Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The advent of Advanced Trauma Life Support (ATLS) brought great improvements to the care of the trauma patient. Standardized methods for assessment and resuscitation of trauma patients have resulted in increased survival and improved outcomes. While recognition of injuries and resuscitative efforts have improved and are the priority in injured patients, there remain outstanding areas where care of the trauma patient can be improved. One such area is the management of the acute pain and its sequalae.
Acute pain is a physiologic defense mechanism that occurs in nearly every patient who is traumatically injured. Left uncontrolled, acute pain can result in multisystemic, homeostatic derangements in endocrine, metabolic, physiologic, psychiatric, and autonomic functions. These derangements have the potential to cause catastrophic downstream morbidity and mortality. Patients with poorly controlled pain can become hypercoagulable, develop cardiovascular and pulmonary complications, and have longer hospital stays. In addition, studies have demonstrated that inadequate analgesia can result in chronic pain and posttraumatic stress disorder (PTSD). Over the past several decades, there has been an increasing reliance on opioids as a mainstay for analgesia of acute pain. However, the need for escalated doses with severe pain results in adverse effects to this class of drugs. Opioid-related adverse drug events (ORADEs) include sedation, confusion, constipation, ileus, nausea, vomiting, respiratory depression, hyperalgesia, and suppression of immune function. Well-intended opioid-based analgesic regimens can impose lasting, catastrophic suffering on patients and their families in the form of abuse and dependence, as well as hyperalgesia effects that last a lifetime.
Since 1999, opioid-related deaths have exponentially increased. In 2018, the number of deaths from opioid overdose exceeded those from motor vehicle accidents in the United States. Unfortunately, as opioids have become more commonly prescribed, the misuse of these drugs has also increased. There is a strong link between prescription opioids and the rise of dependence and addiction. In 2018, 10.3 million people misused prescription opioids with hydrocodone and oxycodone being the highest reported. A survey of heroin users seeking treatment revealed that 75% of these individuals were introduced to opioids through first taking prescription drugs. For these reasons, alternative analgesic strategies designed to relieve pain and reduce opioid exposure are being developed ( Table 1 ).
| Class | Drug | Mechanism of Action | Site of Action | Adverse Effects |
| NSAIDs |
|
COX-1 and COX-2 Inhibition, ↓ PG synthesis, anti-inflammatory | Periphery, dorsal horn, central | Platelet inhibition, renal insufficiency, gastric bleeding, dyspepsia, allergic reaction and bronchospasm, hypertension |
| Celecoxib | COX-2 selective inhibition | |||
| NMDA receptor antagonists |
|
NMDA receptor antagonism, opioid receptors agonist, NE & 5-HT reuptake inhibitor | Dorsal horn, central | Hallucinations, nightmares, diplopia, hypertension, tachycardia |
| APAP | Acetaminophen | Unknown, COX-3 enzyme inhibition | Peripheral, central | Hepatotoxicity |
| Gabapentinoids |
|
GABA receptors | Dorsal horn, central | Dizziness, sedation, peripheral edema |
| α-2 agonists |
|
α-2 receptors, inhibit release substance P | Dorsal horn, central | Hypotension, tachycardia, sedation |
| Glucocorticoids | Dexamethasone | Anti-inflammatory | Peripheral | Hyperglycemia, adrenal suppression |
| Serotonin norepinephrine reuptake inhibitors | Duloxetine | NE & 5-HT reuptake inhibitor | Descending pathway, dorsal horn | Dizziness, nausea, dry mouth, sweating |
| Tricyclic antidepressants |
|
NE & 5-HT Reuptake inhibitor | Descending pathway, dorsal horn | Dry mouth, diplopia, orthostatic hypotension |
| Systemic local anesthetic | Lidocaine | Inhibition of voltage gated Na channels, NMDA antagonism, anti-inflammatory | Periphery, dorsal horn | Cardiac arrhythmia, local anesthetic systemic toxicity |
Over the past decade, multimodal analgesic strategies have demonstrated success in the perioperative arena as a result of efforts made to develop protocols to improve outcomes for surgical patients. Enhanced recover after surgery (ERAS) and Perioperative Surgical Home (PSH) are both practice models using multimodal analgesia protocols for surgical patients. These programs utilize multidisciplinary teams with standardized best-practice, evidence-based treatment protocols for surgical patients. The protocols have been successful in reducing perioperative complications related to surgery and opioid utilization. Multimodal analgesia strategies target multiple sites within the pain pathway in an attempt to eliminate or significantly reduce opioid usage, which results in side effects and delay of recovery.
