Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
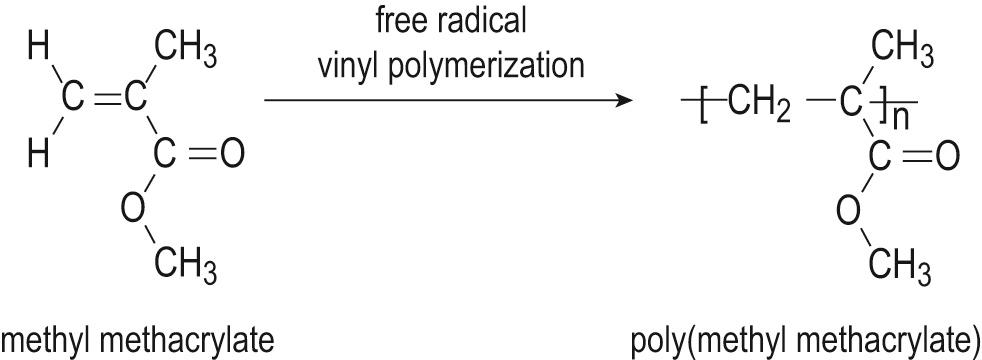
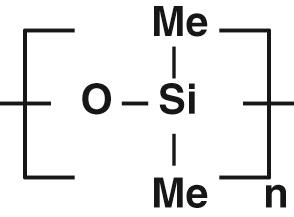
Perhaps the best way of visualising the way in which polymer chains arrange themselves is by taking several pieces of string to represent individual molecules. The usual arrangement is random, with the pieces of string loosely entangled rather than being extended. The interaction and entanglement of the individual molecules in this way gives polymers their characteristic physical properties. By changing the chemical nature of the polymer chain and their arrangement together we can change the physical properties and thus obtain either hard glassy behaviour or, at the other extreme, flexible, elastomeric behaviour. The best example of a hard glassy material is poly(methyl methacrylate) which is formed from methyl methacrylate monomer units ( Fig. 2A.1 ). This figure represents the assembly of ‘n’ methyl methacrylate units to form a PMMA chain ‘n’ units long. The best example of a biomaterial based on the flexible silicon-oxygen backbone is silicone rubber ( Fig. 2A.2 ) which is both elastomeric and highly permeable to oxygen.
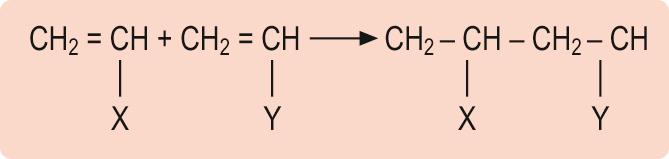
One important way in which a hard glassy polymer can be converted into a flexible material is by the incorporation of a ‘plasticiser’. This is a mobile component, often an organic liquid with a high boiling point that will act as an ‘internal lubricant’. Its presence separates the polymer chains, and allows them to move more freely. A good example is poly(vinyl chloride) or PVC ( Figure 2A.3 X = Y = Cl) which in its unmodified state is a rigid glassy material and will be familiar as the clear corrugated roofing material used on car ports and similar domestic extensions. When a plasticiser is incorporated the material is converted into the flexible material used, for example, as ‘vinyl’ seat coverings in cars and general domestic applications. In these cases, pigments and various processing aids will also have been added in order to enable the polymer to be produced in a variety of colours and textures.
Modest though that might be, in response to the physical trauma of the eyelid-lens interaction and the sliding motion of the upper eyelid over the anterior surface, the result is a possible up-regulation of particular tear or serum derived components and the activation of further host reactive pathways. A reduction in tear flow at the posterior surface and less dynamic interaction with the tissue bed would be expected to produce a quite different level of response with the potential sorbtion of specific biochemical components from this pseudo-closed eye environment. This difference is exemplified by fact that noticeably higher levels of vitronectin deposition (greater than tenfold) have been detected on the posterior, relative to anterior lens surfaces ( ).
Although the lacrimal gland is likely to be the major source of the four principal cations, serum leakage may well influence the calcium and magnesium levels in the posterior tear film. Factors related to the lens and lens material, such as ion permeability, partition coefficient effects and concentration polarisation may also contribute to ion-specific disparities in electrolyte concentrations, particularly at the corneal surface. The existence of a correlation of ion transport with on-eye contact lens movement, first proposed in the patent literature ( ) has recently been re-examined ( ). Interest in the measurement and modelling of sodium ion transport and partition coefficient behaviour of commercial lenses has also come to the fore ( and ). This should lead to more comprehensive studies of electrolyte effects in silicone hydrogel contact lenses, building on earlier studies of these phenomena in conventional hydrogels ( and ).
Because an understanding of the transport processes involved, and thus an ability to influence permeability and permselectivity is important in applications such as reverse osmosis, kidney dialysis, sensors and drug delivery, there have been many attempts to rationalise available data in the form of a universally applicable transport model. The free-volume model takes a partly thermodynamic, partly statistical approach in which the transported species is associated only with the water phase, with its diffusion being dependent upon the probability of it being located next to a suitable hole that is both unobstructed and large enough to accept the permeant. In this free-volume model the flux from high to low concentrations reflects the fact that fewer holes are occupied in the less concentrated regions and the penetrant has a higher probability of jumping to an unoccupied hole in the low concentration regions. The model predicts a linear relationship between ln P and 1/H, where P is the permeability coefficient in the hydrogel and H is the degree of hydration. More sophisticated models relate diffusion coefficient to an array of factors, including the degree of swelling, the radius of the solute, the number averaged molecular weight between crosslinks and function related to the mesh size, taking into account the effects of barriers such as those due to crosslinks and entanglements ( ; ; ; ). Here the chemical composition of the polymer, its water content, and the nature of the solute to be transported interact together to enable transport behaviour to be manipulated in such a way that a degree of specificity and control is achieved.
