Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Editors' comment: When Chesley wrote his original monograph, he, like most of the scientific community, was unfamiliar with the concept of angiogenesis. Even in the multiauthored second edition, in which the endothelial cell hypothesis of preeclampsia was first proposed, the subject “angiogenesis” was not elaborated. This millennium has witnessed a very different story. Starting with preliminary observations of the roles of angiogenic proteins in normal and abnormal placentation a signal paper by Maynard et al., published in the J Clin Invest in 2003 and now cited widely, research into angiogenesis and preeclampsia took off with rocket speed. This publication, followed by an explosion of related research into pro- and antiangiogenic factors, opened the field, which now appears firmly established that these proteins have a role in (1) predicting and diagnosing the disorder, (2) mediating the signs and symptoms of preeclampsia, and (3) potential therapeutic or even preventative use in the disease. In the current edition, the authors update exciting advances in our understanding of the role and application of angiogenic factors in the pathogenesis of preeclampsia and expand their use as biomarkers of pregnancy complications.
Vascular development occurs through angiogenesis and vasculogenesis. , Angiogenesis is the process of neovascular sprouting or branching from preexisting blood vessels, while vasculogenesis is the process of blood vessel generation de novo from angioblast precursor cells.
The human placenta undergoes extensive angiogenesis and vasculogenesis throughout development. Additionally, the developing placenta undergoes a process of vascular mimicry (also referred to as pseudo-vasculogenesis) as cytotrophoblasts convert from an epithelial to an endothelial phenotype (see Chapters 5 and 6 ). When placental vascular development is deranged, the success of the pregnancy is jeopardized and serious complications such as preeclampsia and fetal growth restriction can occur. This chapter will discuss placental vascular development during health and in disease, with an emphasis on the role of placental antiangiogenic factors in the pathogenesis of the maternal syndrome of preeclampsia. Also discussed is our view that preeclampsia, at least the form of the disorder that is associated with adverse maternal/fetal outcomes, is a specific entity whose phenotypes relate to angiogenic imbalance and that measurements of these factors help identify the severe form of the disease and suggest useful management strategies for these patients.
The placenta is a highly vascular organ, containing both embryonic and maternal blood vessels. Thus to understand the role of angiogenesis in successful placentation, a summary of fundamental steps necessary for this critical developmental milestone follows. These include: (1) trophoblast invasion of the superficial maternal decidua; (2) vascularization of the placental bed to establish and maintain fetoplacental nutrient delivery and waste disposal; and (3) subsequent remodeling of the maternal spiral arteries by the trophoblast, enabling robust uteroplacental perfusion. This latter function is critical as placental trophoblast must establish a circulation that permits adequate maternal–fetal exchange. Both vasculogenesis and angiogenesis are involved in these processes.
During embryonic development, the blastocyst differentiates into two cell populations: an outer polarized trophoectoderm and the nonpolarized inner cell mass. The trophoectoderm gives rise to extraembryonic membranes, whereas the inner cell mass is destined to form the embryo proper. These processes are examined in more detail in Chapter 5 .
Extraembryonic mesenchymal cells give rise to cores that penetrate into the center of the cytotrophoblast cores. These mesenchymal cells differentiate into endothelial cells, forming the first capillaries of the placental vasculature. In humans, placental vasculogenesis is evident by approximately 21–22 days postconception. At this stage, cords of hemangiogenic cells are present and some demonstrate primitive lumen formation. These cords further develop so that by approximately day 32 postconception, most villi show the presence of capillary structures. The highly proliferative trophoblasts ultimately invade the entire endometrium, the outer one-third of the myometrium, and the maternal circulation. Hypoxia is an important driving force for trophoblast proliferation.
As trophoblasts invade the uterus, they are exposed to increasing concentrations of oxygen at which time they exit the cell cycle and differentiate. Further development leads to the penetration of cytotrophoblastic cones into the syncytiotrophoblastic mass and the development of lacunae, which eventually become the intervillous space. Continuing growth and differentiation of the trophoblasts lead to branching of the trophoblast villi and the shaping of a placental labyrinth or intervillous space, where the fetal/maternal exchange of oxygen, nutrients, and waste products occurs. Trophoblasts invade maternal tissue with a variable depth of invasion among species; the deepest known being in humans.
The formation of adequate maternal–placental circulation requires remodeling of maternal blood vessels (namely the spiral arteries). In humans, during mid-late first trimester, the trophoblasts invade deeply through the endometrium and into the superficial part of the myometrium, completely remodeling the proximal ends of the maternal spiral arteries. Through the open endings of the maternal vessels that are created by trophoblast invasion, maternal blood is released into the intervillous space, flows around the trophoblast villi, and is drained by spiral veins (see Fig. 9.1 ).