Regional anesthesia (RA) via peripheral nerve blockade and myofascial plane blocks are additional modalities that can be utilized in the trauma patients. With innovations in ultrasound guidance, these procedures have become more widely utilized and provide excellent pain control in patients with extremity injuries or chest wall trauma. Many protocols combine the use of multimodal analgesia (MMA) with RA techniques to improve pain control and decrease opioid utilization.
While these protocols are becoming more standardized for many surgical programs, the management of acute pain in trauma patients remains a challenge. In many institutions, there is a lack of standardized guidelines for treating acute pain from traumatic injury. Nor is there sufficient education for patients, as well as physicians, on pain expectations and the dangers of opioid use. Traumatic injuries and comorbid conditions need to be carefully considered when prescribing any medication. However, as other surgical programs become more widely utilized, such protocols are being adapted to care for trauma patients in an attempt to limit opioid exposure and complications that can occur as a result of their utilization. Recent military experience from implementation of innovative MMA and RA strategies in the combat wounded from the Global War on Terrorism has shown a decrease in acute and chronic pain and their untoward physiologic consequences.
The stress response induced after trauma is a protective mechanism to promote the return to normal steady-state physiology. However, when the stressors remain well after the inciting event, the response can result in a deleterious effect on convalescence. Reducing the stressors and supporting the return to health is paramount to enhancing recovery in trauma patients.
Traumatic injury results in the release of factors known as damage-associated molecular pattern (DAMP) activators or alarmins ( Fig. 1 ). The release of alarmins set in motion multiple intertwined neuronal, inflammatory, and endocrine cascades, mounting the complex stress response to insult. Nociceptors are pain-sensing neurons located in the periphery ( Fig. 1 ). As DAMPs are released from traumatized tissues, transcriptional changes, such as increased expression proinflammatory mediators like tumor necrosis factor-alpha (TNFα) and interleukin (IL)-6, are induced in macrophages. Signaling molecules accumulate and then trigger an action potential, which is transduced to the substantia gelatinosa in the dorsal horn of the spinal cord. Here, chemical neurotransmitters such as Substance P and glutamate are released into the synapse, which activate the action potential in the second-order neurons. The signal travels via the spinothalamic tract to the thalamus, where signaling to a third-order neuron occurs. The signal then travels to the area within the somatosensory cortex that correlates with the location of the injury. The cortex is where the perception of pain occurs.
