Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The protozoan genus Trypanosoma consists of several dozen species. Two of the three species that infect humans are pathogenic, and several other species cause severe and economically important diseases in domestic mammals. Broadly defined, the organisms belonging to this genus are protozoan flagellates of the family Trypanosomatidae, order Kinetoplastida, that pass through different morphologic stages (epimastigote, amastigote, and trypomastigote) in their vertebrate and invertebrate hosts. The criterion of three morphologic stages, however, is not fulfilled by each species in the genus. For example, only Trypanosoma cruzi, the etiologic agent of American trypanosomiasis, or Chagas disease, and one other species multiply in mammalian hosts as intracellular amastigotes similar to those seen in infections caused by organisms belonging to the genus Leishmania. In contrast, African trypanosomes, which cause sleeping sickness in humans and varying degrees of morbidity in wild and domestic mammals, do not have an intracellular form and multiply as trypomastigotes that circulate in the mammalian bloodstream and other extracellular spaces.
The trypomastigote form has a single flagellum originating near the kinetoplast, which is a DNA-containing structure located in the parasite's single, complex mitochondrion. The flagellum runs alongside the body of the parasite and is enveloped in an undulating membrane. It extends beyond the body as a free, threadlike structure. The undulating membrane and the free portion of the flagellum give the organism considerable motility.
According to their course of development in the vector, trypanosomes have been classified into two major groups:
Stercoraria: Multiplication in the mammalian host is discontinuous, taking place in the amastigote stage. Development in the vector (triatomines, or kissing bugs) is completed in the hindgut (posterior station), and mammalian hosts become infected by contaminative transmission. The subgenus Schizotrypanum belongs to this group and includes T. cruzi.
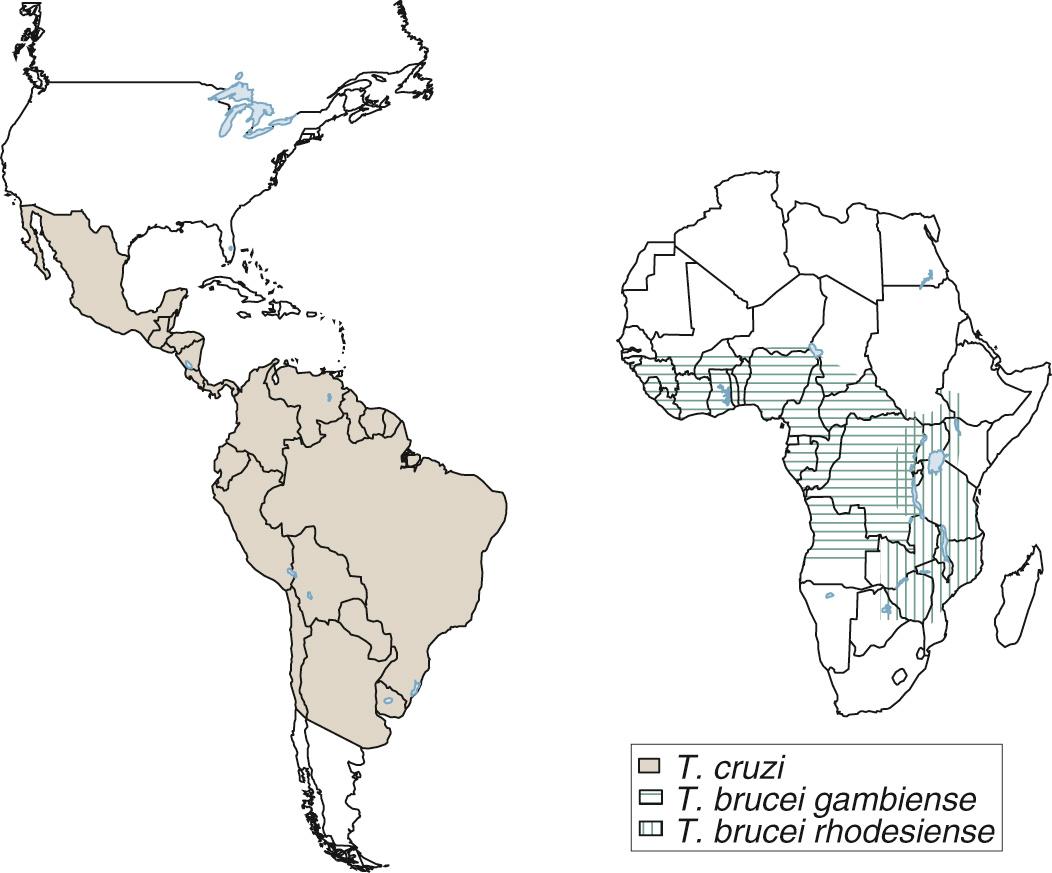
Salivaria: Multiplication in the mammalian host is continuous, taking place in the trypomastigote stage. Development in the vector ( Glossina, or tsetse fly) is completed in the salivary glands (anterior station), and inoculative transmission to mammalian hosts occurs. The subgenus Trypanozoon belongs to this group and includes, among others, the subspecies Trypanosoma brucei brucei, which causes disease in animals but does not infect humans. Trypanosoma brucei gambiense and Trypanosoma brucei rhodesiense, the two causative agents of African sleeping sickness, or human African trypanosomiasis, are also found in this subgenus. As a group, these three subspecies are often referred to as the T. brucei complex. Endemic areas of Chagas disease and African sleeping sickness do not overlap ( Fig. 276.1 ). Moreover, there are such important differences in the transmission, pathogenesis, and clinical course of the two diseases that they have little in common except the genetic and morphologic similarities of the causative agents.
