Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Adnexal tumors or tumorous proliferations are generally classified according to two principles, (1) benign versus malignant and (2) line of differentiation, that is, which normal cutaneous structure the lesion most resembles (hair follicle, apocrine or eccrine gland, or sebaceous gland). Although such a classification scheme seems simple at first glance, the reality is that there is still considerable controversy on how to catalog many lesions. The nomenclature that has evolved historically is sometimes confusing, with many different names for essentially the same type of tumor.
When confronted with adnexal tumors, a pragmatic approach is recommended. The most important issue is to distinguish benign from malignant tumors. In general, benign neoplasms are well differentiated, circumscribed, and noninfiltrative. Malignant tumors tend to be infiltrative or show cytologic atypia.
Subclassification of malignant tumors is important when the tumor types being considered in the differential diagnosis greatly differ in their biologic potential (e.g., low-grade tumors, e.g., microcystic adnexal carcinoma (MAC), which does not metastasize, versus porocarcinoma, which has a propensity for spread to lymph nodes and visceral sites). Subclassification is clinically irrelevant when the biologic potential is the same (eccrine versus apocrine duct carcinoma). Precise classification of benign tumors, although from a clinical care point of view is often irrelevant, is encouraged when possible for the sake of accuracy and because some tumors may alert the clinician to the possibility of a genetic predisposition to internal cancers (e.g., sebaceous neoplasms and Muir-Torre syndrome [MTS]; fibrofolliculoma and Birt-Hogg-Dubé syndrome).
This chapter is not comprehensive. Rather, it provides an overview of common tumors and discusses important aspects in the differential diagnosis.
This rare lesion is characterized by a locally increased number of mature hair follicles.
This rare hamartoma is characterized by numerous dilated hair follicles resembling acne comedones or small infundibular cysts.
Dilated pore and pilar sheath acanthoma and trichofolliculoma tend to occur on the skin of the head and neck and typically present as a solitary small cyst, papule, or small nodule.
Dilated pore of Winer shares with an infundibular cyst the presence of stratified squamous epithelium with granular layer and loosely packed keratin. However, in contrast to a cyst, which is lined by flattened epithelium, rete ridges surround a dilated pore ( Fig. 11-1 ). If the cyst lining epithelium is acanthotic, the term pilar sheath acanthoma ( Fig. 11-2 ) is used.
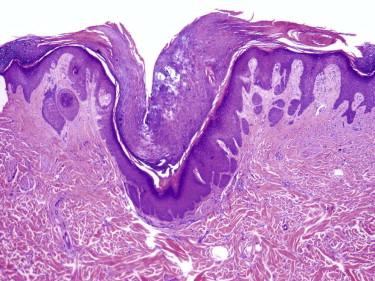
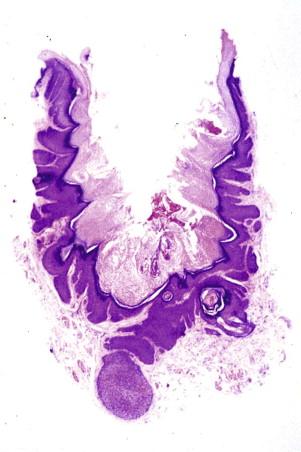
Trichofolliculoma is characterized by a central cystically ectatic follicle, from which several small follicles with variable maturity radiate ( Fig. 11-3 ).
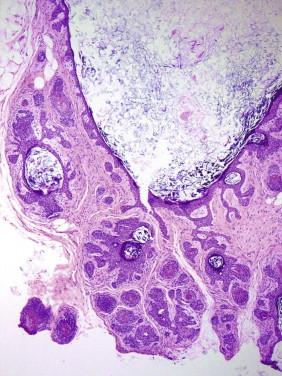
This uncommon benign tumor is composed of a plate-like growth of keratinocytes with several points of attachment to the epidermis and the external root sheath of hair follicles ( Fig. 11-4 ). Its cells may have basaloid features and simulate a superficial basal cell carcinoma (BCC) or the superficial portion of an infundibulocystic BCC. In contrast with a bona fide BCC, an infundibuloma lacks hyperchromatic nuclei, myxoid stroma, or peripheral clefting. On occasion, an infundibuloma may also be difficult to distinguish from an irritated endophytic seborrheic keratosis (SK).
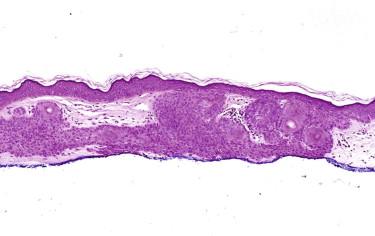
Trichoblastoma (TBL) and trichoepithelioma (TE) are benign follicular fibroepithelial tumors. Many contemporary dermatopathologists consider TE a superficial form of TBL. TE and TBL are used synonymously in the 2006 World Health Organization classification of skin tumors. We share this view; therefore, we describe TE together with TBL herein. Because desmoplastic TE raises diagnostic issues different from TBL, it is discussed separately.
Trichoblastoma typically presents as sporadic, solitary, flesh-colored, small (2- to 8-mm) papules on the face. Rarely, it may form a larger nodule (reported in the literature as giant solitary TE). Uncommonly, multiple TBLs ( Fig. 11-5 ) can occur in families as an autosomal dominant condition or as part of Brooke-Spiegler or Rombo syndrome, with mutations of the CYLD or PATCH genes. TBL is a common secondary neoplasm in nevus sebaceus.
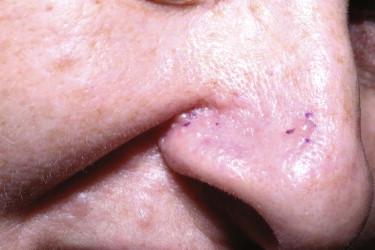
Trichoblastomas are fibroepithelial tumors composed of basaloid epithelial cells with a cellular fibrous stroma distinctly different from the surrounding normal reticular dermis ( Fig. 11-6, A ). A characteristic finding is the presence of papillary-mesenchymal bodies ( Fig. 11-6, B ), which are cuplike proliferations of basaloid cells engulfing fibroblasts, thereby recapitulating the follicular papilla of the hair follicle. Mitotic figures or apoptotic bodies can also be present, but they are not as frequently seen as in BCCs. Stromal calcifications are commonly associated with TBLs.
Superficial lesions of TBL (TEs) typically contain small keratocysts with infundibular keratinization and may form connections to the epidermis. More deeply located tumors usually lack epidermal connections and infundibular cysts.
Trichoblastoma may be inflamed. What has been described in the literature as cutaneous lymphadenoma represents inflamed TBL.
The peritumoral stromal cells are immunopositive for CD34. Bcl-2 is expressed in the basaloid keratinocytes located at the periphery of the cords. Because TBLs are follicular-derived lesions, they usually contain Merkel cells, which can be detected with anti-CK20. A recent marker, CD10, is expressed by the peritumoral spindle cells in TBL and by some of the epithelial tumor cells in BCC. PHLDA1 is primarily expressed in TBL and not in BCC ( Fig. 11-6, C and D ).
The main diagnostic distinction is from BCC. In contrast with BCC, the stroma of TBL shows a tight association with the basaloid epithelial cells. No clefts or retractions are seen between epithelium and stroma, as is characteristic of BCC. Instead, TBL shows a distinct demarcation between its periepithelial stroma and the surrounding dermal collagen. TBL typically contains papillary mesenchymal bodies, which are usually absent in BCC. Mitoses and apoptotic bodies can occur in TBL but are not as common as in BCC. Certain stromal alterations, such as mucin or amyloid deposits, which are typical of BCC, would be unusual for TBL.
