations. Antithrombin’s action is increased over a thousand-fold by the binding of pentasaccharide-chain containing heparins. The pentasaccharide sequence enables the binding of heparins to antithrombin and augments the binding affinity for thrombin and the other clotting factors. Antithrombin is also activated by the glycosaminoglycan heparin sulfate found on the surface of endothelial cells. However, other pathways for the inhibition of thrombin exist and include the binding of thrombin to thrombomodulin and to protein C together with protein S. This inactivates the upstream coagulation proteins, factors Va and VIIIa, and promotes the release of tissue plasminogen activator (TPA).
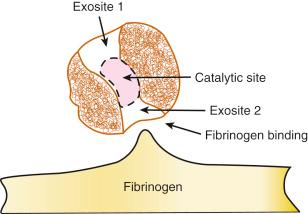
Hence, thrombin plays a central effector role in the vascular response to balloon- and stent-induced vascular injury and remains an important therapeutic target for the prevention of ischemic complications during PCI. A schematic of the structure of the thrombin molecule is presented in Fig. 11.3 . Separate substrate recognition sites are involved in the binding of heparin, fibrinogen, and thrombomodulin, and the catalytic site is responsible for the serine protease activity that is blocked by the direct thrombin inhibitors.
Improvements in interventional techniques and refinements in antithrombotic therapies have led to a substantial decline in the incidence of ischemic complications following PCI. Hence further iterations in antithrombotic strategies can be considered a double-edged sword, with improved prevention of ischemic complications potentially leading to an increase in bleeding complications associated with increased mortality, stroke, nonfatal myocardial infarction (MI), and stent thrombosis. The contemporary definition of PCI-related MI includes a troponin elevation more than five times the 99th percentile in patients with normal values before PCI or a rise greater than 20% if the baseline values are elevated and are stable or falling ( Table 11.1 ). Accompanying the troponin elevation must be at least one of the following three factors: (1) new symptoms or electrocardiographic (ECG) changes, (2) angiographic findings consistent with a procedural complication, or (3) imaging findings consistent with new ischemia. This definition has limitations, however, because the levels of post-PCI troponin elevation whereby long-term prognosis is affected has not been well established. The troponin values in this definition are arbitrarily chosen and lack clinical utility to predict outcome.
| End Point | Definition |
|---|---|
| Myocardial infarction (post PCI) | Troponin elevation more than five times the 99th percentile of the ULN in patients with normal baseline values or a rise more than 20% if baseline values are elevated. In addition, at least one of the following must accompany the troponin elevation: supportive symptoms, new ECG changes, angiographic findings, or imaging findings. SCAI definition: Clinically meaningful MI CK-MB rise more than 10 times the ULN or more than five times the ULN with new pathologic Q-waves in at least two contiguous leads or new persistent LBBB OR In the absence of baseline CK-MB, a cTn rise more than 70 times the ULN or a rise more than 35 times the ULN plus new pathologic Q-waves in at least two contiguous leads or new persistent LBBB |
| Myocardial infarction (post CABG) | Troponin elevation more than 10 times the 99th percentile in patients with normal baseline values; in addition, at least one of the following must accompany the troponin elevation: ECG changes (LBBB or new Q waves), angiographic findings, or imaging findings |
| Myocardial infarction (not periprocedural) | Detection of a rise and/or fall of a cardiac biomarker (preferably troponin) with at least one value above the 99th percentile with at least one of the following: symptoms, ECG changes, imaging findings, or angiographic findings |
| TIMI major bleeding | Intracerebral hemorrhage or any bleeding associated with a more than 5 g/dL fall in hemoglobin or a 15% absolute decrease in hematocrit a |
| TIMI minor bleeding | Any bleeding event associated with a more than 3 g/dL fall in hemoglobin or a 10% absolute decline in hematocrit or a more than 4 g/dL fall in hemoglobin or a 12% absolute decline in hematocrit in the absence of overt bleeding a |
| Major bleeding (REPLACE-2 definition) | Intracerebral hemorrhage or any bleeding event associated with a more than 3 g/dL fall in hemoglobin, a more than 4 g/dL fall in hemoglobin in the absence of overt bleeding, or any red cell transfusion of two or more units a |
| Severe or life-threatening bleeding (GUSTO definition) | Intracerebral hemorrhage or bleeding that causes hemodynamic compromise or requires intervention |
| Minor bleeding (GUSTO definition) | Bleeding that requires transfusion but does not cause hemodynamic compromise |
a All calculations of drops in hemoglobin are adjusted for any transfusion by the Landefeld index.
