Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Dermatopathology requires years of training and practice to attain an acceptable level of diagnostic skill. Many have found this process an exciting and challenging one, well worth the expenditure of time and intellectual effort. To the trainee, there seems to be an endless number of potential diagnoses in dermatopathology, with many bewildering names. However, if a logical approach is adopted, the great majority of skin biopsies can be diagnosed specifically and the remainder can be partly categorized into a particular group of diseases. This learning process can be enhanced under the tutelage of a skilled mentor and by optical mileage, a term used for the self-examination and diagnosis of large amounts of day-to-day material; such cases invariably differ from “classic” examples of an entity found in teaching sets. It should not be forgotten that the histopathological features of some dermatoses are not diagnostically specific, and it may only be possible in these circumstances to state that the histopathological features are “consistent with” the clinical diagnosis.
The interpretation of many skin biopsies requires the identification and integration of two different morphological features—the tissue reaction pattern and the pattern of inflammation . This is a crude algorithmic approach; more sophisticated ones usually hinder rather than enhance the ability to make a specific diagnosis.
Tissue reaction patterns are distinctive morphological patterns that categorize a group of cutaneous diseases. Within each of these histopathological categories there are diseases that may have similar or diverse clinical appearances and etiologies. Some diseases may show histopathological features of more than one reaction pattern at a particular time or during the course of their evolution. Such cases may be difficult to diagnose. In this edition, an attempt has been made to list diseases that characteristically express more than one tissue reaction pattern (presented later).
The pattern of inflammation refers to the distribution of the inflammatory cell infiltrate within the dermis and/or the subcutaneous tissue. There are several distinctive patterns of inflammation (discussed later); their recognition assists in making a specific diagnosis.
Some dermatopathologists base their diagnostic approach on the inflammatory pattern, whereas others look first to see if the biopsy can be categorized into one of the “tissue reactions” and use the pattern of inflammation to further categorize the biopsy within each of these reaction patterns. In practice, the experienced dermatopathologist sees these two aspects (tissue reaction pattern and inflammatory pattern) simultaneously, integrating and interpreting the findings in a matter of seconds. For trainees in dermatopathology, the use of tissue reaction patterns, combined with the mnemonic for diseases with a superficial and deep inflammatory pattern, appears to be the easiest method to master.
The categorization of inflammatory dermatoses by their tissue reactions is considered first.
There are many different reaction patterns in the skin, but the majority of inflammatory dermatoses can be categorized into six different patterns. For convenience, these are called the major tissue reaction patterns . Occasionally, diseases express more than one major pattern, either ab initio or during their evolution. They are dealt with separately in the “Combined Reaction Patterns” section. There are a number of other diagnostic reaction patterns that occur much less commonly than the major group of six but that are nevertheless specific for other groups of dermatoses. These patterns are referred to as minor tissue reaction patterns. They are considered after the major reaction patterns.
There are four patterns of cutaneous inflammation characterized on the basis of distribution of inflammatory cells within the skin:
Superficial perivascular inflammation
Superficial and deep dermal inflammation
Folliculitis and perifolliculitis
Panniculitis.
There are numerous dermatoses showing a superficial perivascular inflammatory infiltrate in the dermis and a limited number in the other categories. Sometimes panniculitis and folliculitis are regarded as major tissue reaction patterns because of their easily recognizable pattern.
A significant number of inflammatory dermatoses can be categorized into one of the following six major reaction patterns, the key morphological feature of which is included in parentheses:
Lichenoid (basal cell damage; interface dermatitis)
Psoriasiform (regular epidermal hyperplasia)
Spongiotic (intraepidermal intercellular edema)
Vesiculobullous (blistering within or beneath the epidermis)
Granulomatous (chronic granulomatous inflammation)
Vasculopathic (pathological changes in cutaneous blood vessels).
Each of these reaction patterns is discussed in turn, together with a list of the dermatoses found in each category.