T. cruzi, the causative agent of American trypanosomiasis, is transmitted by various species of blood-sucking triatomine insects, or kissing bugs ( Fig. 276.2 ).

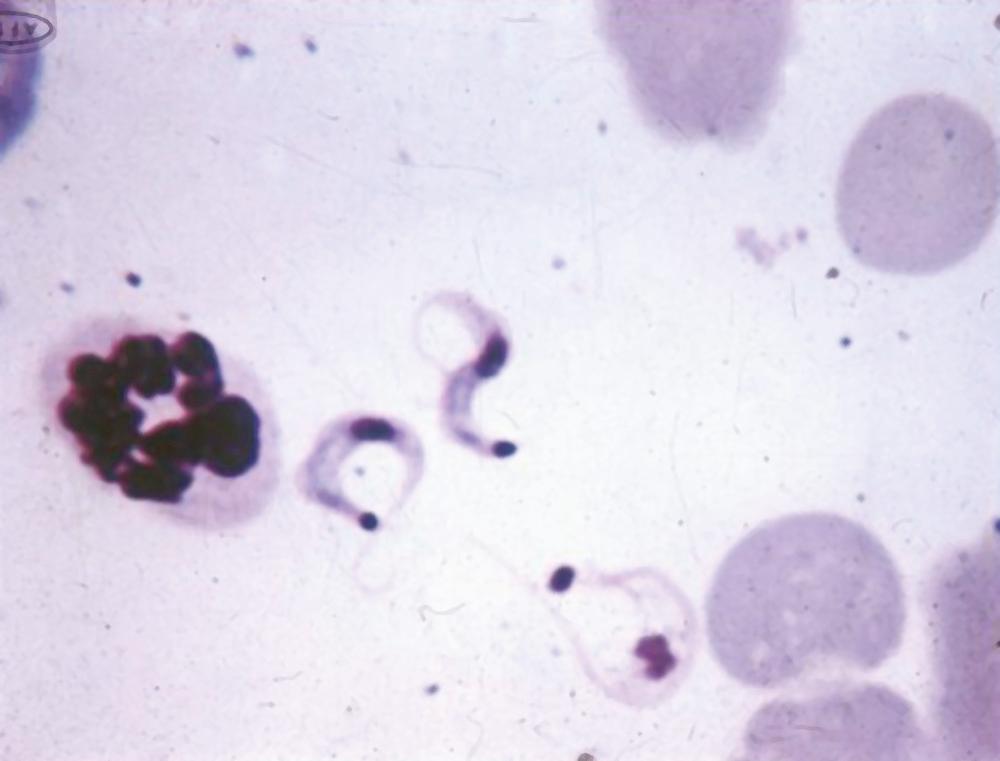
These vectors are found in large numbers in the wild, where they transmit the parasite among many mammalian species that constitute the natural reservoir; in endemic areas, they live in the nooks and crannies of substandard dwellings. The insects become infected by sucking blood from humans or other mammals that have circulating trypomastigotes ( Fig. 276.3 ). The ingested parasites multiply in the midgut of the insects as epimastigotes, which are flagellates of a distinct morphologic type, and in the hindgut transform into infective metacyclic trypomastigotes that are discharged with the feces at the time of subsequent blood meals. Transmission to another vertebrate host occurs when mucous membranes, conjunctivae, or breaks in the skin are contaminated with bug feces containing the infective forms. The parasites then enter a variety of host cell types and multiply in the cytoplasm after transformation into amastigotes. When multiplying amastigotes fill the host cell, they differentiate into trypomastigotes and the cell ruptures. The parasites released invade local tissues or spread hematogenously to distant sites, thus initiating further cycles of multiplication, largely in muscle cells, and maintaining a parasitemia infective for vectors.
Transmission of T. cruzi also occurs through blood transfusions, and historically this typically took place in cities when infected but asymptomatic migrants from endemic rural areas donated blood. Serologic screening of donated blood essentially has eliminated transmission by this route in most endemic countries. T. cruzi can also be transmitted by transplantation of organs obtained from chronically infected persons. Roughly 5% to 10% of infants born to T. cruzi –infected women have congenital Chagas disease. Although some of these infants have severe problems as a result of the infection, most are completely asymptomatic. Numerous laboratory accidents resulting in acute Chagas disease have occurred as a consequence of the ease with which highly infective parasite forms can be produced in the laboratory, but no reports of such events have appeared in recent years. Transmission of T. cruzi by ingestion of food and drink contaminated by infected vectors has also been reported.