Many of the changing facets of the ageing ocular surface including a reduced blink rate ( ), the deterioration of eyelid function ( ), shorter tear break-up times ( ), meibomian gland dropout ( ), lacrimal gland deterioration ( ) and importantly a presumed gland-related change in tear film composition and thus function.
In terms of tear film component changes, the fact that there is already a lack of standardisation of tear collection, sampling, and the analytical protocols and assays makes tear compositional investigation difficult. Added to this the patient populations used, particularly in relation to ageing studies, are not, and cannot be, completely standardised which provides further opportunity for inconsistencies. Examples of compositional changes include a reduction in total protein levels ( ), lipoidal chain modification ( ) and an increase in osmolarity ( ; )-(although some studies found no change in osmolarity with age ( ; )), all with increasing age. However, the effects of age on the other important tear components such as mucins and electrolytes have yet to be adequately investigated.
Dry eye syndrome is an umbrella term for a large number of ocular surface complaints and is a prime example of a compromised tear film and lacrimal functional unit disorder. Subjects with dry eye usually report symptoms including some or all of the following, dryness, discomfort, irritation, itchiness, burning sensation, blurred vision and photophobia. The major etiological causes can mainly be broken down into those arising from either aqueous deficient or excessive evaporation, or it can be a combination of both ( ). Those suffering from dry eye are ultimately at risk of tissue damage and corneal infection (although these may be the original causative factors which escalate overtime). However, it must be noted that sensation and experience of dryness is not itself always an indication of dry eye syndrome. A subject can have a dry eye sensation and not suffer from dry eye syndrome and conversely they can have dry eye syndrome and not experience dryness.
The occurrence and prevalence of dry eye is a large problem in ophthalmology. It has been reported that close to 25% of patients that have visited an eye care practice have reported dry eye symptoms ( ), and it has been the topic of many literature reviews and workshops (e.g. ; ; ; ). Quantifying the overall prevalence of dry eye in the population as a whole has proved difficult to due to the lack of uniformity of its definition and the overall variation in the studies used to assess its prevalence which can vary in race, gender, age and population size ( ).
Measurements from and have not been presented with a definition of the measurement and methodology used and, as a result, may only be viewed as a guideline assessment. It follows that the true cornea with limbus diameter is not known precisely but should approximately be somewhere between 13.7- 14.9 × 12.6 – 13.6 mm if limbus is 1 mm wide. A 2 mm limbal width would push these number to 15.7 – 16.9 × 14.6 – 15.6 mm, which seems somewhat excessive. Indeed, continued biometry research would be welcome!
The rapid refractive changes achieved in orthokeratology (see Chapter 19 ) are believed to occur primarily due to central thinning of the epithelium, together with an approximately equal thickening of peripheral epithelial regions ( , ).
Corneal epithelium thinning can be achieved by:
Pushing cells aside.
Shedding cells.
Flattening cells.
The tension exerted by closed eyelids on a flat fitting, rigid contact lens worn overnight has the ability to alter epithelial thickness. This can be observed on a fresh cadaver eye, where the mouldable, jelly-like properties of the epithelium can be demonstrated together with the plasticity of the structure.
claimed that the sensibilities of touch, cold, warmth and pain may be experienced if the cornea is suitably stimulated.
Temperature-responsive corneal neurones have been identified in animals. Electrical recordings from rabbit anterior segments studied in vitro revealed cold-, mechano-, mechano/heat- and chemosensitive units ( ) with different conduction rates (delta and C fibres) and related to different fibre orientations in the epithelium. For example, fibres running perpendicular to the corneal surface gave C fibre cold or chemosensitive responses. found cold receptors, preferentially located at the periphery of the cornea in cats, which were distinguished from polymodal (mechanical, heat, chemical) receptors.
Sufficient reduction in temperature ( , , ), deprivation of oxygen or the application of metabolic inhibitors such as ouabain ( ), produces reversible swelling and opacification of the cornea, which demonstrates the importance of metabolism in maintaining corneal clarity.
Recovery of normal corneal thickness at body temperature after cooling of excised cornea, whether or not the epithelium is present, demonstrates that deturgescence is substantially the responsibility of the endothelium ( ). A punctate lesion of the corneal endothelium causes a well-circumscribed region of stromal opacification opposite it, suggesting that liquid intake is confined to the traumatised zone by the vigorous activity of the adjacent endothelium ( ).
Ouabain, a specific inhibitor of the sodium–potassium–adenosine triphosphate enzyme enzyme, causes corneal swelling ( ) and prevents reversal of oedema induced by cooling ( ).