The lichenoid reaction pattern (“interface dermatitis”) (see Chapter 4 ) is characterized by epidermal basal cell damage, which may be manifested by cell death and/or basal vacuolar change (known in the past as “liquefaction degeneration”). The basal cell death usually presents in the form of shrunken eosinophilic cells, with pyknotic nuclear remnants, scattered along the basal layer of the epidermis ( Fig. 2.1 ). These cells are known as Civatte bodies. They are undergoing death by apoptosis, a morphologically distinct type of cell death seen in both physiological and pathological circumstances (see p. 50 ). Sometimes the basal cell damage is quite subtle, with only an occasional Civatte body and very focal vacuolar change. This is a feature of some drug reactions.
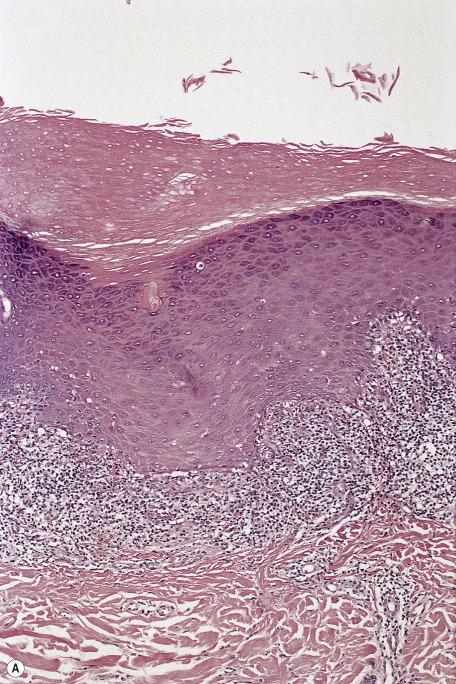
In the United States, the term interface dermatitis is used synonymously with the lichenoid reaction pattern, although it is not usually applied to the subtle variants. Its use in other countries is by no means universal. At other times, it is used for the morphological subset (discussed later) in which inflammatory cells extend into the basal layer or above. The term is widely used despite its lack of precision. It is warmly embraced as a diagnosis, but it is nothing more than a pattern, encompassing many clinical entities with diverse presentations, causes, and treatments.
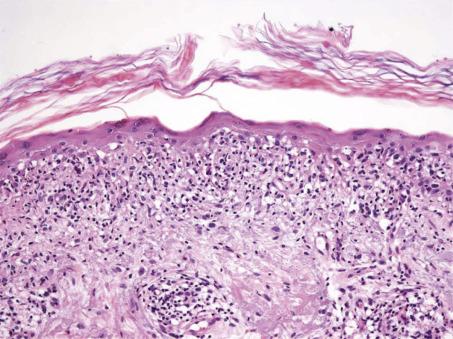
A distinctive subgroup of the lichenoid reaction pattern is the poikilodermatous pattern , characterized by mild basal damage, usually of vacuolar type, associated with epidermal atrophy, pigment incontinence, and dilatation of vessels in the papillary dermis ( Fig. 2.2 ). It is a feature of the various types of poikiloderma (see p. 92 ).
The specific diagnosis of a disease within the lichenoid tissue reaction requires an assessment of several other morphological features, including the following:
The type of basal damage (vacuolar change is sometimes more prominent than cell death in lupus erythematosus, dermatomyositis, the poikilodermas, and drug reactions)
The distribution of the accompanying inflammatory cell infiltrate (the infiltrate touches the undersurface of the basal layer in lichen planus and its variants, early lichen sclerosus et atrophicus, and in disseminated superficial actinic porokeratosis; it obscures the dermoepidermal interface [so-called “interface dermatitis”] in erythema multiforme, paraneoplastic pemphigus, fixed drug eruptions, acute pityriasis lichenoides [PLEVA], acute graft-versus-host disease [GVHD], one variant of lupus erythematosus, and reactions to phenytoin [Dilantin] and other drugs; and it involves the deep as well as the superficial part of the dermis in lupus erythematosus, syphilis, photolichenoid eruptions, and some drug reactions)
The presence of prominent pigment incontinence (as seen in drug reactions, the poikilodermas, lichenoid reactions in dark-skinned people, and some of the sun-exacerbated lichen planus variants, such as lichen planus actinicus)
The presence of satellite cell necrosis (lymphocyte-associated apoptosis)—defined here as two or more lymphocytes in close proximity to a Civatte body (a feature of graft-versus-host reaction, regressing plane warts, subacute radiation dermatitis, erythema multiforme, and some drug reactions).