Trichoblastic proliferations simulating superficial BCC may occur as an inductive phenomenon along the dermal-epidermal junction overlying a dermatofibroma ( Fig. 11-7 ) or along the follicular epithelial interface in association with a blue nevus.
| Basal Cell Carcinoma | Trichoblastoma | |
|---|---|---|
| Papillary mesenchymal body | Usually absent | Usually present |
| Clefts | Between tumor and stroma | Between tumor stroma and surrounding normal dermis |
| Mitotic figures | Common | Rare |
| Mucinous stroma | Common | Rare |
| PHLDA1 expression | Rare | Usually present |
This tumor typically presents as a small (2- to 9-mm), asymptomatic, slowly growing, indurated area, which may have an atrophic, depressed center ( Fig. 11-8 ).
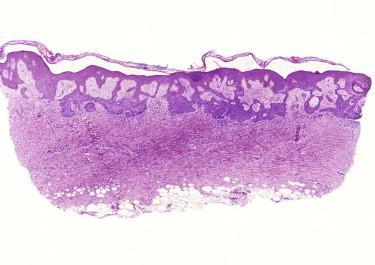
Desmoplastic or sclerosing TE is quite different in appearance from conventional TE or TBL. Although TBL usually forms a spherical nodule, the silhouette of desmoplastic TE is usually that of a superficial dermal plaque. The tumor is characterized by a triad of thin strands of basaloid epithelial cells, keratinous cysts, and a dense fibrous stroma ( Fig. 11-9 ). Additional features may include foreign body giant cell reactions to keratin and calcifications. Focal papillary mesenchymal bodies may be seen.
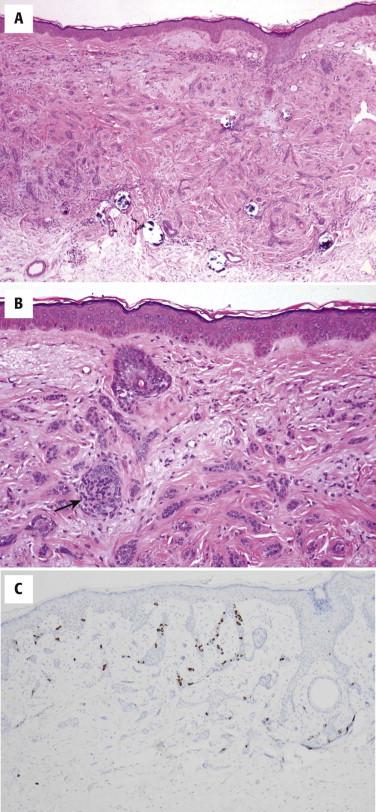
Immunohistochemistry may help because intracytoplasmic lumina may be easier to identify using antibodies for carcinoembryonic antigen (CEA) or BR2 (positive staining argues against desmoplastic TE). CK20-positive Merkel cells are commonly associated with the tumor ( Fig. 11-9, C ). PHLDA1 has recently been reported to be primarily expressed in all types of TBL and TE but not in most BCC.
Desmoplastic TE needs to be distinguished from morpheaform BCC and MAC. Morpheaform BCC tends to show greater asymmetry in its growth and infiltrates more deeply. Furthermore, the degree of nuclear atypia, number of mitoses, or apoptotic bodies is higher in BCC. After foci of more typical features of BCC, such as sheets or small nodules of basaloid cells with peripheral palisading and retraction artifact, are identified, the diagnosis is straightforward. Another helpful feature is the presence of cytologically bland keratocysts. They are commonly seen in desmoplastic TE. Keratocysts may be seen in infiltrative BCCs but rarely in morpheaform variants. If they are present, the tumor cells surrounding the cystic structures are atypical. A possible pitfall is the observation of perineural involvement in desmoplastic TE. Its presence per se does not necessarily imply a diagnosis of carcinoma.
| Desmoplastic Trichoepithelioma | Microcystic Adnexal Carcinoma | Sclerosing Basal Cell Carcinoma | |
|---|---|---|---|
| Papillary mesenchymal bodies | Present | Absent | Absent |
| Keratin cysts | Common | Common | Usually absent |
| Atypia | Absent or mild | Absent or mild | Present |
| Mitoses | Absent or rare | Absent or rare | Common |
| Ducts or tubules | Absent | Usually present | Absent |
| CK20-positive cells | Common | Absent | Absent |
Microcystic adnexal carcinoma or syringoma may also be confused with desmoplastic TE, especially in a small superficial shave biopsy. The presence of papillary mesenchymal bodies excludes syringoma and MAC. On the other hand, if ductal differentiation is identified, desmoplastic TE can be excluded. The identification of CK20- or chromogranin-positive Merkel cells in the tumor is also helpful to support follicular differentiation and to distinguish desmoplastic TE from syringoma or MAC ( Fig. 11-9, C ).
Basaloid follicular hamartoma is a controversial entity. Some consider it a malformative proliferation. Others view it as a genuine tumor, but opinions vary again on whether it is a distinct benign tumor different from TBL and TE or an incipient infundibulocystic BCC. Basaloid follicular hamartoma may be an incidental histologic finding or present clinically as a small papule. Hereditary and nonhereditary, localized and generalized variants have been described.
Basaloid follicular hamartoma is typically described and illustrated as a small dermal proliferation of thin anastomosing strands and branching cords of basaloid cells, which are often adjacent to or attached to hair follicles. Small keratocysts may be seen. The cytology of the basaloid cells is bland, and mitotic figures are absent or rare. As judged from published cases, lesions reported as basaloid follicular hamartoma seem to represent a spectrum of basaloid epithelial proliferations ranging from TBL and TE and tumors indistinguishable from BCCs, in particular infundibulocystic variants of BCC, to a subset of lesions for which a separate designation of basaloid follicular hamartoma may be reasonable.
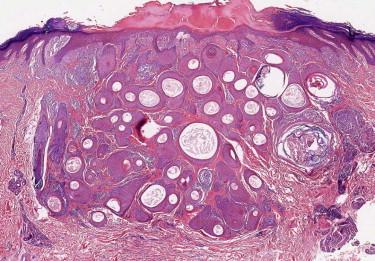
Trichoadenoma is a rare tumor. It typically occurs on the face and is histologically characterized by a circumscribed small superficial dermal nodular aggregate of well-formed keratinous cysts ( Fig. 11-10 ). On a small shave biopsy, one needs to be careful with regard to a definitive diagnosis of trichoadenoma because MAC may show similar features in its superficial portion.
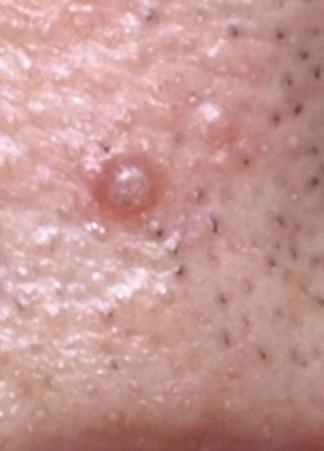
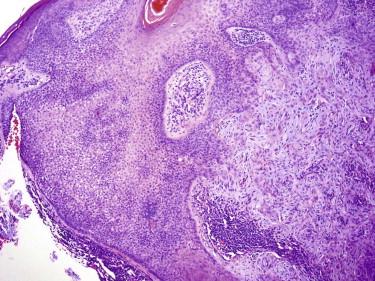
Tricholemmomas are solitary lesions typically present on the face of young individuals ( Fig. 11-11 ). Most of them are sporadic solitary lesions. Multiple lesions are a marker for Cowden syndrome (CS; see the following).