In acute Chagas disease, the inflammatory lesion caused by T. cruzi at the site of entry is called a chagoma. Local histologic changes include intracellular parasitism of muscle and other subcutaneous tissues, interstitial edema, lymphocytic infiltration, and reactive hyperplasia of adjacent lymph nodes. Trypomastigotes released when infected host cells rupture can often be detected by microscopic examination of fresh blood. Muscles, including the myocardium, are the most heavily parasitized tissues. Myocarditis may develop in association with patchy areas of infected cells and necrosis.
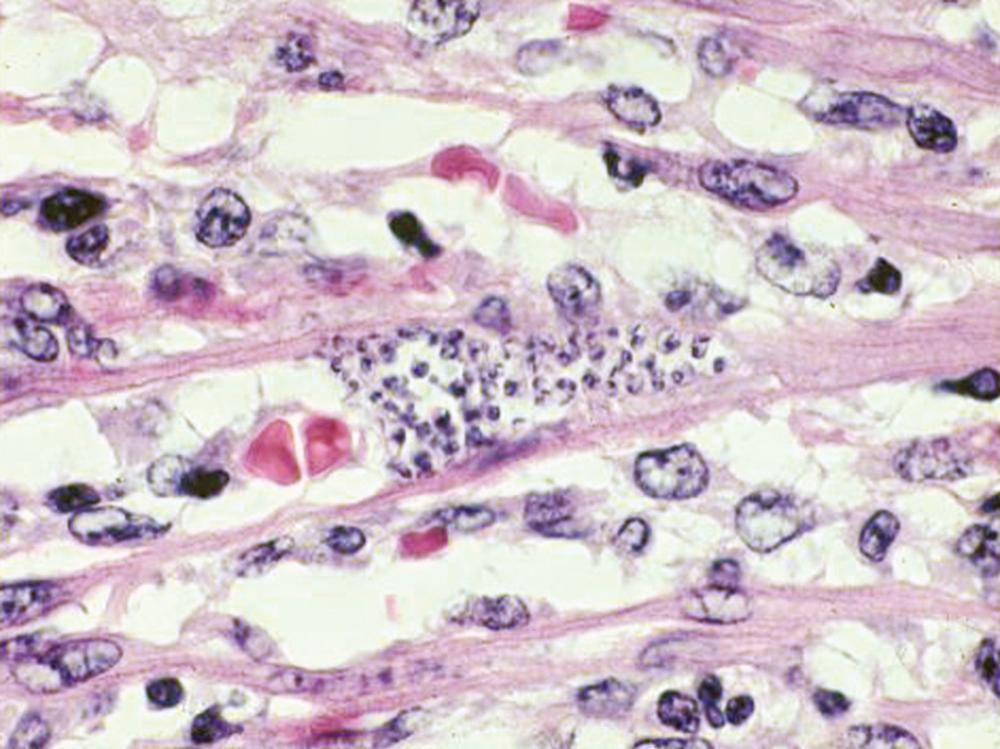
The characteristic “pseudocysts” seen in sections of infected tissues are aggregates of intracellular amastigotes ( Fig. 276.4 ). Lymphocytosis accompanies the high parasitemias of the acute illness, and mild elevation of transaminase levels is occasionally seen. In some patients, parasites can be found in the cerebrospinal fluid.
The heart is the organ most commonly affected in chronic Chagas disease. Gross examination of the hearts of chronic chagasic patients who died of heart failure reveals marked bilateral ventricular enlargement, often involving the right side of the heart more than the left. Thinning of the ventricular walls is common, as are apical aneurysms and mural thrombi. Widespread lymphocytic infiltration is present, accompanied by diffuse interstitial fibrosis and atrophy of myocardial cells. Parasites are rarely seen in stained sections of myocardial tissue, but studies using polymerase chain reaction (PCR) assays have demonstrated the presence of parasites in areas of focal inflammation.
Pathologic changes are also common in the conduction system of chronic chagasic hearts. Dense fibrosis and chronic inflammatory lesions most frequently involve the right branch and the left anterior branch of the bundle of His, but lesions of this type are found in other parts of the conduction system as well.
The striking features apparent on gross examination of the esophagus or colon of a patient with chronic Chagas disease of the digestive tract (megadisease) are the enormous dilatation and muscular hypertrophy of the affected organs. On microscopic examination, focal inflammatory lesions with lymphocytic infiltration are seen. A marked reduction in the number of neurons in the myenteric plexus is also apparent, and periganglionic and intraganglionic fibrosis in the presence of Schwann cell proliferation and lymphocytosis is found. In most patients the clinical effects of this parasympathetic denervation are confined to the esophagus or the colon, or both, but similar lesions have been observed in the biliary tree, the ureters, and other hollow viscera.
The pathogenesis of the cardiac and gastrointestinal lesions of chronic Chagas disease was debated for many years. Starting in the early 1990s, however, convincing evidence has accumulated indicating that the persistence of parasites in heart muscle stimulates a chronic inflammatory process that often results in rhythm disturbances and cardiomyopathy.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here