Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Amniotic fluid surrounds the developing embryo throughout gestation, protecting the fetus and umbilical cord from trauma while allowing mobility to facilitate structural growth and development. The underlying forces that regulate amniotic fluid are exceedingly complex and dynamic, synchronizing volumes within relatively narrow ranges and providing an indirect measure of fetal well-being. Understanding the pathways of water and solute exchange provides the physiologic basis of the pathologic conditions of oligohydramnios and polyhydramnios.
Several pathways for movement of amniotic fluid and solutes exist, including fetal swallowing, urination, pulmonary secretions, intramembranous movement between fetal blood and the placenta, and transmembranous movement across the amnion and chorion. To understand how variations in amniotic fluid volume can affect the fetus, we must first understand fluid dynamics.
Amniotic fluid contains 98%-99% water, with its solute composition changing throughout gestation. In early pregnancy, the factors that affect amniotic fluid volume are not well known. What is known is that the osmolarity of amniotic fluid and maternal plasma are similar, suggesting that amniotic fluid is a transudate of maternal plasma originating from either placental surfaces or coming through fetal skin. The fetal skin remains nonkeratinized until week 22-25, allowing it to act as a membrane through which amniotic fluid can readily pass. As the fetal skin becomes keratinized and renal function matures, the hypotonic fetal urine causes the amniotic fluid osmolality to decline from 290 mOsm/kg in the first trimester to approximately 255 mOsm/kg near term. Conversely, concentrations of creatinine, urea, and uric acid from the fetal urine increase progressively in the amniotic fluid. Low concentrations of proteins (principally albumin) are found in late pregnancy and provide a minor source of nutrition for the developing fetus. Near term, the amniotic fluid contains increased particulate matter from desquamated fetal skin and gastrointestinal cells, hair, vernix caseosa, stem cells, and occasionally meconium.
Surface exchange is the main component of fluid dynamics in early pregnancy before skin keratinization. Beyond 24 weeks, the surfaces in the mouth and nose can additionally act to exchange fluid, but this is not considered a major source of amniotic fluid volume regulation. A fundamental source of amniotic fluid, particularly in the second half of pregnancy, is the fetal renal system. This is evidenced by the almost complete lack of amniotic fluid in fetuses with renal agenesis or bladder outlet obstructions. Secretions of the respiratory tract, fetal swallowing, and transport within and across the fetal membranes also contribute to amniotic fluid volume ( Fig. 24.1 ).
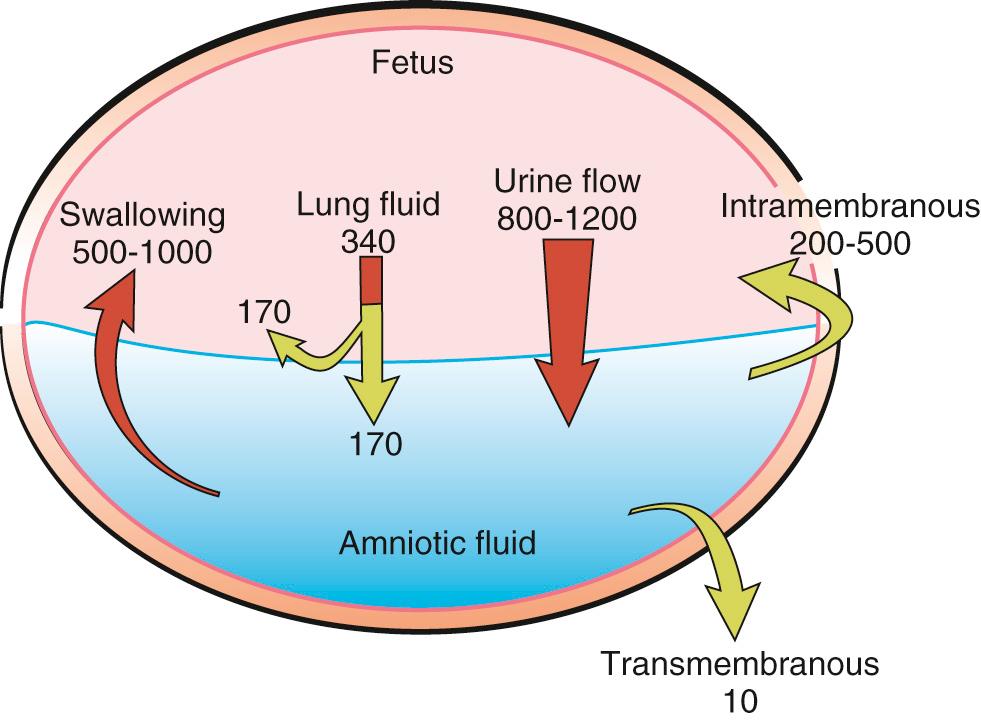
Confirmation of a functioning renal system first starts around 8-11 weeks, when urine is initially observed in the fetal bladder. The dilute urine that is produced is thought to cause the observed drop in osmolality and sodium concentration in the amniotic fluid that persists until delivery. As pregnancy advances, urinary byproducts are observed at two to three times the concentration found in fetal plasma. Estimates of fetal urine output have been made by three-dimensional (3D) measurements of bladder volume at timed intervals and have been found to range from 7-70 mL/hour from 24 weeks until delivery in two separate studies. Fetal urine production is characteristically decreased in pregnancies with abnormalities of placental function that result in intrauterine growth restriction (IUGR). However, no correlation is observed between fetal weight and urine production in normal pregnancies. In normal pregnancies, low fetal urine output is not associated with lower Apgar scores, pH less than 7.25, or late decelerations in labor.
