Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The Achilles tendon is formed by a coalescence of fibers from the gastrocnemius and soleus muscles. This complex spans both the knee and ankle joints, making it more susceptible to injury than muscles that span a single joint. The Achilles tendon is notably susceptible to injury with concomitant knee extension and ankle dorsiflexion. The medial and lateral heads of the gastrocnemius originate from the medial and lateral femoral condyles, respectively. The soleus muscle originates from the posterior proximal tibia and fibula. More distally, the medial and lateral gastrocnemius and soleus tendons coalesce to form the triceps surae complex. The Achilles tendon then rotates 90 degrees such that the medial gastrocnemius position is more posterior and superficial. This rotation may result in torque stresses that can increase the risk of tendinopathy. After passing distal to the posterior superior calcaneal tuberosity, the Achilles tendon inserts into the posterior and plantar calcaneal tuberosity about halfway between the dorsal and plantar aspects of the calcaneus.
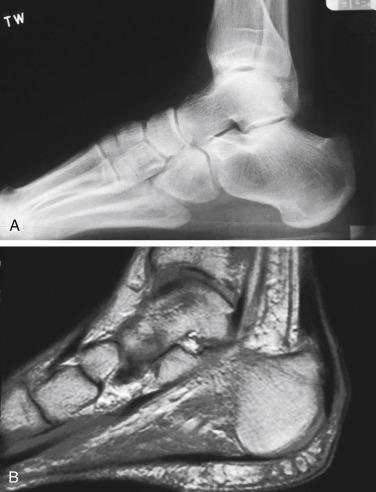
The retrocalcaneal bursa lies between the distal Achilles tendon and the posterior superior calcaneal tuberosity. It is horseshoe shaped and sits around the insertion of the Achilles, which has more fibers centrally and proximally. Anteriorly it is composed of fibrocartilage, whereas posteriorly it blends with the paratenon and commonly connects to the posterior Achilles tendon. The pre-Achilles bursa lies superficial to the Achilles between the Achilles and the skin. These bursae, composed of synovium, provide lubrication to assist with tendon gliding and to minimize tendon irritation. A large, sometimes abnormal prominence of the posterior superior calcaneus, Haglund’s deformity, may create repetitive frictional irritation on the Achilles tendon that can lead to tendinopathy ( Fig. 9.1A–B ).
The Achilles tendon is the strongest and longest tendon in the body, measuring approximately 12 to 15 cm in length. Although it is the main plantarflexor of the ankle, it also functions to invert the heel during late stance phase and thereby locks the transverse tarsal joint for push-off along with the posterior tibial tendon. It is subject to forces up to 10 times body weight during running, experiencing up to 7000 N of force. , ,
The blood supply to the Achilles tendon is segmental and is predominantly derived from anterior branches of the paratenon. Additional sources include intratendinous vessels, the posterior tibial artery, and distal osseous and periosteal branches. A relative zone of hypovascularity exists within 2 to 6 cm proximal to the calcaneal insertion, corresponding to the site of most Achilles tendon ruptures and noninsertional tendinopathy. ,
Similar to other tendons, the Achilles is composed of predominantly type I collagen. Collagen fibrils are bundled into fascicles, which contain elastin, lymphatics, and neurovascular structures and are held together by the endotenon. The epitenon surrounds the group of fascicles, forming the structural unit of the tendon. The paratenon further surrounds the epitenon and consists of an inner parietal layer, lying directly on the epitenon, and an outer, visceral layer. The paratenon, containing a small amount of fluid between its layers, facilitates glide and minimizes posterior adhesion formation.
The nomenclature used to describe tendon disorders has been confusing, as multiple terms are used to describe the same disease process. Tendinitis is the term that has traditionally been used to mean chronic pain and dysfunction of tendons with inflammation as the main underlying etiologic factor. However, histologic examination has demonstrated the absence of an acute inflammatory process or minimal inflammation as compared to what was originally anticipated. On the contrary, several other studies have demonstrated that in the early stages of tendon damage, there is an acute phase of tendon injury that involves inflammatory cells. , Tendinosis describes a chronic degenerative process within tendons that lacks an inflammatory process. The term tendinopathy encompasses a broad spectrum of acute and chronic disease processes affecting tendons. It is most likely that inflammatory and chronic degenerative processes both play integral roles during tendon pathology. For the purpose of this chapter, the term tendinopathy will be used to encompass acute and chronic degenerative stages of tendon pathology.