Tricholemmomas are usually small, circumscribed tumors composed of one or more lobules of cuboidal cells with central, round nuclei and lightly pink or clear cytoplasm ( Fig. 11-12 ). There is typically a small popular exophytic component as well as an endophytic part extending into the upper reticular dermis. The lobules tend to connect to the epidermis or follicular infundibulum at several points. At the periphery of the lobules is a layer of columnar basaloid cells with nuclear palisading resembling the outer root sheath. They are usually surrounded by a hyaline pink band reflecting thickened basement membrane material. Clear cell changes are common and reflect abundant cytoplasmic glycogen. Keratinization and squamous eddies may be present.
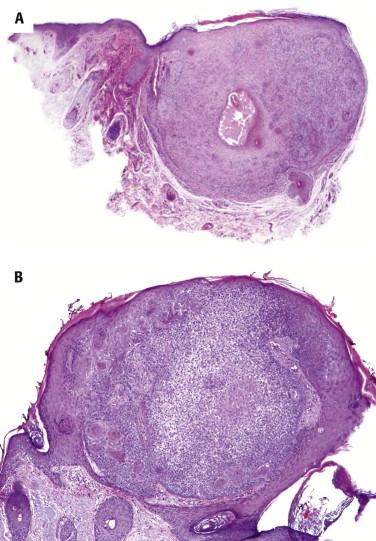
A variant of tricholemmoma shows a hyalinized or desmoplastic stroma ( Fig. 11-13 ), in which the tumor cells often display an irregular and seemingly infiltrative growth pattern (desmoplastic tricholemmoma).
Tricholemmoma typically strongly label for CD34 by immunohistochemistry. A periodic acid-Schiff (PAS) and PAS-diastase (PAS-D) stain can help highlight the presence of glycogen (positivity with PAS is removed after treatment with diastase).
Tricholemmomas share many features with inverted follicular keratosis. However, the latter lacks hyaline membranes, and clear cell or basaloid features are less common. Tricholemmomas show overlap with warts affecting hair follicles, thereby inducing a trichilemmal hyperplasia (so-called trichilemmal warts). Such warts typically have a papillomatous exophytic component ( Fig. 11-14 ). On a partial superficial biopsy, tricholemmoma, especially when irritated and inflamed, may be confused with basal cell or even squamous cell carcinoma (SCC). Attention to the bland cytology of the tumor cells should avoid this pitfall. In problematic cases, immunohistochemistry for CD34 can help because strong labeling is only seen in tricholemmomas but not basal or SCC. Tricholemmoma differs from a trichilemmal carcinoma by the lack of atypia and a benign, that is, noninfiltrative, silhouette.
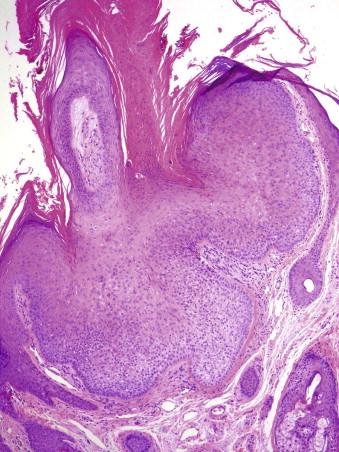
Inverted follicular keratosis typically occurs on the central face in middle-aged individuals. It is clinically indistinct and often biopsied to rule out BCC or a wart.
Typically, there is a wedge-shaped proliferation of keratinocytes with a lobulated circumscribed peripheral growth pattern. Coarse keratohyaline granules and whorls of squamous cells (squamous eddies) are usually present. Histologically, the differential diagnosis includes SCC and tricholemmoma. Inverted follicular keratosis differs from SCC by the lack of an infiltrative growth pattern. It differs from tricholemmoma by the lack of prominent peripheral palisading and basement membrane thickening.
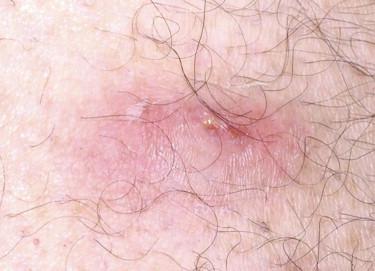
Pilomatricomas manifest usually as solitary, small (but rarely up to 5 cm in size), tan, firm nodules on the face and upper extremities, although they may occur in almost any hair-bearing area ( Fig. 11-15 ). The majority of lesions arise during the first two decades of life, but they can also develop in elderly individuals. Familial occurrence is rarely reported, particularly in association with myasthenia gravis or Gardner's syndrome.
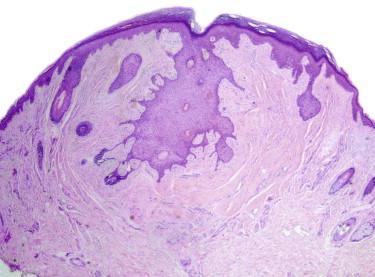
Pilomatricomas tend to present as well-circumscribed nodules in the dermis or superficial subcutis (or both) ( Fig. 11-16 ). A typical pilomatricoma is composed of basaloid (matrical) cells with mitotic figures and “ghost” cells (polygonal, deeply eosinophilic cell remnants, with a centrally located clear area corresponding to the absent nucleus). There is often an associated foreign body–type giant cell reaction. Calcifications are commonly found. Some pilomatricomas contain melanophages in the stroma and dendritic melanocytes within the clusters of basaloid cells (pigmented pilomatricoma or melanocytic matricoma). The early (proliferative) stage of pilomatricomas has a higher proportion of basaloid cells (often with numerous mitoses). In a later stage, the basaloid component may be minimal or absent. Tumors in the proliferative stage have also been termed proliferating tricholemmomas .

Pilomatricomas show nuclear and cytoplasmic labeling for β-catenin.
Pilomatricoma needs to be distinguished from pilomatrix carcinoma. The main feature that helps in differentiating between them is the silhouette of the tumor. Whereas nodular circumscribed lesions should be considered as benign, infiltrative lesions may recur or, rarely, metastasize. Pilomatrix carcinomas typically show a high proportion of basaloid cells or features indistinguishable from SCC. Cytologic atypia and necrosis are often present in larger tumors.
On occasion, pilomatricoma also needs to be distinguished from BCC, which may show focal matrical differentiation with ghost cells. Identification of classic features of BCC in parts of the tumor is helpful in this regard. Hybrid cysts of patients with Gardner's syndrome may show mixed features of pilomatricoma and other follicular cysts.
Both fibrofolliculomas and trichodiscomas may be single or multiple, a few millimeters in diameter, yellow-white, on the head and neck. Multiple lesions are associated with Birt-Hogg-Dubé syndrome (see the following).
Fibrofolliculomas have elongated, interlacing trabeculae of basaloid cells surrounded by fibromyxoid stroma. A dilated hair follicle may be seen in the center of the lesion. Trichodiscomas typically appear as perifollicular or parafollicular elliptical nonencapsulated angiofibromatous stromal proliferation ( Fig. 11-17 ).