Fluid is excreted from the fetal pulmonary system. Approximately 300-400 mL of fluid is secreted per day, driven by chloride ion exchange across the pulmonary epithelium. It has been demonstrated in fetal sheep by Brace et al. that half of secreted lung fluid enters the amniotic fluid, and the other half is swallowed upon exiting the trachea. Amniotic fluid is prevented from entering the lungs by the closed larynx, producing a new efflux of fluid from the fetal lungs. This was confirmed in an investigation by Liley et al. in which the intra-amniotic injection of contrast media resulted in, with the exception of a few cases, undetectable contrast in the fetal or neonatal lung. The net amount of lung fluid entering the fetal amniotic fluid is approximately 150-200 mL per day. As further evidence of secreted lung fluid being found in amniotic fluid, the phospholipids found in amniotic fluid, which are produced by pulmonary cells, are found in the amniotic fluid. Although meconium staining is not infrequent, with rates in the literature of 7%-27% based on the population, meconium aspiration past the trachea is rare, less than 1%-5%, and is usually associated with accompanying neonatal hypoxia. Rarely, the aspiration of amniotic fluid uncontaminated by meconium has been reported as a cause of neonatal death confirmed at autopsy.
Analogous to renal agenesis being associated with low amniotic fluid, disruption in fetal swallowing is associated with excess amniotic fluid volume. The human fetus demonstrates swallowing around the same time that urine production begins, at 8-11 weeks, and the swallowed volume increases with gestational age. The ovine model has been used historically to approximate human fetal development. Studies evaluating the volume of amniotic fluid swallowed by the fetus were done primarily in sheep models and were found to range between 210 and 1085 mL/day. Ovine studies have also shown that the amount of amniotic fluid swallowed daily in late gestation is correlated with the amniotic fluid volume, suggesting that the fetus can react and attempt to regulate its amniotic fluid surroundings. This attempted regulation is not thought to be a major controller of volume, although the fetus is able to modulate its swallowing. In primate studies, esophageal ligation initially resulted in polyhydramnios, but the polyhydramnios did not persist across gestation, and at the time of delivery the amniotic fluid volume was normal. Observations of swallowed amniotic fluid volume in near-term fetuses range from 210-840 mL per day (average 565 mL). This volume is equivalent to 5%-10% of the fetal body weight.
Excess fluid not removed by fetal swallowing is reabsorbed via an intramembranous pathway. More specifically, this is the reabsorption of fluid and solutes from the amniotic compartment to the fetal blood via the amnion. This pathway is capable of absorbing large quantities of fluid. As the fluid requirements of the fetus increase with increasing gestation, water flow from the amniotic cavity to the fetal circulation via the fetal membranes increases up to 400 mL/day. This is thought to be regulated at least partially by aquaporins found in the human chorioamniotic membranes and the placenta. Aquaporins are small (26- to 34-kDa) cell membrane proteins, which regulate water flux across the cell membranes, have been found to be adaptive to abnormal amniotic fluid levels, and may represent a possible future therapeutic target for abnormal amniotic fluid volume regulation. The osmotic gradient between the amniotic fluid and fetal blood also drives fluids and solutes from the amniotic fluid into the fetal blood. The flow of solutes and fluid, although bidirectional, is not necessarily equal. Vascular endothelial growth factor (VEGF) may also have an impact on intramembranous absorption of fluid.
Transmembranous movement of amniotic fluid describes the transport of water and solutes from the maternal circulation to the amniotic compartment via the placenta. Overall, this has been found to be an insignificant contribution to amniotic fluid dynamics.