Achilles tendinopathy is common among athletes, affecting nearly 18% of runners. Repetitive impact-loading activities (overuse) such as jumping are responsible for the majority of cases. Other predisposing extrinsic risk factors include overuse, training errors, environmental conditions, poor footwear, and improper training techniques (excessive running, sudden increase in intensity, uphill running). Other intrinsic predisposing factors include poor extremity biomechanics (foot pronation, cavus foot, genu varum), increased age, increased body mass index, gender, prior history of tendon disorders, medical comorbidities, and genetics. , Another risk factor includes the previous use of fluoroquinolone antibiotics. Athletes commonly affected by tendinopathy are involved in running, jumping, dancing, tennis, racquetball, basketball, and soccer
Repetitive impact-loading activities
Abnormal lower-extremity biomechanics (foot pronation/supination, stiff joints, genu varum)
Improper training techniques (intensity, frequency, duration, speed, terrain)
Poor footwear selection
Fluoroquinolone antibiotics
Puddu et al. classified Achilles tendinopathy into three clinical categories. Peritendinitis is characterized by inflammation affecting the paratenon. Peritendinitis with tendinosis refers to both inflammation involving the paratenon and degeneration of the Achilles tendon. Tendinosis reflects Achilles degeneration. Clain and Baxter later created an anatomic classification, separating tendinopathy into insertional disorders, affecting the area of the enthesis, and noninsertional disorders, commonly affecting the tendon 2 to 6 cm proximal to the calcaneus.
Whereas noninsertional tendinopathy occurs more often in younger, more active athletes, insertional Achilles tendinopathy develops more often in those athletes who are older, less active, and sometimes overweight. Although bilateral insertional tendinopathy will most likely be due to mechanical overuse, it may be associated with inflammatory disorders, including seronegative spondyloarthopathies. ,
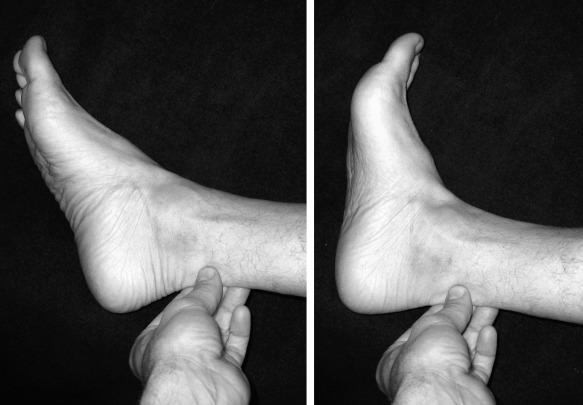
Noninsertional tendinopathy can present with peritendinitis. The Achilles tendon itself is less involved. In chronic cases, adhesions may form between the paratenon and tendon, leading to more profound pain and tenderness. Pain is noted most often at the initiation of activity (start-up pain) and improves with continued exercise. Acute pain typically resolves with rest. In chronic cases, however, the pain may persist and significantly impair further athletic participation. On examination, a localized, increased diameter that more commonly affects the medial side is appreciated with palpation of the tendon. Tenderness, and at times crepitus, is noted throughout all ankle range of motion ( Fig. 9.2 ). Radiographs generally are unremarkable.
Peritendinitis with tendinopathy represents further inflammation with associated intratendinous degeneration. Pain is more marked and constant. The tendon is thickened and infrequently has palpable intrasubstance calcifications ( Fig. 9.3 ). The painful arc sign may help to distinguish between tenderness associated with peritendinitis and that associated with tendon degeneration. Tenderness related to peritendinitis will be constant in location as the ankle is brought through a range of motion, whereas tenderness associated with tendinopathy will change position with ankle motion.
