Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Acne vulgaris is the most common skin problem in the United States, affecting nearly 80% to 85% of individuals at some point between 11 and 30 years of age. , Although not a serious disease, acne may be the source of permanent scarring and, even more importantly, psychosocial morbidity and decreased emotional well-being. Withdrawal from society, depression, and decreased self-esteem may occur in individuals with more significant disease. Girls and boys with acne have more feelings of uselessness and lower body satisfaction compared with those without acne, and these features are compounded by the fact that acne typically presents in adolescents, who tend to be psychologically vulnerable and sensitive to modification in their physical appearance. Acne severity has been shown to be directly correlated with extent of embarrassment and lack of enjoyment of or participation in social activities. Patients with even mild to moderate acne have demonstrated high scores on the Carroll Rating Scale for Depression and an increased prevalence of suicidal ideation (SI). In addition, patients with severe acne may have poorer academic performance and higher unemployment rates. Issues such as these highlight that the potential benefits of acne therapy extend far beyond the simple cosmetics of the disease.
Pediatric acne is the terminology applied to acne occurring from birth through 11 years of age; adolescent acne describes patients from age 12 to adulthood. The contemporary classification of pediatric acne based on age of onset of the disease is shown in Fig. 8.1 .

Acne vulgaris is seen routinely by primary care physicians, and so familiarity with the treatment of this condition is vital. The majority of acne patients with mild to moderate disease respond well to traditional therapies; patients with severe or recalcitrant disease may merit treatment with agents most appropriately prescribed by dermatologists. Although the underlying cause of acne vulgaris remains unknown, considerable data concerning its pathogenesis have accumulated in recent decades to allow a rational and therapeutically successful approach to its management. There is no single “gold standard” therapy for acne, and treatment regimens must be individualized and occasionally fine-tuned to achieve the optimal response.
The tendency to develop acne is often familial and is felt by some to be inherited as an autosomal dominant trait. However, because of the high prevalence, the genetics of the disease remain unclear. Adolescent acne usually begins around the time of onset of puberty, occurring earlier in girls than boys. However, the onset of acne may occur significantly earlier, and it may be the first sign of pubertal maturation in girls, correlating with increasing levels of dehydroepiandrosterone (DHEA), an adrenal androgen. Natural history studies show that in 80% of patients the incidence and severity of acne decline by the early 20s, although as many as 50% of adults older than 25 years are affected and a minority may have persistence into middle age. , An important consideration is the timing of onset of puberty, which has followed a downward trend for many years. Studies from the mid-twentieth century onward suggest that breast and pubic hair development in American girls is occurring at younger ages, with Black girls disproportionately represented. As a result the regional definitions of precocious puberty have been revised such that puberty is currently considered precocious if it occurs before 8 years of age in European girls, before 7 years of age in American White girls, and before 6 years of age in Black girls. A similar trend has not been identified in boys, in whom the cut-off age for precocious puberty continues to be 9 years. ,
An understanding of acne pathogenesis helps the practitioner conceptualize and formulate the therapeutic plan. Four interrelated processes, including hyperkeratinization, androgen stimulation, bacterial infection, and inflammation, are traditionally believed to be involved in this multifactorial disease. A pictorial representation of acne pathogenesis is shown in Fig. 8.2 . Individual acne lesions usually begin with obstruction of the pilosebaceous unit (composed of the hair follicle and the sebaceous gland). These units are localized primarily to the face and trunk, and these “follicular plugs” (or microcomedones) are caused by excessive numbers of desquamated epithelial cells from the wall of the follicle in combination with excessive amounts of sebum, the oily, lipid-rich substance produced by the sebaceous glands. Sebum production is stimulated by adrenal and gonadal androgens and rises to maximum levels during late adolescence. These microcomedones enlarge into comedones, which may be open (blackheads) or closed (whiteheads). The reason for the black appearance of open comedones is unclear; one hypothesis suggests compaction and oxidation of the keratinous material at the follicular opening results in this finding. Melanin pigment may also play a role. ,
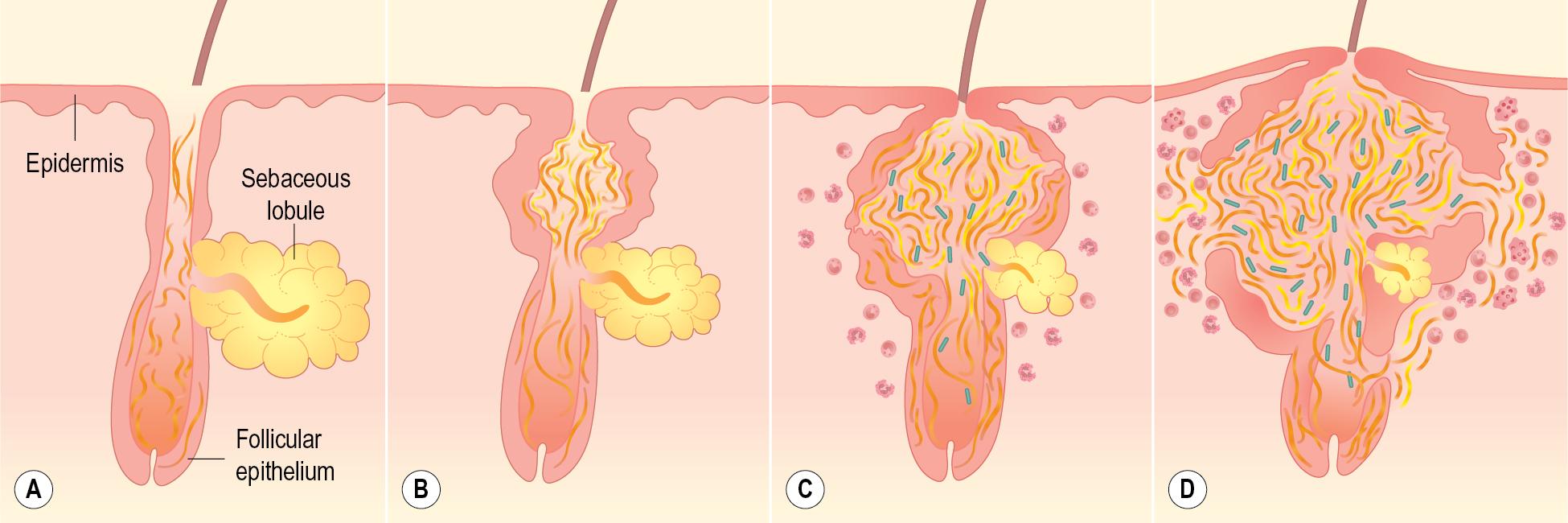
Propagation of acne lesions occurs when Cutibacterium acnes (formerly Propionibacterium acnes), a resident anaerobic organism, proliferates in this environment of sebum and follicular cells with the production of chemotactic factors and proinflammatory mediators, which contribute to inflammation. Hypersensitivity to this organism may also play a part in the more severe forms of acne. Observations regarding the potential mechanisms of C. acnes induced inflammation have suggested activation of toll-like receptor signaling, stimulation of antimicrobial peptide activity, induction of matrix metalloproteinase production, and inflammasome activation. The clinical counterpart to these infectious and inflammatory events are papules and pustules. With continued inflammation and macrophage recruitment, larger lesions (cysts and nodules) may result. Finally, sequelae of active acne lesions include dyspigmentation (usually hyperpigmentation or, occasionally, hypopigmentation) and scarring. Box 8.1 summarizes the various types of acne lesions.
Active lesions (increasing severity)
Microcomedone (may not be clinically apparent)
Open comedone (blackhead)
Closed comedone (whitehead)
Papule
Pustule
Nodule
Cyst-like nodule
Sequelae
Dyspigmentation (usually hyperpigmentation)
Scarring (sometimes keloidal)
Although the pathway in Fig. 8.2 has traditionally been used to explain acne pathogenesis, ongoing research continues to shed light on these processes. Subclinical inflammation, even before the formation of the microcomedone, may be present in acne as demonstrated by upregulated levels of inflammatory cytokines and cells in uninvolved skin from acne patients. Another study supporting this hypothesis used digital photographic images to follow the evolution of acne lesions and showed that nearly 30% of inflammatory lesions arose from normal-appearing skin (as opposed to comedones). Further elucidation of the mechanisms that give rise to acne lesions will enable better targeting of acne therapies.
In addition to the established pathogenic factors discussed previously, there may be other triggers or exacerbating conditions that contribute to acne vulgaris. These other factors are less well understood and in some cases controversial. Stress appears to be a common trigger for acne, possibly via increased activation of the hypothalamic–pituitary–adrenal axis and the resultant increase in androgen production. Acute worsening of acne has been documented in college students during examination periods and correlated with an increased perceived stress score. Mechanical factors such as skin occlusion from sports gear (i.e., helmets, chin straps, shoulder pads) may exacerbate the condition. Applications topically applied to hair and skin, especially pomades and greasy ointments, may contribute to physical obstruction of the pilosebaceous unit and worsen the disease. Several medications may worsen acne, including anabolic steroids, progestins, lithium, isoniazid, hydantoin, and gold. Pathologic androgen excess is often associated with acne (see later), and although endocrinologic evaluation is not indicated for the majority of patients, it should be considered in patients who have additional signs as listed in Box 8.2 . Individuals with moderate to severe keratosis pilaris appear to have lower prevalence and severity of facial acne vulgaris.