The evaluation of amniotic fluid volume is important during the routine ultrasound evaluation of the fetus, because variation above or below normal is associated with increased perinatal morbidity and mortality. Amniotic fluid volume can be measured directly at the time of cesarean delivery (with corrections made for blood contamination) or by dye-dilution techniques using para-amino hippurate. The direct measurement can only be done on a single occasion at the time of cesarean delivery, making it impractical for serial assessments of amniotic fluid volume, and the dye-dilution technique is invasive (requires an amniocentesis), necessitates laboratory support to calculate the amniotic fluid volume, and is also impractical for the clinical determination of amniotic fluid volume. The relative accuracy of this indicator dilution technique has been validated by comparing the actual amniotic fluid volume measured at cesarean delivery with the determined by dye-dilution technique, showing only a 7% difference between the dye-determined and direct-measurement volumes ( r = .99, P <.001). Magnetic resonance imaging has also been evaluated as a means for estimating amniotic fluid volume ; however, this is an impractical approach to everyday screening. Although the aforementioned techniques to measure amniotic fluid volume are accurate, they are time consuming, cumbersome, and may require laboratory support; therefore, the volumes are usually estimated by ultrasound.
Sonographic techniques to estimate amniotic fluid volume include subjective (qualitative) estimation and semiquantitative methods: measurement of maximum vertical or single deepest pocket (SDP) and the amniotic fluid index (AFI). Advantages of sonographic estimates include that they are simple to perform; easy to teach to residents, midwives, and nurses; and are reproducible. In a study comparing all three measurements in singleton pregnancies with dye-dilution technique across different operator experiences, the accuracy of subjective and objective (sonographic) estimates was not affected by operator experience and was similarly accurate in identifying normal amniotic fluid volume. The disadvantage is that they are two-dimensional (2D) representations of a complex, 3D structure with limited significance relative to clinical outcome. Although each has been studied extensively, no technique is universally considered superior at predicting perinatal outcome. In the same study, the accuracy of estimates of all three techniques ranged from 65%-70%, and the three sonographic estimates were similar, ranging from 59%-67%. Alarmingly, none of the techniques were accurate in the identification of abnormal volumes (oligohydramnios, or low amniotic fluid, and polyhydramnios, or high amniotic fluid). The accuracy in the identification of normal volumes in twin pregnancies was dismal, ranging from 7%-29%.
Subjective estimation of amniotic fluid is based on the overall sonographic visual impression of the amniotic fluid volume by an experienced sonographer, established by visualizing echolucent pockets between the fetal parts and uterine wall without measurements. Qualitative estimation of the fluid is characterized as normal, increased, reduced, or absent.
