Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Acute mesenteric ischemia of the small bowel has four major causes: (1) arterial embolism, (2) arterial thrombosis, (3) nonocclusive mesenteric ischemia, and (4) mesenteric venous thrombosis ( Table 26-1 ). Less common causes include aortic dissection, spontaneous dissection of the celiac or superior mesenteric artery (SMA), and vasculitis. The common end result is an acute reduction in splanchnic blood flow that can lead to bowel necrosis.
| Cause | Onset of Presentation | Associated Features |
|---|---|---|
| SMA embolism | Acute | Myocardial infarction, arrhythmia, ventricular aneurysm, valvular disease, prior embolic event |
| SMA thrombosis | Acute or acute on chronic | Atherosclerotic disease, hypercoagulable state |
| Nonocclusive mesenteric ischemia | Acute, failure to thrive | Critical illness, hypotension, myocardial infarction, sepsis, disseminated intravascular coagulation, vasopressors |
| Mesenteric venous thrombosis | Acute (<4 wk) Chronic (≥4 wk) |
Recent surgery, hypercoagulable state, oral contraceptive use |
Acute mesenteric ischemia affects approximately 1% of patients with acute abdomen and increases in incidence with age. Mortality rates exceed 60%, exacerbated by delays in diagnosis.
Arterial embolism, the most frequent cause of acute mesenteric ischemia, is implicated in up to 50% of cases and most often involves the SMA. The embolus is usually of cardiac origin; hence, myocardial infarction, arrhythmia, valvular disease, and ventricular aneurysm are important risk factors.
Acute SMA thrombosis is the second most common cause of acute mesenteric ischemia, accounting for 25% of cases, usually in the setting of severe atherosclerosis. Hypercoagulability is another important risk factor.
Nonocclusive mesenteric ischemia, implicated in 20% of patients with acute mesenteric ischemia, has a mortality rate as high as 70%. It is most often seen in patients older than age 50 years with reduced cardiac output, hypovolemia, or hypotension. A low-flow state results in diffuse mesenteric vasoconstriction, which may be exacerbated in patients on vasopressor therapy. The incidence of nonocclusive mesenteric ischemia is declining, presumably owing to advances in critical care medicine, including the use of vasodilator therapy.
Mesenteric venous thrombosis accounts for approximately 18% of cases of acute mesenteric ischemia. It has a poor prognosis with a long-term survival of 30% to 40%. Mesenteric venous thrombosis is considered acute when symptoms are present for less than 4 weeks. Risk factors include recent surgery, trauma, inflammatory disorders such as pancreatitis, and hypercoagulable states.
The clinical presentation of acute mesenteric ischemia is nonspecific and may mimic other common disease processes such as small bowel obstruction or pancreatitis. The presentation is also a function of the specific pathologic process that is compromising splanchnic blood flow—SMA embolism or thrombosis, nonocclusive mesenteric ischemia, or mesenteric venous thrombosis (see Table 26-1 ). Because of the nonspecificity of manifestation, delays in diagnosis are common and many cases progress to bowel infarction.
Acute mesenteric ischemia classically manifests as acute abdominal pain that is disproportional to the findings on physical examination. Other findings may include dehydration, tachycardia, and altered mental status. Nonspecific laboratory findings such as an elevated serum lactate level and leukocytosis may be found with all major causes of acute mesenteric ischemia.
Both SMA occlusion and thrombosis may manifest acutely. Patients with SMA thrombosis, however, may report an antecedent history of postprandial pain, weight loss, and aversion to food. Because these symptoms are associated with chronic mesenteric ischemia secondary to atherosclerosis, there may be an “acute on chronic” manifestation, in which there may be a better developed collateral circulation than in patients with SMA embolism.
Patients with nonocclusive mesenteric ischemia are usually older, critically ill, intubated, or on vasopressor or digitalis therapy and may be unable to communicate symptoms. Clinicians must have a high index of suspicion, particularly in patients with unexplained failure to thrive.
Patients with acute mesenteric venous thrombosis are more typically symptomatic than patients with chronic mesenteric venous thrombosis and may present with slowly progressive diffuse abdominal pain and distention. Bloody ascites, dehydration, hemoccult-positive stool, and hypotension may be associated findings.