While corneal deturgescence can be remarkably sensitive to bicarbonate concentrations ( ), the relative effect on the actual transendothelial fluid pump appears to be far less ( ). found that the hydroscopic properties of the corneal stroma can also be bicarbonate and pH sensitive, and that chloride ions might also play an important role ( ). From such a perspective, the leaky membrane of the corneal endothelium could play a role in regulating the anion composition of the corneal stroma in addition to transporting water out ( ).
observed passive diffusion of labelled amino acid through the endothelium. The rapid turnover of epithelial cells demands a considerable utilisation of amino acids in the synthesis of protein and, as expected, the concentration was high. It was actively accumulated by the epithelium only after it had appeared in the stroma, suggesting that none was taken up from the tears despite their rich amino acid content ( ). It appears, therefore, that epithelium has a very low permeability to amino acids and glucose.
exposed the eyes of rabbits to a pure oxygen atmosphere for 3–3.5 hours and found that the lactic acid concentration in the cornea was reduced by a third, indicating an increase in aerobic glycolysis. Using a nitrogen atmosphere for the same period of time, lactic acid concentration was increased by a third, indicating a decrease in aerobic glycolysis.
concluded that the rabbit cornea also utilises aqueous oxygen. The lactic acid concentration decreased from a value of unity to 0.7, 3–3.5 hours after introducing an oxygen bubble into the anterior chamber. Barr and his co-workers found an oxygen gradient reducing from the front surface of the cornea to the anterior chamber in rabbits and concluded that atmospheric oxygen normally diffuses through the full thickness of the cornea to the anterior chamber ( , ).
The basic secretion of tears is largely derived from the lacrimal gland, with additional contributions from the accessory lacrimal glands: the mucous conjunctival glands and the sebaceous tarsal glands. A conjunctival gland or goblet cell is shown in Chapter 3 , Fig. 3.21 in the process of releasing mucin granules, containing MUC 5AC, into the tear film. The lacrimal gland is classified as a serous gland, but its cytology and histochemistry suggests some variety of output ( , ). In the palpebral accessory lacrimal glands, the epithelial cells lining the ducts leading to the openings along the palpebral conjunctiva contain secretory granules. These granules have a different electron density from those found within the acinar secretory cells ( ). It is possible that the ductal epithelial cells produce the mucoid secretion from this and the other lacrimal glands.
The eye remains moist if the lacrimal gland is congenitally absent, when the gland is removed or if its motor nerve supply is interrupted, but this radically reduces basic tear secretion ( ).
Superimposed on the basic secretion are the phasic increments in secretion induced reflexly by mechanical or chemical irritation and by psychogenic factors. To distinguish between the two, the words ‘lacrimation’ (to describe reflex tears) and ‘weeping’ (for psychogenic tears) have been suggested.
Healthy tears are very important in keeping the anterior surface in good health and asymptomatic. Insufficiencies in producing adequate amounts of well structured tears is the cause of dry eye disease. The Dry Eye WorkShop (DEWS) report states as a definition “Dry eye is a multifactorial disease of the tears and ocular surface that results in symptoms of discomfort, visual disturbance, and tear film instability with potential damage to the ocular surface. It is accompanied by increased osmolarity of the tear film and inflammation of the ocular surface”. According to about 5 million Americans over the age of 50 have significant dry eye complaints but tens of millions more people are affected to a lesser degree. This is especially true for contact lens wearers where the tears need to maintain lens hydration and therefore the assesment of tears has become an important part of clinical practice especially for contact lens wearers.
considered the precorneal film to consist of a triple-layered structure with a central watery phase made up of the serous secretion of the lacrimal gland and constituting the bulk of the film. A thin superficial oily layer was thought to issue from the tarsal glands and a deep layer of mucoproteins from the conjunctival glands.
The classic tri-lamina structure described has essentially survived the test of time. Although, it is now realised that the composition of the tears is a little more complex than this, as is illustrated in Chapter 3 , Fig. 3.22 . Mucins with different functions have been identified. The glycocalyx with its membrane-spanning, disadhesive mucins, MUC1, MUC4, and MUC16 is the wetting agent that lines the fatty, membranous ocular surface ( ). Primarily MUC 5AC but also MUC 2 are gel-forming mucins dispersed in the aqueous phase of the tears ( ). These are located deeper in the tears and are believed to help stabilise tears. The presence of a thin superficial lipid layer is suggested by the coloured interference fringes, which may be observed with a biomicroscope or Tearscope (see Chapter 5 ). demonstrated that, in rabbits, tear film evaporation increases at least 10-fold in the absence of tarsal gland secretion which is approximately that expected of water in the absence of a surface lipid film. This amount of increased evaporation is approximately that expected of water in the absence of a surface lipid film. showed that in human tear thinning rate increases 5 times and in some 30 times when the lipid layer is washed away.
The lipid layer extends from its sources at the openings of the tarsal glands at the lid margins to cover the tear film, and provides a stable interface between the aqueous layer and air. It is approximately 0.1 µm thick ( ), varying substantially with the size of the palpebral aperture and time of exposure. It compresses and thickens as the eyelids close, thinning gradually following a blink. In contrast to the rest of the tear film, the lipid layer does not take part in the flow of tears from lateral to medial canthi and is essentially an independent part of the tear film. Lipid does not normally enter the conjunctival sac.
Specular reflection of white light, from the lipid layer of the tears generates first order coloured interference fringes when the lipid thickness exceeds one quarter of the wavelength of light; i.e., approximately 100 nm, for a minimum visible wavelength for blue light of approximately 400 nm.” This is because of the white light which is made up of different wavelengths being differentially refracted and split into the colours of the spectrum – the shortest visible wavelength for will be around 400 nm (blue). The laws of physical optics dictate that interference fringes will become visible when the thin film is more than  of a wavelength thick. The thinnest that this can happen is at
of a wavelength thick. The thinnest that this can happen is at  of the shortest wavelength ie.
of the shortest wavelength ie.  of 400 which is 100 nm. This means blue is the first colour you tend to see by interferometry if fringes are visible. To create yellow fringes, you would need the lipid layer thickness to be nearer 140 nm, corresponding to
of 400 which is 100 nm. This means blue is the first colour you tend to see by interferometry if fringes are visible. To create yellow fringes, you would need the lipid layer thickness to be nearer 140 nm, corresponding to  of the wavelength of yellow light.
of the wavelength of yellow light.