More recently, definitions of clinically meaningful postprocedural MI have been proposed that are more in line with levels of myocardial injury used to define post-coronary artery bypass graft (CABG) MI. These so-called clinically relevant values in patients with normal baseline biomarkers include a post-PCI creatinine kinase myocardial band (CK-MB) elevation 10 or more times the upper limit of normal (ULN) and a lower threshold five or more times the ULN in the presence of new Q-waves or left bundle branch block (LBBB). For patients with elevated CK-MB prior to PCI that is either stable or falling , an increment 10 or more times the ULN from the most recent level is considered diagnostic. The definition for those whose CK-MB level is not stable or falling includes a further rise above the most recently measured value of 10 or more times the ULN plus new ST elevation or depression plus signs consistent with a clinically relevant MI, such as new or worsening heart failure or hypotension.
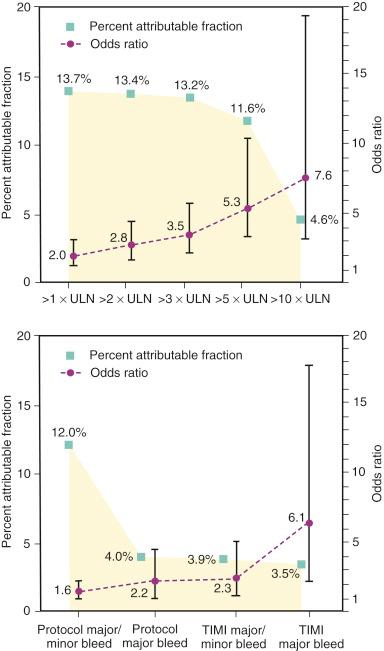
Historical definitions of PCI-related MI have included CK elevation (two times the ULN) but underestimated the incidence of periprocedural MI by 40% to 50%. In an analysis of patients enrolled in the Randomized Evaluation in PCI Linking Angiomax to Reduced Clinical Events (REPLACE)-2 study, CK-MB elevation greater than or equal to three times the ULN was associated with a 3.5-fold excess risk of mortality at 12 months and accounted for 13.2% of all mortality seen by 12 months ( Fig. 11.4 ).
In one study, an isolated post-PCI troponin elevation, compared with no elevation, in patients with normal preprocedural values carried a small but increased risk of 30-day mortality (0.3% vs. 0.1%); however, this was not an independent predictor of long-term mortality. Pre-PCI troponin elevation—as well as age, diabetes, and a history of congestive heart failure and stroke—were all independent predictors of long-term mortality.
Non–CABG-related bleeding within 30 days of PCI is strongly associated with increased mortality, stroke, nonfatal MI, and stent thrombosis ( Table 11.2 ). Factors that contribute to these outcomes include reduced myocardial oxygen delivery as a result of hypotension and anemia, cessation of anti-thrombotic and anti-ischemic agents, increased myocardial oxygen demand from the systemic inflammatory response syndrome, and the adverse effects of the storage lesion in the transfusion of red blood cells. The bleeding rates after PCI in patients with acute coronary syndromes (ACSs) have been estimated to be as high as 10%, and various risk calculators have been developed. However, the definition of bleeding in the major PCI trials has varied over the last 20 years, which makes comparisons among trials more difficult. Many trials use their own definitions of bleeding, along with the commonly used thrombolysis in myocardial infarction (TIMI) and global utilization of streptokinase and tissue plasminogen activator for occluded coronary arteries (GUSTO) definitions, both originally developed to monitor bleeding complications in fibrinolytic trials. This lack of uniformity has made it more difficult to judge the effectiveness of anticoagulant therapies among trials. More recently, however, an Academic Consortium definition of bleeding has been created in an attempt to standardize trial data and better evaluate the safety and efficacy of antithrombotic therapies ( Table 11.3 ).