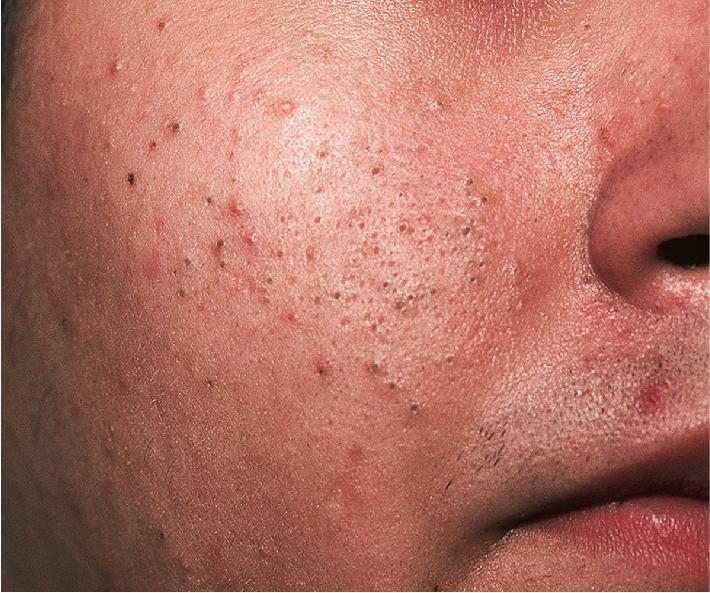
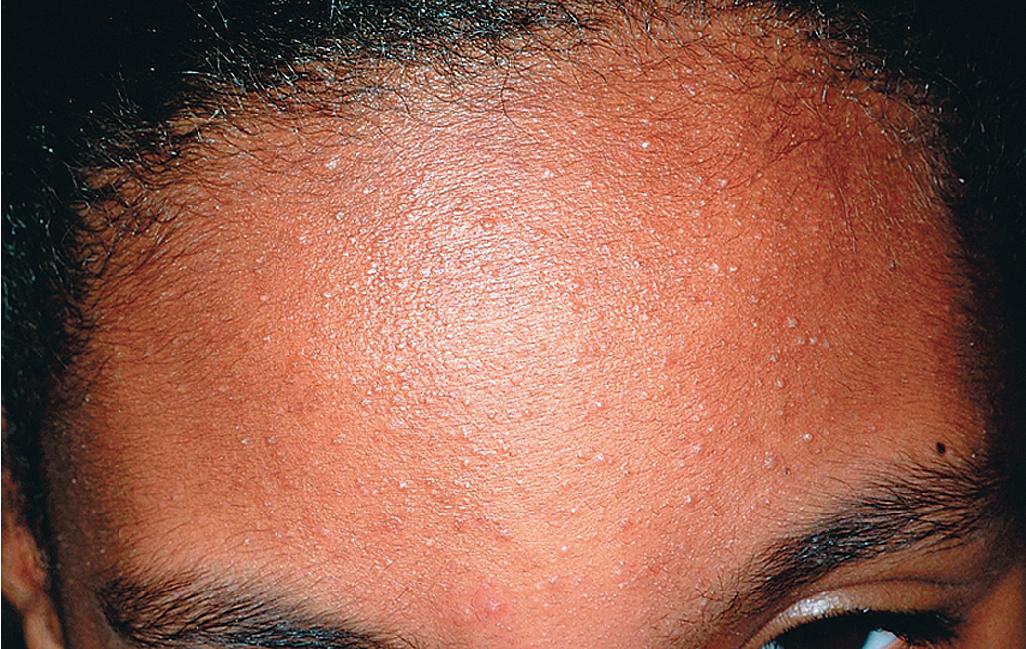
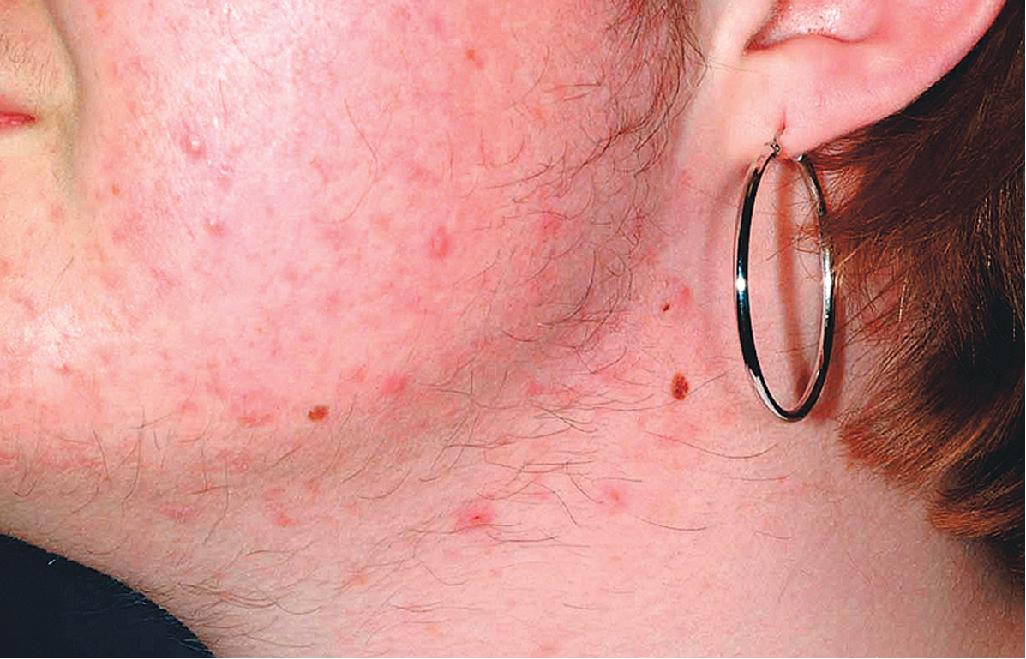
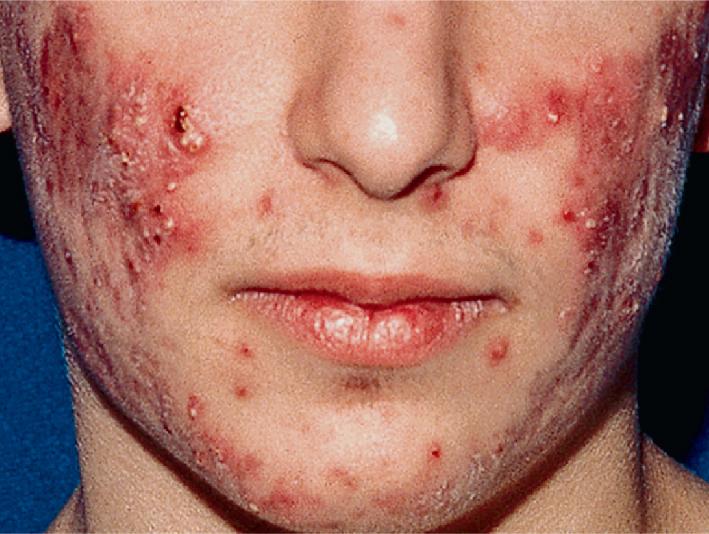
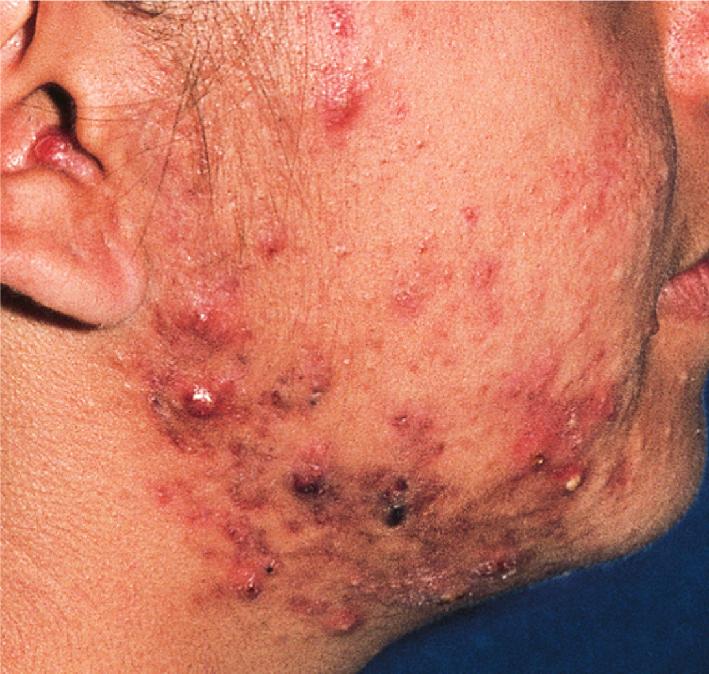
The role of diet in the pathogenesis of acne vulgaris remains controversial, and controlled studies have refuted the value of dietary restrictions. For many years the elimination of various foods such as chocolate, soft drinks, milk, ice cream, fatty foods, shellfish, and iodides was recommended. The misconception that iodine is injurious to patients with acne vulgaris originated with the concept that iodides administered orally as a medication occasionally initiate a papulopustular acneiform eruption. The concept that chocolate exerts an adverse effect on acne has been challenged, and one controlled double-blind study found that it failed to affect either the course of the disease or the production and composition of sebum. However, the possibility that diet plays a role in the pathogenesis or exacerbation of acne continues to be addressed. A review of two non-Westernized populations (one in Papua New Guinea and the other in Paraguay) revealed an astonishing difference in acne prevalence compared with individuals in Westernized societies. One potential explanation asserted for these differences is the diets of these populations, which are composed mainly of minimally processed plant and animal foods and are nearly devoid of the carbohydrates typical in Western diets, which may result in high insulin levels. Hyperinsulinemia, in turn, may promote the development of acne by its ability to increase androgen production. It remains unclear whether adhering to a diet with a low glycemic load (LGL) can effect a change in acne, although several studies seem to suggest an association. Studies have suggested increased sex hormone–binding globulin, reduced androgen levels, and reduced acne lesion development in young men who ate an LGL diet, although the associated weight loss in this cohort and the lower saturated fat content and higher fiber level in the LGL diet are considered by some as potential confounders. , In a cross-sectional study of 50 nonobese adults with acne vulgaris, high glycemic index and high glycemic load diet was positively associated with acne vulgaris, and an inverse correlation between glycemic index and adiponectin (a hormone derived from subcutaneous fat that exhibits antiinflammatory effects) was noted. The association between milk consumption and development of acne has been explored, with several studies (some of which were limited by their retrospective and/or questionnaire-based design) suggesting a positive association, possibly related to a combination of exogenous hormones and growth factors and stimulation of endogenous hormones. Interestingly, skim milk was consistently associated with acne in most (but not all) of these studies, suggesting that fat content alone may not explain the acne-causing ability of milk in the studied cohorts. , Other dietary factors that have been studied with regard to acne pathogenesis include omega-3 fatty acids, antioxidants, zinc, vitamin A, and dietary fiber. , Acne presents with a combination of the various lesion types previously discussed: comedones, papules, pustules, cysts, nodules, scarring, and dyspigmentation. The earliest lesion type is usually the comedone, and mild acne may be limited to these lesions ( Figs. 8.3 and 8.4 ). With increasing severity, patients develop inflammatory lesions, including papules and pustules ( Figs. 8.5 and 8.6 ). Nodules and cyst-like nodules may be present in patients with moderate to severe disease ( Figs. 8.7 and 8.8 ) and may result in permanent acne scarring ( Figs. 8.9 and 8.10 ). Dyspigmentation is a common sequela, especially in patients with darker complexions ( Figs. 8.11 and 8.12 ). The primary sites for acne lesions correlate with the areas most concentrated with sebaceous glands: the face, chest, back, and shoulders. In patients with regressing acne lesions, macular erythema ( Fig. 8.13 ) and hyperpigmentation are commonly seen. Although patients commonly regard these as active lesions and “scars,” they usually imply effectiveness of the therapy, and their temporary nature should be explained to the patient. The scarring that occurs in patients with the more severe papulopustular and nodulocystic forms of acne develops because of fibrous contraction after the inflammatory phase. In addition to sharply punched-out pits and craters, hypertrophic or keloidal scars may also develop. The severity of the scarring depends on the depth and degree of the inflammation and on the patient’s individual susceptibility.
