Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
In patients with a history of myocardial infarction (MI) who experience episodes of sustained ventricular tachycardia (VT), surgical treatment was the mainstay of procedural therapy before the advent of catheter ablation. Initial experience with aneurysmectomy showed only moderate success rates for long-term arrhythmia control. Further refinement of surgical technique with the removal of subendocardial tissue surrounding the aneurysm increased success rates even further, emphasizing the importance of targeting critical portions of the arrhythmia circuitry not necessarily within the dense scar. Given the highly invasive nature of the procedure in patients with typically impaired cardiac function, however, a less invasive approach was desirable.
The introduction of implantable cardioverter defibrillators (ICDs) for the treatment of ventricular arrhythmias (VAs) in patients with ischemic heart disease (IHD) was a turning point in arrhythmia management. Before long, ICDs became the cornerstone therapy for the prevention of sudden cardiac death (SCD) in patients with IHD. Nevertheless, ICD shocks, although potentially life-saving, were associated with impaired quality of life and increased mortality. Antiarrhythmic drugs (AADs) were used for arrhythmia prevention and shock reduction; however, these agents often have limited efficacy and significant side effects. ,
With the advent of catheter-based mapping and ablation technologies, catheter ablation assumed an increasingly important role in VT management. The ability to characterize circuits via mapping and entrainment demonstrated scar-related reentry as the most common mechanism of monomorphic VT in patients with prior MI. Operators were now able to deliver energy to target critical components of these arrhythmia circuits via a minimally invasive approach. In the setting of IHD and drug-refractory VT, ablation has been shown to reduce arrhythmia recurrence and the need for ICD therapies. Catheter ablation has also been shown to be effective in the treatment of VT storm in patients with structural heart disease (SHD) receiving chronic AAD therapy. In a recent trial comparing escalation of AAD therapy with catheter ablation in patients with IHD, ablation was shown to better reduce the composite endpoint of death, VT storm, and appropriate ICD shocks.
Trials are presently ongoing to compare initiation of AAD therapy with catheter ablation as first-line treatment for patients with VT. In addition, studies on optimal ablation targets, ablation endpoints, and the use of different mapping and ablation technologies are ongoing in this rapidly evolving and exciting field.
Preoperative assessment before catheter ablation should include electrocardiographic characterization of all VAs, preprocedural cardiac imaging, and risk stratification (assessment of heart failure status, hemodynamic stability, renal function) coupled with medical optimization as required.