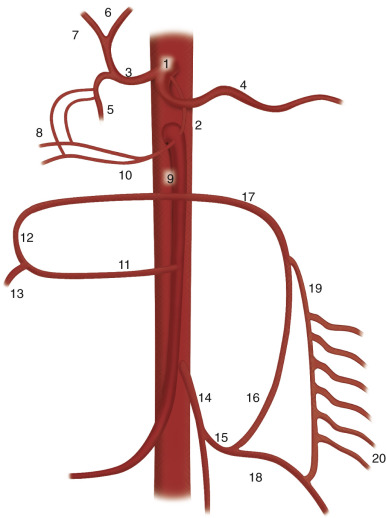
The arterial supply of the small bowel is predominantly from the celiac axis and SMA. The inferior mesenteric artery and the internal iliac arteries may become important contributors in the setting of arterial disease.
The celiac artery gives origin to the left gastric artery, and then it bifurcates into the splenic and common hepatic arteries ( Figure 26-1 ). The gastroduodenal artery is a branch of the common hepatic artery and gives off, among other vessels, the superior pancreaticoduodenal arteries.
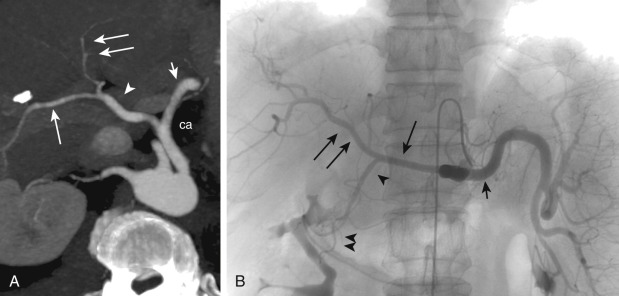
The SMA arises from the anterior aspect of the aorta at the level of the L1 vertebral body ( Figure 26-2 ) and supplies the duodenum, small bowel, and colon proximal to the splenic flexure. The inferior pancreaticoduodenal artery arises from the SMA as the first branch and anastomoses with the superior pancreaticoduodenal artery, forming the pancreaticoduodenal arcades ( Figure 26-3 ). The middle colic artery may arise from the right side of the SMA to supply the transverse colon. It bifurcates into a right branch, which anastomoses with the right colic artery, and a left branch, which anastomoses with the ascending branch of the left colic artery ( Figure 26-4 ). Multiple jejunal and ileal arteries as well as the right colic artery supplying the ascending colon arise from the SMA. More distally, the SMA terminates in the ileocolic artery, which supplies the terminal ileum, cecum, and ascending colon.
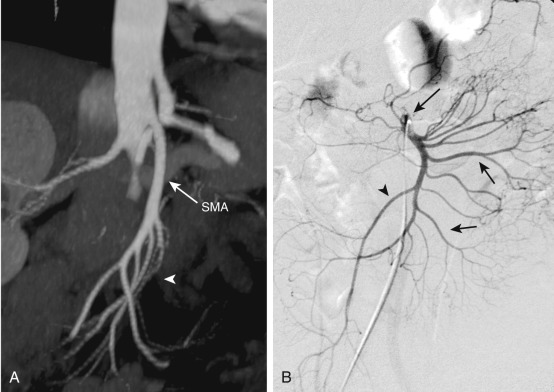
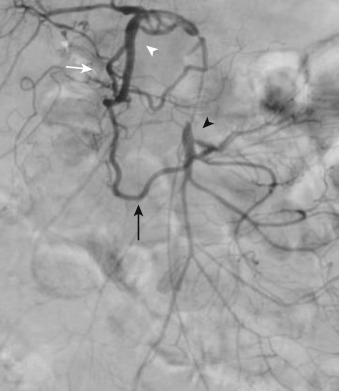
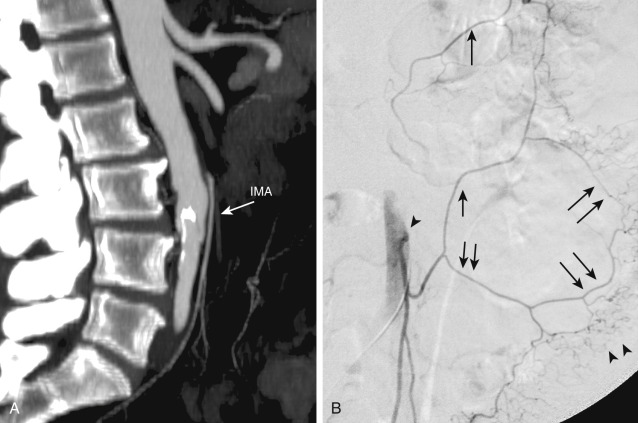
The inferior mesenteric artery arises from the left aspect of the aorta at the level of the L3 vertebral body (see Figure 26-4 ). It divides into the left colic, sigmoid, and superior hemorrhoidal arteries, which supply the descending colon, sigmoid colon, and rectum, respectively. The ascending branch of the left colic artery anastomoses with the left branch of the middle colic artery, a branch of the SMA. The superior hemorrhoidal artery anastomoses with branches of the anterior division of the internal iliac arteries.