In general, no further surgery is necessary after an unequivocal diagnosis of a benign follicular tumor except for cosmetic reasons. Other than surgical excision, locally destructive modalities (laser, cryotherapy, or conservative surgery) are available to remove the lesion(s).
In doubtful, incompletely excised lesions—for example, when a TBL is favored based on a small shave biopsy but a BCC cannot entirely be ruled out—it seems prudent to recommend conservative complete removal of the lesion.
Although BCC is discussed in many textbooks as a tumor of the epidermis, it is now widely accepted to represent a tumor with predominant follicular differentiation; therefore, it is included in this chapter. Trichoblastic carcinoma may be a more accurate term for BCC, but we continue to use the term BCC because clinicians and patients are familiar with it.
Basal cell carcinoma is a very common neoplasm. It mainly occurs on sun-exposed skin, in general in any hair-bearing area, but may also occur at sites with limited or no sun exposure. Patients with xeroderma pigmentosum, who have a diminished capability for repairing sun-induced mutations, develop a large number of both basal cell and SCCs early in life. Multiple BCCs or SCCs develop early in life in patients with the basal cell nevus syndrome or Bazex syndrome.
Mutations of the PTCH gene have been identified in the majority of syndromic and sporadic BCCs.
Approximately 900,000 new individuals with basal cell carcinoma (BCC) per year in the United States
Occurs in all races but most often in fair-skinned people
Usually in patients older than 40 years of age
More often in men than in women (male : female ratio, 1.6 : 1)
Exposure to ultraviolet light
Syndromes: basal cell nevus syndrome; Bazex syndrome; xeroderma pigmentosum
History of radiation treatment
Flesh-colored, erythematous, or pigmented papule or nodule; may be ulcerated
Indurated plaque (sclerosing BCC)
Usually only local growth; may be locally destructive with significant morbidity depending on location and size
Exceedingly rarely can metastasize (preferred sites: lymph nodes, lung, and bones)
Surgical: saucerization, standard full-thickness excision, Mohs micrographic surgery, curettage with or without electrodesiccation, cryosurgery
Medical: topical 5-fluorouracil, imiquimod
Radiation
The most common appearance is that of a papule or nodule, which may be eroded or ulcerated (ulcus rodens). Papules of BCC may clinically be confused with a nevus, fibroma, or folliculitis. BCC may also manifest as an erythematous patch ( Fig. 11-18 ), papule, nodule, or plaque ( Fig. 11-19 ), which is often eroded, ulcerated ( Fig. 11-20 ), or indurated ( Fig. 11-21 ). It may be difficult or impossible to distinguish BCC clinically from Bowen's disease or irritated or lichenoid keratoses. At times pigmentation may be present ( Fig. 11-22 ), and BCC may simulate a nevus or melanoma.
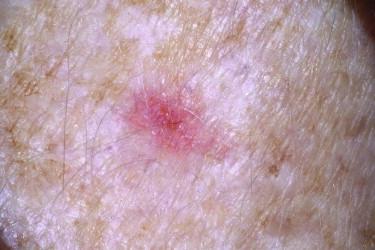
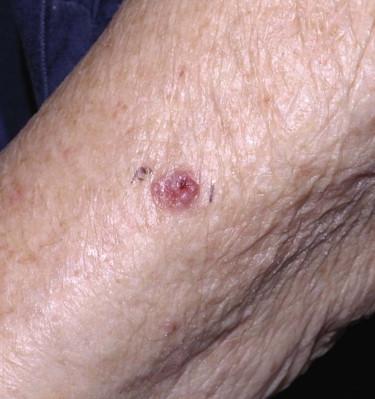
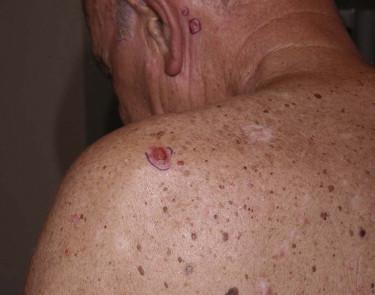
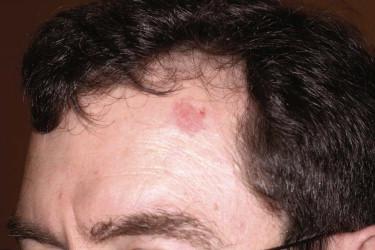
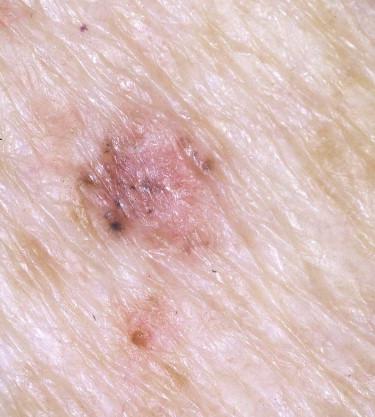
Basal cell carcinoma is histologically characterized by the presence of basaloid cells, peripheral palisading, mitotic figures, apoptotic bodies, myxoid stroma, and peritumoral clefting ( Fig. 11-23 ). Among these features, the presence of myxoid stroma and peripheral clefting has been suggested to be the most helpful ones to distinguish BCC from other basaloid tumors, such as TBL. A number of secondary features may occur in BCCs, such as dystrophic calcifications, amyloid deposition, or inflammatory reactions with or without features of partial regression.
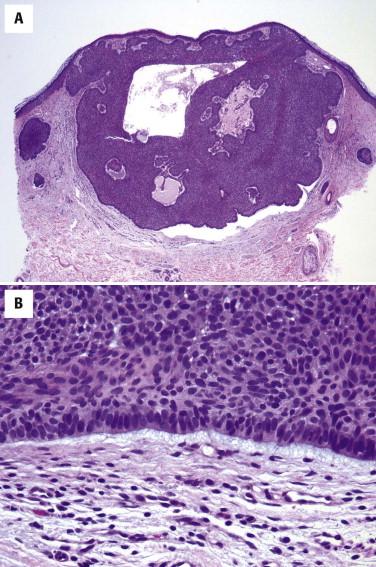
Several patterns of BCC have been described. One pattern may predominate or be present in pure form. Many times, however, a combination of patterns is seen. Furthermore, within these patterns, many additional variations may be noted (e.g., presence of melanin pigment, squamous metaplasia, stromal fibrosis). The main patterns include the following.
Basaloid cells with elongated hyperchromatic nuclei and scant cytoplasm
Peripheral palisading
Clefts between tumor and surrounding stroma
Mucinous alteration of the surrounding stroma
Positive for Ber-EP4, 34BE12, MNF116
Negative with Cam5.2, AE1 : AE3 *
* Basal cell carcinoma with squamous metaplasia may be positive for AE1 : AE3.
Melan-A often demonstrates melanocytes within the tumor
Trichoblastoma
Merkel cell carcinoma (small biopsy sample with crushed cells)
Squamous cell carcinoma with basaloid features (versus squamatized basal cell carcinoma [BCC])
Sebaceous carcinoma
Microcystic adnexal carcinoma (versus sclerosing BCC)
The superficial pattern is characterized by the presence of aggregates of basaloid tumor cells, which are still attached to the epidermis or adnexal structures ( Fig. 11-24 ). Such basaloid nests are typically multifocal and separated from each other by intervening areas of normal epidermis (also known as skip areas). We have recently shown that measurement of the depth of invasion (similar to Breslow thickness) helps predict response to topical therapy (i.e., imiquimod). In that large series, lesions thinner than 0.4 mm did not recur.