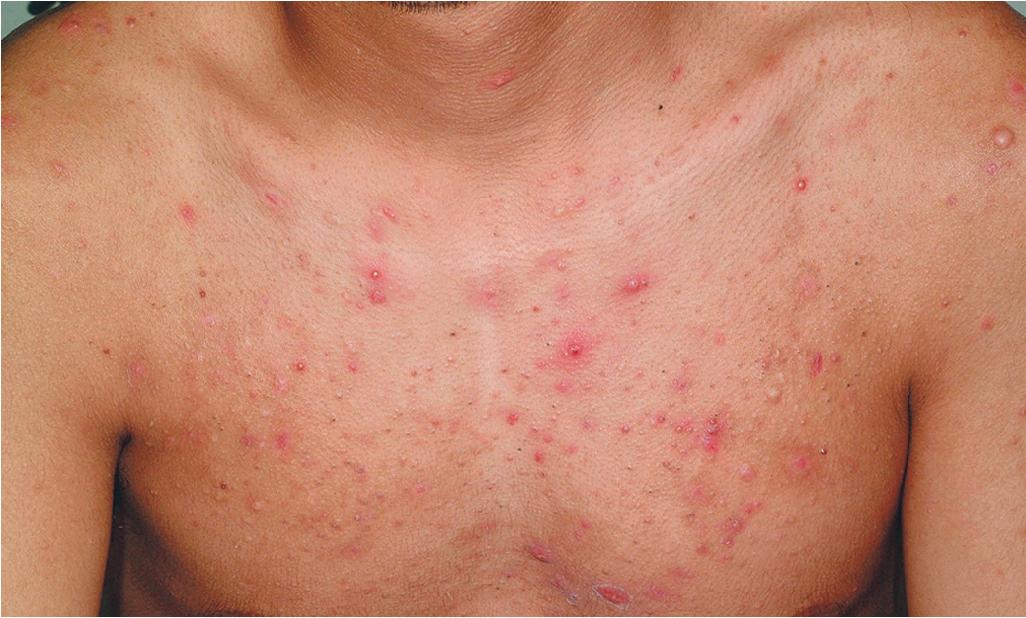
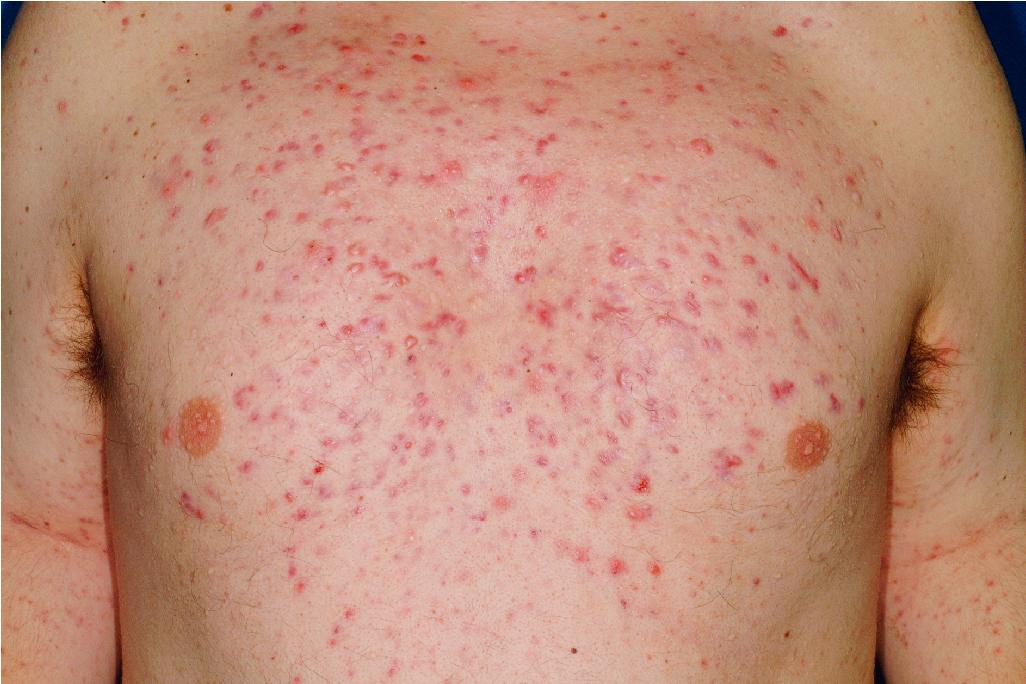
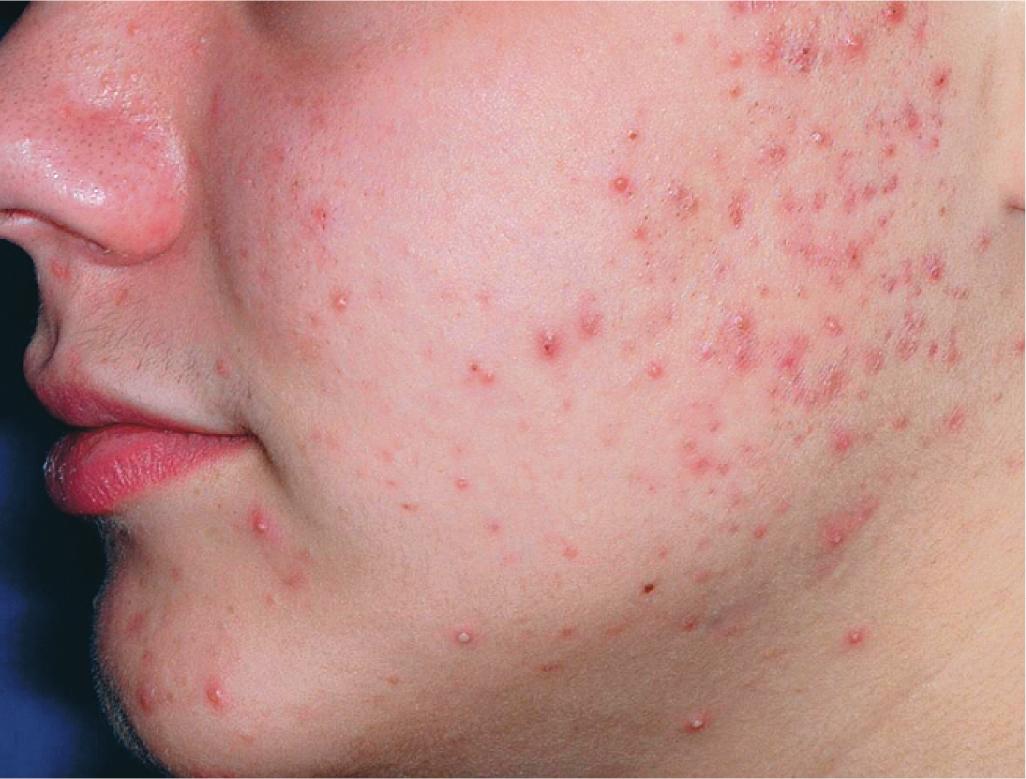
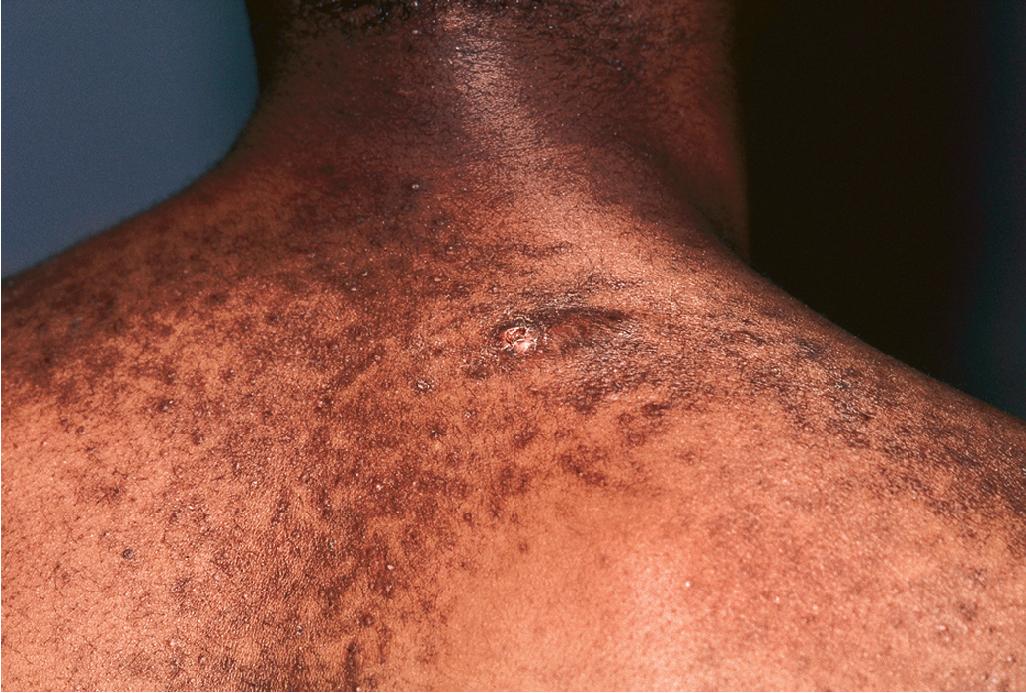
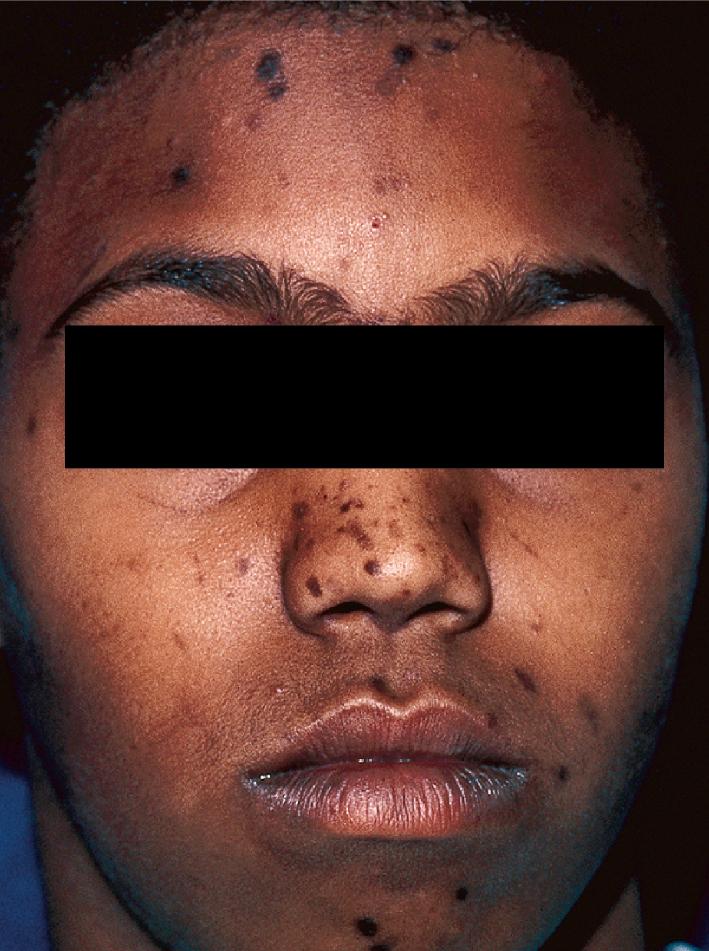
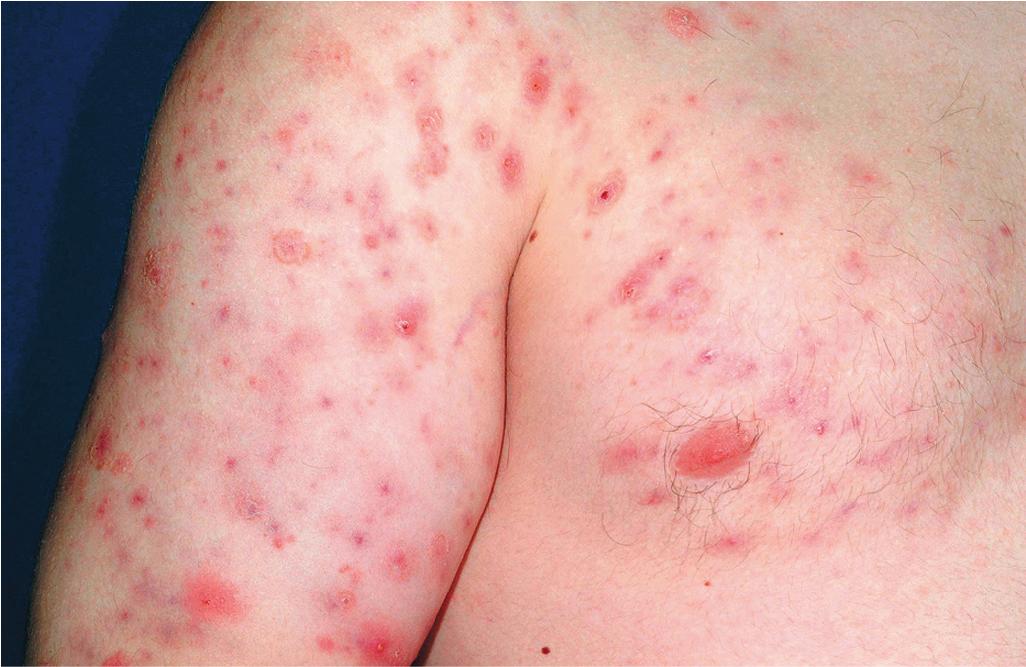
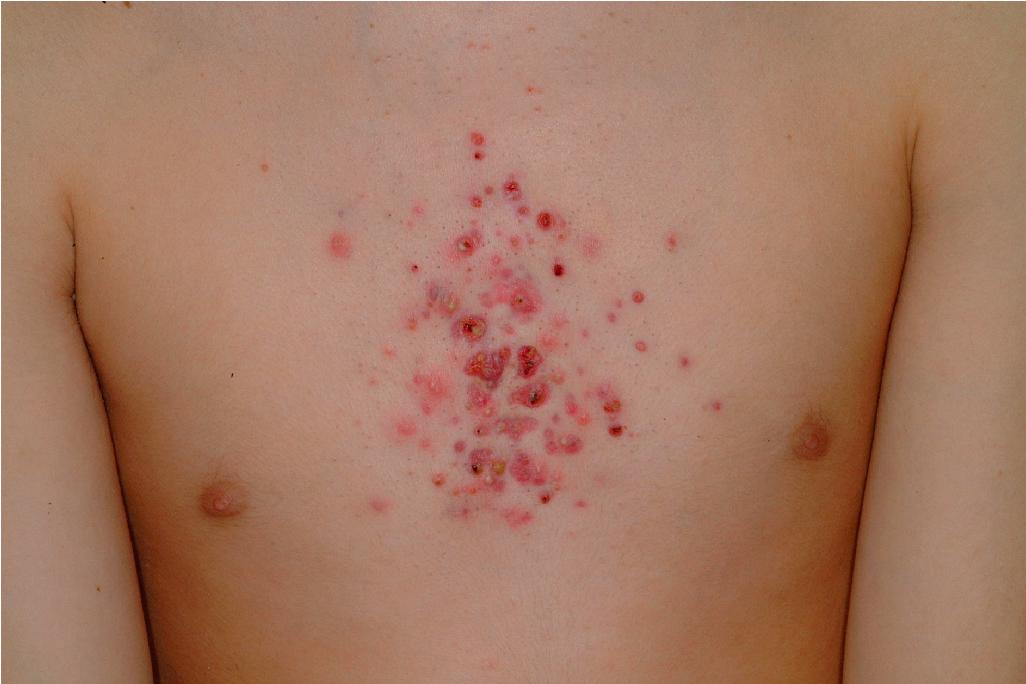
Acne fulminans is a rare and severe form of acne vulgaris, occurring primarily in boys and young men. Patients present with an acute onset of painful, pustular, nodular, and occasionally ulcerative acne lesions concentrated on the chest ( Fig. 8.14 ), shoulders, back, and face. Fever, leukocytosis, polyarthralgia, and elevation in inflammatory markers may also be present, and the skin lesions usually result in permanent scarring. Osteolytic bone lesions may be present, especially in the clavicle, sternum, and long bones. Ironically, acne fulminans has most often been ascribed to treatment with oral isotretinoin. Systemic corticosteroids are the mainstay of therapy, with the addition of isotretinoin after the acute inflammatory stage has subsided. A recent acne fulminans consensus treatment guideline recommends temporary discontinuation of isotretinoin while oral corticosteroids are started, with reintroduction at a very low dose (0.1 mg/kg per day) once lesions begin to heal over, with very gradual isotretinoin dose escalation. Other reported therapies include etanercept, infliximab, anakinra, cyclosporine, and dapsone.