The overall thickness of the dynamic tear film have proved difficult to determine. Measurements range from 3-60 µm ( , , and ) and increases in thickness at the lid margins where a meniscus of tear fluid, the marginal tear strip, is formed. However, thinning may occur along the line joining the film with the meniscus. Tear fluid flows medially within the marginal tear strips, hence the alternative name of lacrimal river or rivus lacrimus.
There is little tear flow across the cornea between blinks. The limited flow and the uniform thickness are the consequences of the ‘framing’ function of the eyelids which is eliminated if the eyelids are withdrawn from the eyeball, causing a spotty drying of the precorneal film.
Apposition of the eyelid margins during blinking permits replenishment of lipid from the tarsal glands, which is then spread across the surface of the reconstructed film as the eyelids part. Similarly, mucus is spread over the corneal and conjunctival epithelium by the massaging action of the eyelids. Spreading of the tear film is facilitated by the movement of the eyelids across the cornea. In the presence of a contact lens, the interfacial tension at both lens surfaces has also to be considered. The desirable establishment of low interfacial tension at the two surfaces of the contact lens is partly related to the lens material and condition.
Tear liquid is essential for the maintenance of the normal optical properties of the cornea. Without it, corneal metabolic processes are impaired, as noted earlier, with consequent loss of transparency. The tears transport atmospheric oxygen and ions to the cornea and provide an excellent refracting surface. Tear liquid is the lubricant for eyelid movement over the cornea and the medium for flushing away foreign matter, which potentially endangers the optical and physical integrity of the cornea.
Tears have antibacterial properties. Lysozyme, the enzyme originally described by in tear liquid, provides a degree of protection against certain Gram-positive bacteria lodged in the conjunctival sac. Claims for the presence of a non-lysozyme antibacterial factor (NLAF) in tears with far greater activity than lysozyme are broadly agreed. Lactoferrin is present in abundance in tears ( ) and may have an anti-inflammatory function ( ). suggested that lactoferrin is effective in disrupting the cell membrane of Gram-negative bacteria (Pseudomonas aeruginosa, Escherichia coli and Proteus).
Among the antibody proteins, IgA and IgG are prominent in tears but other immunoglobulins are also present (see also Chapter 4 ). reported that the tear film concentration of secretory phospholipase A2 is sufficiently high to kill Gram-positive bacteria (Listeria monocytogenes and Staphylococcus aureus) but was not bactericidal against Gram-negative bacteria (E. coli, Salmonella typhimurium and P. aeruginosa).
Antimicrobial peptides (AMPs) are known to have antimicrobial actions and have been identified in tears ( ). It is not certain whether they are present in high enough concentration to be effective antimicrobially but may achieve active levels when the eye is challenged. Animal models have indicated that AMPs such as defensin and cathelicidin can reduce the severity of infection from bacteria and fungi. These peptides are secreted by ocular surface epithelium and consist of a number of different types with varying antimicrobial activity ( ).
found the daily output of tears to be less than 1g. This has been questioned because of the probable inaccuracy of his measuring technique. calculated an average rate of 14 g of tear secretion daily, but results were similar to those of Schirmer. subsequently reported a daily tear output of 15–30 g measured after instilling dye. The instillation of a stain (rose Bengal and/or fluorescein) will elevate the recorded figure for basic tear secretion, both by its own bulk and by its irritant effect.
Tear output measurements range from 1g/day ( ; ) to 15-30g/day (Norn, 1965). reported that the volume of tears present in the undisturbed eye is approximately 6-8 µl. found that full-term infants produced a mean basal tear production of 7.3 (± 3.2) mm as measured with the Schirmer test.
In a study of full-term infants, encountered shedding of tears as early as 5.5 hours of age and as late as 84 days. The onset of lacrimation occurred during the first 4 weeks in a majority of infants and in most it occurred first with crying associated with hunger and pain. used nasal irritants to determine the onset of reflex lacrimation in infants and found that it occurred in all but 13% during the first few weeks of life.
With advancing years, basic tear secretion decreases gradually: the difference between the sexes is negligible (Norn 1965) except in early adult life, when females have a higher rate of secretion ( , ). Sex differences concerning psychogenic tears or weeping are clearly a separate matter.