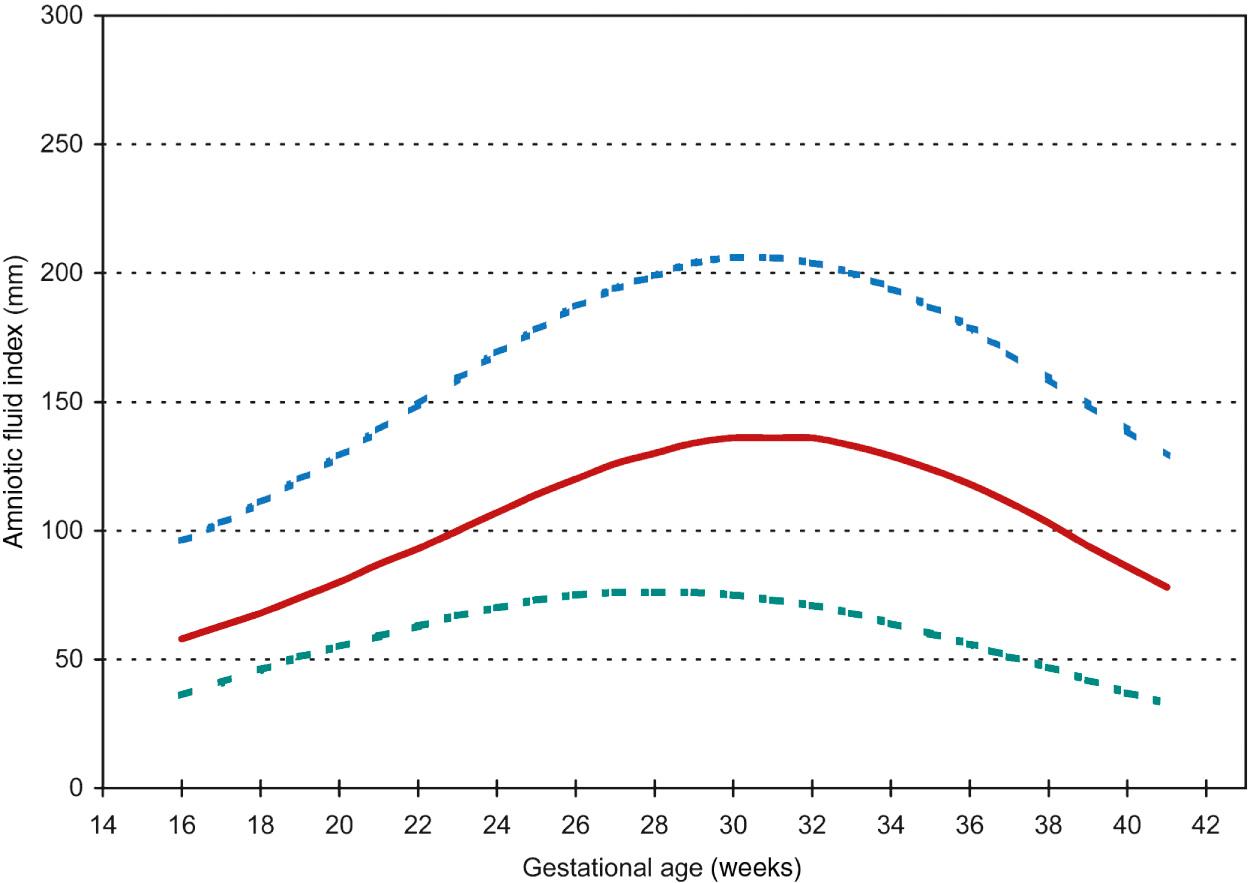
Amniotic fluid index is the summation of the vertical diameter of the largest pocket in each of the four quadrants, as proposed by Phelan and Rutherford. It uses the maternal umbilicus as the central reference point and requires the transducer to be oriented in the longitudinal axis of the maternal abdomen and be kept perpendicular to the floor during scanning. The AFI correlates closely with actual amniotic fluid volume as determined by dye-dilution techniques for normal fluid volume, although it loses its predictive value at the upper and lower ends of the extreme. Moore and Cayle established the mean and outer boundaries (5th and 95th percentiles, respectively) for the AFI from 16-42 weeks’ gestation in a study of 791 normal pregnancies. Magann et al. also established 5th-95th percentiles, respectively, as 4.2-14.9 cm for AFI at gestational age 37-41 weeks. Both can be seen in Fig. 24.2 . The mean AFI is approximately 12-14 cm throughout most of pregnancy, declining after 33 weeks. Above the 95th percentile is approximately 20 cm and below the 5th percentile is approximately 7 cm. The absolute maximum normal AFI is less than 24 or 25 cm. Typical limits for amniotic fluid assessment by AFI are defined in Table 24.1 .
| Amniotic Fluid Volume | AFI Value | Patients |
|---|---|---|
| Polyhydramnios | ≥25 cm | 2% |
| Normal | >8 to <25 cm | 76% |
| Moderate oligohydramnios | ≥5 to ≤8 cm | 20% |
| Severe oligohydramnios | <5 cm | 2% |
The single deepest vertical pocket, as originally described by Chamberlain, is simply found by identifying the largest pocket of amniotic fluid after a global assessment and taking the largest vertical measurement with a horizontal measurement of 1 cm. At gestational age 37-41 weeks, the 5th-95th percentile was 2.3-6 cm for SDP according to nomograms by Magann et al. Typical limits for amniotic fluid assessment by SDP are shown in Table 24.2 .
| Amniotic Fluid Volume | SDP Value | Patients |
|---|---|---|
| Polyhydramnios | ≥8.0 cm | 3% |
| Normal | >2 to <8 cm | 94% |
| Moderate oligohydramnios | ≥1 to ≤2 cm | 2% |
| Severe oligohydramnios | <1 cm | 1% |
As stated, the ideal test for sonographic assessment of amniotic fluid volume has not been firmly established. The ideal semiquantitative sonographic technique should be one that correctly identifies the patients at risk for adverse outcome. Amniotic fluid index, SDP, and a two-diameter pocket (horizontal x vertical measurements in cm 2 ) were compared in a study of 50 normal pregnancies between the gestational ages of 14 and 41 weeks. The prevalence of oligohydramnios as defined by AFI less than or equal to 5 cm, SDP less than 2 cm, or two-diameter pocket less than 15 cm 2 were significantly different ( P <.0001) for the three techniques (8%, 1%, and 30%, respectively). The prevalence of polyhydramnios as defined by AFI greater than 24 cm, SDP greater than 8 cm, or two-diameter pocket greater than 50 cm 2 was also diagnosed with significantly different frequencies ( P <.0001) (0%, 0.7%, and 3%, respectively). Outcome was not assessed in this study.