Before any attempt is made to treat acne, patient education is vital. Important considerations include explaining the nature and natural history of acne vulgaris, dispelling acne myths regarding causes and treatments, highlighting the need for adherence with therapy and appropriate follow-up care with the physician, reviewing the correct usage of medications and the potential side effects, and emphasizing the expected gradual improvement in symptoms with therapy. Patient education materials are very useful to this end and will help increase adherence with treatment. Such materials can be created and personalized by the physician or purchased from national organizations such as the American Academy of Dermatology ( www.aad.org ) or the American Academy of Pediatrics ( www.aap.org ). Box 8.3 lists some clinical pearls for increased adherence in acne therapy.
Use written action plans for topical and systemic medication use.
Keep the treatment regimen as simplified as possible; use of fixed-dose combination agents (when possible) may facilitate this goal.
Consider the vehicles of prescribed agents and tolerability by the individual patient.
Take time to educate the patient regarding expectations (length of time to see improvement, likelihood of mild irritation early in the course of therapy, potential side effects).
Review appropriate application of topical medications.
Reinforce the importance of ongoing use (maintenance therapy), once improved.
Consider psychologic comorbidities and encourage open discussion regarding acne-associated stressors.
Acne treatment in the past was hindered by mythical concepts regarding its causes, a more limited armamentarium of treatment options, and a lack of appreciation for the dire psychosocial consequences that can result for the acne patient. With the current level of understanding of acne pathogenesis, the ever-expanding array of therapeutic approaches, and recognition of both the physical and emotional benefits of successful therapy, every patient with acne who desires treatment should be able to benefit from physician intervention. The primary care provider is optimally situated to offer therapy to patients with mild and moderate disease, and case-based education has been shown to significantly increase pediatric provider knowledge on treating acne in accordance with published evidence-based recommendations. For those with recalcitrant or severe involvement, referral to a dermatologist is often necessary. Although acne therapy is a combination of art and science, a solid understanding of the various classes of medication and their proper use in both monotherapy and combination therapy provides the foundation for designing treatment plans. A conceptual framework is shown in Table 8.1 for an acne treatment algorithm. Contemporary treatment algorithms are also available in the published literature.
| Therapy | SEVERITY (LESION TYPE) | ||||
| Mild (Comedonal) | Mild (Inflammatory/Mixed) | Moderate (Inflammatory/Mixed) | Severe (Inflammatory/Mixed) | Severe (Nodular/Scarring) | |
| Initial therapy options * , † | Topical retinoid BP Salicylic acid cleanser |
BP/retinoid combo BP/antibiotic combo Antibiotic/retinoid combo + BP |
BP/retinoid combo BP/antibiotic combo ± topical retinoid Antibiotic/retinoid combo + BP ± oral antibiotic |
BP/retinoid combo + oral antibiotic BP/antibiotic combo + topical retinoid + oral antibiotic Antibiotic/retinoid combo + BP + oral antibiotic |
Isotretinoin |
| Alternative therapy options * , † , ‡ | Add BP or retinoid if not already prescribed BP/antibiotic combo BP/retinoid combo Antibiotic/retinoid combo |
Substitute another combo product Add missing component (e.g., topical retinoid, BP, topical antibiotic) Change type, strength or formulation of topical retinoid |
Substitute another combo product Add missing component (i.e., topical retinoid, BP, topical antibiotic, oral antibiotic) Change type, strength or formulation of topical retinoid Consider hormonal therapy § for female patients Consider oral isotretinoin |
Consider changing oral antibiotic Consider isotretinoin Consider hormonal therapy § for female patients |
Consider hormonal therapy § for female patients |
| Maintenance therapy | Topical retinoid or BP/retinoid combo | Topical retinoid or BP/retinoid combo | Topical retinoid or BP/retinoid combo | Topical retinoid or BP/retinoid combo | Topical retinoid or BP/retinoid combo |
* If combination products not available to patient, consider substitution of individual components as separate prescriptions.
† Topical dapsone may be considered in place of topical antibiotic.
‡ If needed as determined by physician assessment and patient satisfaction.
Topical therapies are first-line treatment for patients with mild acne and are very useful as part of a combination therapy regimen for patients with moderate or severe acne. Prescription-strength topical agents are summarized in Table 8.2 . Benzoyl peroxide (BP) and topical retinoids (vitamin A acids), although potential irritants, appear to be the most effective topical agents. Based on current understanding of acne pathogenesis, these two products offer a highly effective therapeutic approach that can be tailored to each patient. Although success in the management of acne vulgaris can be achieved by the use of these agents alone, the therapeutic effect can be increased substantially by their use in combination, despite the fact that they are applied at separate times.
| Drug | Brand * | Formulation |
|---|---|---|
| ANTIMICROBIALS | ||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Amzeek | 4% foam |
| Sulfacetamide |
|
|
| Azelaic acid | Azelex | 20% cream |
| RETINOIDS | ||
| Adapalene | Differin ‡ |
|
| Adapalene/BP † |
|
|
| Tazarotene | Tazorac |
|
| Fabior | 0.1% foam | |
| Tretinoin | Retin-A |
|
|
|
|
| Tretinoin/clindamycin † |
|
|
* Listed are examples; this list is not exhaustive. Many of these preparations are available in generic form as well.
Daily facial cleansing, preferably in the morning and at bedtime, is a useful step in acne management. The goal of cleansing is to remove excess oil, dirt, and makeup. Use of a gentle, soap-free, pH-balanced cleanser or acne wash is recommended. It is important that the acne patient be appropriately educated that harsh scrubbing and use of abrasive devices or sponges may increase inflammation and actually worsen acne or increase the risk of scarring.
BP and salicylic acid are the active ingredients in most over-the-counter (OTC) acne products. In fact most BP acne products are now limited to OTC status, excepting some prescription combination products. Salicylic acid helps reduce sebum and decrease comedones and is found in various concentrations as a gel, lotion, wash, or cream. It may be an alternative comedonal therapy for those who cannot tolerate topical retinoids (now considered the mainstay of comedonal acne therapy). Glycolic acid, one of the α-hydroxy acids, is found naturally in fruits and yogurt and may be useful in decreasing hyperkeratosis associated with acne.
BP is traditionally the most commonly used topical preparation for acne vulgaris and, as noted earlier, is available as an OTC product and in some select prescription (primarily fixed-dose combination) products. It is a powerful antimicrobial, useful for reducing colonization of C. acnes, and also has some comedolytic and antiinflammatory effects. BP is available in a variety of strengths and comes as a gel, wash, cream, lotion, and even shaving cream. It is usually used once or twice daily. BP products may bleach colored clothing or linens, and patients should be warned of this possibility. Combination products of BP with topical antibiotics (erythromycin or clindamycin) are readily available, and these agents appear to be more effective than either ingredient used alone. Combination BP/topical retinoid products are also available. These agents are discussed in more detail in the paragraphs that follow.
Potential side effects of BP include irritant contact dermatitis and rarely allergic contact dermatitis. , Irritant dermatitis is particularly problematic when the product is used excessively or in association with abrasive soaps or astringents or both. Therapy must be individualized and initiated gradually, particularly in fair-skinned or atopic individuals. BP should be applied as a thin film and rubbed in gently, gradually increasing the frequency and strength of the preparation as tolerance is developed (generally over several weeks). As with most topical agents, a small portion of the product is generally sufficient for application to the entire face. If irritation or excessive dryness develops, the preparation may be discontinued for several days and then restarted more sparingly, and/or noncomedogenic facial emollients may be used (in small amounts).
Topical retinoids (see Table 8.2 ) are a very effective class of medication used in the treatment of acne. These drugs normalize the keratinization process within the follicles, which reduces obstruction and therefore the formation of microcomedones and comedones. Some topical retinoids may also have antiinflammatory activity. These agents are thus both comedolytic and anticomedogenic and, as such, function to help prevent new acne and treat existing lesions. They have also been shown to have a beneficial effect on secondary lesions, such as scarring and pigmentation. The prototype drug in this class is tretinoin, an acid of vitamin A. Newer-generation agents include adapalene and tazarotene. Tazarotene was used initially in the treatment of psoriasis and subsequently received approval for the treatment of acne vulgaris. Although success in the management of acne can often be achieved by the use of topical retinoids alone, the therapeutic effect can be substantially increased by their use in combination with BP or a topical antibiotic. A tretinoin/clindamycin combination ( Table 8.3 ; see Table 8.2 ) has been found to improve both comedonal and inflammatory acne, and an adapalene/BP combination product was found to be more effective than either agent alone and placebo.
| Active Agents | Brand(s) | Formulation |
|---|---|---|
| Clindamycin/BP | BenzaClin Duac Acanya Onexton |
1%/5% gel 1%/5% gel 1.2%/2.5% gel 1.2%/3.75% gel |
| Erythromycin/BP | Benzamycin Benzapak Actipak |
3%/5% gel 3%/5% gel 3%/5% gel |
| Adapalene/BP | Epiduo Epiduo Forte |
0.1%/2.5% gel 0.3%/2.5% gel |
| Clindamycin/tretinoin | Ziana Veltin |
1.2%/0.025% gel 1.2%/0.025% gel |
Topical retinoids are generally applied once nightly and should be applied sparingly. A useful analogy for the patient is to use a “pea-sized” or “chocolate chip–sized” amount of product for a full-face application. The traditional recommendation was for retinoids to be applied to a dry face, with 30 to 45 minutes of “air drying” allowed after washing and before applying the product. However, this appears to be unnecessary with the newer formulations of tretinoin, adapalene, and tazarotene. The primary side effect of topical retinoids is irritation, which can be minimized by appropriate application and use. In general the cream forms are less drying than the gels, solutions, or pledgets, although again, newer formulations have minimized this effect, even with the gel vehicle. In patients with sensitive skin, good initial choices for retinoid therapy are tretinoin cream (in a lower strength), adapalene cream, or one of the micro preparations. If the product is tolerated well and more effect is desired, the patient can then be advanced to higher strengths, a gel or pledget formulation, or tazarotene. Another popular strategy used to decrease the incidence of irritation is to have the patient begin therapy 3 nights weekly, increasing as tolerated over a few weeks to nightly application. Because retinoids may degrade significantly when mixed with BP, such combination therapy should be used only in the form of preformulated combination products (see Tables 8.2 and 8.3 ) or, if used as individual agents, they should be applied at different times.
Increased sun sensitivity is another potential side effect of topical retinoids, and patients should be counseled regarding the appropriate use of a sunscreen with a sun protection factor of 30 or higher, especially if they are also using a photosensitizing oral agent. The risk of teratogenicity with use of the topical retinoids is controversial but remains a concern given the well-established teratogenic potential of the oral retinoid, isotretinoin. There are sporadic reports of congenital malformations in infants born to women who used topical tretinoin during pregnancy, but controlled human studies are lacking for this drug, which is classified as a pregnancy category C (studies in animals have revealed adverse effects on the fetus and there are no controlled studies in women or studies in women and animals are not available), as is adapalene (tazarotene, however, is classified as pregnancy category X). The limited transdermal uptake, lack of alteration of plasma retinoid levels, epidemiologic data, and margin of safety when compared with known teratogens all suggest the unlikelihood of tretinoin being a human developmental toxicant. However, until a formal consensus exists, the potential risks of topical retinoids should be discussed in detail with women of childbearing potential, and their use should probably be avoided during pregnancy.
Topical antibiotics (see Table 8.2 ) exert their effect primarily by decreasing the population of C. acnes , and they may also have antiinflammatory effects. The most commonly used topical antibiotics for acne are clindamycin, erythromycin, and sulfacetamide. These agents are most useful for inflammatory acne, and in patients with mixed inflammatory and comedonal disease they should be used in combination with another agent that has comedolytic properties (e.g., BP or retinoid). These agents are usually well tolerated and are applied once or twice daily. As with other topical agents, lotions and creams are less drying than solutions, pledgets, and gels. Monotherapy with topical antibiotics should be discouraged, given the greater development of bacterial resistance, which can be lessened by the concomitant use of a preparation containing BP (see later).