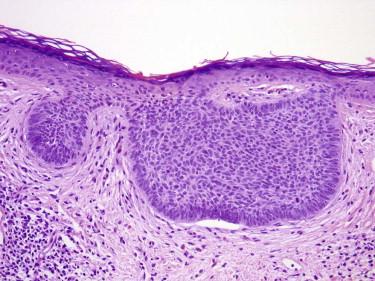
The nodular pattern is characterized by nodular aggregates of tumor cells in the dermis, at least focally detached from the epidermis and involving the reticular dermis ( Figs. 11-23 and 11-25 ). The nodules vary in size and shape. Some writers have proposed to distinguish BCC with large dermal nodules from those with multiple small nodules, which have been referred to as the micronodular pattern ( Fig. 11-26 ). The actual size of these nests required to distinguish micronodular from nodular BCC is subjective. Some authors recommend considering micronodular lesions as those that have nests smaller than 0.5 mm in diameter; such a definition would include a majority of BCCs. We render a diagnosis of micronodular BCCs when there are numerous similarly sized nests (occupying at least 25% of the base of the lesion) and these nests are smaller than a medium vessel (≈0.2 mm).
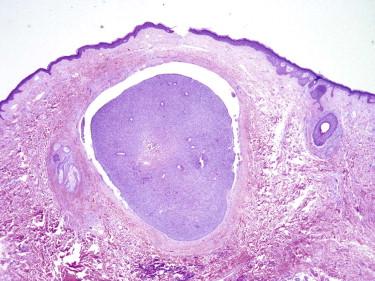
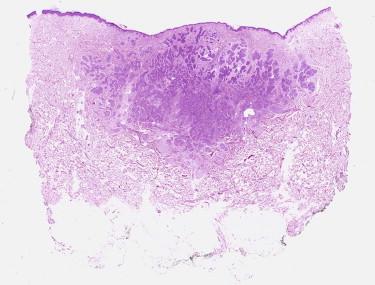
Although nodular BCCs are invasive carcinomas, the term infiltrative is generally applied to describe a tumor with less of a solid nodular growth pattern (or none of it) and a predominance of small often irregularly shaped cell aggregates, narrow strands of cells, multiple micronodules, or even single cells in the stroma. The haphazard distribution of such small tumor cell aggregates together with ill-defined interfaces with the surrounding stroma gives the tumor its infiltrative silhouette ( Fig. 11-27 ). Not all infiltrative BCCs are morpheaform, that is, sclerosing in a strict sense. We reserve the terms morpheaform, sclerodermoid, and sclerosing for BCCs that are associated with prominent stromal fibrosis ( Fig. 11-28 ). In this variant of BCC, the tumor cell aggregates are typically compressed into very narrow cords. Large solid nodules are usually absent or rare.
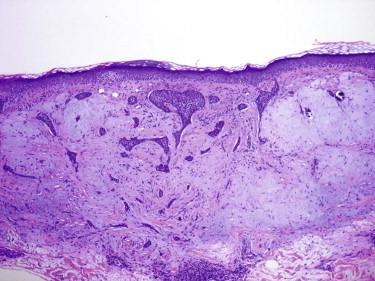
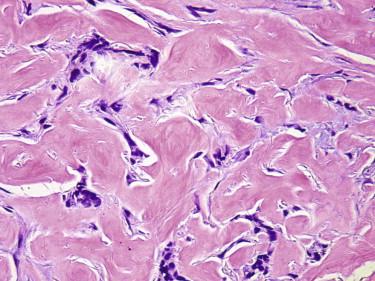
The fibroepitheliomatous pattern (fibroepithelioma of Pinkus) is characterized by cords and columns of basaloid cells with a fenestrated growth pattern associated with prominent fibroplasia ( Fig. 11-29 ).
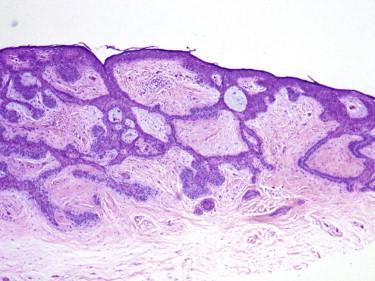
Infundibulocystic (aka hamartomatoid ) BCC is usually a small indolent lesion characterized by the presence of small keratocysts and bland basaloid cells, often arranged in small cords or a racemiform growth pattern ( Fig. 11-30 ). This lesion has features intermediate between BCC and TBL, including immunohistochemical findings such as CD34 expression by the peritumoral stromal cells. It may be associated with conventional BCC ( Fig. 11-30, B ). Multiple lesions of infundibulocystic BCC are often seen in patients with basal cell nevus syndrome.
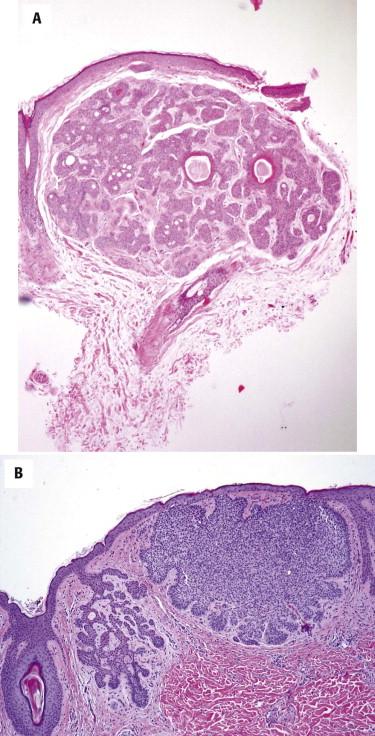
Miscellaneous additional phenotypic variations of BCC exist, such as the following.
Basal cell carcinomas may show a cribriform or pseudoglandular architecture (usually with an overall nodular growth pattern) associated with mucinous stroma simulating at first glance an adenoid cystic carcinoma ( Fig. 11-31 ).
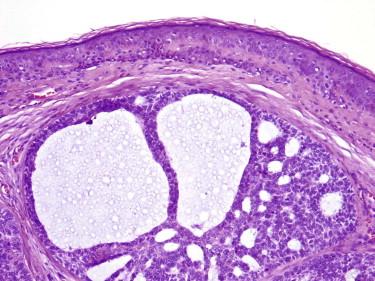
Any pattern of BCC can be pigmented (most often superficial BCCs and nodular BCCs). The histologic correlate of a clinically dark brown nodule is usually the presence of melanophages in the stroma ( Fig. 11-32 ). However, melanin pigment may also be present in the carcinoma cells. On occasion, one also finds dendritic melanocytes within the tumor compartment (melanocytes and Langerhans cells). Collisions of BCC and melanocytic proliferations (most often nevi, at times melanoma—usually lentigo maligna melanoma in situ) may also give rise to the clinical impression of pigmented BCC.
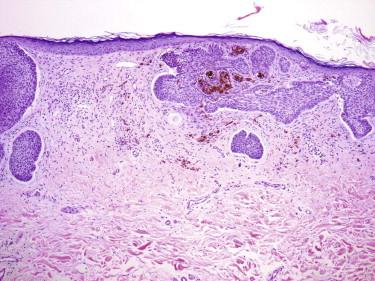
At times, a BCC may show squamous differentiation, especially superficially underneath an ulcer. Squamous differentiation may also occur in deeper infiltrative parts of the tumor, especially in recurrent tumors. BCCs with squamous differentiation have also been referred to as metatypical BCCs ( Fig. 11-33 ). In a partial tissue sample, a squamatized BCC may be indistinguishable from SCC.