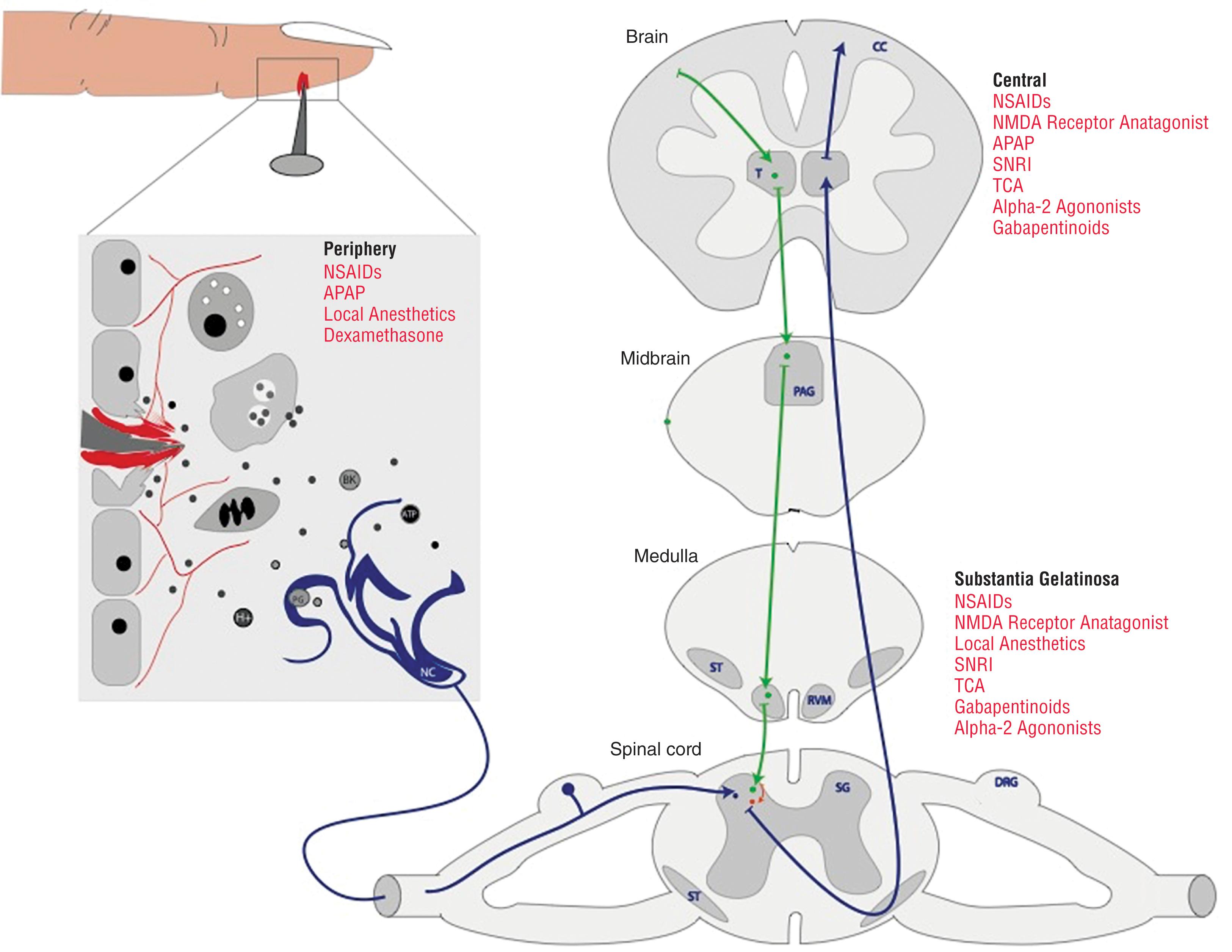
Repeated stimulation of the nociceptor leads to a wind-up phenomenon, known as central sensitization. When this occurs, synaptic efficacy is increased, and modulation from the descending pathway is lessened. The second-order neurons become hyperexcitable leading to hypersensitivity, hyperalgesia, and allodynia, where there is pain response disproportionate to the stimuli. Glutamate and substance P act to promote excitability at these synapses. When central sensitization persists beyond the time it takes for the tissues to heal, chronic or persistent pain syndromes can occur.
Modulation of the pain signal occurs along the entire pain pathway. The descending pathway acts to modulate pain signals from the ascending pathway. The descending pathway neurons arise in midbrain from regions rich in norepinephrine (NE) and serotonin (5-HT). It also receives input from the ascending pathway, the sensory cortex, and limbic nuclei. The fibers descend down the spinal cord to the substantia gelatinosa of the dorsal horn where they synapse with inhibitory interneurons by releasing 5-HT or NE. Inhibitory interneurons, in turn, synapse with the nociceptive neurons and second-order neurons ( Fig. 1 ). Endogenous opioids, cannabinoids, NE, γ-aminobutyric acid (GABA), and adenosine modulate inhibitory effects on these synapses. Enkephalins, endorphins, and dynorphins are released by the interneurons, which reduce the action potentials that are propagated to the second-order neurons of the ascending pathways, thus decreasing the pain signals to the CNS.