Topical erythromycin has been used for decades in the treatment of acne and is usually well tolerated. The addition of zinc to erythromycin may increase its therapeutic efficacy. Topical erythromycin has fallen out of favor with many acne experts given its reduced efficacy compared with clindamycin, secondary to resistance of cutaneous organisms. Topical clindamycin is another well-tolerated antibiotic preparation for acne. Although pseudomembranous colitis is usually associated with oral clindamycin, there are rare reports of this complication with topical application of this agent. , Therefore if a patient using topical clindamycin develops persistent diarrhea, this rare complication should be considered. Topical sulfonamide preparations (mainly sulfacetamide) are also available by prescription and may be particularly useful in patients who also show evidence of rosacea. These medications are often well accepted, although some patients may find their odor offensive. Patients with allergies to oral sulfa drugs should not use topical sulfonamides. Topical dapsone, a newer arrival in the acne antibiotic market, appears to be most effective against inflammatory lesions, although its mechanism may not be related to C. acnes reduction. Topical dapsone may result in clinically insignificant mild hemolysis in patients with glucose 6-phosphate dehydrogenase deficiency.69 More recently, minocycline foam has been approved as another topical option.
The landscape of topical acne therapy has evolved significantly in recent decades, with the increasing availability of fixed-dose combination therapies (see Table 8.3 ). These agents are approved for mild to moderate acne vulgaris, and they offer the advantages of improved adherence and complementary mechanisms of action of the individual active ingredients. Available fixed-dose combination products include those combining BP and clindamycin, BP and adapalene, clindamycin and tretinoin, and BP and erythromycin. Each of these products, to receive US Food and Drug Administration (FDA) approval, was demonstrated to have superior efficacy over placebo as well as the individual active ingredients (“monads”) in large, multicenter clinical trials.
With increasing use of topical antibiotics for the treatment of acne vulgaris, the development of resistant strains of C. acnes is being observed. Cross-resistance of C. acnes to erythromycin and clindamycin is now widespread. In one study, erythromycin-resistant organisms were isolated from 51% of patients treated with oral erythromycin but also from 42% of patients treated with topical clindamycin. Antibiotic-resistant propionibacteria may be transmissible between acne-prone individuals as well as between patients and their physicians. Data suggest that the concomitant use of BP with topical (as well as oral) erythromycin or clindamycin diminishes the emergence of resistant bacterial strains. , ,
Azelaic acid is a naturally occurring dicarboxylic acid produced by the fungal organism Pityrosporum ovale . It has antibacterial and anticomedonal properties and is felt by some to be equal in efficacy to BP or tretinoin. The effectiveness of azelaic acid may be increased by using it in combination with other topical medications such as retinoids, antibiotics, or BP. Another benefit of this agent is its ability to decrease hyperpigmentation caused by acne. Azelaic acid is marketed as a 20% cream and is applied twice daily. Side effects are rare but include burning, tingling, mild erythema, and pruritus.
Physicians are commonly consulted regarding the safety of acne treatment in women who are pregnant, may become pregnant, or are breastfeeding an infant. Although there are no long-term studies on the use of topical antibiotics during pregnancy, erythromycin seems to be safe. Because it is estimated that approximately 8% of topically applied clindamycin can be absorbed, this drug is probably best avoided for women who are breastfeeding, are pregnant, or are contemplating pregnancy. Although approximately 5% of topically applied BP is absorbed through the skin, it is rapidly metabolized to benzoic acid, enters the dermal blood vessels as benzoate, and is then transported to the kidneys and excreted in the urine. Thus BP appears to be safe during pregnancy, although controlled studies are lacking. The use of tretinoin is discussed earlier in this section.
Oral antibiotics ( Table 8.4 ) are the most common form of systemic therapy used in the treatment of acne vulgaris, despite the fact that the only agent with FDA-approval for this indication is extended-release minocycline. These agents are quite effective and are best reserved for patients with moderate or severe inflammatory acne. They are useful in decreasing C. acnes, free fatty acids found in sebum, and inflammation (via their inhibitory effect on neutrophil chemotaxis). Oral antibiotics generally require 4 to 8 weeks to achieve their maximum effect, and it is usually necessary to continue therapy for several months, with gradual tapering as tolerated once the disease activity has diminished. The most commonly used antibiotics for treating acne are the tetracycline-class drugs (tetracycline, minocycline, doxycycline, and, more recently approved, sarecycline) and erythromycin. Less commonly, clindamycin, trimethoprim-sulfamethoxazole, azithromycin, and cephalexin are used. These medications are usually given on a twice-daily basis.
| Drug | Usual Dosage * | Comments/Side Effects |
|---|---|---|
| COMMONLY USED | ||
| Tetracycline | 250–500 mg | Dental staining <9 years Dairy products decrease absorption GI upset, photosensitivity, teratogenic, PTC, VVC, IBD |
| Minocycline | 50–100 mg 55, 65, 80, 105, 115 mg ER (1 mg/kg once daily) |
Dental staining <9 years Dairy products decrease absorption Vertigo (lower incidence with ER), GI upset, blue-gray skin pigmentation, severe drug reactions with hepatitis/pneumonitis, lupus-like reactions, SJS, teratogenic, hepatitis, PTC, VVC, IBD |
| Doxycycline | 50–100 mg 75, 100, 150 mg ER, once daily 20 mg (“subantimicrobial dose”) |
Dental staining <9 years Dairy products decrease absorption Photosensitivity, photoonycholysis, GI upset (rare), teratogenic, PTC, VVC, IBD |
| Erythromycin | 250–500 mg | GI upset (common), VVC, drug–drug interactions, prolongation of the QT interval; no longer recommended by most experts given increased resistance |
| LESS COMMONLY USED | ||
| Trimethoprim-sulfamethoxazole | 80/400 mg, 160/800 mg | Severe drug reactions, SJS, bone marrow suppression, hepatitis, GI upset, VVC, fixed drug eruption; routine use for acne strongly discouraged |
| Azithromycin | Varied; often pulse dosing | GI upset, occasional drug reactions |
| Clindamycin | 75–150 mg | Pseudomembranous colitis, GI upset, drug reactions, VVC |
| Cephalexin | 250–500 mg | GI upset, drug reactions, VVC |
* Usually given twice daily for acne unless otherwise noted in table.
The safety of long-term antibiotic therapy for acne has been a concern of patients, parents, and physicians. Although the risk-to-benefit ratio of the most commonly used medications is favorable, each agent carries its own inherent potential toxicities, some of greater concern than others. The development of C. acnes resistance has been demonstrated for most of the agents used in this setting, especially erythromycin and clindamycin, in which case cross-resistance patterns may emerge. , General guidelines for oral antibiotic use in acne include avoiding the oral agent if topical agents will suffice, continuing treatment for no longer than is necessary, reusing the same drug when possible (if restarting oral therapy is required), and avoidance of concomitant oral and topical treatment with dissimilar antibiotics. In addition, it is recommended that treatment be limited to the shortest possible duration, with reevaluation in 3 to 4 months, in an attempt to minimize bacterial resistance and that concomitant therapy with BP and/or a retinoid be used and continued for maintenance after completion of systemic therapy. , In patients who do not respond to oral antibiotics, the possibility of resistant strains should be considered. Overall, the commonly used oral antibiotics for acne are well tolerated, side effects are uncommon, and routine laboratory testing is unnecessary. Concomitant use of antibiotics and oral contraceptive agents may result in decreased efficacy of the latter, and this possibility should be discussed with female patients.
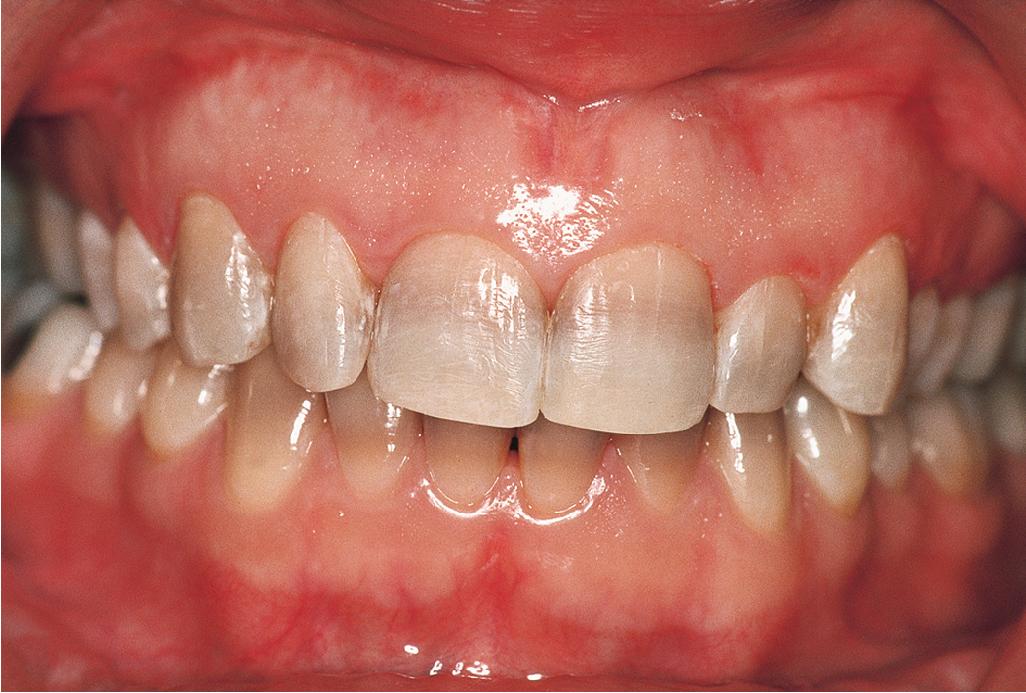
Tetracycline-class antibiotics (especially doxycycline and minocycline) are the gold standard of oral antibiotic therapy for acne. These agents have a long track record of safety and efficacy, although side effects are possible. The possibility of dental staining ( Fig. 8.15 ) in children younger than 9 years of age precludes their use in this population, who only occasionally have moderate to severe acne. Tetracycline-class antibiotics should also not be used in pregnant women, given the deposition of the drug in developing teeth as a result of its chelating properties and the formation of a tetracycline–calcium orthophosphate complex.
The most common side effects of the tetracyclines are photosensitivity and gastrointestinal upset. The risk of phototoxicity is greatest with tetracycline and doxycycline and extremely rare with minocycline. Pseudotumor cerebri is a rare side effect, the risk being greatest when isotretinoin and a tetracycline are taken concomitantly. Tetracyclines cannot be taken with dairy products or antacids, which impair absorption. In addition, tetracycline (but not doxycycline or minocycline) absorption is impaired in the presence of food, and thus the medication must be taken 1 hour before or 2 hours after a meal. This feature may result in decreased adherence among active teenagers. Tetracyclines should be used with caution in patients with renal disease.