Subsequently, two prospective randomized trials comparing the use of SDP versus AFI during modified biophysical profiles in complicated pregnancies were performed. Chauhan and colleagues studied 1080 women and found that AFI identified oligohydramnios in 17% of pregnancies compared with 10% identified in SDP-assessed pregnancies ( P = .002). However, there was no difference in delivery mode, NICU admission, umbilical artery pH, or Apgar score. Magann and colleagues had similar findings, showing oligohydramnios was identified in twice as many patients using AFI as compared with SDP in 537 pregnancies (38% vs. 17%, P <.001), resulting in a doubling of inductions of labor (30% vs. 15%, P <.001) and cesarean sections for fetal distress (13% vs. 7%, P <.05). There was, however, no improvement of perinatal outcome. This was confirmed in two meta-analyses comparing the measurement of AFI to SDP. Neither sonographic test was superior to the other. Both poorly identified abnormal volumes when compared with dye-determined or directly measured amniotic fluid volume. However, AFI overdiagnosed clinically insignificant oligohydramnios, leading to more intervention without a difference in outcome, suggesting that the single deepest pocket may be a superior sonographic assessment for antenatal testing of the at-risk pregnancy.
Use of color Doppler has not been shown to aid in identification of pregnancies with adverse outcomes. Using dye-determined volume as the benchmark, Magann et al. compared grayscale ultrasonography to Doppler color ultrasonography and found that Doppler not only overdiagnosed oligohydramnios but labeled 37% of women with normal AFV as having oligohydramnios.
Sahin et al. attempted to apply the Cavalieri method, a mathematical approximation of volumes, to estimate amniotic fluid volume. Their results correlated with concurrent AFI measurements, and although this is a promising theory, it is complicated to perform and has not been correlated with pregnancy outcome or a directly measured volume.
Three-dimensional ultrasound technology has found relevance in the evaluation of the fetus; however, it has not yet found application in the assessment of amniotic fluid. There has been only one attempt to assess third-trimester amniotic fluid volume with 3D ultrasonography. It was concluded that 3D volume datasets are reliable for determining volume; however, it was determined subjectively by the sonographer based on five volume acquisitions and compared with the value obtained during the 2D ultrasound; there was no attempt at calculating a volume based on a gold standard. Interestingly, in the first trimester, growth charts have been developed for gestational sac volume and embryonic volume, the ratio of which correlates positively with gestational age. Clinical applications for this data have not yet been developed, but normal versus abnormal early pregnancies are being investigated to determine if these parameters can help predict early adverse outcomes such as miscarriage.
Gravid women accumulate approximately 6 L of additional fluid volume during pregnancy. Most of this fluid is associated with the conceptus: 2800 mL in the fetus, 400 mL in the placenta, and 700-800 mL of amniotic fluid. The remainder of the fluid is associated with the maternal uterus (800 mL), breasts (500 mL), and maternal blood volume expansion (850 mL). Throughout gestation, amniotic fluid volume is highly regulated, gradually increasing in the first trimester, stabilizing in the second trimester, and decreasing late in the third trimester while remaining in a relatively narrow range of volumes.
Normality can be defined in two ways. Mathematically it can represent the majority of the population as expressed by area under the curve in a Gaussian distributed population. Typically the lower and upper 5% are excluded as “abnormal.” Normality can also be defined in terms of outcome. Undesired outcomes are tracked, and measurements associated with these outcomes are defined as “abnormal.” This second definition is more clinically useful but much more difficult to define.
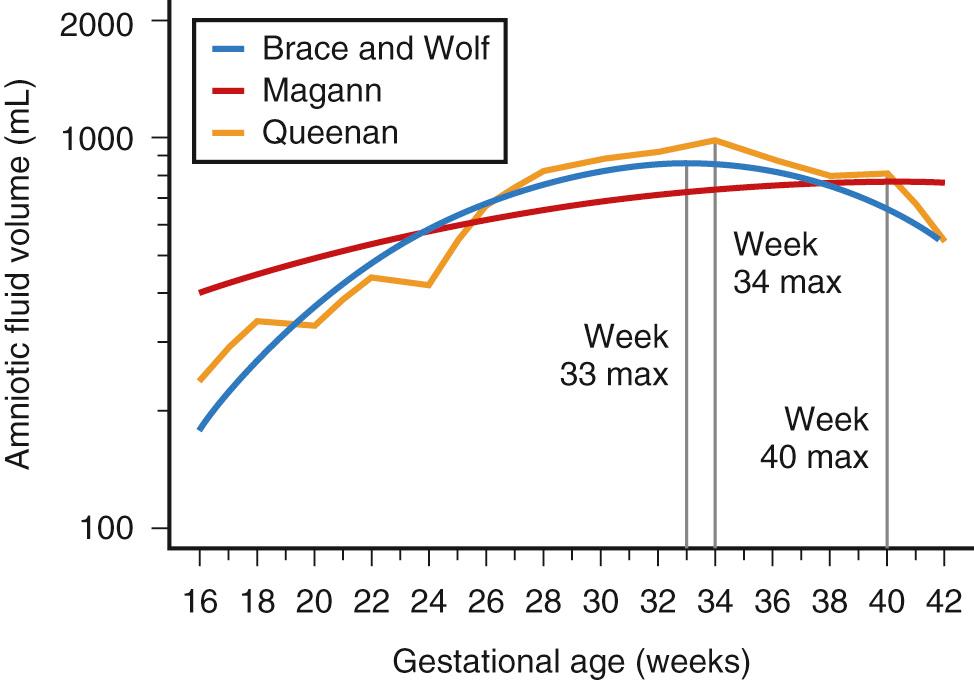