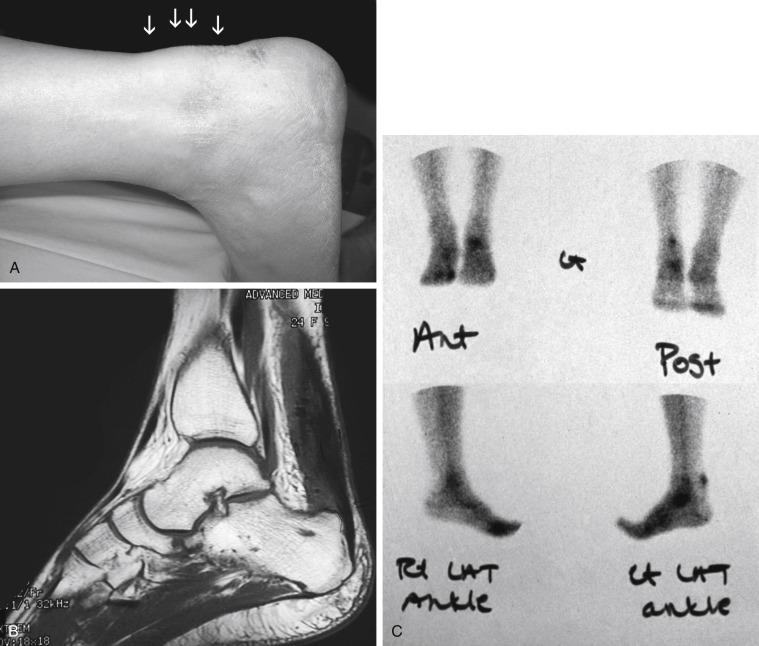
Isolated tendinopathy, or noninflammatory atrophic degeneration, is associated with normal aging and typically is accelerated by overuse. Most affected are middle-aged, recreational athletes. With repetitive trauma, microtears develop within the tendon, mostly in the hypovascular zone, leading to further fibrosis and degeneration. These athletes complain of weakness in push-off, with pain and swelling localized to the area approximately 2 to 5 cm proximal to calcaneus. Whereas ankle dorsiflexion commonly is limited, tendon elongation may develop with an associated increase in passive ankle dorsiflexion. Pathologic examination reveals fatty degeneration with disorganized collagen. Calcific deposits may be present
Paratendinitis: release paratenon
Tendinopathy: degree of width involved <50%, ellipse and repair; >80%, ellipse and augment
1–3 cm gap after debridement:V-Y
3–5 cm gap after debridement: turndown
>5 cm gap after debridement: turndown with consideration for FDL or FHL tendon transfer
FDL, flexor digitorum longus; FHL , flexor hallucis longus
Insertional tendinopathy is an inflammatory reaction within the Achilles tendon affecting the enthesis, or tendon insertion onto the calcaneus. This disorder more commonly affects older, heavier, and less active athletes but can be seen in competitive athletes as well. An abnormally enlarged, bony prominence may aggravate this condition. There is a high association with Haglund’s deformity and retrocalcaneal bursitis, but unlike these disorders, insertional tendinopathy involves the tendon itself. This most often results from chronic overuse and poor training habits. Improper techniques include inadequate stretching, rapid increase in training, running on harder surfaces, and heel running. Although pain initially follows exercise, particularly uphill running, symptoms may become more persistent despite activity modification.