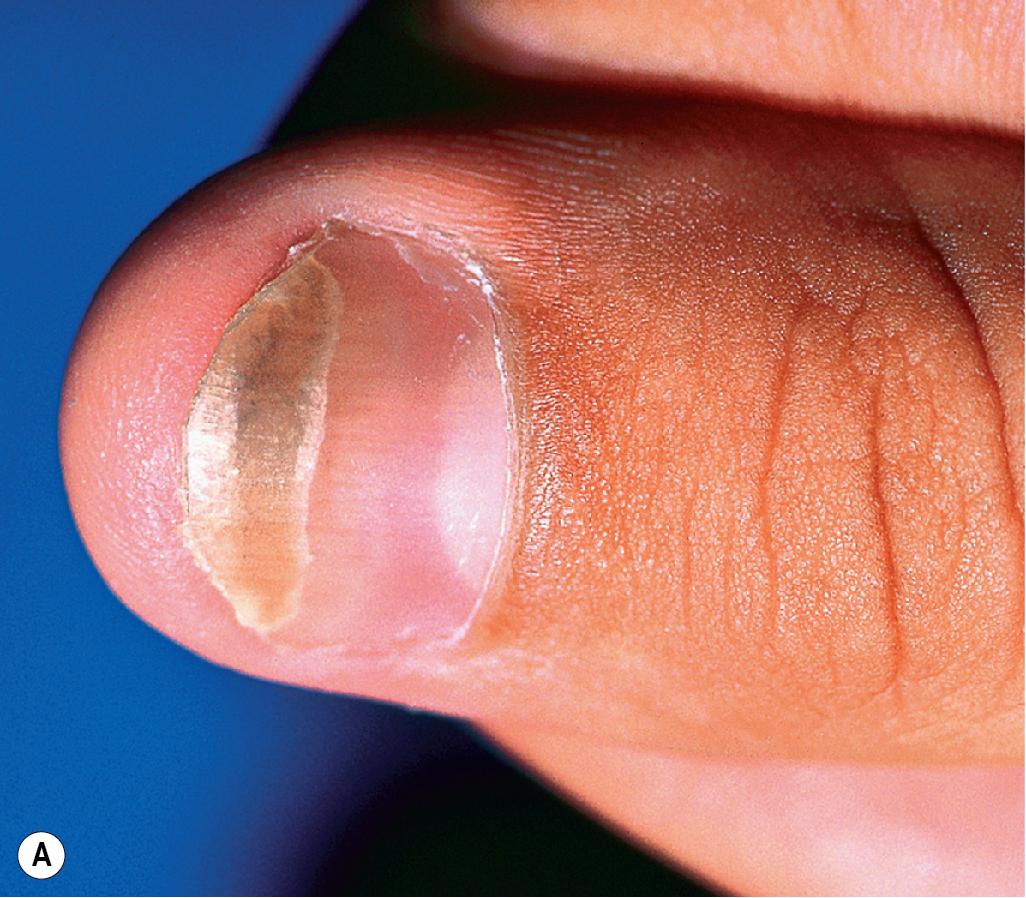
Doxycycline has the benefit of sustained absorption in the presence of food but may result in severe photosensitivity reactions. One such type of reaction is “photoonycholysis,” which presents with erythema and separation of the nail plate from the nailbed ( Fig. 8.16 ). These patients usually also have an associated photosensitivity rash on exposed areas of skin. Pill esophagitis is another potential side effect of oral doxycycline and can usually be prevented by taking the medication with a full glass of water and avoiding recumbency for at least 1 hour after dosing. In a retrospective cohort study of patients with acne who were treated with oral tetracycline-class antibiotics, the hazard ratio (HR) for developing inflammatory bowel disease (IBD) was increased for all agents in this class but most notably for doxycycline (HR of 1.63). However, a definitive causal link between tetracycline-class antibiotics and development of IBD remains to be proven. Off-label use of subantimicrobial-dose doxycycline hyclate (20-mg tablets taken twice daily) has been reported in acne vulgaris and has been shown to significantly reduce the number of lesions without any detectable antimicrobial effect on skin flora or alteration of resistance patterns. An extended-release formulation of doxycycline is available and can be administered once daily.
Minocycline is the most widely prescribed systemic antibiotic for the management of acne in the United States, Canada, and the United Kingdom, and it is estimated that 65% of prescriptions for this drug are for acne. It is particularly effective against C. acnes and also has antiinflammatory properties. Similar to doxycycline, an extended-release formulation of minocycline is available and may improve adherence as well as decrease the incidence of vestibular side effects. Although the side effect profile is somewhat similar to that of other tetracyclines, minocycline rarely causes photosensitivity. Other side effects more specific to minocycline are headache, dizziness, vertigo, and a blue-gray pigmentation of mucosae (see Fig. 11.64 A ) or at sites of previous inflammation such as acne scars.
Occasional yet potentially serious toxicities may be associated with minocycline, including autoimmune hepatitis, drug-induced lupus (DIL), serum sickness–like reaction (SSLR), and drug reaction with eosinophilia and systemic symptoms (DRESS). DRESS (see Chapter 20 ) is characterized by a diffuse erythematous rash, facial (especially periorbital) edema, lymphadenopathy, fever, and internal organ involvement (most commonly the liver). It tends to occur within 6 to 8 weeks of starting the drug and is similar to the hypersensitivity reaction occasionally seen with the aromatic anticonvulsants (phenytoin, carbamazepine, phenobarbital, lamotrigine). SSLR (see Chapter 20 ) presents with fever, urticarial rash, periarticular swelling with arthralgia, and occasional nephritis and lymphadenopathy. A comparative study reviewing adverse events of tetracycline, minocycline, and doxycycline revealed that these reaction patterns (DIL, SSLR, and DRESS, also known as drug hypersensitivity reaction or drug hypersensitivity syndrome ) are all more common with minocycline than with the other two medications. Also of note is the fact that SSLR and DRESS usually occur within 2 months of beginning treatment, whereas DIL may be delayed for up to 2 years. Like doxycycline, minocycline use has been linked to a possible increased risk of IBD, but this link remains definitively unproven.
Autoimmune hepatitis, DIL, arthritis, vasculitis, and development of autoantibodies have been unified under the moniker of minocycline-induced autoimmunity (MIA). A study of 27 children with MIA revealed a mean duration of treatment of 13 months before the diagnosis. The majority of patients had polyarthralgia or polyarthritis, mostly of the hands and feet. Other features included constitutional symptoms, livedo reticularis, Raynaud phenomenon, elevation of liver enzymes, and elevated antinuclear antibody titers. Importantly, 26% of patients had chronic autoimmunity, manifested mostly as persistent arthritis. Many patients with minocycline-associated DIL have no skin eruption. Polyarteritis nodosa has been reported, and positivity for antinuclear antibody and perinuclear antineutrophil cytoplasmic antibody supports the diagnosis. , Antihistone antibodies, traditionally considered pathognomonic for DIL, are often negative in patients with minocycline-associated disease. In a study of a cohort of patients with minocycline-induced liver injury, 80% of whom were female with a median age of 19 years, a markedly increased carrier frequency (compared with population controls) for human leukocyte antigen (HLA)–B*35:02 was found, suggesting increased risk in those carrying this HLA allele.
Although these major types of tetracycline class drug reactions are rare overall, their possibility should be discussed with the patient, especially when minocycline is being considered. Many experts consider doxycycline to be a safer alternative, even in patients who have a history of MIA.
Erythromycin was traditionally a useful antibiotic for acne vulgaris, but its use is often limited by the fairly high incidence of gastrointestinal disturbance, even with the enteric-coated preparations. Otherwise it has few side effects and carries the distinct benefit of no phototoxicity. However, the high prevalence of erythromycin-resistant C. acnes organisms further limits its clinical utility. Other antibiotics occasionally used for acne include clindamycin, trimethoprim-sulfamethoxazole, azithromycin, and cephalexin. Prolonged use of oral clindamycin is not desirable because of the risk of pseudomembranous colitis caused by the toxin liberated by Clostridium difficile, which is able to grow in large numbers in the intestinal tract of some patients who receive this drug. Trimethoprim-sulfamethoxazole can be very effective for acne, but it is not recommended as a first-line agent given the severe and potentially life-threatening reactions (i.e., DRESS, Stevens–Johnson syndrome, and toxic epidermal necrolysis) and bone marrow suppression that may occur.
The most common complication of antibiotic therapy in adult female patients is vaginal candidiasis, but it rarely occurs in teenagers. This complication is proportionately more common in women who take combined oral contraceptives (COCs) concomitantly with their systemic antibiotics. Another concern in patients who are also taking COCs is the possibility of the oral antibiotic diminishing the effectiveness of the contraceptive. Although this possibility remains controversial, it should be discussed with patients and alternative forms of birth control considered during the period of treatment. Patients on long-term antibiotic therapy may rarely develop a condition termed Gram-negative folliculitis, caused by superinfection of the pilosebaceous units with Gram-negative organisms. This condition should be considered in the acne patient presenting with a pustular eruption and who has a history of antibiotic treatment for 3 to 6 months. It is manifested by a pustular folliculitis (often with lesions situated around the nose and mouth) or deep nodulocystic lesions topped by pustules and recalcitrant to the acne regimen. The organisms causing Gram-negative folliculitis include Escherichia coli , Pseudomonas aeruginosa , Serratia marcescens , Klebsiella spp., and Proteus mirabilis . It may be related to impaired host responses in cell-mediated or humoral immunity. Gram-negative folliculitis may respond to a change in antibiotics to reduce Gram-negative organisms, but it responds more consistently to isotretinoin. ,
Hormonal therapies are another option for the treatment of acne in female patients. The primary goal of these therapies is to oppose the effects of androgens on the sebaceous glands. Hormonally driven acne may have some distinguishing clinical features, including larger and deep-seated inflammatory nodules in the mandibular and anterolateral neck regions, menses-associated flares, and associated signs of hyperandrogenism such as hirsutism and menstrual irregularities. If polycystic ovary syndrome (PCOS) is suspected, initial laboratory testing should include serum total and free testosterone, DHEA sulfate, and luteinizing hormone and follicle-stimulating hormone levels, as well as the 17-hydroxyprogesterone level if late-onset congenital adrenal hyperplasia is suspected. Abnormal findings should prompt referral to an endocrinologist.
Hormonal acne therapies include androgen receptor blockers, adrenal androgen production blockers, ovarian androgen production blockers, and enzyme inhibitors, and they may be prescribed in consultation with a gynecologist or endocrinologist. This group of medications may have other beneficial effects on the hirsutism, androgenetic alopecia, and menstrual irregularities in some female patients. COCs contain both an estrogen and a progestin, and those with a lower intrinsic androgenic activity type of progestin (desogestrel, norgestimate, and drospirenone) are most desirable for acne therapy. Drospirenone, a novel progestin, has both antiandrogenic and antimineralocorticoid activity and is reportedly better tolerated in terms of weight gain and mood changes. Norgestimate and ethinyl estradiol (Ortho Tri-Cyclen); norethindrone, ethinyl estradiol, and ferrous fumarate (Estrostep Fe); drospirenone and ethinyl estradiol (Yaz); and drospirenone, ethinyl estradiol, and levomefolate calcium (Beyaz) are FDA-approved for use in the treatment of acne vulgaris. COCs may be the first-line treatment in female patients with skin manifestations of hyperandrogenemia. Risks of COCs include venous thromboembolism, myocardial infarction, and ischemic or hemorrhagic stroke, although these risks seem to be more clinically significant in patients with other risk factors such as tobacco use, diabetes mellitus, and hypertension. Box 8.4 lists contraindications to treatment with COCs. Antiandrogenic agents include spironolactone, flutamide, cyproterone acetate, gonadotropin-releasing hormone antagonists, and 5-α-reductase inhibitors. Spironolactone, which is used primarily as a diuretic in the treatment of hypertension, competes for androgen receptors on target cells and inhibits 5-α-reductase. It is usually started at 25 to 50 mg/day, and maintenance doses, which vary by the individual, range from 25 to 200 mg/day. Potential side effects of spironolactone therapy include menstrual irregularities (in as many as half of treated women), nausea, dizziness, polyuria, and hyperkalemia, although the latter has been shown to be rare in otherwise healthy women. , In a study of 27 women with severe acne who were treated with spironolactone and a combined contraceptive (drospirenone and ethinylestradiol), this combination was demonstrated to be effective and safe, without hyperkalemia or other serious side effects.
Pregnancy
Breast cancer (current or past)
Breastfeeding (<6 months postpartum)
History of the following:
Diabetes with end organ damage or if <35 years of age
Deep vein thrombosis or pulmonary embolism
Hypertension (uncontrolled)
Major surgery with prolonged immobilization
CVA
Ischemic heart disease
Valvular heart disease with high risk
Hypercholesterolemia
Migraine headache and >35 years of age (or <35 years of age and focal neurologic symptoms)
Liver disease (hepatitis, cirrhosis, liver tumor)
Smoking and >35 years of age
Known thrombogenic mutations
Systemic lupus with antiphospholipid antibodies
CVA , Cerebrovascular accident.
Isotretinoin (13- cis retinoic acid; Absorica, Amnesteem, Claravis, Myorisan, Sotret, Zenatane) is a derivative of vitamin A that is highly effective for recalcitrant nodulocystic acne, and the availability of this agent revolutionized the treatment of severe disease. Although the precise mechanism of action is unknown, its effects appear to be related to marked inhibition of sebum synthesis, lowering of C. acnes concentration, inhibition of neutrophil chemotaxis, and comedolytic and antiinflammatory effects. Isotretinoin therapy often, but not always, results in complete and permanent remission of acne vulgaris. Preteens and young teenagers seem to have the highest rate of relapse. In patients who experience a recurrence of disease, however, the flares may be milder and more responsive to therapies used before isotretinoin therapy. Repeat courses may occasionally be necessary. Although approved by the FDA for treatment of severe acne, in recent guidelines for management of acne vulgaris, the expert working group agreed that indications for isotretinoin therapy include moderate acne that is either treatment resistant or that produces physical scarring or significant psychosocial distress.
Patients usually start by taking 0.5 mg/kg per day of isotretinoin, increasing the dosage to 1 mg/kg per day, usually for around 16 to 24 weeks. A lower starting dosage is often used in patients with severe acne to prevent exacerbation. Cumulative dosing goals, ranging from 120 to 150 mg/kg for the entire course of treatment, have been advocated because they may decrease the rate of relapse. , Early in the course of isotretinoin therapy, there may be a temporary flare of lesions, and patient education regarding this possibility is vital. Although isotretinoin is truly a miracle drug for severe forms of acne, it has several potential side effects and should only be prescribed by physicians experienced in its use.