The diseases showing the lichenoid reaction pattern are listed in Table 2.1 .
| Lichen planus |
| Lichen planus variants * |
| Lichen nitidus |
| Lichen striatus |
| Lichen planus–like keratosis |
| Lichenoid drug eruptions * |
| Fixed drug eruptions * |
| Erythema multiforme and variants * |
| Superantigen ID reaction * |
| Graft-versus-host disease * |
| Subacute radiation dermatitis * |
| Eruption of lymphocyte recovery |
| AIDS interface dermatitis |
| Lupus erythematosus * |
| Dermatomyositis |
| Poikiloderma congenita(le) * |
| Kindler's syndrome |
| Congenital telangiectatic erythema (Bloom's syndrome) |
| Lichen sclerosus et atrophicus |
| Dyskeratosis congenita |
| Poikiloderma of Civatte |
| Pityriasis lichenoides * |
| Persistent viral reactions |
| Perniosis |
| Polymorphic light eruption (pinpoint type) |
| Paraneoplastic pemphigus |
| Lichenoid purpura |
| Lichenoid contact dermatitis |
| Still's disease (adult onset) |
| Late secondary syphilis |
| Porokeratosis |
| Drug eruptions |
| Phototoxic dermatitis |
| Prurigo pigmentosa |
| Erythroderma |
| Mycosis fungoides |
| Regressing warts and tumors |
| Regressing pityriasis rosea |
| Lichen amyloidosis |
| Vitiligo |
| Lichenoid tattoo reaction |
From a morphological standpoint, the psoriasiform tissue reaction (see Chapter 5 ) is defined as epidermal hyperplasia in which there is elongation of the rete ridges, usually in a regular manner ( Fig. 2.3 ).
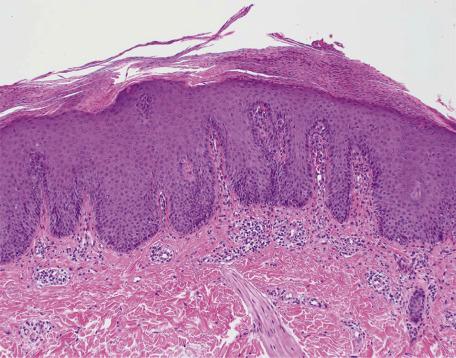
It is acknowledged that this approach has some shortcomings because many of the diseases in this category, including psoriasis, show no significant epidermal hyperplasia in their early stages. Rather, dilated vessels in the papillary dermis and an overlying suprapapillary scale may be the dominant features in early lesions of psoriasis. Mitoses are increased in basal keratinocytes in this pattern, particularly in active lesions of psoriasis.
The psoriasiform reaction pattern was originally defined as the cyclic formation of a suprapapillary exudate with focal parakeratosis related to it. The concept of the “squirting dermal papilla” was also put forward with the suggestion that serum and inflammatory cells escaped from the blood vessels in the papillary dermis and passed through the epidermis to form the suprapapillary exudate referred to previously. This “concept,” although outmoded, is useful in considering early lesions of psoriasis in which dilated vessels and surface suprapapillary scale are often the only features. The epidermal hyperplasia that also occurs was regarded as a phenomenon secondary to these other processes .