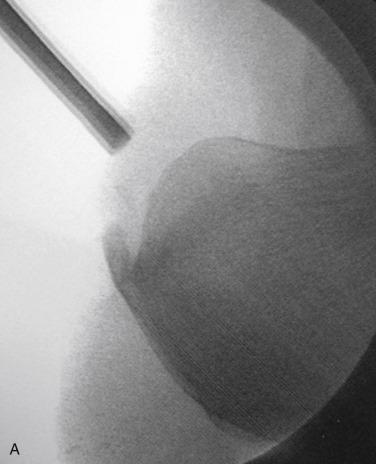
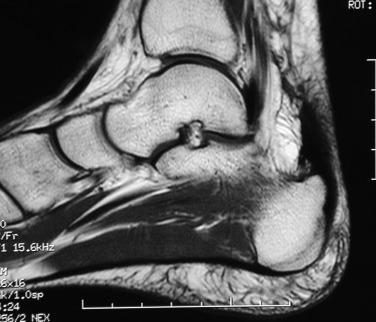
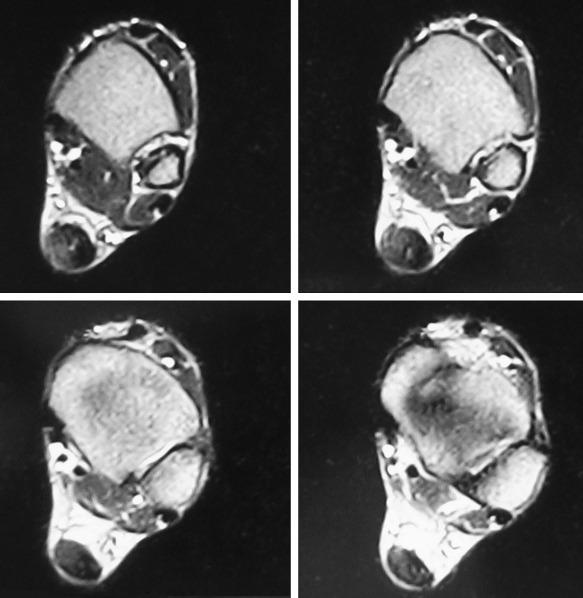
Pain, swelling, and warmth are noted specifically at the enthesis. In athletes, there often is a localized area of pain with a small spur. Ankle range of motion is painful, with dorsiflexion typically limited because of a tight or painful Achilles tendon. External irritation from a shoe’s heel counter plays less of a role in provoking symptoms in athletes with Achilles tendinopathy than in retrocalcaneal bursitis and Haglund’s deformity. This is because the insertion lies within the center of the heel cup and tends not to get rubbed unless the tendinopathy extends more proximally near where the edge hits the shoe. Radiographs generally reveal calcifications or a bony spur at the most distal aspect of the Achilles insertion ( Fig. 9.4A ). Magnetic resonance imaging (MRI) will show thickening of the tendon with longitudinal striations of fibrosis or splits where the tendon attaches to the calcaneus. Often there will be localized bony edema in the calcaneus at this junction ( Fig. 9.4B ).
Haglund is credited with first describing the presence of a prominent posterolateral superior calcaneal tuberosity in 1927. This enlarged superolateral tuberosity predisposes the precalcaneal bursa to be compressed between it and any tightly fitting shoe heel counter, possibly leading to skin irritation and inflammation. Because of the association of footwear, this disorder also has been referred to as a “pump bump” and “winter heel.” Although there is a frequent association with retrocalcaneal bursitis and insertional Achilles tendinopathy, Haglund’s deformity initially generally does not involve the Achilles tendon.
Poorly fitting shoes in conjunction with this genetic bony prominence generally are responsible for the development of symptoms of Haglund disorder. Other predisposing risk factors include the presence of a cavus foot and hindfoot varus. In rare cases, childhood apophyseal trauma may be a cause. In the nonathletic population, repetitive injury or trauma may result in bone overgrowth. Most affected are young women who wear fashionable high-heeled shoes. In the athletic population, we have observed this condition more commonly in males who participate in running sports. Long-distance runners are susceptible to this condition and the other Achilles tendon disorders (see Figs. 9.1 and 9.5 ).
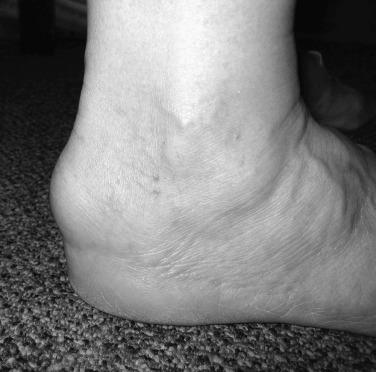
On examination, the affected heel has a swollen, red, and tender posterior prominence, predominantly on the lateral side of the calcaneus. The Achilles tendon itself is not tender. Numerous radiographic measurements have been used to quantify the size of the posterosuperior prominence. These techniques generally are not used by orthopaedists as they do not always correlate with the clinical findings.