The marginal artery, defined as the artery closest to and parallel with the mesenteric margin of the intestine, gives off the vasa rectae, which are small vessels that supply the bowel wall. In the colon, this artery is termed the marginal artery of Drummond (see Figure 26-4 ). The middle colic artery may serve as the marginal artery for much of its distribution.
The rich arterial supply of the bowel provides ample opportunity for collateral development in the setting of mesenteric arterial stenosis or occlusion ( Figure 26-5 ). The superior and inferior pancreaticoduodenal arteries can provide important collaterals between the celiac axis and the SMA (see Figure 26-3 ). In addition, a persistent fetal arterial communication between the celiac axis and the SMA (often referred to as the arc of Buehler) may exist in up to 2% of patients. Collateral pathways between the superior and inferior mesenteric arteries primarily involve communication between the left and middle colic arteries via the arc of Riolan ( Figure 26-6 ), located centrally in the mesentery, and the marginal artery of Drummond, located peripherally. The internal iliac arteries also may provide collateral circulation to the principal mesenteric vessels.
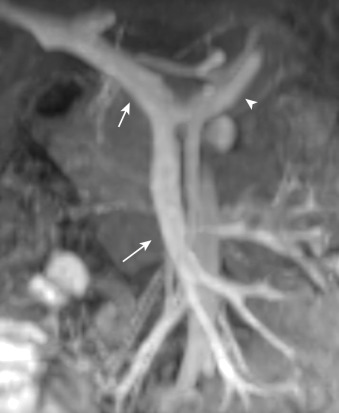
The superior mesenteric vein is usually a single vessel that drains the small intestine and the ascending and transverse colon. Within the mesentery, the superior mesenteric vein courses anteriorly and to the right of the SMA ( Figure 26-7 ) and joins the splenic vein to form the main portal vein. The inferior mesenteric vein, which drains the left colic, sigmoid, and superior hemorrhoidal veins, usually terminates in the splenic vein or the superior mesenteric vein. Portal-to-portal collaterals may develop in the setting of chronic superior mesenteric vein occlusion; these are often submucosal and are prone to bleeding. Portal-to-systemic collaterals (varices) also may develop in portal hypertension.
Mesenteric blood flow is regulated by perfusion pressure, oxygen demand, alpha- and beta-adrenergic stimulation, and humoral factors such as vasopressin. At baseline, 20% to 25% of mesenteric capillaries may be open with considerable reserve against changes in blood flow. Although the bowel can tolerate a considerable reduction in mesenteric perfusion, when there is a prolonged mismatch between demand and supply, ischemia will ensue. Prolonged ischemia may result in tissue damage secondary to reperfusion injury, resulting in increased microvascular permeability. Ultimately, compromise of the intestinal mucosal barrier may occur, mediated by oxygen free radicals.
SMA emboli are most often of cardiac origin, with approximately one third of patients having a history of embolic events. Arterial emboli typically involve the SMA because of the oblique angle of its origin, with approximately 15% occluding the origin. A majority of emboli, however, will lodge distally in the SMA at branch points, and the distribution of ischemic bowel in many cases will therefore involve the distal jejunum and ileum, while sparing the proximal jejunum. Large emboli that initially occlude the SMA origin may propagate distally and potentially obstruct collateral flow from the celiac artery and inferior mesenteric artery. The clinical presentation is acute, with insufficient time to develop a collateral perfusion.
The most common site of arterial thrombosis is the origin of the SMA. Because of progressive atherosclerotic disease, collateral circulation may have developed and thus symptoms occur only when there is disease of multiple mesenteric arteries and major collateral vessels or when thrombosis occurs with insufficient collateral support. Acute hemodynamic compromise, dehydration, or hypercoagulability in the setting of visceral artery stenosis may prompt the thrombotic event. In SMA thrombosis, a greater length of bowel may become ischemic or progress to infarction than with SMA embolus.