Amniotic fluid volume has been defined in both these manners. The 5th and 95th percentiles have been defined for AFI, SDP, and dye-determined techniques across gestational ages, as fluid has been found to vary significantly throughout pregnancy. Nomograms for amniotic fluid in normal pregnancies were developed by Queenan et al. in 1972 by dye-determined methods, Brace and Wolfe by dye-dilution and direct measurement, and Magann et al. by dye-dilution and direct measurement. ( Fig. 24.3 ). Two of these studies showed that amniotic fluid volume increases steadily in early gestation and remains relatively stable between 22 and 38 weeks, averaging 750-800 mL. Thereafter, amniotic fluid declines by 8% per week, with a mean volume of approximately 500 mL at 40-42 weeks. The third nomogram found that amniotic fluid volume continues to increase throughout gestation, confirming a mean volume of approximately 800 mL at term. A more recent publication evaluated 1,190 normal amniotic fluid volumes (by dye-dilution and direct measurement) across gestation, comparing four statistical methods to construct gestational age-specific reference intervals for amniotic fluid. The investigators determined that quantile regression because of its flexibility and robustness, especially when data is sparse, is the preferred statistical technique to construct gestational age-specific reference for normal amniotic fluid volume ( Table 24.3 ).
| EGA (weeks) | 2.5th | 5th | 50th | 95th | 97.5th |
|---|---|---|---|---|---|
| 10 | 10 | 11.67071 | 33 | 54.5 | 54.5 |
| 11 | 14.67523 | 17.77286 | 46.78348 | 80.00315 | 85.11197 |
| 12 | 20.88759 | 26.02843 | 64.63046 | 114.4997 | 128.7214 |
| 13 | 28.86455 | 36.72243 | 87.08291 | 159.8753 | 188.7096 |
| 14 | 38.76757 | 50 | 114.5418 | 217.9379 | 268.4339 |
| 15 | 50.65874 | 65.81518 | 147.2026 | 290.2362 | 370.8499 |
| 16 | 64.47283 | 83.89999 | 185 | 377.8607 | 498.0732 |
| 17 | 80 | 103.7623 | 227.5713 | 481.246 | 650.9382 |
| 18 | 96.88325 | 124.716 | 274.2441 | 600 | 828.619 |
| 19 | 114.6324 | 145.9388 | 324.053 | 732.7873 | 1028.385 |
| 20 | 132.6541 | 166.5515 | 375.784 | 877.2853 | 1245.544 |
| 21 | 150.2943 | 185.7025 | 428.0441 | 1030.229 | 1473.61 |
| 22 | 166.8892 | 202.647 | 479.3488 | 1187.546 | 1704.68 |
| 23 | 181.8158 | 216.8096 | 528.2168 | 1344.573 | 1930 |
| 24 | 194.5394 | 227.8216 | 573.2642 | 1496.338 | 2140.636 |
| 25 | 204.6493 | 235.533 | 613.2876 | 1637.871 | 2328.18 |
| 26 | 211.8822 | 240 | 647.3289 | 1764.527 | 2485.396 |
| 27 | 216.1299 | 241.4547 | 674.7173 | 1872.273 | 2606.735 |
| 28 | 217.433 | 240.2633 | 695.0874 | 1957.925 | 2688.672 |
| 29 | 215.9638 | 236.8802 | 708.3736 | 2019.309 | 2729.829 |
| 30 | 212 | 231.8045 | 714.7848 | 2055.337 | 2730.905 |
| 31 | 205.8945 | 225.5429 | 714.7645 | 2066 | 2694.441 |
| 32 | 198.0441 | 218.5812 | 708.9409 | 2052.289 | 2624.451 |
| 33 | 188.8609 | 211.3661 | 698.0733 | 2016.051 | 2526 |
| 34 | 178.7474 | 204.2954 | 683 | 1959.804 | 2404.752 |
| 35 | 168.0774 | 197.7177 | 664.5898 | 1886.54 | 2266.555 |
| 36 | 157.1828 | 191.9366 | 643.7017 | 1799.513 | 2117.087 |
| 37 | 146.3463 | 187.2225 | 621.1525 | 1702.055 | 1961.572 |
| 38 | 135.7982 | 183.8267 | 597.6943 | 1597.407 | 1804.597 |
| 39 | 125.7176 | 182 | 574 | 1488.587 | 1650 |
| 40 | 116.2365 | 182.0158 | 550.6572 | 1378.3 | 1500.832 |
| 41 | 107.4454 | 184.1975 | 528.1678 | 1268.871 | 1359.384 |
| 42 | 99.39999 | 188.9548 | 506.9539 | 1162.223 | 1227.244 |
A study of more than 15,000 patients done by Shanks et al. showed that AFI less than the 5th percentile versus a cutoff of 5 cm better predicts fetuses at risk for newborn intensive care (NICU) admission, although this study has been criticized for its retrospective design and the variable intervals between the time the measurement was obtained and the time of delivery. Additionally, an investigation of 291 pregnancies with dye-determined volumes in which the third and fifth percentiles of the AFI were assessed to determine if either of these percentiles was superior to the fixed cutoff of less than 5 for the AFI to detect the 75 pregnancies with oligohydramnios, discovered that both the percentiles (third and fifth) and the fixed cutoff (<5) poorly identified oligohydramnios, but neither was superior to the other.