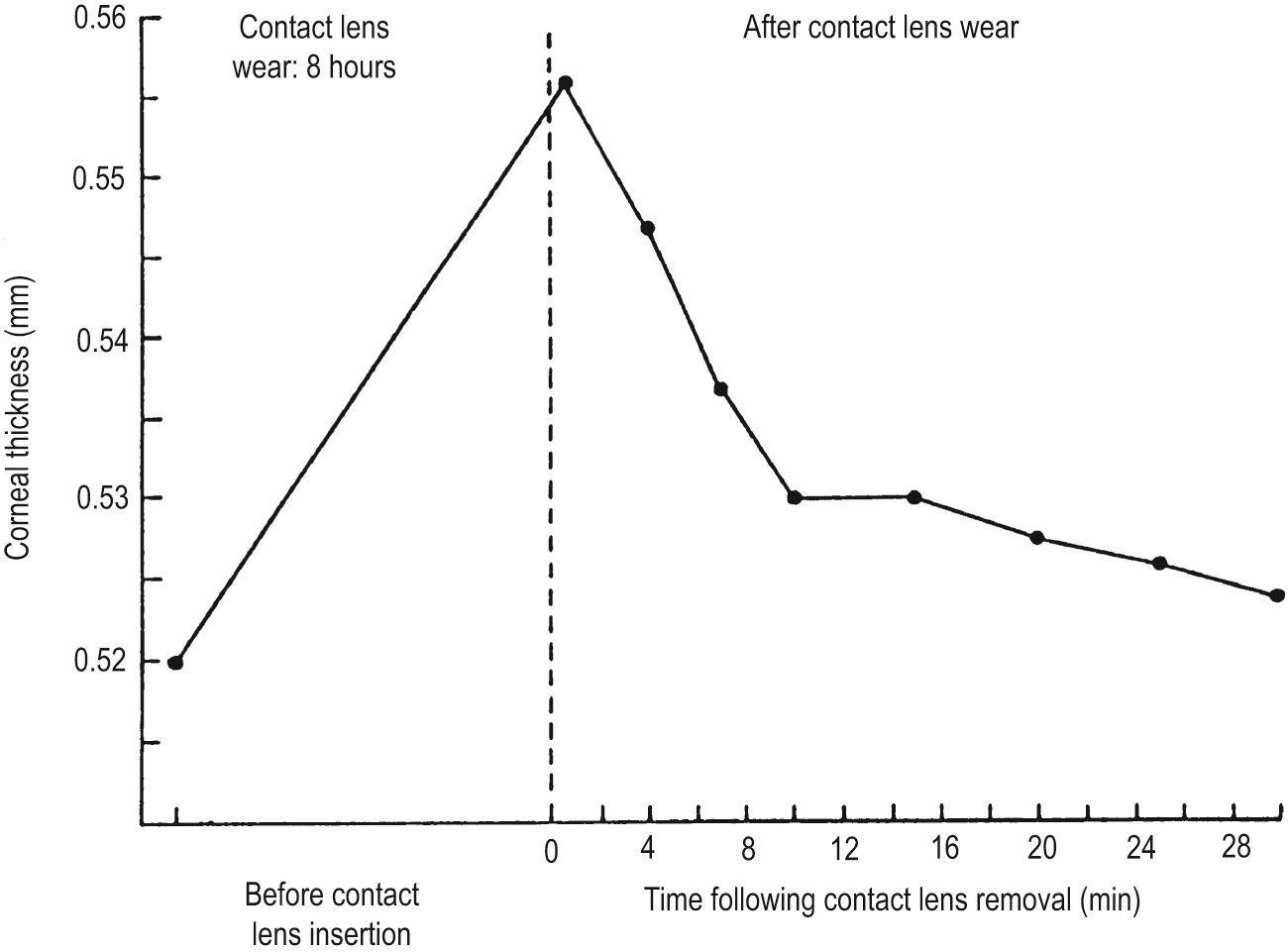
Interference with normal corneal metabolism produces thickening and reduction of transparency of the cornea. The thickening observed with lens use varies between individuals and depends on the type and material of the lens. Numerous early studies of the effect of PMMA lens wear produced figures of corneal thickness increase of 4–8%, usually measured after 8 hours wear. Fig. 3A.1 shows a typical result and adds a temporal profile of recovery following lens removal. Epithelial swelling may contribute to thickening.
Hiti et al. (2001) showed microwave ovens to be effective against Acanthamoeba (trophozoite and encysted forms) in as little as three minutes, while at 10% output, they were effective against bacteria in fifteen minutes (Kastl and Maehara, 2001) with minimal effects except with high water or tinted lenses (Harris et al., 1990; Crabbe and Thompson, 2001a and b).Using a no-rub MPS (ReNu® Multipurpose Solution), Crabbe and Thompson (2004) reported flocculation of solution components and foaming, leading to loss of solution and they concluded that lenses could be disinfected in 10-15 seconds, provided surfactants were avoided.
Raised temperature and therefore pressure, results from carrying a ‘sealed’ case close to the body, while reduced pressures and temperatures occur in aircraft. If the contents leak, there is:
Loss of solution.
Loss of antimicrobial action as external contaminants are ‘ingested’.
Partial lens dehydration.
A mess.
It is likely that the modes-of-action of different silver-containing lens cases vary from one another. Microblock shows good action against Gram-negative organisms, such as Pseudomonas aeruginosa and Serratia marcescens , while i-clean is effective against Staphylococcus aureus ( ). While broad-spectrum activity is the ultimate goal, that difference in case performance gives the practitioner options to suit a particular wearer. The Microblock lens case has also been shown to be effective outside the laboratory in two clinical trials ( and ). also showed that biofilm prevention with silver-doped cases is enhanced by wet-case storage during lens wear. Although those studies were not designed to determine whether antimicrobial cases reduced microbially driven adverse events, there was no increase in adverse events due to the liberated silver, indicating that, in the short-term at least, there was no safety issue. One area of concern in the development of silver-impregnated contact lens cases has been long-term systemic toxicity and ocular argyrosis. Ocular argyrosis manifests as a blueish-black discoloration of the conjunctiva, lacrimal sac, and cornea due to long-term silver exposure. The levels of contact lens case silver-ion exposure are much lower than that resulting in argyrosis from the use of Arygrol , a mucous membrane soothing agent, from which the condition takes its name ( ). Although organism resistance to silver is low and unlikely to be a clinical threat currently, the increased use of silver in medical devices warrants surveillance of silver as an antimicrobial agent.