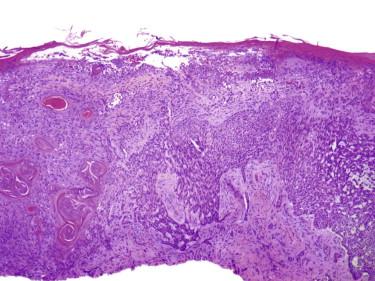
This BCC is characterized by the presence of tumor cells with glycogen-rich clear cytoplasm ( Fig. 11-34, A ).
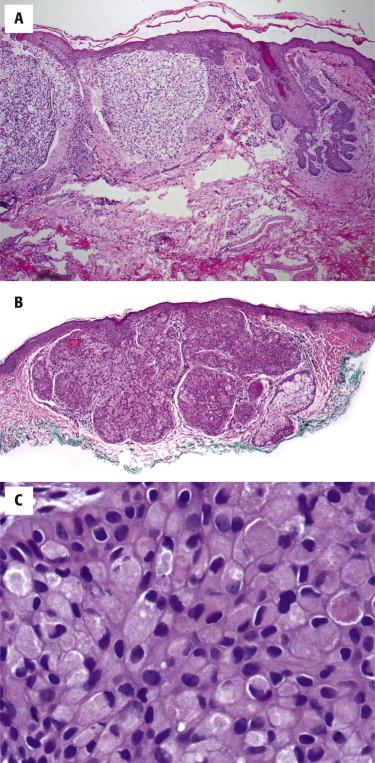
This BCC is characterized by a pink appearance because the tumor cells contain abundant granular eosinophilic cytoplasm ( Fig. 11-34, B and C ). Because of its pink appearance, it may be confused with SCC. Ancillary studies, such as Berep4 and CD68 or NKI/C3, help in establishing the correct diagnosis.
These are tumors with focal duct formation in the context of a classic BCC. Eccrine differentiation is a rare phenomenon. More often, one can observe preexisting sweat ducts, which may be ectatic, engulfed by BCC ( Fig. 11-35 ). These lesions may have an aggressive behavior with distant metastases.
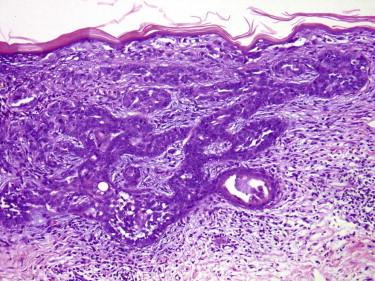
Sebocytes may get entrapped in a BCC (BCC may grow into and populate a sebaceous lobule). This should not lead to a diagnosis of sebaceous carcinoma.
Occasionally, a tumor with features otherwise typical of BCC shows focal shadow cells similar to what is seen in bona fide pilomatrix tumors ( Fig. 11-36 ).
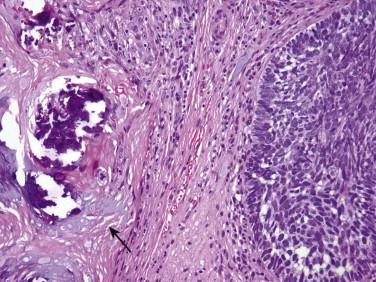
Occasionally, a BCC shows marked nuclear pleomorphism ( Fig. 11-37 ). To date, there is no evidence that this is a clinically significant finding.
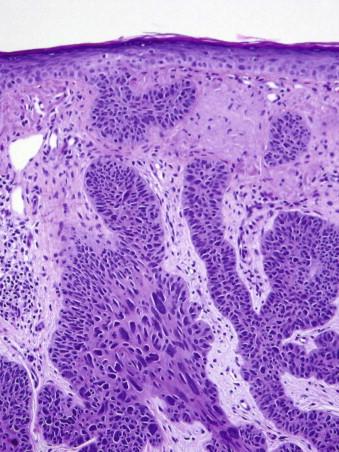
Basal cell carcinomas may collide with a number of epithelial and nonepithelial tumors. Common associations include BCC and SK ( Fig. 11-38 ) or BCC and melanocytic nevus ( Fig. 11-39 ).
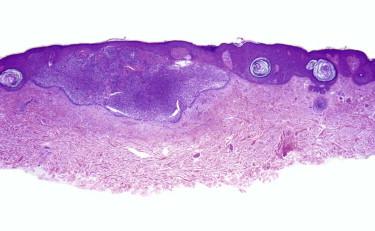
Rarely, a tumor may show features of both BCC and sarcoma ( Fig. 11-40 ). Such tumors tend to be large and more aggressive. These tumors have been reported as carcinosarcoma, sarcomatoid BCC, or high-grade trichoblastic carcinosarcoma.
![FIGURE 11-40, Basal cell carcinosarcoma (sarcomatoid basal cell carcinoma [BCC]) is a biphenotypic tumor with an epithelial component in the appearance of a BCC and an associated malignant spindle cell component with mesenchymal features. FIGURE 11-40, Basal cell carcinosarcoma (sarcomatoid basal cell carcinoma [BCC]) is a biphenotypic tumor with an epithelial component in the appearance of a BCC and an associated malignant spindle cell component with mesenchymal features.](https://storage.googleapis.com/dl.dentistrykey.com/clinical/AdnexalTumors/39_3s20B9780323261913000113.jpg)
Immunohistochemistry may on occasion be helpful for the distinction of BCC from histologic mimics thereof. BCCs are typically immunoreactive for the following epithelial markers: 34BE12, MNF116, and Ber-EP4. They are usually negative with Cam5.2, AE1:AE3, epithelial membrane antigen (EMA), and CK7. Focal labeling with AE1:AE3 may be seen in the squamatized portion of a BCC. CD10 is expressed by some of the epithelial tumor cells. Immunoreactivity with Cam5.2 or anti-CK7 may be found when eccrine ducts are entrapped by BCC or in BCCs with focal eccrine differentiation. BCCs are negative for CK20, but a subset of BCCs is (usually only weakly) positive for chromogranin or synaptophysin.
The histologic diagnosis of BCC is usually straightforward. However, problems can arise from histologic overlap with other basaloid epithelial neoplasms and from insufficient histologic sampling.
In the differential diagnosis of BCC from TBL or desmoplastic TE, features that favor BCC include the lack of papillary mesenchymal bodies, presence of clefts between tumor and immediately surrounding stroma, cytologic atypia, readily identifiable mitotic figures, and mucinous stroma (see also TBL and desmoplastic TE).
Basal cell carcinomas with squamous metaplasia may be difficult to distinguish from SCC, especially in a superficial shave biopsy, in which the squamous component predominates. At times, the BCC may only be fully recognized when more of the tumor is sampled and typical areas of BCC are identified. Likewise, biopsy of the deep portion of a recurrent BCC may reveal features of SCC. In these cases, the diagnosis of BCC rests on identification of typical areas of BCC in the same or prior biopsy. If on an initial superficial biopsy a reliable distinction between a squamatized BCC and SCC cannot be made, it is best to acknowledge it and defer to review of the excised tumor for final classification. Immunohistochemical studies may be helpful if immunoreactivity is found with BER-EP4. Positive staining with AE1:AE3 does not prove the presence of SCC because the squamatized portion of a BCC is often immunoreactive for this marker.