In nonocclusive mesenteric ischemia, diminished mesenteric arterial flow results from reduced perfusion pressure or vasoconstriction rather than from a physical impediment to blood flow. This reduced perfusion pressure resulting from various causes such as heart failure, hypotension, sepsis, disseminated intravascular coagulation, vasoconstrictive medications, and/or major surgery, eventually causes diffuse mesenteric vasoconstriction via autoregulatory mechanisms.
Mesenteric vein thrombosis, which is often associated with recent surgery, hypercoagulable state, or inflammatory disorders, usually begins in the venous arcades and can propagate to the superior mesenteric vein and portal vein. The inferior mesenteric vein is less frequently affected. Venous obstruction results in hypovolemia and hemoconcentration, arteriolar vasoconstriction, and reduced arterial inflow, ultimately leading to hemorrhagic bowel infarction. Infarcted bowel is segmental, and the transition between normal and ischemic bowel is typically more gradual compared with other causes of acute mesenteric ischemia.
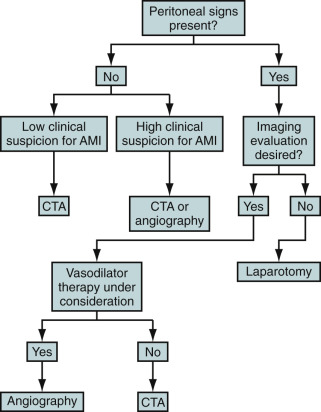
Because acute mesenteric ischemia may rapidly progress to bowel infarction, patients with peritoneal signs should proceed to emergent laparotomy. Nevertheless, imaging can play an important role in the diagnosis of SMA embolism and operative planning.
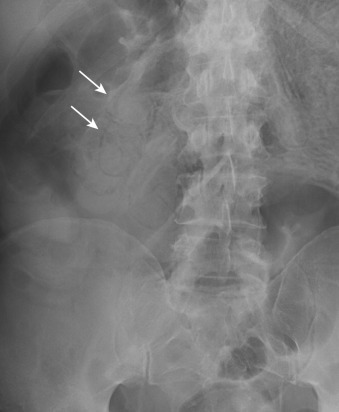
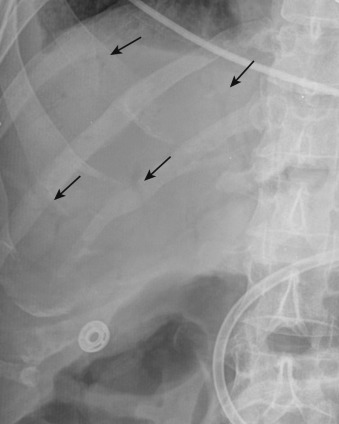
Plain radiography is nonspecific in all causes of acute mesenteric ischemia and may be normal in one fourth of cases. Plain radiographs may demonstrate dilated fluid-filled bowel loops, suggesting a nonspecific ileus, thumbprinting from focal submucosal hemorrhage, or separation of bowel loops as a result of mesenteric thickening. Pneumatosis ( Figure 26-8 ), mesenteric or portal venous gas ( Figure 26-9 ), and pneumoperitoneum indicate bowel infarction. Plain radiography may help exclude some other causes of abdominal pain such as bowel obstruction.
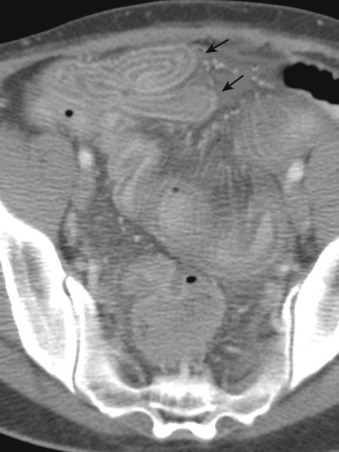
CT may demonstrate nonspecific fluid-filled dilated bowel with wall thickening, ascites, and mesenteric edema. CT also may demonstrate a water-halo sign, in which two concentric rings of different attenuation are seen in the bowel wall: either a high-attenuation outer ring with gray attenuation inner ring or vice versa. Similarly, a target sign ( Figure 26-10 ) may be seen, in which a central ring of gray attenuation is interposed between two rings of higher attenuation. Inflammatory bowel disease may produce a similar appearance.