Potential adverse effects of isotretinoin are numerous and are summarized in Table 8.5 . Laboratory monitoring during therapy is typically limited to fasting serum lipid profiles and hepatic function studies (in addition to monthly pregnancy tests for female patients of childbearing potential). Recent studies have suggested that monitoring of lipid and hepatic panels may be indicated less frequently, especially if findings are normal when tested 2 months into therapy. , A risk management program, iPLEDGE ( www.ipledgeprogram.com ), was approved by the FDA and drug manufacturers and implemented in March 2006. This mandatory distribution program, which requires prescribers, patients, pharmacies, drug wholesalers, and manufacturers in the United States to register and comply, was created in an effort to prevent the use of isotretinoin during pregnancy because of its teratogenic effects (see later). Under this system, prescribers must record two negative pregnancy tests before a female patient can commence therapy with isotretinoin. The iPLEDGE program represents the most rigorous risk management program in history for such a widely prescribed drug. Overall the risk of major side effects from isotretinoin (assuming pregnancy is avoided) is quite low. Unfortunately, a retrospective cohort study found no significant decrease in fetal exposure in female patients of childbearing age compared with the prior risk management program (System to Manage Accutane-Related Teratogenicity [SMART]).
| Side Effect | Comment |
|---|---|
| Dry skin and nasal membranes | Occasional epistaxis |
| Dry eyes | Most problematic for contact-lens wearers; may develop conjunctivitis, chalazion, hordeolum |
| Cheilitis | May be quite severe |
| Photosensitivity | |
| Decreased night vision | |
| Corneal opacities | |
| Alopecia | Diffuse thinning; generally reversible but can be the start of androgenetic alopecia |
| Headache | |
| Pseudotumor cerebri | Most common with concomitant use of tetracycline-class antibiotic |
| Musculoskeletal pain | |
| Hyperostosis | Mainly seen with prolonged use |
| Elevated triglycerides, cholesterol | Rare pancreatitis |
| Hepatitis | |
| Inflammatory bowel disease | Controversial; current evidence insufficient to prove an association or causal relationship |
| Rhabdomyolysis | |
| Teratogenicity | See text for discussion |
| Depression, suicidal ideation | See text for discussion |
Isotretinoin is a known human teratogen and is a pregnancy category X drug. Major fetal malformations have been observed in exposed infants, including abnormalities of the skull, ear, eye, central nervous system, and heart. As little as one pill can have devastating consequences on the developing fetus. To qualify for therapy, females of childbearing potential must have two negative serum or urine pregnancy tests and commit to either abstinence or use of two forms of contraception, one of which must be a primary form (COC, intrauterine device, injectable or implantable hormonal birth control product, partner’s vasectomy, or tubal ligation), for at least 1 month before initiation of therapy and for 1 month after discontinuing therapy. Abstinence is most desirable for female patients on isotretinoin therapy. A signed patient consent form is required. Unfortunately, even with dedicated pregnancy prevention programs and education regarding isotretinoin-associated teratogenicity, inadequate birth control efforts continue to be practiced by some female patients being treated with isotretinoin. Ongoing fetal exposure to isotretinoin has been demonstrated to be an international problem, highlighting the need for more effective strategies that incorporate considerations for cultural differences.
The risks of isotretinoin-related depression and SI have come to the forefront in recent years. The exact association, if any, remains controversial, and arguments both supporting and refuting the potential for mood disturbance have been made. Epidemiologic data have demonstrated a lower suicide rate in 12- to 18-year-old isotretinoin users compared with the annual rate of suicide for that population in the United States. Critical literature reviews have failed to confirm an association, and although retinoid receptors are widely distributed in the brain, there is no known pharmacologic mechanism to account for isotretinoin-induced psychiatric symptoms. In one systematic literature review it was noted that studies comparing depression before and after treatment did not show a significant increase and that, surprisingly, some revealed a trend toward fewer or less severe depressive symptoms after isotretinoin therapy. A meta-analysis of the literature confirmed these observations, finding a lower prevalence of depression after isotretinoin treatment of acne, with decrease in mean depression scores. In a cross-sectional questionnaire-based study exploring the relationship of SI, mental health problems, and social functioning to acne severity among adolescents, SI was reported two to three times more often in those with increasingly severe acne compared with those with little to no acne. Mental health problems and poorer school functioning were also associated with substantial acne, and the authors concluded that adverse events such as depression and SI that have been ascribed to acne therapies may actually reflect the burden of the disease rather than a medication effect. Given these observations and others, it is recommended that isotretinoin patients (a population at greater risk for psychiatric comorbidity) be appropriately screened and monitored for symptoms of depression and SI, with prompt referral to a mental health professional if concerns arise.
The potential association between isotretinoin use and IBD, which was first suggested in the mid-1980s, continues to be debated. A case-control study of more than 8000 cases of IBD compared with nearly 22,000 controls revealed a strong association between previous isotretinoin exposure and ulcerative colitis, but not Crohn’s disease, with the risk increasing in a dose-dependent manner. In another case-control study and meta-analysis, there was no increased risk ratio for IBD in patients who had been treated with isotretinoin. In a retrospective population-based cohort study in Canada, no significant association between isotretinoin use and IBD was found, although in a secondary analysis of individuals limited to ages 12 through 19 years, a weak IBD association was noted for both isotretinoin and topical acne medications. In two other studies, including a meta-analysis and a large single-center retrospective study, there was no increased risk of IBD identified with isotretinoin exposure. , Hence, as supported by the position statement of the American Academy of Dermatology, current evidence is insufficient to prove either an association or a causal relationship between isotretinoin use and IBD in the general population. Until more is known, however, it is prudent to mention this possible association to patients and parents and to monitor patients appropriately during therapy.
It is a common notion that skin surgery or other procedures should be delayed after isotretinoin therapy, given the traditional belief that the agent impairs normal wound healing or may result in abnormal scarring. However, in a systematic review of the literature over a 35-year period, there was insufficient evidence to support delaying skin surgery, fractional ablative or nonablative laser procedures, or a variety of cosmetic procedures (manual dermabrasion, superficial chemical peels, laser hair removal) in patients either receiving or having recently completed isotretinoin treatment.
There are a variety of miscellaneous treatments for acne vulgaris, and these are listed in Table 8.6 . , Box 8.5 lists examples of therapeutic approaches based on acne type and severity.
| Treatment | Comment |
|---|---|
| Comedone extraction | Performed with comedone extractor |
| Injections | Intralesional triamcinolone injected into large cysts or nodules |
| Light therapy | Blue light (may photoinactivate C. acnes ) Red light (may have antiinflammatory effect) Combination blue-red light Intensed pulse light (IPL) Photodynamic therapy (PDT) Photopneumatic therapy |
| Laser therapy | KTP, pulsed dye laser (rarely used) |
| Resurfacing laser (including both nonablative and ablative resurfacing lasers); useful for acne scarring | |
| Dermabrasion/dermasanding | Useful for acne scarring |
| Collagen injection | Useful for acne scarring |
| Chemical peels | Useful for acne scarring and hyperpigmentation |
| Punch grafts/tissue augmentation | Useful for acne scarring |
| Trichloroacetic acid | Useful for atrophic acne scars |
| Radiation therapy | Outdated modality |
| Topical tea tree oil | Other herbal agents including oral barberry extract, oral ayurvedic compounds, gluconolactone solution |
Topical retinoid once daily, or
Topical retinoid/BP combination product once daily, or
BP wash once daily
BP cleanser daily and topical retinoid once daily, or
BP/topical antibiotic combination product in am and topical retinoid in pm, or
BP cleanser daily and topical retinoid/topical antibiotic combination product once daily
Oral antibiotic and topical retinoid once daily, or
Oral antibiotic and topical retinoid/BP combination product once daily, or
Oral antibiotic, BP wash, spironolactone and topical retinoid once daily (female patient), or
COC, oral antibiotic and topical retinoid/BP combination product once daily (female patient)
Isotretinoin
BP, Benzoyl peroxide; COC, combined oral contraceptive.
*These vignettes are therapeutic examples only and not intended to be specific recommendations; acne therapy should always be tailored to the specific patient and severity of involvement.
Although acne is usually a disorder of adolescents and young adults, neonates and infants are occasionally affected. Neonatal acne has its onset within the first weeks of life and may be congenital in up to 20% of newborns. Infantile acne (see Infantile Acne section), on the other hand, usually presents between 1 and 12 months of life and tends to be more severe and more persistent. Both types occur more often in boys than in girls.
Neonatal acne typically presents with mildly erythematous papules involving the face and scalp and less commonly the chest and back ( Fig. 8.17 ). Comedones occasionally may be present, although these tend to be more characteristic of infantile acne. Neonates presenting with more pustular lesions may have a variant referred to as neonatal cephalic pustulosis (see below). Neonatal acne should be distinguished from other potentially papulopustular disorders of the neonate, including infection (bacterial, candidal, or viral), erythema toxicum neonatorum, miliaria (prickly heat), eosinophilic folliculitis, and milia (typically noninflamed white papules rather than pustules).
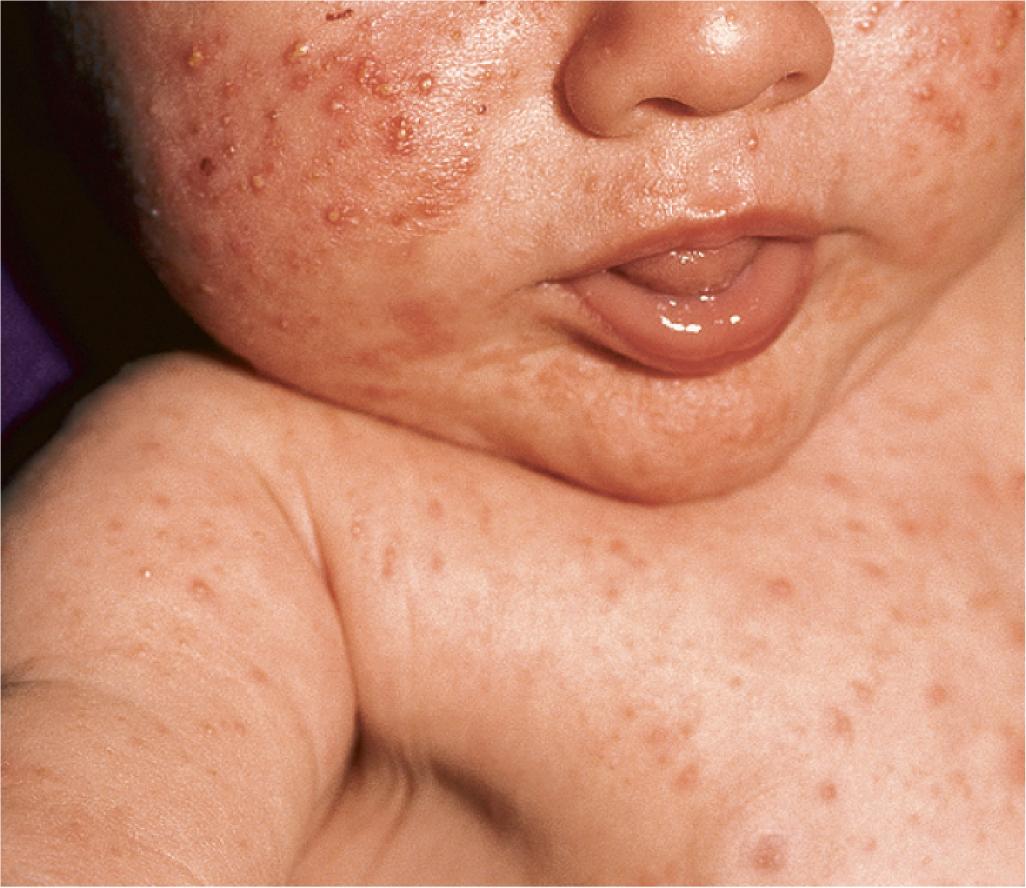
The cause of neonatal acne is believed to relate to increased production of DHEA by the fetal adrenal glands. Another factor may be androgens, both delivered transplacentally and manufactured by the neonatal testicles. Testicular androgen production serves as a stimulus to primed adrenal glands, which may partially explain the increased incidence of neonatal acne in male babies. In babies with neonatal acne, a family history of acne may or may not be present.