Sodium salicylate , a non-steroidal anti-inflammatory drug (NSAID) similar to aspirin, was shown to reduce Pseudomonas aeruginosa and Staphylococcus epidermidis biofilm formation on plastic contact lens cases ( ) in the laboratory. Further studies have not been forthcoming.
Selenium bonded covalently to contact lens cases generates short-lived superoxide free radicals that inhibit bacterial proliferation and colonisation ( ) and it can inhibit the formation of biofilms in contact lens cases by Staphylococcus aureus ( ). Selenium's advantages over silver as an antimicrobial lens case additive are:
it does not need to leach out of the polymer to have an effect
less likely to cause allergies
less expensive ( ).
It is likely, however, that the reasons for non-compliance are not the same for all wearers and that will affect how they are best managed. For some it may be lack of time or complacency while for others it may be a lack of knowledge or an awareness of, but disregard for, the risks. For many wearers, re-education about risks with non-judgemental, sympathetic but authorative interactions with practitioners is likely to improve compliance ( and ) as has been shown with counselling patients against eye rubbing ( ). A recent large-scale survey of compliance has found that time since last visit to a practitioner was associated with non-compliance in a multivariate analysis ( ).
Following the recall of Complete MoisturePlus in the mid 2000s, the number of AK cases in the US fell. However, the decrease recorded did not reach the level reported prior to the product launch ( ) suggesting there is some residual increased risk that was not present in the early 2000s.
Reports from two centres in the UK indicate an increase in cases of AK. While those cases occurred more commonly with a particular contact lens solution ( and ), the findings are difficult to interpret, as there is no data to indicate what the market penetrance of the solution in question was in those regions. Such reports highlight a need for well-designed case-control studies to establish independent risk factors for AK.
Since the mid-2000s, a much-discussed topic has been solution induced corneal staining (SICS), a term coined originally by the late Prof Brien Holden. Although the levels of corneal surface compromise were less than with previous contact lens solutions containing preservatives such as mercury-based thiomersal ( ), reports emerged of low-grade ocular surface staining with certain combinations of lens care product and type of contact lens. published a paper on asymptomatic corneal sodium fluorescein (NaFL) staining following SiHy lens wear using multipurpose solution containing Poly HexaMethylene Biguanide (PHMB) as a preservative/disinfecting agent. Two patterns of staining were noted, an overall diffuse pattern, and a peripheral annular ring adjacent to the limbus. It is believed that the latter is a result of a low tear reservoir within the annulus formed by the contact lens conforming/flattening onto the sclera as determined by the lens-anterior eye fitting relationship. That aligns with the ‘uptake and release’ theory of SICS in which the preservative is either absorbed into the lens matrix or adsorbed onto the lens surface, and then released into the tear film subsequently ( ). They also showed that the peak staining times for different lens-solution combinations vary.
Soon after those first reports of SICS were aired, , in an endeavour sponsored by Alcon (before its merger with CIBA Vision), produced an evolving website, www.stainggrids.com , on which their results, purporting to indicate the proportion (% area) of the cornea stained after a 2-hour provocative test, appeared for a large number of solution-lens combinations. Subsequently, the Institute for Eye Research (IER) (now known as the Brien Holden Vision Institute), produced the IER matrix, from studies sponsored by CIBA Vision. The IER matrix shows the proportion of contact lens wearers exhibiting the characteristic SICS staining patterns per month during the first three months of lens wear ( and ). The general results of these two staining grids were at odds with each other but the methodology and measures were not comparable directly. What are the facts and the evidence?
Fluorescein has been used by the ophthalmic professions since 1882 as a routine way of determining ocular surface integrity. Recently, the role of fluorescein in SICS has been questioned. claim that the role of NaFL in SICS with PHMB was the combination of the positively-charged ion of PHMB and the negatively-charged fluorescein and the appearance of SICS was the adherence of those entities to the corneal surface. That was termed preservative-associated transient hyperfluorescence (PATH). That hypothesis was tested in a liposome-based model of the corneal epithelial surface in vitro . One of the limitations of that model is the artificial environment employed compared with multicellular models and in vitro models. Using a rabbit model, showed that staining represents fluorescein inside epithelial cells. More recently, a study by demonstrated that SICS also results in staining of corneal epithelial cells and that the transient nature of SICS is associated with an upregulation of surface cell shedding. Those studies counter the PATH theory of benign binding of a fluorescein complex to epithelial cells.
The question of whether SICS is associated with symptoms remains unanswered with some studies suggesting it is associated ( and ) while others say it is likely asymptomatic ( and ). It is likely that symptomatology of SICS is patient-dependent and may well depend on the type and sensitivity of the survey questions.
A range of antimicrobial strategies including: NSAIDs, selenium, melimine (a synthetic antimicrobial peptide), and furanones (or fimbrolides, substances that disrupt bacterial communications and were originally isolated from marine algae). Development of silver-impregnated contact lenses are more advanced with clinical trials to investigate their safety completed satisfactorily ( ). It has also been shown that these lenses do not alter the ocular surface microbiota ( ). Studies to show effectiveness (i.e. a decrease in adverse events) need larger wearer numbers as adverse events are rare. To date such trials have not been performed.
found an evaporative component in as many as 86% of dry eye cases and reported a four-fold increase in tear evaporation in the human eye in vivo , in the absence of a clinically visible or continuous lipid layer.
This can be described according to its proteome (proteins and peptides), lipidome (lipids), mucins and glycocalyx, and other components.