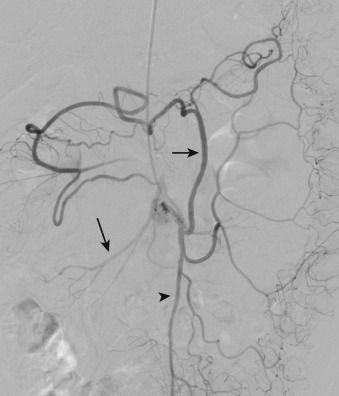
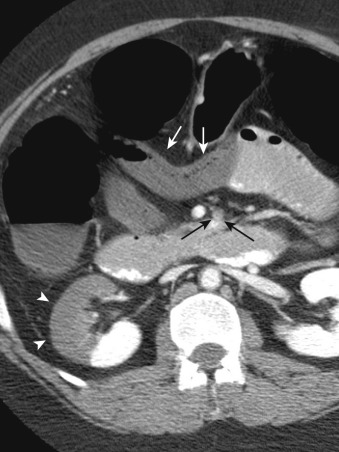
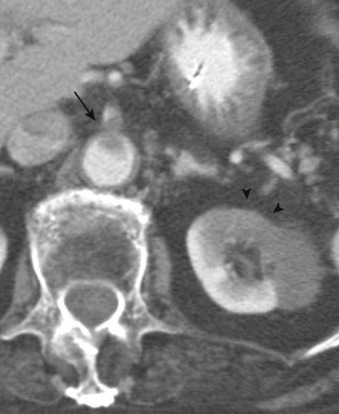
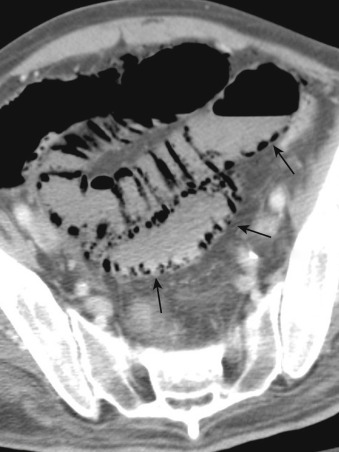
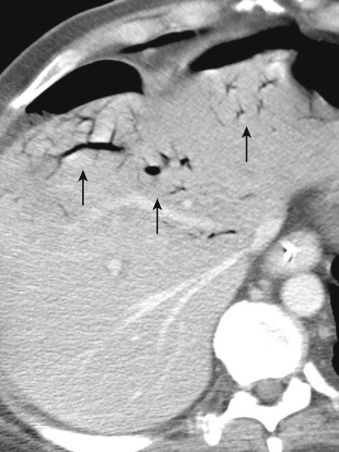
More specific findings include lack of bowel wall enhancement and infarcts of other visceral organs such as the kidney ( Figure 26-11 ). CT angiography (CTA) increases the accuracy of the detection of an SMA embolus and should include initial noncontrast images for evaluation of vascular calcifications. CTA will demonstrate a filling defect within or nonopacification of the SMA ( Figures 26-11 to 26-13 ), although sensitivity diminishes with more distal emboli. Few to no collateral vessels are demonstrated.
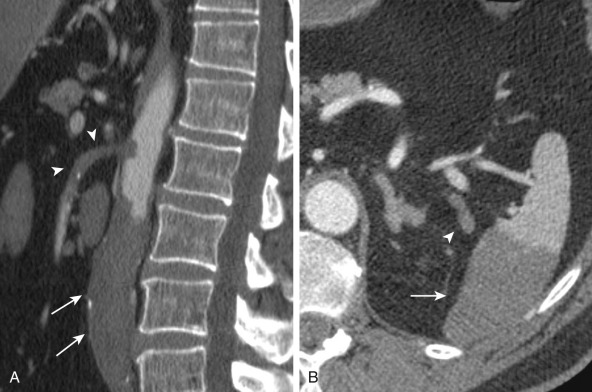
Late-stage findings such as pneumatosis ( Figure 26-14 ), pneumoperitoneum, and mesenteric and portal venous gas factors ( Figure 26-15 ) are markers of bowel infarction and necrosis. CT is helpful to evaluate other causes of abdominal pain as well.
Gadolinium-enhanced MR angiography (MRA) can be used to demonstrate SMA emboli but is not the first-choice imaging modality in acute mesenteric ischemia, given long examination times that can delay treatment.
Although duplex sonography may detect occlusion at the origin of the SMA, more distal emboli may be missed. An occluded vessel will appear dilated and may contain echogenic debris with absent Doppler flow. Linear echogenic foci of intramural or portal venous gas may be detected and are signs of bowel infarction. Ultrasonography is operator dependent and can be limited by bowel gas, body habitus, and patient noncooperation.
There is no role for scintigraphy in the evaluation of SMA embolism.