Multifetal pregnancies are at a higher risk of perinatal morbidity and mortality than singleton pregnancies. When evaluating the health of the amniotic fluid in twin pregnancies, singleton growth curves currently provide the best predictors of adverse outcomes, and the evaluation of fluid with singleton nomograms is frequently being used. This approach appears reasonable because in the only evaluation of volume in third-trimester diamniotic twin pregnancies, the amniotic fluid volume, or each sac, was similar to that of normal singleton pregnancies. Morin and Lim performed a literature review with the Diagnostic Imaging Committee of the Society of Obstetricians and Gynaecologists of Canada and suggest that the SDP should be used in each sac with standard definitions for singleton pregnancies, although they state that there is not enough evidence to suggest that one method is more predictive than the others of adverse pregnancy outcome. The AFI has been used to estimate amniotic fluid volume in twins; however, when using the summated AFI (measurement of all four quadrants as is done in singletons but without taking membrane placement into consideration), the measurement poorly predicts dye-determined oligohydramnios and polyhydramnios.
In a single prospective observational study of 299 diamniotic twin pregnancies, SDP was found to be constant between 17 and 37 weeks, with an increase in fetal labor intolerance and neonatal morbidity in twin pregnancies with polyhydramnios. Alternate fixed cutoffs of less than 2.2 cm for a low amniotic fluid volume and greater than 7.5 cm for a high amniotic fluid volume were suggested in diamniotic twin pregnancies to identify pregnancies at risk for adverse intrapartum and neonatal outcomes ( Table 24.4 ).
| EGA (weeks) | 2.5th | 5th | 10th | 50th | 90th | 95th | 97.5th | Twin Sacs |
|---|---|---|---|---|---|---|---|---|
| <17 | 2.02 | 2.11 | 2.54 | 3.96 | 6.48 | 6.86 | 6.88 | 22 |
| 17-19 | 2.13 | 2.14 | 2.64 | 4.07 | 6.56 | 6.97 | 7.02 | 143 |
| 20-22 | 2.22 | 2.23 | 2.79 | 4.22 | 6.6 | 7.04 | 7.21 | 276 |
| 23-25 | 2.23 | 2.28 | 2.93 | 4.38 | 6.67 | 7.19 | 7.45 | 322 |
| 26-28 | 2.29 | 2.37 | 3.06 | 4.53 | 6.8 | 7.44 | 7.79 | 390 |
| 29-31 | 2.43 | 2.46 | 3.2 | 4.67 | 6.91 | 7.69 | 8.09 | 323 |
| 32-34 | 2.44 | 2.51 | 3.32 | 4.8 | 6.98 | 7.76 | 8.27 | 283 |
| 35-37 | 2.41 | 2.54 | 3.39 | 4.9 | 7.01 | 7.81 | 8.39 | 129 |
Hernandez et al. performed a retrospective review of 1951 twin pregnancies, both dichorionic and monochorionic, with twin–twin transfusion syndrome excluded. Polyhydramnios was identified in 18% of pregnancies, defined as mild (SDP 8–9.9 cm), moderate (SDP 10–11.9 cm), and severe (SDP ≥12 cm). Pregnancy outcomes were reviewed, and polyhydramnios was not associated with preterm delivery, fetal growth restriction, NICU admission, or neonatal death in either twin. The incidence of major anomalies became more common in increasing polyhydramnios in both monochorionic and dichorionic pregnancies with a prevalence of almost 20% in severe polyhydramnios. Severe polyhydramnios was significantly associated with stillbirth in monochorionic pregnancies (27%, P <.001).