Retrocalcaneal bursitis refers to inflammation affecting the bursa immediately anterior to the Achilles tendon. As with Haglund’s deformity and Achilles tendinopathy, this condition is common in running athletes. In the general population, as with insertional tendinopathy, those most commonly affected are older, less active recreational athletes. As the disorder becomes chronic, the bursa enlarges and may become adherent to the Achilles tendon. A prominent posterosuperior bony projection may be present.
Athletes typically complain of pain with activities that force the ankle into dorsiflexion, particularly uphill running, and thereby compress the inflamed bursa between the posterosuperior calcaneus and the Achilles tendon. Schepsis et al. described the two-finger squeeze test, in which pain is noted when two fingers compress medially and laterally immediately superior and anterior to Achilles insertion. This area will be warm with a notable soft-tissue bulge. Pain is elicited with passive dorsiflexion. Radiographs may show a subtle soft tissue fullness and loss of the retrocalcaneal soft-tissue shadow, as well as the presence of a posterosuperior bony prominence. MRI demonstrates the bursal enlargement anterior to the Achilles tendon above its insertion in the retrocalcaneal region ( Fig. 9.6 ).
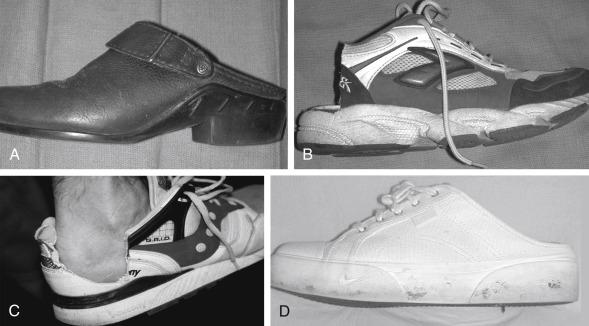
The treatment for Achilles tendinopathy is nonoperative at first with surgical intervention for recalcitrant cases. Initial treatment should include anti-inflammatory medications, gentle Achilles stretching and activity modification. Relative rest with limitations on intensity, duration, or frequency of training and concomitant institution of nonstressful cross training (exercise bike, pool running, elliptical trainer) should be helpful. An open-back shoe may benefit those with no heel counter pressure ( Fig. 9.7 ). A padded heel sleeve can be comfortable. If there is no response, a heel lift (one-fourth to three-eighths inch), night splint, or temporary immobilization in neutral or slight plantarflexion with a removable walking boot or cast may be required. If the athlete has notable foot pronation, a semirigid orthotic may improve overall foot biomechanics by supporting the medial arch.
Intratendinous corticosteroid injections should be avoided because local use of these injections has been associated with tendon attrition and potential rupture. Although there is no strong evidence of similar deleterious effects after paratendinous corticosteroid injections, there are similar worries with an injection in the bursa. It would be advisable to immobilize the ankle temporarily after a retrocalcaneal injection because the retrocalcaneal bursa has a direct communication to the Achilles and may make it structurally vulnerable. , In general, we advise against corticosteroid injections.
For refractory peritendinosis we have found that brisement may provide symptomatic relief in a third to half of total cases. Brisement consists of injecting 5 to 10 ml of sterile saline or local anesthetic agents into the Achilles tendon sheath; this may forcibly disrupt any adhesions between the paratenon and Achilles tendon. Repeating the injections two to three times over several weeks may be necessary to achieve success. ,
After initial symptoms resolve, it is imperative to correct predisposing factors, including improper technique, excessive training, inappropriate footwear, and poor flexibility. In addition, it may be critical to temporarily or permanently eliminate provocative, more rigid, and less compliant surfaces and terrain.
Reported results of nonoperative treatment of insertional and noninsertional Achilles tendinopathy have been generally successful. Studies have found that 70% to 90% of patients have found symptomatic improvement after corrections in their footwear, training habits, and mechanics. , There are, however, fewer predictable results with nonsurgical management in those with chronic tendinopathy and in the older athlete, as a result of greater degenerative tendon involvement.
In our experience, athletes with isolated Haglund’s deformity can be managed with footwear modification about 50% of the time. The presence of a prominence does not mandate surgery. We have seen improvement in about 30% of patients with retrocalcaneal bursitis and in about 25% of those with insertional tendinopathy.