Diseases showing the psoriasiform reaction pattern are listed in Table 2.2 .
| Psoriasis |
| Psoriasiform keratosis |
| AIDS-associated psoriasiform dermatitis |
| Pustular psoriasis |
| Reiter's syndrome |
| Pityriasis rubra pilaris |
| Parapsoriasis |
| Lichen simplex chronicus |
| Benign alveolar ridge keratosis |
| Subacute and chronic spongiotic dermatitides |
| Erythroderma |
| Mycosis fungoides |
| Chronic candidosis and dermatophytoses |
| Inflammatory linear verrucous epidermal nevus (ILVEN) |
| Norwegian scabies |
| Bowen's disease (psoriasiform variant) |
| Clear cell acanthoma |
| Lamellar ichthyosis |
| Pityriasis rosea (“herald patch”) |
| Pellagra |
| Acrodermatitis enteropathica |
| Glucagonoma syndrome |
| Secondary syphilis |
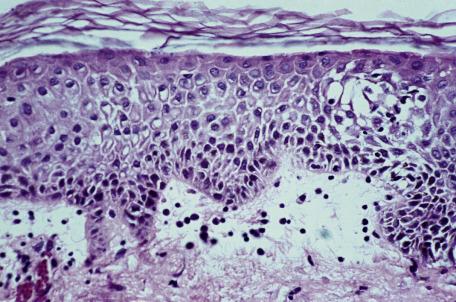
The spongiotic reaction pattern (see Chapter 6 ) is characterized by intraepidermal intercellular edema (spongiosis) . It is recognized by the presence of widened intercellular spaces between keratinocytes, with elongation of the intercellular bridges ( Fig. 2.4 ). The spongiosis may vary from microscopic foci to grossly visible vesicles. This reaction pattern has been known in the past as the “eczematous tissue reaction.” Inflammatory cells are present within the dermis, and their distribution and type may aid in making a specific diagnosis within this group. This is the most difficult reaction pattern in which to make a specific clinicopathological diagnosis; often a diagnosis of “spongiotic reaction consistent with …” is all that can be made.

The major diseases within this tissue reaction pattern (atopic dermatitis, allergic and irritant contact dermatitis, nummular dermatitis, and seborrheic dermatitis) all show progressive psoriasiform hyperplasia of the epidermis with chronicity ( Fig. 2.5 ). This change is usually accompanied by diminishing spongiosis, but this will depend on the activity of the disease. Both patterns may be present in the same biopsy. The psoriasiform hyperplasia is, in part, a response to chronic rubbing and scratching.

Six patterns of spongiosis can be recognized:
Neutrophilic spongiosis (where there are neutrophils within foci of spongiosis)
Eosinophilic spongiosis (where there are numerous eosinophils within foci of spongiosis)
Miliarial (acrosyringial) spongiosis (where the edema is related to the acrosyringium)
Follicular spongiosis (where the spongiosis is centered on the follicular infundibulum)
Pityriasiform spongiosis (where the spongiosis forms small vesicles containing lymphocytes, histiocytes, and Langerhans cells)
Haphazard spongiosis (the other spongiotic disorders in which there is no particular pattern of spongiosis).
The diseases showing the spongiotic reaction pattern are listed in Table 2.3(a) .
| Neutrophilic spongiosis |
| Pustular psoriasis/Reiter's syndrome |
| Prurigo pigmentosa |
| IgA pemphigus |
| Infantile acropustulosis |
| Acute generalized exanthematous pustulosis |
| Palmoplantar pustulosis |
| Staphylococcal toxic shock syndrome |
| Neisserial infections |
| Dermatophytosis/candidosis |
| Beetle (Paederus) dermatitis |
| Pustular contact dermatitis |
| Glucagonoma syndrome |
| Amicrobial pustuloses |
| Periodic fever syndromes |
| Eosinophilic spongiosis |
| Pemphigus (precursor lesions) |
| Herpetiform pemphigus |
| Pemphigus vegetans |
| Bullous pemphigoid/cicatricial pemphigoid |
| Herpes gestationis |
| Idiopathic eosinophilic spongiosis |
| Eosinophilic, polymorphic, and pruritic eruption |
| Allergic contact dermatitis |
| Protein contact dermatitis |
| Atopic dermatitis |
| Arthropod bites |
| Eosinophilic folliculitis |
| Incontinentia pigmenti (first stage) |
| Drug reactions |
| ID reaction |
| Still's disease |
| Wells’ syndrome |
| Miliarial spongiosis |
| Miliaria (may look pityriasiform on random section) |
| Follicular spongiosis |
| Infundibulofolliculitis |