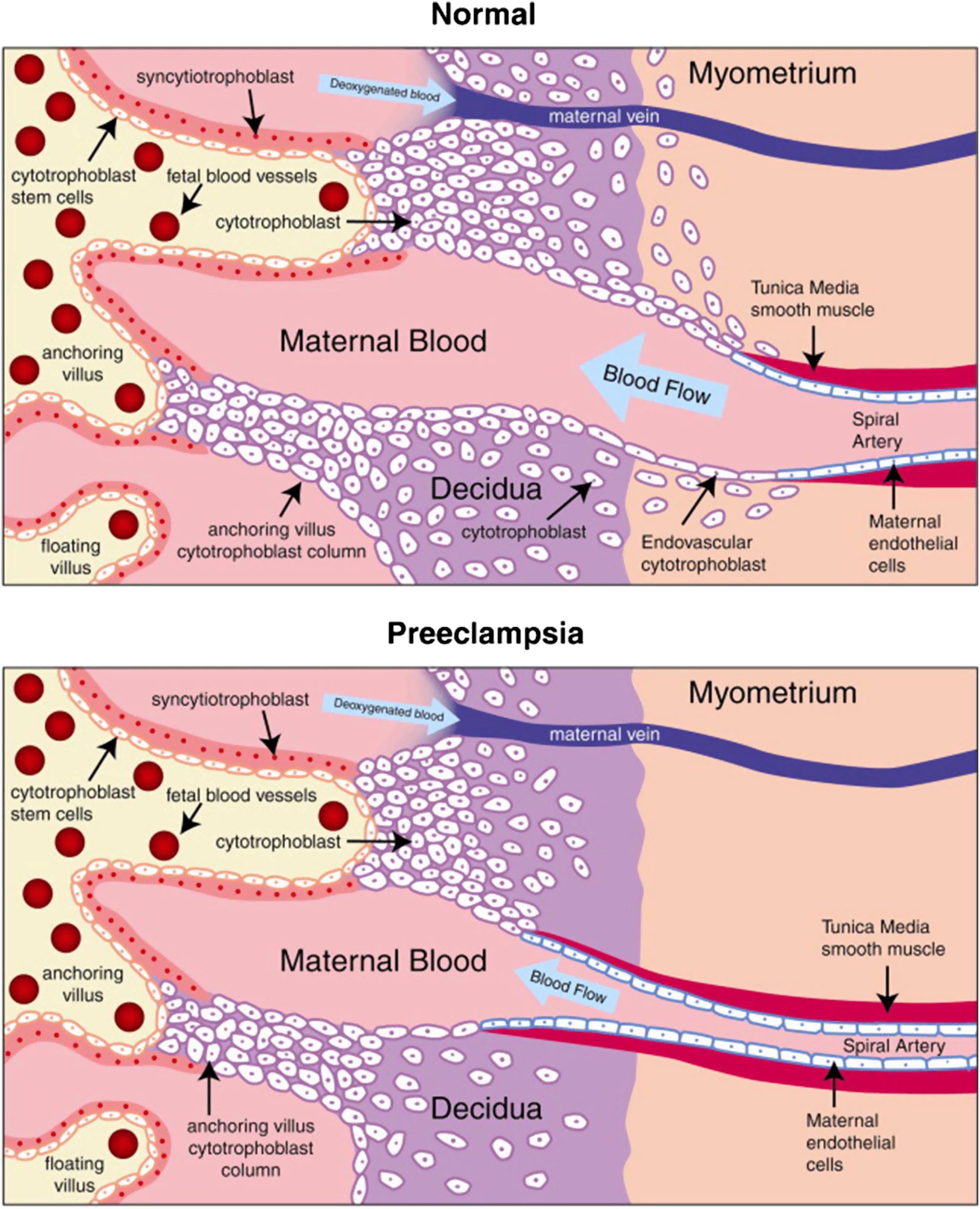
When cytotrophoblasts invade maternal spiral arteries, they replace the luminal endothelial cells, a process common in all species with hemochorial placentation. During this process, the endovascular cytotrophoblasts convert from an epithelial to endothelial phenotype, a process referred to as pseudovasculogenesis or vascular mimicry ( Fig. 9.1 ; see also Chapters 5 and 6 ). Thus cytotrophoblast stem cells lose epithelial markers, such as E-cadherin and α 6 β 3 integrin, and gain endothelial markers, such as vascular endothelial-cadherin (VE-cadherin) and α v β 3 integrin. ,
In addition to replacing maternal endothelial cells, cytotrophoblasts also remodel the highly muscular tunica media of spiral arteries, a process that is dependent on the enzyme membrane metalloproteinase 9 (MMP-9). , This transforms the maternal high-resistance vessels into larger low-resistance capacitance vessels. Uterine blood flow during pregnancy increases more than 20-fold; and the functional consequence of spiral artery remodeling maximizes the capacity of the maternal–placental circulation by providing sufficient blood supply for placenta and fetus at low blood pressure. Of note, spiral arteries from both the implantation and nonimplantation regions display these physiological changes.
The mechanisms underlying these changes remain unknown. Placental oxygen tension has been suggested to be one of the major regulators of cytotrophoblast migration and differentiation. It has also been hypothesized that decidual natural killer (NK) cells and/or activated macrophages play a role in this vascular remodeling. ,
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here