In recent years a neonatal pustular eruption that presents in a similar fashion to neonatal acne has been described and has been termed neonatal cephalic pustulosis. This disorder presents with multiple facial papules and pustules ( Fig. 8.18 ), and direct examination of pustule smears may reveal yeasts of Malassezia furfur or Malassezia sympodialis . , In a 1996 series of 13 neonates with such a pustular eruption, smears were notable for M. furfur in 8 of the patients, all of whose conditions cleared rapidly after application of ketoconazole cream. Colonization of neonates with Malassezia begins at birth and increases over the first few weeks of life. However, the exact role of Malassezia in this setting is poorly understood, with inconsistent findings in subsequent reports. The association between neonatal cephalic pustulosis and neonatal acne remains unclear.
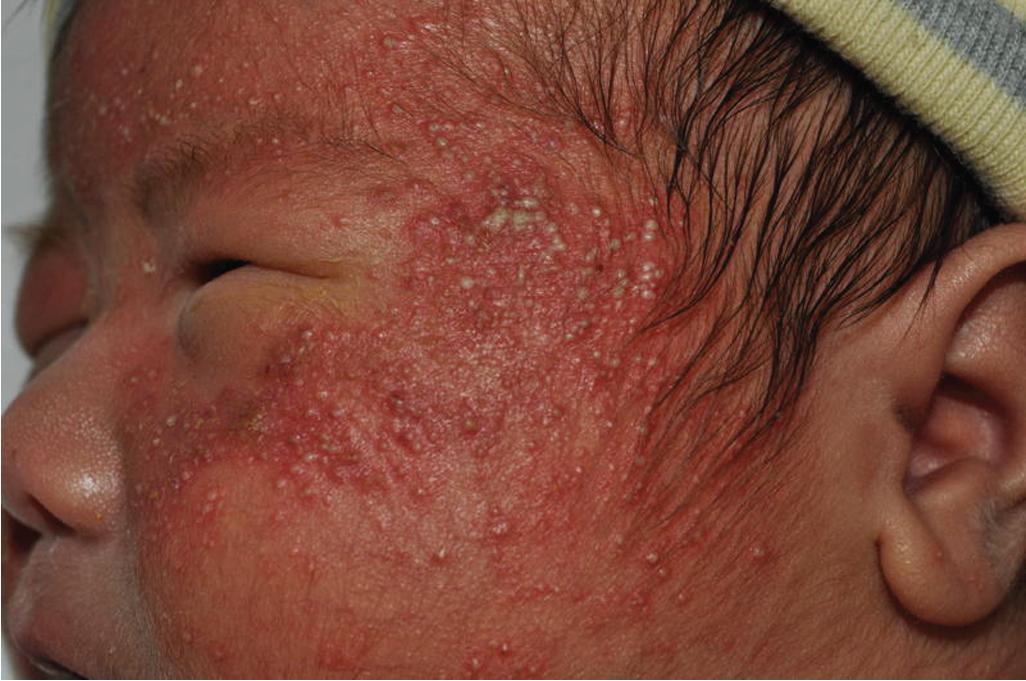
Neonatal acne usually regresses spontaneously over 3 to 6 months, only occasionally persisting longer. In mild cases, therapy is generally unnecessary; daily cleansing with a gentle soap and water may be all that is required. Exogenous oil such as baby oils, creams, ointments, and lotions may aggravate the condition and should be avoided. For infants with more inflammatory or persistent disease, a low-strength (2.5% to 5%) BP gel or a topical antibiotic (e.g., erythromycin gel) are reasonable treatment options. In the otherwise-well neonate with a significantly pustular facial eruption (i.e., more suggestive of neonatal cephalic pustulosis), topical antifungal therapy can be considered. If there is a significant comedonal component or nodules are present, the infant more likely has infantile acne (see Infantile Acne section), and other therapeutic options may be necessary, including oral antibiotics.
Infantile acne presents between 1 and 12 months of life. Clinically the infant most often has open and closed comedones ( Fig. 8.19 ), but inflammatory papules and pustules occasionally occur ( Fig. 8.20 ). They may also develop larger papules and nodules with the potential for scarring ( Fig. 8.21 ), even without comedonal lesions. Lesions occur most commonly on the face, but the neck, back, and chest may also be involved. Most experts consider treatment of infantile acne vital to prevent scarring and permanent facial disfigurement.
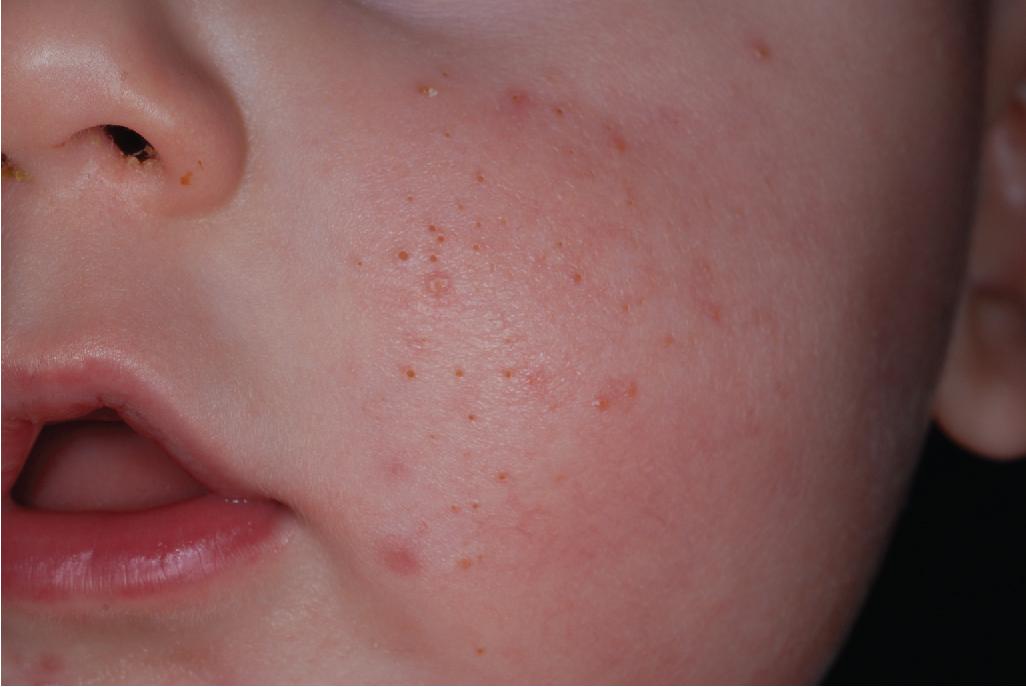
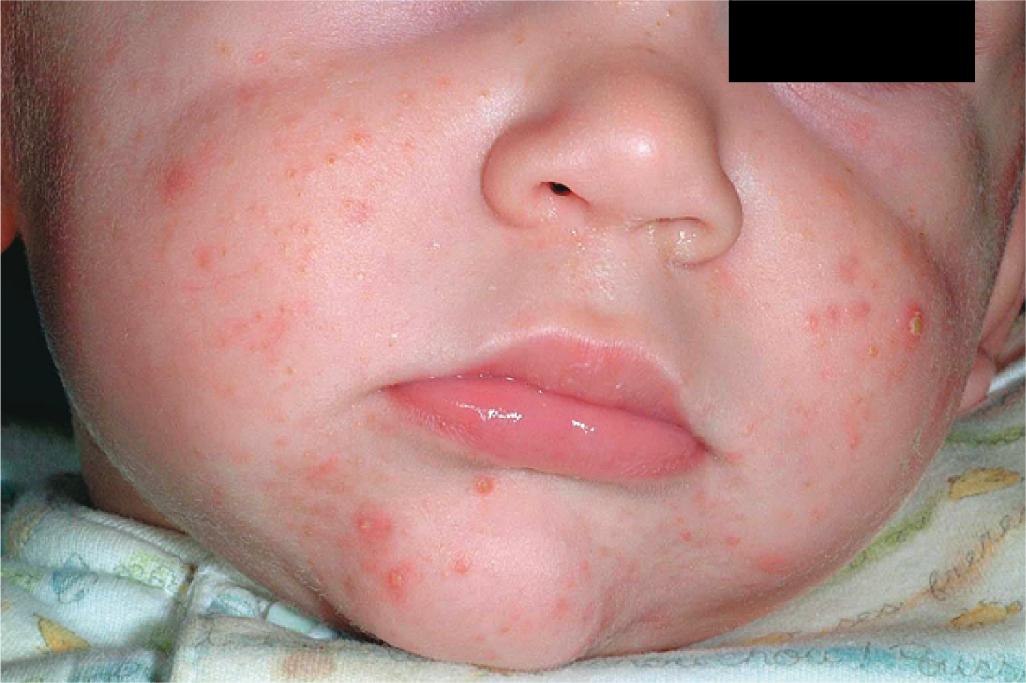
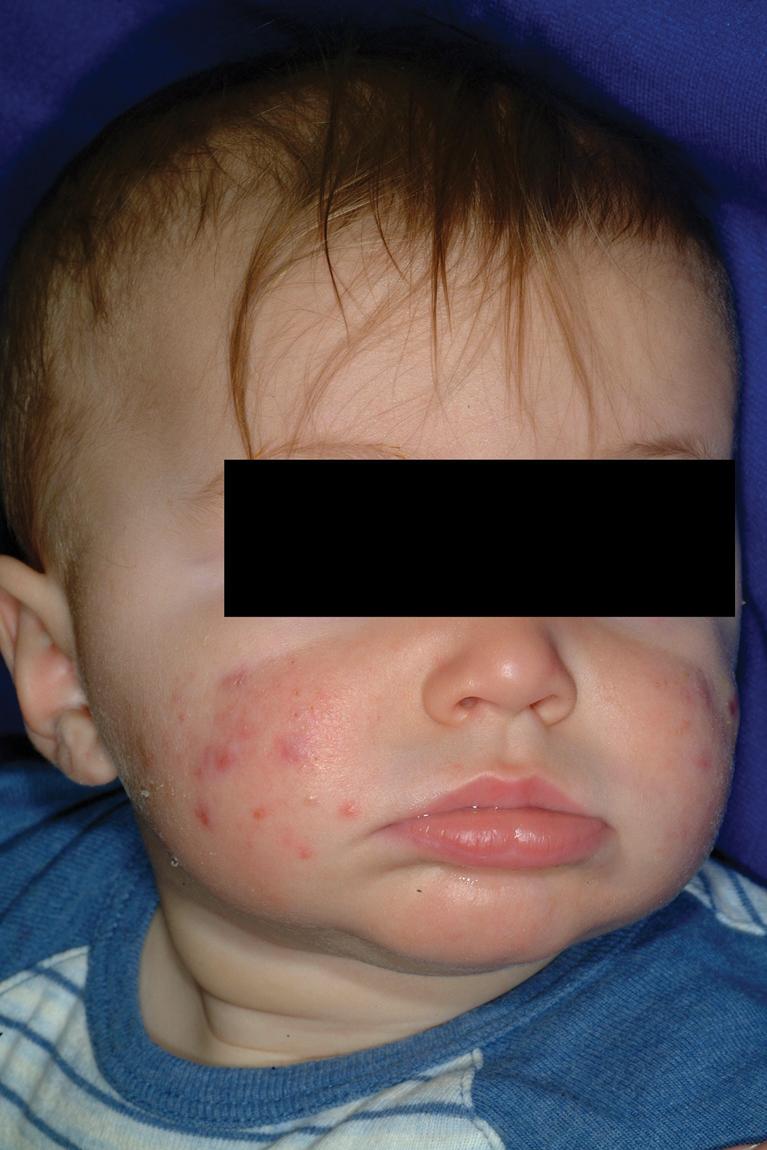
The patient with infantile acne should have a concomitant growth assessment (with charting of height and weight) and examination for any features of precocious puberty or androgen excess, including axillary odor, clitoromegaly, presence of axillary or genital hair, breast development, and increased muscle mass. Left-hand radiography for bone age may be a useful screening test. If any concerns are noted, an evaluation for hyperandrogenism should be performed ( Box 8.6 ).
Serum testosterone (free and total)
Luteinizing hormone (LH)
Follicle-stimulating hormone (FSH)
Dehydroepiandrosterone sulfate (DHEAS)
17-Hydroxyprogesterone (17-OHP)
Prolactin
Radiographic bone age
Treatment for infantile acne depends on the severity of the clinical lesions. Mild to moderate disease is usually treated with a combination of BP, topical antibiotics, and/or topical retinoids. The latter should always be included in patients with more comedonal disease. For disease that does not respond to topical measures, oral erythromycin is an excellent first-line antibiotic therapy, and the ethylsuccinate form is generally well tolerated. Refractory involvement or the presence of nodules or scarring suggests the need to consider oral isotretinoin therapy. Intralesional steroids have also been used to treat larger nodules.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here