The use of extracorporeal shock wave therapy (ESWT) for the treatment of Achilles tendinopathy has not been conclusively studied. The most information on shock wave therapy comes from upper extremity tendinopathy (e.g., tennis elbow) and plantar fasciitis. Shock wave therapy works by creating a pressure change that propagates rapidly through a medium. When transmitted through a water medium, it can either directly create high tension at a given structure or indirectly create microcavitations. Theories behind its analgesic effect in orthopedic applications include an alteration of the permeability of neuron cell membranes and induction of an inflammatory-mediated healing response by increasing local blood flow. Studies have also shown that ESWT decreases tenocytes expression of matrix metalloproteases and interleukins. Studies on ESWT on Achilles tendinosis have shown a success rate of approximately 30%–40%. , A systematic review by Al-Abbad et al. demonstrated evidence for the effectiveness of ESWT in the treatment of chronic insertional and noninsertional Achilles tendinopathies with a minimum of 3 months follow-up before undergoing surgery. Furthermore, they demonstrated that ESWT in conjunction with eccentric loading shows superior results. In our experience, we have found a success rate of approximately 50% in athletes. Even with this lower success rate, we try ESWT and then wait 3 months before surgery given the minimal side effects. Depending on the immediate results, we may allow sports play with only 1 or 2 weeks off. If the athlete is in midseason, then this modality is his or her best chance to resume play. If the athlete is at the end of the season, then we may try shock wave therapy and a boot brace for 2 to 6 weeks and then allow the athlete to resume impact activities. After the season, when there is more time for recovery, decisions regarding further treatment can be made. Contraindications to ESWT quoted in the literature include pregnancy, coagulopathies, bone tumors, bone infection, and skeletal immaturity.
Saggini et al. noted successful outcomes after two treatments with no complications using shock wave therapy on Achilles tendinopathy. Several later studies reported promising results after ESWT with those affected with chronic Achilles tendinopathy. The cost of shock wave treatment can be an important consideration because the therapy may not be covered by insurance. With lower-energy shock wave machines, three treatments are used, at a total cost of $500 to $1500. The higher-energy machines usually require one treatment ($1000 to $3000) but this typically involves a regional or general anesthesia given the magnitude of the pain. Authors (LCS and JPT) do not generally recommend ESWT for those patients with bony prominences (e.g., Haglund’s or large insertional spurs).
Injection of autologous platelet-rich plasma (PRP), bone marrow concentrate or allogenic factors—derived from decellularized placental/chorion/amniotic tissue—can also be considered for resistant cases (See Chapter 29 ). For the PRP, patients need to have their blood drawn and equipment is needed to process the blood and harvest the platelets via centrifugation or filtration. Costs can be between $500 and $2000. The techniques of the platelet isolation and the subsequent quantity of platelets and white blood cells (WBCs) vary. Clinical studies have not been able to demonstrate a clinically significant improvement of tendon healing with PRP injection. , However, platelet-rich plasma has been shown to stimulate cell proliferation, enhance the activity of growth factors like vascular endothelial growth factor (VEGF) and platelet-derived growth factor (PDGF), and increase total collagen production in a controlled laboratory study.
For the bone marrow concentration, the patients require some pre-medication and may benefit from sedation in the operating room or procedure room. The bone marrow is harvested in 5ml aliquots via multiple trajectories from the iliac crest. Typically a specialized chamber is used to perform centrifugation to separate the red blood cells (RBCs) from the plasma and the platelet rich cellular buffy coat. Unlike in PRP, the concentrate contains endothelial progenitor cells, mesenchymal signaling cells, hematopoietic stem cells, and other progenitor cells in addition to the platelets. For the allogenic factor injections, the materials and methods also are diverse. For both, the method of injection and post injection rehabilitation vary as do the results in the literature. When there is a bony element that causes a mechanical prominence (e.g., large Haglund’s or spurring in the insertional Achilles tendinopathy) the results can be less than 30% successful. If there is only the bursitis or tendinopathy, the results can be up to 75% for PRP or growth factors or 90% for bone marrow concentrate in the authors’ (LCS and JPT) experience.