Assessment of the baseline sinus rhythm electrocardiogram (ECG) will give important information as to the arrhythmic substrate. Surface Q waves or lack of R wave progression in the precordial leads is indicative of prior MI and may hint to the origin of the clinical VT.
If available, the 12-lead ECGs of all clinical VTs are useful for characterizing the VT exit sites. The onset of the QRS vector during VT will point away from the exit site; thus identification of the limb lead vector and the transition in the precordial leads can aid in localizing the target area for ablation. An algorithm for identifying the VT exit site and some examples are shown in Fig. 86.1 .
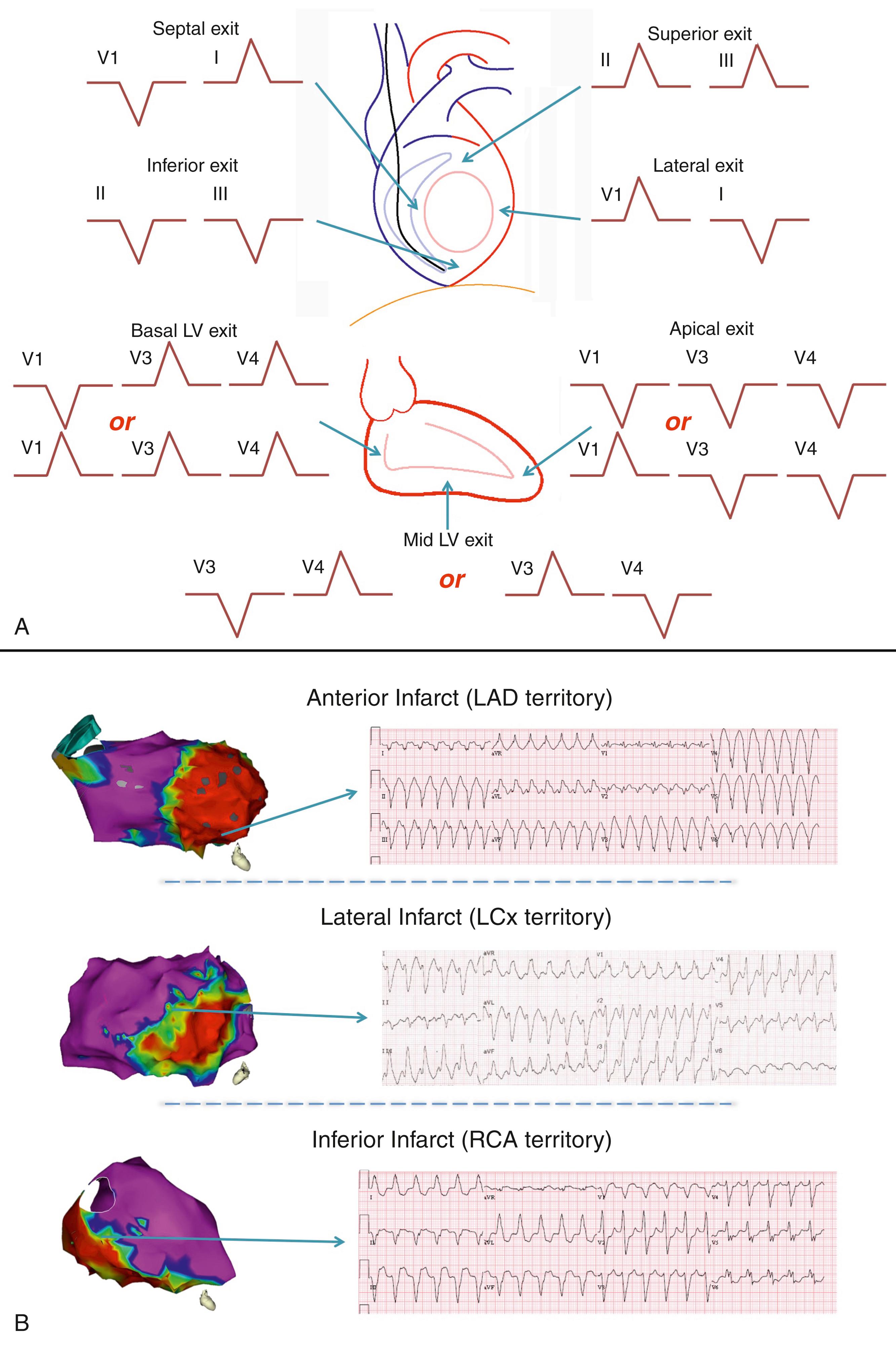
In the absence of ECGs of the clinical VT, ICD electrograms (EGMs) of the clinical event can be used to confirm that the spontaneous clinical VT is the same as the induced VT in the electrophysiology lab. In addition, pace mapping during the procedure and comparing paced EGMs to clinical VT EGMs can be used to estimate (with a spatial resolution of approximately 5 cm) the catheter proximity to the VT exit site.
Of note, patients with coronary artery disease may also exhibit a nonischemic substrate/etiology for VT, as suggested by the presence of multiple VTs of basal origin. Further testing, such as magnetic resonance imaging (MRI), may help elucidate a nonischemic cause of VT in patients with IHD if this is suspected. This is especially important for procedural planning because epicardial access is more commonly required in patients with a nonischemic VT etiology.
Preprocedural imaging is vital both for substrate characterization and as a component of risk assessment. Assessment of the coronary circulation (via coronary angiography or cardiac computed tomography [CT]) can help identify any critical stenosis that may need to be addressed before ablation. Coronary assessment can also demonstrate occlusions responsible for the territory of tissue damage leading to the arrhythmia.
Echocardiography is also important for the assessment of ventricular function, assessment of aortic valve anatomy (before retrograde aortic left ventricular [LV] access), and exclusion of intracardiac thrombi. If the patient is in atrial fibrillation and the anticoagulation status is in question, exclusion of left atrial appendage thrombus is also required (in case of electrical cardioversion during the procedure). In the presence of LV thrombus, a period of anticoagulation is favored to resolve the thrombus before the ablation procedure. If the procedure is deemed urgent, however, ablation in the presence of LV thrombus has been reported.
In the presence of peripheral vascular disease, imaging can be performed to guide decisions regarding retrograde aortic access via the femoral artery versus transseptal LV access via the femoral vein. The presence of mechanical valves may further limit ventricular access and alternative approaches may be required. ,