Proteome: in excess of 1000 proteins, or fractions of proteins, have been identified within the human tear film ( ). The total tear protein content (3.5 – 9.5 mg/ml) is similar in basal tears (non-stimulated) and reflex tears (stimulated) but the concentration of proteins increases significantly (16-18 mg/ml) in the closed eye state ( ). The tear film contains three types of proteins:
regulated proteins, which have a level of production that varies according to flow rate
constitutive proteins, which have a constant rate of production
serum-derived proteins. During reflex tearing, constitutive proteins (e.g., secretory IgA).and serum-derived proteins (e.g., albumin).decrease in concentration, but the concentration of regulated proteins such as lysozyme, lactoferrin and lipocalin-1, remain relatively stable ( ).
The tear fluid contains a wide range of tightly-regulated inflammatory mediators, including complement, arachadonic acid metabolites, and a range of cytokines) ( , ). Proteinase activity occurs through gelatinolytic and collagenolytic proteases within the tear film, including matrix metalloproteinase-9 (MMP-9), which is up-regulated in inflammatory ocular surface disease, such as dry eye disease.( )
Lipidome: Meibum contains non-polar wax esters, cholesterol esters, diesters, triacylglycerol, with a lesser proportion of cholesterol, fatty acids and polar lipids. Polar lipids account for up to 15% of the total lipids and these include phospholipids, ceramides, cerebrosides and long chain (O-acyl)-ω- hydroxyl fatty acid (OAHFA).( ) The origin of the polar lipids that interface with the aqueous tear component remains controversial with reports suggesting a source distinct from the meibomian glands ( ).
Mucins and glycocalyx: Mucins are high molecular weight proteins that form a protective film over the epithelial cell surface.( ) Glycosylation of these proteins grants them hydrophilicity. There are two types, described as transmembrane mucins and secreted mucins. Those produced by the corneal and conjunctival epithelia are transmembrane mucins that mostly remain bound to the apical surfaces of the cells. These mucins (MUC1, MUC4 and MUC16) interact with galectins on the epithelial surface to form a glycocalyx barrier that resists adhesion ( ). Secreted mucins (MUC5AC).are produced by the conjunctival goblet cells and are distributed throughout the aqueous phase of the tears.( ) Their concentration is highest adjacent to the glycocalyx and decreases towards the lipid layer.
Other components: The tears further contain antioxidant components, which protect against oxidative stress,( ) and white blood cells (polymorphonuclear leucocytes and neutrophils). Exogenous components include cosmetic products, contact lens wetting agents, and topical medications and their preservatives ( ).
Reduced stability of the tear film is observed most dramatically with rigid contact lenses but occurs with all types ( , Morris et al., 1997). While lens dehydration might play some role in the loss of stability in soft lens wearers, the main factor is believed to be altered lipid layer structure, which leads to increased tear evaporation and ultimately to lens surface dewetting.( ).
Accompanying decreased tear film stability and, in particular, the adversely affected lipid layer during contact lens wear, is a corresponding and significant increase in tear evaporation rate ( , , ). The increase in tear evaporation rate is observed to be comparable across all lens types ( ).
Lipidome: Studies evaluating compositional changes in the tear film with contact lens wear are limited. It has been suggested that increased tear cholesterol ester levels are associated with poor lipid layer thickness and lens discomfort.( ) Levels of polar lipids, and specifically phospholipids, decrease in contact lens wear ( ) which may be related to the increased levels of the lipolytic enzyme, phospholipase A2.( ) Lipid components from the tears that deposit on the surface of contact lenses can also induce lens intolerance from their degradation.( )
Proteome: Total protein concentration and the levels of lysozyme and lactoferrin are not significantly altered during contact lens wear.( , ) Albumin levels also stay relatively constant, except in the closed eye environment such as with orthokeratology, where a rise in albumin levels is observed.( ) Inflammatory mediator levels are generally observed to increase during contact lens wear, in a length of wear-dependent fashion.( )
Mucins and glycocalyx: Both rigid and soft contact lens wear have been associated with reduced MUC5AC levels, indicating a reduction in secreted mucins from the goblet cells.( ) Differences in experimental approach and lens material are thought to explain the conflicting reports of upregulation and downregulation of transmembrane mucins, in in vitro and in vivo studies, respectively.( ) The ocular surface glycocalyx undergoes thinning and compression due to the direct physical insult of the contact lens in situ .( ) Multipurpose contact lens solutions can further adversely affect the glycocalyx, affecting mucin expression and shedding.( ).
A meta-analysis by indicated that there was no significant difference in MGD prevalence between lens wearers and non-lens wearers.
The possible association between contact lens wear and meibomian gland dropout is unclear, with conflicting reports ( ) Contact lens wear appears to be accompanied by changes to meibomian gland morphology ( , ) with emerging evidence suggesting these changes may occur during the early years of lens wear and that prolonged lens exposure may not be associated with progressive morphological alterations ( ).
Variable degrees of correlation between measures derived from the Phenol red thread and Schirmer tests have been reported, suggesting that these indirect measures of tear secretion are may be measuring different aspects of tear production ( ).
This may result from a range of factors, including hypoxia, trauma/mechanical effects, toxicity, inflammation, exposure, allergy and/or infection ( ). The pattern and location of staining can be useful for deducing the potential aetiology. For example, exposure keratitis typically produces a band of inferior arcuate staining that occurs in association with incomplete blinking. Another common form of exposure keratitis can occur in rigid corneal lens wearers, whereby localised corneal staining occurs along the horizontal meridian, at three and nine o'clock, due to desiccative damage at the lens edge.