| Atopic dermatitis (follicular lesions) |
| Apocrine miliaria |
| Eosinophilic folliculitis |
| Follicular mucinosis |
| Infectious folliculitides |
| Perioral dermatitis |
| Pityriasiform spongiosis |
| Pityriasis rosea |
| Pityriasiform drug reaction |
| Erythema annulare centrifugum |
| Allergic contact dermatitis |
| Nummular dermatitis |
| Lichen striatus (uncommonly) |
| Gianotti–Crosti syndrome (sometimes) |
| Other spongiotic disorders |
| Irritant contact dermatitis |
| Allergic contact dermatitis |
| Nummular dermatitis |
| Sulzberger–Garbe syndrome |
| Seborrheic dermatitis |
| Atopic dermatitis |
| Papular dermatitis |
| Pompholyx |
| Unclassified eczema |
| Hyperkeratotic dermatitis of the hands |
| Juvenile plantar dermatosis |
| Vein graft donor–site dermatitis |
| Stasis dermatitis |
| Autoeczematization (ID reaction) |
| Dermal hypersensitivity reaction/urticarial dermatitis |
| Pityriasis rosea |
| Papular acrodermatitis of childhood |
| Spongiotic drug reactions |
| Autoimmune progesterone dermatitis |
| Estrogen dermatitis |
| Chronic superficial dermatitis |
| Perioral dermatitis |
| Blaschko dermatitis |
| Psoriasis (spongiotic and site variants) |
| Light reactions (particularly polymorphic light eruption) |
| Dermatophytoses |
| Arthropod bites |
| Grover's disease (spongiotic variant) |
| Toxic shock syndrome |
| PUPPP |
| Herpes gestationis (early) |
| Erythema annulare centrifugum (not always pityriasiform) |
| Figurate erythemas |
| Pigmented purpuric dermatoses |
| Pityriasis alba |
| Eczematoid GVHD |
| Allograft rejection |
| Eruption of lymphocyte recovery |
| Lichen striatus |
| Lichen simplex chronicus |
| Sweet's syndrome |
| Erythroderma |
| Mycosis fungoides |
| Acrokeratosis paraneoplastica |
A seventh pattern, which is really a variant of haphazard spongiosis, combines epidermal spongiosis with subepidermal edema ( Fig. 2.6 ), which can vary from mild to severe, even forming subepidermal blisters. Its causes are listed in Table 2.3(b) .
| Arthropod bites and bite-like reactions in lymphoma |
| Cercarial dermatitis/larva migrans |
| PUPPP |
| Autoeczematization |
| Superantigen ID reaction |
| Allergic contact dermatitis (“dermal type”) |
| Contact urticaria, papular urticaria |
| Dermal hypersensitivity/urticarial dermatitis |
| Erysipelas, erysipeloid |
| Dermatophytoses |
| Prebullous pemphigoid |
| Sweet's syndrome |
| Wells’ syndrome |
| Miliaria rubra |
| Pompholyx |
| Polymorphic light eruption |
| Spongiotic drug reactions (including estrogen/progesterone dermatitis) |
In the vesiculobullous reaction pattern, there are vesicles or bullae at any level within the epidermis or at the dermoepidermal junction (see Chapter 7 ). A specific diagnosis can usually be made in a particular case by assessing three features—the anatomical level of the split, the underlying mechanism responsible for the split, and, in the case of subepidermal lesions, the nature of the inflammatory infiltrate in the dermis.
The anatomical level of the split may be subcorneal, within the stratum malpighii, suprabasal, or subepidermal. The mechanism responsible for vesiculation may be exaggerated spongiosis, intracellular edema and ballooning (as occurs in viral infections such as herpes simplex), or acantholysis. Acantholysis is the loss of coherence between epidermal cells. It may be a primary phenomenon or secondary to inflammation, ballooning degeneration (as in viral infections of the skin), or epithelial dysplasia. In the case of subepidermal blisters, electron microscopy and immunoelectron microscopy could be used to make a specific diagnosis in most cases. In practice, the subepidermal blisters are subdivided on the basis of the inflammatory cell infiltrate within the dermis ( Fig. 2.7 ). Knowledge of the immunofluorescence findings is often helpful in categorizing the subepidermal blistering diseases.