Certain clinical risk factors have been shown to correlate with acute hemodynamic compromise during VT ablation and have been published as the PAINESD score (chronic obstructive pulmonary disease, age >60 years, ischemic cardiomyopathy, New York Heart Association [NYHA] class III–IV, ejection fraction [EF] <25%, VT storm, and diabetes mellitus; Fig. 86.2 ). A higher score has been shown to correlate to worse procedural outcome, VT recurrence, and mortality. ,
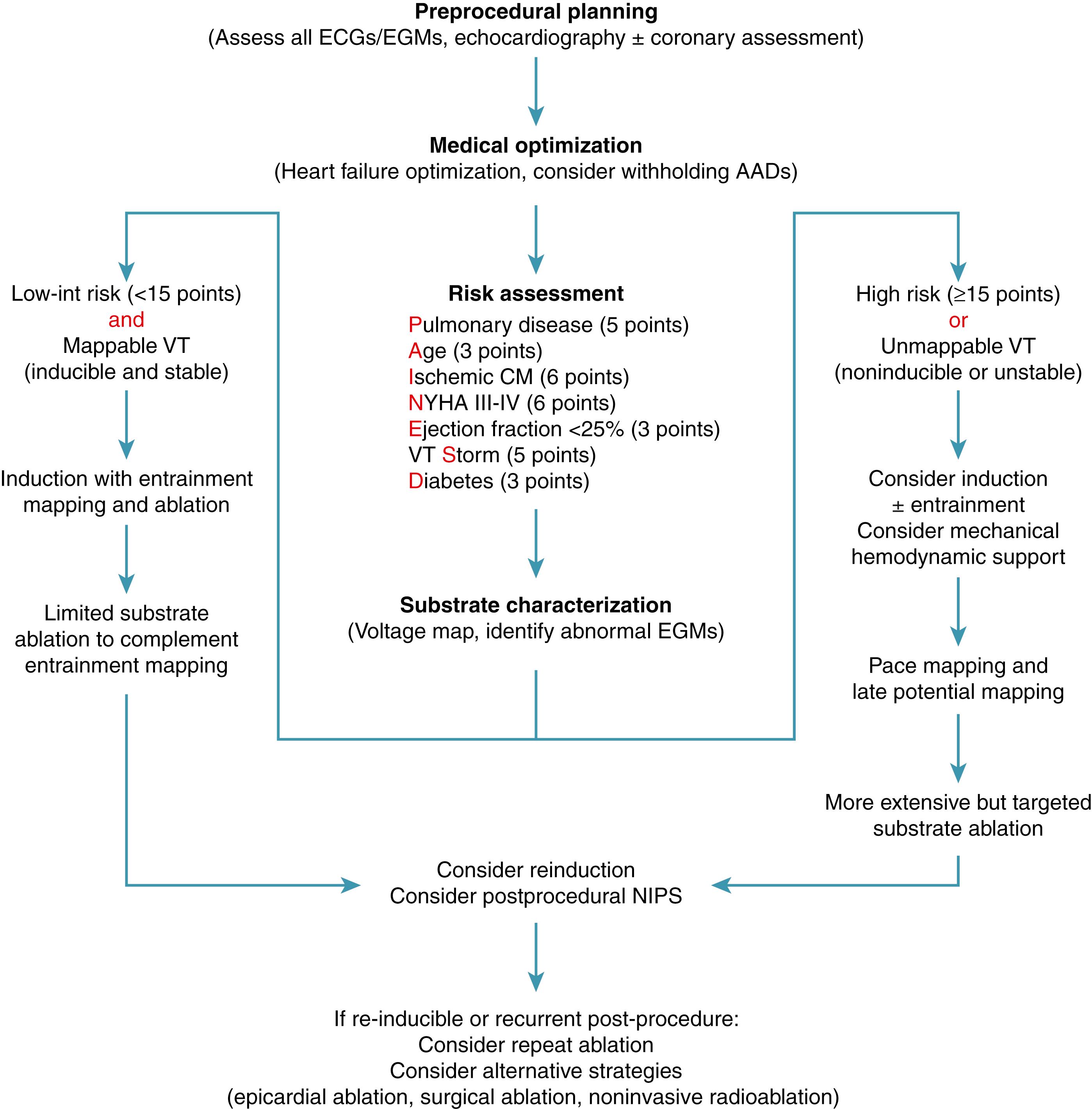
The ability to identify patients at higher risk for periprocedural complications is important to optimize clinical status before the procedure and assess the optimal ablation strategy. Reducing procedure time, the use of mechanical circulatory support, avoidance of general anesthesia, and potential avoidance of periods of hypotension caused by VT induction/mapping are all potential strategies to reduce procedural risk.
Efforts should be made to optimize hemodynamic status before proceeding with ablation. Medical management may involve IV diuresis or ultrafiltration. In patients with cardiogenic shock, hemodynamic mechanical support may be required before or during catheter ablation. , Tools for mechanical support may include intraaortic balloon pumps (IABPs), percutaneous left-ventricular assist devices (pLVADs), or extracorporeal membrane oxygenation (ECMO). There are potential complications and increased costs associated with the use of such devices, and these must be weighed against the benefits. To date, no improvement in arrhythmia outcomes with these devices has been documented ; however, there may be a role in supporting hemodynamic status and reducing procedural complications related to hemodynamic compromise. , ,
When possible, withholding AADs for a minimum of five half-lives before the procedure is optimal for the induction and targeting of all potential VTs. For patients at high risk for VT during the interim or patients requiring amiodarone, bridging with IV lidocaine in a monitored setting has been our standard practice.
In the setting of IHD, the arrhythmia circuit responsible for VT is made up of viable myocytes separated by fibrosis, allowing for the slow conduction needed to facilitate reentry. , Regardless of approach, the goal of ablation is to abolish the viable myocytes within and around the scar tissue that are critical to VT maintenance. The vast majority of VT circuits in patients with a postinfarction substrate are accessible from the endocardial surface, without the need for epicardial mapping or ablation.
The LV can be accessed either retrogradely across the aortic valve via the femoral artery or transseptally across the mitral valve via the femoral vein. In the presence of aortic valve disease, significant peripheral vascular disease, or atheroma in the ascending aorta, transseptal access is preferred. On the other hand, in the presence of mitral valve pathology, retrograde access may be preferred. In some cases, to facilitate access to specific parts of the ventricle, both routes may be used simultaneously.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here