| Myocardial Infarction | Non-CABG Major Bleeding | ||||
|---|---|---|---|---|---|
| Clinical Factor | OR (95% CI) | P Value | Clinical Factor | OR (95% CI) | P Value |
| Biomarker elevation | 1.66 (1.40–1.97) | <.001 | Male sex | 0.42 (0.36–0.50) | <.001 |
| Family history of CAD | 1.48 (1.27–1.73) | <.001 | Anemia (Hct <39% in males and < 36% in females) |
1.96 (1.63–2.36) | <.001 |
| Age per 5 years | 1.08 (1.04–1.11) | <.001 | Age per 5 years | 1.14 (1.10–1.19) | <.001 |
| ST-segment deviation ≥1.0 mm | 1.38 (1.18–1.61) | <.001 | ST-segment deviation ≥1.0 mm | 1.31 (1.10–1.54) | <.01 |
| Prior MI | 1.28 (1.08–1.50) | <.001 | Prior PCI | 0.70 (0.58–0.84) | <.01 |
| Creatinine per 0.1 mg/dL | 1.08 (1.05–1.10) | <.001 | |||
| White cell count per 109/L | 1.08 (1.05–1.11) | <.001 | |||
| Prior CVA | 1.60 (1.21-2.10) | <.001 | |||
| Type | Definition |
|---|---|
| 0 | No bleeding |
| 1 | Bleeding that is not actionable (no investigations, treatment, or hospitalization is required); may include episodes of self-discontinuation of medical therapy by the patient |
| 2 | Any overt actionable sign of hemorrhage that does not fit criteria for type 3, 4, or 5 but does at least one of the following: (1) requires nonsurgical medical intervention, (2) leads to hospitalization or increased level of care, or (3) prompts evaluation |
| 3 | 3a: Overt bleeding with Hb drop of 3-5 g/dL, a any transfusion with overt bleeding 3b: Overt bleeding with Hb of ≥5 g/dL, a cardiac tamponade, bleeding that requires surgical intervention or vasoactive agents 3c: Intracranial hemorrhage (does not include microbleeds or hemorrhagic transformation), intraocular bleed that compromises vision |
| 4 | CABG-related bleeding |
| 5 | 5a: Probable fatal bleeding 5b: Definite fatal bleeding |
Unfractionated heparin (UFH) is a heterogeneous group of glycosaminoglycans of various lengths (5000 to 30,000 Da) that exhibit a high affinity for antithrombin. This binding augments antithrombin’s enzymatic inactivation of thrombin, FXa, and FIXa, but its effects on thrombin are the most pronounced. UFH is considered an indirect antithrombin because its action requires the simultaneous binding to antithrombin and thrombin. Up to two-thirds of UFH preparations contain shorter molecules (<18 saccharides) that lack sufficient length to simultaneously span antithrombin and thrombin, and they do not exhibit antithrombin activity. However, the anti-FXa effects of heparin are not dependent on simultaneous binding of both antithrombin and FXa, and therefore antithrombin effects are observed across a wider range of saccharide chain lengths.
The elimination of UFH is initially through rapid but saturable metabolism within endothelial cells and macrophages (zero-order kinetics) followed by slower renal clearance (first-order kinetics). The plasma half-life depends on the dose administered and is approximately 1 hour at doses of 100 IU/kg. In the context of excessive dosing or bleeding, UFH can be reversed by the administration of protamine; however, the clinical efficacy and safety of this strategy has not been well established.
Limitations of UFH include the activation of platelets, a dependence on antithrombin levels, nonspecific binding to plasma proteins, and an inability to inhibit clot-bound thrombin. In addition, direct binding to platelet factor IV can contribute to heparin-induced thrombocytopenia (HIT) in 1% to 3% of treated patients. Platelet activation by heparin is evidenced by an increase in the expression of platelet surface adhesion molecules.
The low-molecular-weight heparins (LMWHs) are produced by chemical or enzymatic depolymerization of UFH, which results in heparin fragments with a mean molecular weight approximately 30% of most UFH preparations. However, the molecular size of the heparin molecules still varies; therefore anticoagulant characteristics remain heterogeneous, although they are more predictable when compared with heparin. The principal effect of the LMWHs is the inhibition of anti-FXa via antithrombin. In comparison with UFH, the LMWHs have a longer half-life and demonstrate a more consistent dose response. Reduced platelet activation and fewer platelet factor IV interactions result in less HIT. Clearance is by renal excretion, however, and the biologic half-life is increased in people with renal failure ( Table 11.4 ).
| Property | Unfractionated Heparin | Enoxaparin | Fondaparinux | Bivalirudin |
|---|---|---|---|---|
| Mean molecular weight (Da) | 15,000 | 5000 | 1728 | 2180 |
| Dependence on antithrombin | Yes | Yes | Yes | No |
| Anti-FXa, anti-FIIa activity | Yes | 2-4 | No anti-FXa activity | No anti-FXa activity |
| Half-life | ∼60 min | ∼240 min | 17–21 h | 25 min |
| Bioavailability | + to +++ | ++++ | ++++ (Subcutaneous) | ++++ |
| Subcutaneous absorption | ++ | ++++ | ++++ | − |
| Binding to plasma proteins | +++ | + | + | − |
| Binding to platelets/macrophages | ++ | + | − | − |
| Antigenicity/HIT | ++ | + | −/+ (Very rare) | − |
| Clearance | Renal | Renal | Renal | Renal/proteolysis |
| Protamine neutralization | ++++ | ++ | − | − |
In one study, adequate levels of anti-FXa where observed among patients 2 to 8 hours following subcutaneous dosing of enoxaparin 1 mg/kg twice daily, as well as in those receiving an additional 0.3 mg/kg intravenous (IV) dose 8 to 12 hours following subcutaneous dosing at 1.0 mg/kg. Other investigators have suggested that doses as low as 0.5 mg/kg of IV enoxaparin may be safe and efficacious and may also enable easier sheath management, although a quarter of the patients in this study also received a GP IIb/IIIa inhibitor. Some evidence suggests that enoxaparin may be reversed by the IV administration of protamine, but these data are limited.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here