On occasion, collision tumors (BCC and SCC at the same site) or biphasic tumors (“true” basosquamous carcinomas) may occur that show both typical features of BCC and SCC within the same tumor nodule. Biphenotypical carcinomas usually have a predominant basal cell appearance on the initial biopsy, but with subsequent recurrences, the squamous component tends to dominate and dictate the clinical behavior.
Basal cell carcinomas with solid infiltrative or adenoidal patterns may be confused with adenoid cystic or basaloid sweat gland carcinoma. Identification of true tubule formation and attention to the overall growth pattern or immunohistochemistry are helpful. BCCs are positive for BER-EP4 or 34BE12 but negative for CK7, Cam5.2 antigens, and EMA. Sweat gland tumors, including adenoid cystic carcinomas, tend to be positive for the latter three markers.
Morpheaform BCC may be confused with a sclerosing basaloid proliferation of MAC. Sclerosing BCC tends to lack the keratocysts or small luminal structures within basaloid cells often seen in MAC and usually shows a higher degree of cytologic atypia.
The most problematic diagnostic pitfall is the failure to recognize Merkel cell carcinoma (MCC), which may clinically and histologically simulate the appearance of a BCC. Attention to cytologic detail should allow for a distinction: the tumor cells of MCC usually lack the palisading arrangement of BCC. In contrast they show a salt and pepper chromatin pattern and many more mitotic figures and apoptotic bodies than BCCs. In difficult cases, immunohistochemistry for CK20 (positive in MCC; negative in BCC) allows for a definitive diagnosis. Staining for chromogranin or synaptophysin is not as helpful because a subset of BCCs shows focal expression for these neuroendocrine markers.
Frequently, initial levels of a clinically suspected BCC fail to reveal tumor cells. However, certain clues should prompt the histopathologist to order deeper levels. Frequently, they do indeed reveal foci of tumor. In general, deeper levels are a prudent course of action if no histologic finding is identified, which could explain the clinical lesion (usually that of a papule), such as a dermal melanocytic nevus, folliculitis, sebaceous hyperplasia, or fibrous papule. Clues to the possible presence of adjacent BCC include inflammation and fibrosis as well as “pseudocysts” (empty spaces, from which tumor cells seemingly “may have fallen out,” a phenomenon also referred to as “stroma interrupted”).
Basal cell carcinoma is usually a slow-growing tumor. Some variants of BCC, such as infundibulocystic BCC, tend to be indolent, with limited potential for tissue destruction over many years. Other so-called aggressive variants, which include infiltrating, morpheaform, and micronodular BCCs, may cause severe tissue destruction.
Based on a number of clinical and pathologic features, patients with BCCs are often classified into low- and high-risk categories.
The vast majority of superficial and nodular BCCs are cured by excision. Narrow excisions suffice. There is no need for wide margins. For early superficial lesions, local destructive modalities, such as cryotherapy, may be adequate. For facial lesions, surgery with frozen section examination is the treatment of choice. Postoperative or intraoperative complete circumferential peripheral and deep margin assessment with frozen or permanent sections is critical for large and infiltrative lesions. Radiation therapy is also an option for patients who are reluctant or unable to undergo surgery or in the case of positive margins or extensive perineural invasion.
In very rare cases, usually after repeated local recurrences, large and deeply infiltrative BCCs may metastasize to lymph nodes or distant sites, such as the lung, and become lethal.
For biopsies, the essential task for the pathologist is to diagnose the presence or absence of BCC. Some clinicians may request subtyping of BCC. Knowledge of the subtype may be helpful, for example, for a Mohs surgeon to have a visual image of what to expect on frozen sections, especially if the slides of the prior biopsy cannot readily be made available. On shave or saucerization excisions, it is also helpful to comment on the margin status. Otherwise, patients with a small BCC may undergo unnecessary subsequent excisions for tumors that were already completely removed by the initial biopsy.
For an excision of a large BCC, in addition to the diagnosis and margin status, it also seems prudent to report the size of the lesion; to indicate which anatomic levels are involved (dermis only, subcutis, muscle, or bone); and to document the histologic patterns, the presence of perineural invasion, including caliber and location of the largest involved nerve, or the presence of squamous differentiation.
Pilomatrix carcinoma is a rare tumor of middle-aged and elderly individuals. The head and neck area or extremities are most often affected. The tumors may present as papules or nodules. They may be large (larger than 5 cm) and are commonly ulcerated.
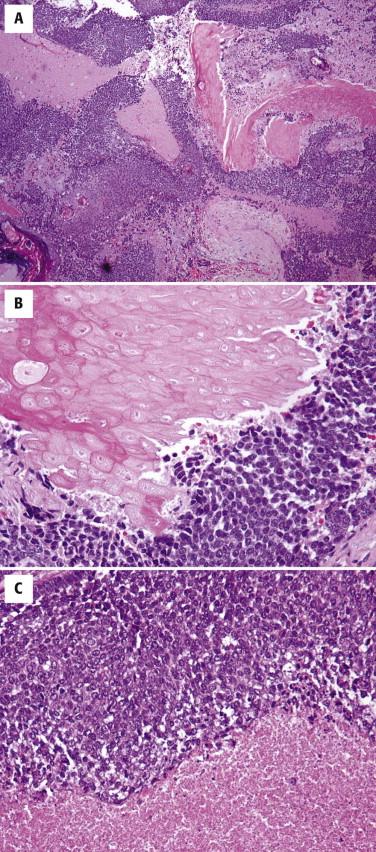
The silhouette of the tumors is asymmetric and infiltrative with an overall lobular growth pattern. The tumors are composed of two major cell types: (1) basaloid cells with matrical features are arranged in lobules and sheets or cords and bands and (2) “ghost” or “shadow” cells as seen in pilomatricoma. Foci of necrosis are usually present ( Fig. 11-41 ). Mitotic figures among basaloid cells are easily identified and may be numerous (areas with up to 10 mitoses per high power field are not uncommon). The degree of nuclear atypia is variable. Some pilomatrix carcinomas show areas histologically indistinguishable from SCC.
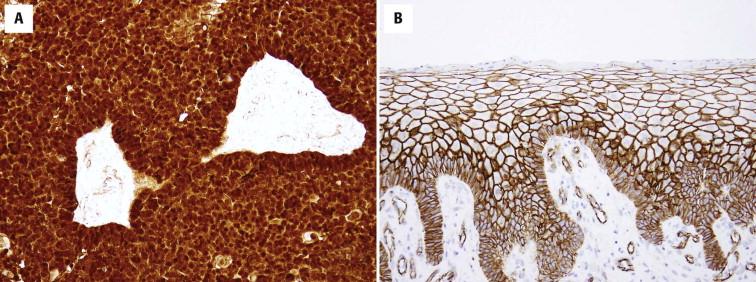
Pilomatrix tumors contain β-catenin mutations and show a distinct cytoplasmic and nuclear labeling of tumor cells ( Fig. 11-42 ) in contrast to the membranous labeling of normal epidermis or SCCs.
Pilomatrix carcinoma needs to be distinguished from pilomatricoma, BCC, and SCC. Pilomatrix carcinoma differs from pilomatricoma primarily by its infiltrative growth pattern and the presence of cytologic atypia. Furthermore, carcinomas tend to be larger and are more likely to have necrosis and features of squamous metaplasia simulating the appearance of SCC.
Basal cell carcinoma, which may show focal matrical differentiation, differs from pilomatrix carcinoma (and pilomatricoma) by the presence of typical features of BCC, such as palisading nuclear arrangement, stromal mucin, clefts, or connections to the epidermis or non-neoplastic follicular structures.