Assessing relative amniotic fluid volume in twin pregnancies is also important because disparity in amniotic fluid volume is a significant component of twin oligohydramnios-polyhydramnios sequence (TOPS), of which twin–twin transfusion syndrome (TTTS) is the extreme endpoint. Twin–twin transfusion syndrome carries with it a three- to fivefold increase in mortality and morbidity. It is often uncovered with ultrasound evidence of discordant fetal weights and amniotic fluid in monochorionic/diamniotic twins, although it has been documented in monochorionic/monoamniotic pregnancies. It may also present sonographically as a “stuck twin,” with the fetus appearing to be suspended to the uterine wall with no intervening membrane visible, or there may be tightly adhered membranes that make a fetus appear wrapped in plastic wrap ( Fig. 24.4 ). It is recommended that evaluation for twin–twin transfusion in these pregnancies should begin in the second trimester with weekly surveillance if discordant and evaluation every 2 weeks for concordant pregnancies.
Deviation of amniotic fluid volume outside the normal range is associated with increased pregnancy loss. As early as the first trimester, low gestational sac fluid volume is predictive of increased rates of spontaneous abortion, 94% versus 8% in patients with normal sac size. In the second and third trimesters, departure above or below normal volumes increases the perinatal mortality (PNM) rate ( Fig. 24.5 ). When diagnosis was made in the third trimester, patients fared better, although there was still a 15% PNM rate.
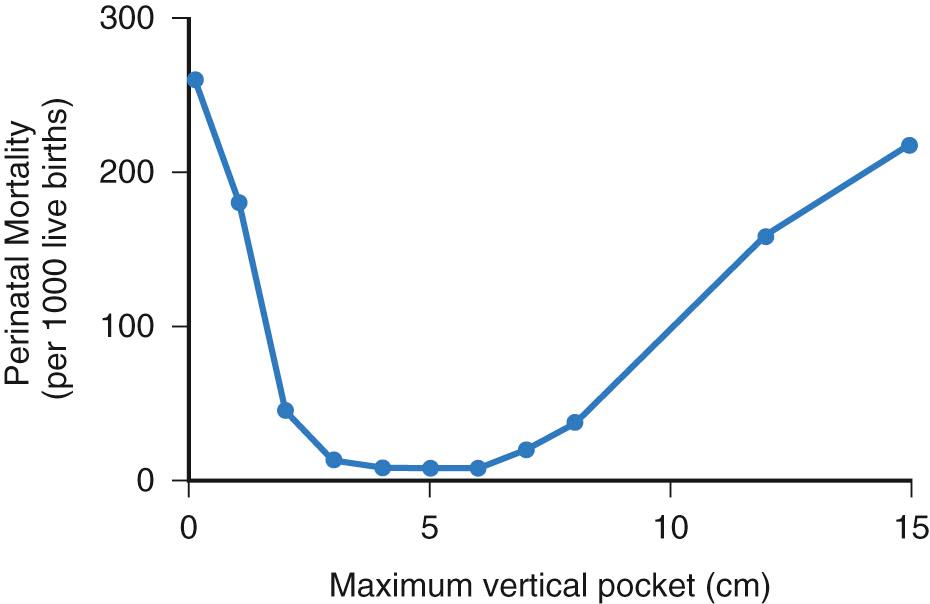
Oligohydramnios has been defined as an amniotic fluid volume that is less than 200 mL or 500 mL and occurs in 1%-2% of pregnancies. It is sonographically defined as an SDP less than 2 cm, AFI less than 5 cm, or a subjectively low volume. Borderline fluid has been defined as an AFI between 5 and 8 cm or 5 and 10 cm and has been associated with fetal malformations if diagnosed at age 24 and 34 weeks.
Low amniotic fluid can be caused by underproduction or loss or can be idiopathic. Underproduction can be the result of absent or dysfunctional kidneys, urinary tract obstruction, uteroplacental insufficiency, maternal medications, or maternal dehydration. Loss is caused by rupture of membranes. Box 24.1 lists conditions associated with oligohydramnios. The clinical management of oligohydramnios should be directed toward diagnosing and alleviating remediable underlying conditions. Appropriate work-up for this abnormality is a review of the maternal history for evidence of rupture, anatomic evaluation of the renal system and bladder, and assessment of placental function and fetal growth and pulmonary status.
Chromosomal abnormalities
Congenital anomalies
Genitourinary (renal agenesis, polycystic or multicystic dysplastic kidneys, ureteral or urethral obstruction)
Intrauterine growth restriction
Intrauterine fetal demise
Postmaturity
Premature rupture of membranes (occult or overt)
Preterm
Prolonged
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here