Surgical intervention is considered after approximately 3 months of nonoperative treatment measures. The surgical technique is chosen on the basis of location of pathology. For a symptomatic athlete with a normal tendon, determined by physical examination and possibly by MRI, we generally try to avoid a procedure that may irritate or traumatize the tendon. A medial or lateral approach 5 to 10 mm anterior to the Achilles tendon and paralleling its course is best in these cases because it runs through thicker skin and subcutaneous tissues. If the tendon is involved, this approach also provides excellent access to the tendon with the creation of full thickness flaps down to the level of the paratenon. Debridement of the tendon depends on the location of the clinical and radiographic findings – above, at, or below the insertion of the tendon. In general, the points of tenderness dictate where the exposure must occur. Thus, if there is more medial and central Achilles tendinopathy, the tendon in the medial and central aspect must be elevated off the calcaneus, the tendon must be debrided there, and the underlying bone must be resected and recontoured.
For noninsertional tendinopathy, the choice of procedure is based on whether the disease involves the paratenon, tendon, or both. In peritendinitis, all adhesions are excised, and the surgeon also performs a limited resection of any thickened paratenon. The extremity is immobilized for 3 to 5 days, followed by a range-of-motion program to limit the recurrence of scar formation ( Fig. 9.8 ).
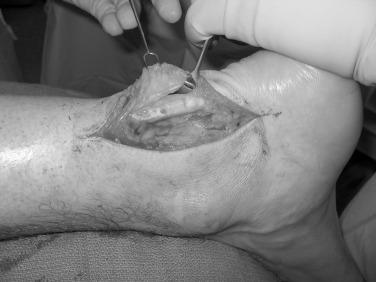
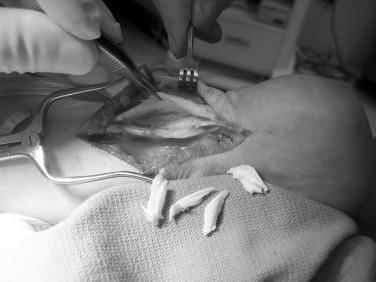
When there is tendinopathy and peritendinitis, elliptical excision of the tendon and longitudinal paratenon release is performed. Maffulli et al. have reported a success rate of approximately 70% after percutaneous longitudinal tenotomy of the middle third of the Achilles tendon. In this technique a no. 11 or no. 15 blade is introduced posteriorly through the skin and tendon. With the blade held stationary, the ankle is dorsiflexed, and the tendon moves, allowing it to be cut longitudinally. Next the blade direction is reversed 180 degrees and the ankle is plantarflexed. The process is repeated through four additional incisions in the zone of the degenerative tendon ( Fig. 9.9 ). This creates a controlled tendon trauma and triggers a healing response.

The type of procedure chosen for treatment of tendinopathy depends on many factors, the largest, in our experience, being the extent of tendon involvement, determined by clinical findings, ultrasound, or MRI. When less than 50% of the tendon is involved, we longitudinally ellipse the diseased tendon; when more than 80% of the tendon is involved, a debridement and tendon augmentation (e.g., turndown) or transfer is recommended ( Fig. 9.10 ). When there is between 50% and 80% involvement, the decision is determined by the patient, the sport, and the surgeon’s preference.
For tendinopathy, typically the degenerative portion of the Achilles tendon is debrided and the paratenon is released. If less than 50% of the tendon width is debrided, then the remaining section of intact longitudinal tendon should be strong enough to withstand stresses.
Typically, a medial incision is made just anterior and parallel to the border of the tendon that is thickened, and the paratenon is entered. On the basis of maximal tenderness, MRI, or ultrasound localization of the degenerative zone of the tendon, an elliptical longitudinal excision of the diseased tendon is performed, leaving intact the anterior and posterior surfaces of the tendon. Essentially the zone of ellipsed tissue should include the degenerative fibers and the thickened tendon ( Fig. 9.11 ). The tendon then is repaired with internally placed, nonabsorbable sutures with buried knots. The subcutaneous tissues are apposed, followed by closure of the skin. The leg is immobilized for 3 to 5 days in a splint, followed by range-of-motion exercises, strengthening, and nonimpact activities. A boot brace is worn for 6 to 12 weeks during ambulation to unload the healing tendon. Jogging and running may be introduced at 3 months, depending on the extent of involvement and the nature of the patient’s athletics.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here