Squamous cell carcinomas are best distinguished from pilomatrix carcinomas by the presence of a precursor lesion (SCC in situ), connection of invasive keratinizing tumor to the skin surface, and lack of ghost cells.
| Pilomatricoma | Proliferating Pilomatricoma | Pilomatrix Carcinoma | |
|---|---|---|---|
| Clinical findings | |||
| Gender | F > M | F > M | M > F |
| Location | Head and neck, extremities | Head and neck | Head and neck, trunk, extremities |
| Size | Small (0.5-3 cm) | Medium (1-5 cm) | From 0.5 to >20 cm |
| Histologic features | |||
| Ulceration | Rare | Rare | Common |
| Growth pattern | Nodular | Nodular | Infiltrative |
| Shadow cells | Present | Present | Present |
| Basaloid cells | Variable | Prominent | Prominent |
| Atypia | None or mild | None or mild | Present |
| Squamous metaplasia | Rare | Rare | Common |
| Necrosis | Rare | Rare | Common |
| Mitoses | Present in basaloid cells | Present in basaloid cells | Present in basaloid and squamatized cells |
| Prognosis | Rare local persistence | Rare local recurrence | Frequent local recurrence Rare distant metastasis |
Pilomatrix carcinomas are slowly growing, locally aggressive tumors. They are usually cured by excision, but rare tumors metastasize to lymph nodes and visceral sites. Too few cases have been studied to establish prognostic parameters.
Trichilemmal carcinoma is a rare tumor. Not enough bona fide cases have been reported to allow a clear description of associated clinical and histologic features. Reported cases suggest a spectrum of papules and plaques clinically overlapping with basal cell and SCC. Trichilemmal carcinomas are described in the literature as infiltrative dermal tumors with outer root sheath differentiation, typically showing a clear, glycogen-rich cytoplasm. However, the presence of a clear cytoplasm is not distinctive because it can be seen in several other neoplasms. For a superficial dermal clear cell carcinoma to be convincingly acceptable as a trichilemmal carcinoma, the tumor either must be associated with a benign precursor (i.e., a trichilemmoma) or show continuity with the outer sheath epithelium of a coexisting hair follicle with minimal or no intraepidermal extension. Otherwise it is difficult and quite arbitrary to distinguish a trichilemmal carcinoma from a clear cell variant of squamous cell or BCC. Additional features suggestive of trichilemmal carcinoma include peripheral cell palisading and trichilemmal keratinization (i.e., absent or minimal granular cell layer, presence of dense keratin material). It is currently not known whether the distinction of trichilemmal carcinoma from a clear cell variant of SCC (which may colonize a hair follicle) is relevant for patient care, that is, whether trichilemmal carcinomas behave any differently from SCCs of similar size and depth of invasion.
Proliferations of the apocrine or eccrine gland apparatus are characterized by the presence of ducts, that is, lumina lined by epithelial cells. Although the presence of the classic apocrine feature, picturesquely described as epithelial snouts or decapitation secretion or other manifestation of apocrine differentiation, such as the presence of plasmacytoid epithelial cells or mucin, allows the pathologist to designate a glandular or ductal proliferation as apocrine, the lack of such features does not exclude an apocrine derivation. Because it is often difficult or impossible to distinguish histologically apocrine from eccrine neoplasms and such a distinction has no or little clinical relevance in the management of tumors, we place no emphasis on it in our presentation of glandular and ductal proliferations.
This rare lesion is characterized by a circumscribed area of hyperhidrosis. Histologically, these lesions have localized increased number and size of eccrine glands. The variant called eccrine angiomatous hamartoma has, in addition to the eccrine glands, adipose tissue and small capillaries.
An apocrine nevus is characterized by an increased number of apocrine glands. It may occur as a rare solitary lesion but is most often found as part of a nevus sebaceus.
These are clinically indistinct tumors, typically presenting as flesh-colored papules or nodules. Some may arise in association with nevus sebaceus.
These are benign tumors with glandular differentiation (presence of ducts, lumina) ( Figs. 11-43 to 11-45 ). They tend to be centered in the dermis and are well circumscribed. They may show tubular, cribriform, papillary, micropapillary, or mixed growth patterns. The ductal lining is bilayered, with luminal cuboidal secretory cells and peripheral cuboidal or flat myoepithelial cells. The epithelial cells may be small or large cuboidal or flat. The cytoplasm may range from dense pink to a clear cell appearance. Focal squamous or mucinous metaplasia may occur. The stroma of adenomas has various degrees of fibrosis and may be hyalinized.
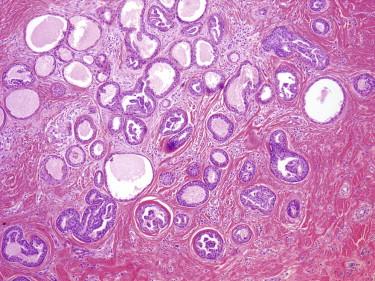
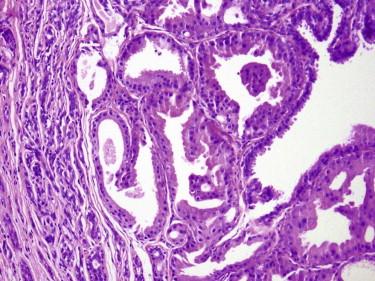
Adenomas can be classified further based on the predominant growth pattern (e.g., tubular, cribriform, or papillary adenomas, the latter being identical to hidradenoma papilliferum). At the current time, we see little value in detailed subclassification of benign sweat gland adenomas. The important issue is simply to recognize them as being benign. On the other hand, some subtypes of sweat gland adenomas have become such established and familiar entities (e.g., hidradenoma papilliferum, nodulocystic hidradenoma) that they are discussed separately here.
The important issue is to distinguish adenomas (circumscribed lesions) from well-differentiated adenocarcinomas. In the majority of cases, this distinction is straightforward because many carcinomas are large and infiltrative and show significant cytologic atypia. However, in some instances, such as digital lesions, ductal adenocarcinomas may be deceptively “bland” with regard to their cytologic appearance (see aggressive papillary digital adenoma—adenocarcinoma).
This lesion is typically found in the vulva and perianal region as a solitary papule or small nodule but may occur anywhere, especially along the “milk line.”
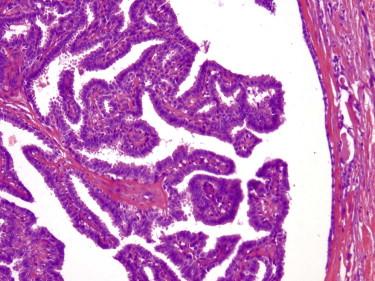
Hidradenoma papilliferum is a well-circumscribed dermal nodule composed of both solid and cystic regions. The hallmark of this lesion is the papillary architecture, with a layer of luminal secretory cells surrounded by myoepithelial cells ( Fig. 11-45 ). Occasionally, there are large cuboidal cells identical to those seen in apocrine metaplasia of mammary glands. Because identical lesions can be seen in other anatomic locations, an alternative name for all of these lesions may be papillary apocrine adenoma.
In contrast with syringocystadenoma papilliferum, hidradenoma papilliferum does not show prominent plasma cell infiltrates.
Hidradenomas are solitary skin-colored or reddish papules or nodules ( Fig. 11-46 ). They may be large, measuring up to 2 cm or more in diameter. They can occur at any anatomic location and any age.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here