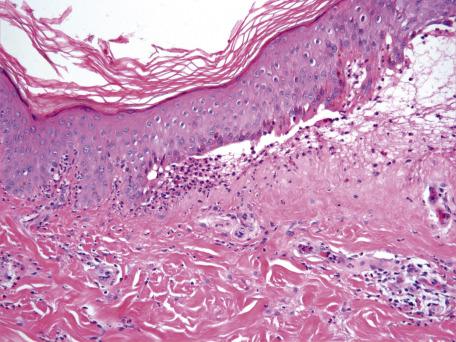
Table 2.4 lists the various vesiculobullous diseases, based on the anatomical level of the split and, in the case of subepidermal lesions, the predominant inflammatory cell within the dermis.
| Intracorneal and subcorneal blisters |
| Peeling skin syndrome |
| Adult Still's disease |
| Impetigo |
| Staphylococcal “scalded skin” syndrome |
| Dermatophytosis |
| Pemphigus foliaceus and erythematosus |
| Herpetiform pemphigus |
| Subcorneal pustular dermatosis |
| IgA pemphigus |
| Infantile pustular dermatoses |
| Acute generalized exanthematous pustulosis |
| Miliaria crystallina |
| Intraepidermal (stratum malpighii) blisters |
| Spongiotic blistering diseases |
| Palmoplantar pustulosis |
| Amicrobial pustulosis of autoimmune diseases |
| Erosive pustular dermatosis of leg |
| Viral blistering diseases |
| Epidermolysis bullosa simplex (localized type) |
| Friction blister |
| Suprabasilar blisters |
| Pemphigus vulgaris and vegetans |
| Paraneoplastic pemphigus |
| Hailey–Hailey disease |
| Darier's disease |
| Grover's disease |
| Acantholytic solar keratosis |
| Subepidermal blisters with little inflammation |
| Epidermolysis bullosa |
| Porphyria cutanea tarda and pseudoporphyria |
| Bullous pemphigoid (cell-poor variant) |
| Burns and cryotherapy |
| Toxic epidermal necrolysis |
| Suction blisters |
| Blisters overlying scars |
| Bullous solar elastosis |
| Bullous amyloidosis |
| Waldenström's macroglobulinemia |
| Drug reactions |
| Kindler's syndrome |
| Subepidermal blisters with lymphocytes |
| Erythema multiforme |
| Paraneoplastic pemphigus |
| Bullous fixed drug eruption |
| Lichen sclerosus et atrophicus |
| Lichen planus pemphigoides |
| Polymorphic light eruption |
| Fungal infections |
| Dermal allergic contact dermatitis |
| Bullous leprosy |
| Bullous mycosis fungoides |
| Subepidermal blisters with eosinophils * |
| Wells’ syndrome |
| Bullous pemphigoid |
| Pemphigoid gestationis |
| Arthropod bites (in sensitized individuals) |
| Drug reactions |
| Epidermolysis bullosa |
| Subepidermal blisters with neutrophils * |
| Dermatitis herpetiformis |
| Linear IgA bullous dermatosis |
| Mucous membrane pemphigoid |
| Ocular cicatricial pemphigoid |
| Localized cicatricial pemphigoid |
| Deep lamina lucida (anti-p105) pemphigoid |
| Anti-p200 pemphigoid |
| Bullous urticaria |
| Bullous acute vasculitis |
| Bullous lupus erythematosus |
| Erysipelas |
| Sweet's syndrome |
| Epidermolysis bullosa acquisita |
| Subepidermal blisters with mast cells |
| Bullous urticaria pigmentosa |
| Miscellaneous blistering diseases |
| Drug overdose–related bullae |
| Methyl bromide–induced bullae |
| Etretinate-induced bullae |
| PUVA-induced bullae |
| Cancer-related bullae |
| Lymphatic bullae |
| Bullous eruption of diabetes mellitus |
* Varying admixtures of eosinophils and neutrophils may be seen in cicatricial pemphigoid and late lesions of dermatitis herpetiformis.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here