There is no role for positron emission tomography with computed tomography (PET/CT) in the evaluation of SMA embolism.
Angiography is the gold standard for the detection of SMA embolism, with sensitivity exceeding 90%. Lateral aortography is used to evaluate the origins of the artery and celiac axis ( Figure 26-16 ). Anteroposterior aortography is useful to assess the aorta, renal arteries, and distal mesenteric vessels. The angiographic diagnosis of acute embolus is made when a filling defect is demonstrated that at least partially obstructs the artery, with absence of collateral vessels. Selective arteriography of the SMA can be performed in the absence of disease at its origin to assess for distal occlusion and may be accompanied by catheter-directed intervention, if appropriate.
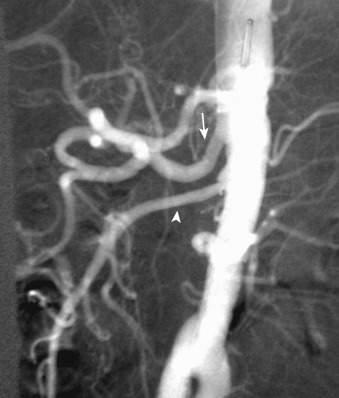
Angiography, however, is invasive and time consuming and is associated with potential nephrotoxicity and other procedure-related morbidities.
An imaging algorithm is provided in Figure 26-17 . See also Table 26-2 .
Abdominal pain out of proportion to physical findings
Filling defect within the SMA
Lack of bowel wall enhancement
Infarcts of other visceral organs
Late findings: Pneumatosis, portomesenteric venous gas, pneumoperitoneum
| Modality | Accuracy | Limitations | Pitfalls |
|---|---|---|---|
| Radiography | Poor | Sensitivity 30% Nonspecific |
May not suggest disease until late stage of bowel infarction/perforation |
| CT | Sensitivity 64%-82% | Radiation | Distal emboli may be missed. |
| MRI | Although sufficient comparative data are lacking, sensitivity and specificity may be comparable to that for CT for vessel findings but less accurate for bowel findings. | Long examination times High cost |
Suboptimal evaluation of stented vessels Tailored examination less able to assess abdomen for other pathologic process compared with CT |
| Ultrasonography | Accuracy diminishes with more distal emboli and is operator dependent. | Patient body habitus Bowel gas Patient cooperation |
Distal emboli may be missed. |
| Nuclear medicine | No role in the evaluation of acute mesenteric ischemia | Poor spatial resolution | |
| PET/CT | No role in the evaluation of acute mesenteric ischemia | Radiation High cost |
|
| Angiography | Sensitivity 90% | Radiation Invasive |
SMA thrombosis may manifest as “acute on chronic” symptoms. Atherosclerotic disease may affect several mesenteric vessels, with thrombotic occlusion of the SMA precipitating the acute clinical event.
Plain radiography of the abdomen is nonspecific in all major causes of acute mesenteric ischemia.
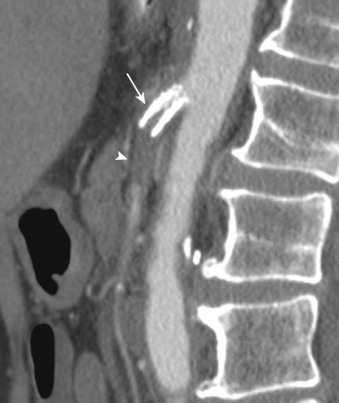
CT and CTA are useful in the evaluation of SMA thrombosis. Bowel findings overlap with that of SMA embolism. As previously noted, CTA should initially include noncontrast images so as to evaluate for vascular calcifications that may otherwise be obscured. Stenosis or occlusion may be demonstrated in the origin of the superior mesenteric, inferior mesenteric, or celiac arteries and collateral vessels may be identified. Thrombosis of a previously stented mesenteric artery also may cause acute mesenteric ischemia ( Figure 26-18 ). Three-dimensional (3D) image postprocessing can be useful in diagnosis and in planning revascularization.
Contrast-enhanced MRA may similarly demonstrate narrowing or cutoff of the SMA. Collateral vessels may be demonstrated. However, MRA is not advocated for the evaluation of acute mesenteric ischemia because long examination times can delay treatment.
Duplex sonography may demonstrate elevated peak systolic velocities in the proximal SMA, indicating stenosis, or lack of flow, suggesting occlusion. As with SMA embolism, ultrasonography is not advocated in the evaluation of acute mesenteric ischemia.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here