There is a significant literature relating to the association between corneal staining and specific combinations of contact lens materials and contact lens disinfection solutions. At present however, the association between corneal staining and contact lens comfort remains to be clarified.
Ocular demodecosis and blepharitis are associated with each other, although there lacks clarity over cause and effect.( ) The pathogenic mechanisms of Demodex are not fully understood but are considered to include cellular consumption, lipolytic enzyme production and direct tissue micro-trauma, which stimulate epithelial hyperplasia and reactive hyperkeratinisation.( , ).
The Blephasteam® is a proprietary latent heat device in which moist heat is delivered, without simultaneous compression of the lid tissues, within a sealed goggle system. Improvements in symptoms and signs of MGD,( , ) as well visual quality,( ) conjunctival hyperaemia,( ) and tear evaporation rate,( ) have been noted following treatment.
, , ) have reported improvemnts from intensed pulsed light (IPL). In the clinical setting, therapeutic gland expression is typically performed immediately following IPL treatment ( ).
Increasing blink rate can improve tear film stability and symptoms in VDU users ( )
The polar lipid pattern of meibomian gland secretions is influenced by the dietary intake of ω-3 EFAs ( ). Furthermore, in a small pilot study, supplementation with short-chain ω-3 EFAs for 12 months was shown to reduce the saturated fatty acid content of meibum in people with MGD, which was associated with improvements in signs and symptoms ( ). There are now several published clinical trials investigating the use of EFA supplementation for treating dry eye, with growing evidence to support this modality for reducing ocular surface inflammation and improving tear stability. Further research is needed to clarify the optimal daily dose to recommend to patients, the necessary duration of treatment and the ideal supplement composition.
Lipid-containing lubricants have been shown to be useful for improving some clinical signs of dry eye, including dryness symptoms, lipid layer structure, TBUT and tear evaporation ( ).
The safety and efficacy of topical cyclosporine is well established for dry eye disease ( ). However, there have only been a few, small studies that have considered the potential application of cyclosporine-A for treating contact lens discomfort, with conflicting findings. Additional research is required to clarify the potential role of this therapy for modulating tear film integrity, with the specific intent of enhancing contact lens comfort.
This graph compares the spectacle magnification for a spectacle lens (blue line) and a contact lens (green line) based only on their power and vertex distance (to the entrance pupil), ignoring magnification due to lens thickness, with that of a typical contact lens including the magnification due to lens thickness and front surface power.
Spectacle magnification = (that produced by the prescription and vertex distance, the Power Factor) × (that produced by the thickness of the lens and the power of the front surface, The Shape factor)
A thin lens means that we ignore the shape factor.
These graphs therefore show the difference the RGP, Soft and thin contact lenses relative to a thin spectacle lens, all giving the same effective correction at the cornea. Hence there is no thin lens shown on the graph
Fig. 7.12 (see Chapter 7 ) shows typical RGP and soft lenses to demonstrate how little magnification the thickness and front surface power of the lens makes.
The results from showed that the other higher order Zernike aberrations (see , or text books on optics for further details) averaged to approximately zero, but this was not confirmed in a very large study by .
The values of aberrations found by and when lenses were immersed in a saline cell may not be replicated when the lens is on the eye. Conversely, since the thickness profile of a contact lens does not change when it flexes onto the eye, suggested that the wavefront aberration for a contact lens in air or saline would show similar results when on an eye.
Negative powered lenses would be expected to show negative spherical aberration and therefore reduce the average ocular aberration, while positive lenses would be expected to increase it. Using theoretical simulations of vision and the results of aberrometry measurements on 1074 myopic eyes and 50 hypermetropic eyes, predicted that more than half the population would have had a better VA by ![]() a line (logMAR) and 68% by more than one letter with lenses designed to correct the average spherical aberration. compared contact lenses with a spherical and a new aspherical design and found that the aspherical design reduced the mean spherical aberration to near zero irrespective of lens power. The -7.00 spherical design made the overall spherical aberration negative, the -5.00 power reduced the mean aberration to near zero, while both the -3.00 and +3.00 powers increased the positive spherical aberration.
a line (logMAR) and 68% by more than one letter with lenses designed to correct the average spherical aberration. compared contact lenses with a spherical and a new aspherical design and found that the aspherical design reduced the mean spherical aberration to near zero irrespective of lens power. The -7.00 spherical design made the overall spherical aberration negative, the -5.00 power reduced the mean aberration to near zero, while both the -3.00 and +3.00 powers increased the positive spherical aberration.
To give an idea of the design of these lenses, power profiles of bifocal and progressive power contact lenses, together with some single vision lenses, has been published by . made aberration and contrast sensitivity measurements and suggested that the two brands of lens that they tried did not provide sufficient myopic shift in refraction and increased depth of focus to give full contrast sensitivity in near vision. This was at the 5 mm artificial pupil size used, so the smaller pupils that might be met in presbyopic wearers might give poorer vision. Improving near performance might, however, be at the expense of general image quality.
investigated the modulation transfer function for several types of multifocal and progressive power lens. They concluded that the reduced contrast in the image would make little difference to high contrast acuity. investigated, amongst other factors, the performance of bifocal lenses as a function of the relative sizes of central and peripheral optic zones, and decentration, but found their results too complicated for simple summary.
A number of computer-assisted objective grading techniques have been described to quantify retinal vasculature. Edge detection and colour extraction techniques have been shown to be the most stable to changes in image luminance and to correlate well to traditional grading scale images ( ).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here