Neuropathic pain arises from direct injury of the peripheral or CNS. Lesions in the neuron lead to physical and functional changes with the end result of central sensitization. Increased expression of ion channels (voltage gated Na channels) and N -methyl- d -aspartate (NMDA) receptors lead to spontaneous firing of action potentials, known as ectopic discharges. Repeated action potentials from the periphery leads to accumulation of glutamate and substance P in the substantia gelatinosa. These changes cause neuroplastic postsynaptic modifications in second-order neurons resulting in allodynia and hyperalgesia. Then, when a nerve injury occurs, inhibitory GABAergic interneurons are lost, resulting in an increased pain sensation.
Within the pain pathway are multiple opportunities to suppress pain through various modalities. In the past, analgesic strategies for acute pain in trauma patients have been dominated by unimodal opioid administration. MMA strategies are aimed at multiple targets throughout the pain pathway, instead of only opioid receptors. By using several different interventions, lower doses of medications may be employed, decreasing medication-associated adverse events. In addition, these strategies can reduce or even eliminate the use of opioids to hasten recovery and avoid ORADEs. Utilization of nonsteroidal anti-inflammatory drugs (NSAIDs), acetaminophen (APAP), gabapentinoids, antidepressants, NMDA receptor antagonists and RA can produce excellent analgesia by targeting several receptors within the nociceptive pathway.
During injury, damaged tissue and immune cells convert phospholipids to prostaglandins (PGs) via the arachidonic acid (AA) pathway. Nociceptor receptors bind PGs triggering action potentials in the ascending pathway. PGs also contribute to localized edema and inflammation. The AA pathway is catalyzed by cyclooxygenase (COX) isoenzymes COX-1 and COX-2. COX-1 is a constitutively expressed enzyme that maintains physiologic balance in tissues such as the stomach and kidneys, as well as in platelets. COX-2 expression is induced in areas of inflammation, increasing the PG levels in tissues and promoting inflammation.
NSAIDs are potent analgesics that also have antipyretic and anti-inflammatory activity. These medications work by inhibiting the formation of PG. Nonselective (ns) NSAIDs, such as ibuprofen, ketorolac, and meloxicam, inhibit both COX-1 and COX-2 enzymes, whereas selective NSAIDs, such as celecoxib, inhibit only COX-2 enzymes. NSAIDs are available as oral, topical, parenteral, and rectal preparations.
While NSAIDs provide excellent opioid-sparing analgesia, careful consideration of the patient’s injuries and comorbidities should weigh in the decision to utilize these medications in trauma patients. Inhibition of COX-1 by nsNSAIDs blocks the formation of thromboxane A2 (TXA2) in platelets, impairing platelet function. Avoidance of nsNSAIDs may be considered in trauma patients who may be at risk for bleeding from their injuries in the setting of platelet dysfunction. Selective COX-2 inhibitors do not reduce TXA-2 production and may be considered for such patients. In addition to platelet dysfunction, COX-1 inhibition can result in dyspepsia, gastrointestinal bleeding, acute renal failure, hypertension, and exacerbations of myocardial and cerebrovascular events.
For trauma patients with musculoskeletal injuries, nsNSAIDs have been often avoided out of concern for impairment of the healing of long bone fractures. Much of this theory is now being reconsidered. In a study looking at humeral shaft fractures, both nsNSAIDs and opioid administration in the third month after the injury was associated with nonunion. When the data were further analyzed, it was realized that patients were receiving these therapies because they were suffering pain from already nonhealing fractures. A meta-analysis also concluded that the risk of nonunion was not associated with NSAID exposure when high-quality studies were reviewed. Given the nature and complexity of trauma patients, each patient should be evaluated on an individual basis for the appropriateness of NSAID therapy.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here