Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Rickettsia prowazekii is the only rickettsial species that can cause devastating, naturally occurring epidemics capable of killing a substantial proportion of human populations infested with body lice. Epidemics are associated with conditions that prevent bathing and washing of clothes in hot water, such as war and poverty, natural disasters such as earthquakes and floods, displacement of populations, jails, and lack of hygiene. Historical accounts of typhus epidemics in Mexico, associated with times of extreme poverty and hunger, have been correlated with drought through the study of tree-ring data to infer annual precipitation. A continued problem in impoverished, louse-infested populations, epidemic typhus threatens to reemerge as it did during the Civil War in Burundi, where an estimated 100,000 persons developed typhus in 1997.
Based on his observations of an Italian epidemic in 1528, epidemic typhus was described vividly by Hieronymus Fracastorius as a previously unknown, life-threatening disease in which a petechial rash appeared 4 to 7 days after the onset of fever and was accompanied by stupor and delirium. The word typhus is derived from the Greek typhos, meaning “smoky” or “hazy,” which describes the state of confusion accompanied by stupor. Luis de Toro described the same course and signs of an illness that occurred in soldiers on the Iberian Peninsula in 1557, also noting winter seasonality and association with contact with clothing of the ill. The term exanthematic typhus was proposed by Boissier de Sauvages in 1760 and differentiated the disease from typhoid. During an epidemic of typhoid fever in Philadelphia in 1836, Gerhard distinguished these diseases by the presence of intestinal lesions in typhoid patients. In 1909, Charles Nicolle experimentally established the fact that typhus was a transmissible infection with the human body louse as its vector. Investigations from 1910 to 1922 by Ricketts, von Prowazek, da Rocha-Lima, and Wolbach used microscopy, xenodiagnosis in lice, and histochemistry to establish that the agent was a bacterium that proliferated in human endothelial cells and louse gut epithelium but could not be cultured axenically.
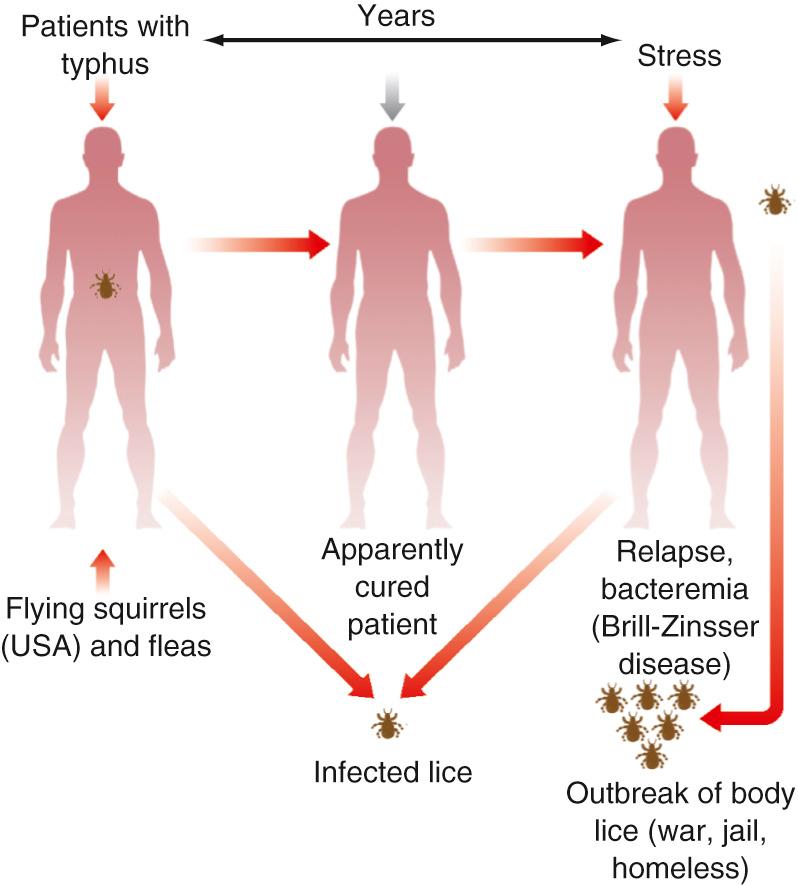
In 1896 and 1910, Nathan Brill described a series of patients in New York with a febrile illness that was shown by Hans Zinsser in 1934 to be a rickettsial infection. Zinsser hypothesized correctly that the illness is a recrudescence of long-latent R. prowazekii, now called Brill-Zinsser disease. The distinction of Rickettsia typhi as a separate agent that causes an endemic, clinically similar disease transmitted to humans by fleas from a zoonotic cycle involving rats was established by the work of Neill (1917), Mooser (1928), Maxcy (1929), and Dyer (1931). The feared specter of epidemic louse-borne typhus receded with the following: (1) the success of a killed Rickettsia vaccine in preventing the deaths of allied soldiers during World War II; (2) the effectiveness of insecticides in curtailing epidemics by louse control; and (3) the effective treatment of illness with tetracyclines and chloramphenicol. Currently, epidemics of typhus are increasingly recognized, lice resistant to various insecticides have been detected, antibiotic-resistant rickettsiae have been developed, and aerosol-transmitted, weaponized R. prowazekii has emerged as a biothreat. Lack of direct human-to-human transmission is a limitation of use as a bioterror weapon. R. prowazekii has been isolated from Amblyomma and Hyalomma ticks in Ethiopia and in Amblyomma tenellum ticks in Mexico. The presence of R. prowazekii DNA has also been found within Ixodes ricinus ticks in the Netherlands. Moreover, in 1975, R. prowazekii was identified in a highly prevalent zoonotic cycle in flying squirrels in the eastern United States.
The causative agent of louse-borne typhus, R. prowazekii , is an obligately intracellular, small (1 × 0.3 µm) coccobacillus. Its 1.1-Mb genome has undergone considerable reduction, contains a subset of the genes of Rickettsia conorii , and exhibits little variation among isolates . R. prowazekii’s conserved structure necessitates its ability to manipulate host cell processes, as demonstrated for small GTPases. Many biosynthetic functions are provided by the organism's milieu in the resource-rich host cell cytosol, to which it has adapted by means of evolutionary selection for transport mechanisms for adenosine triphosphate (ATP), amino acids, phosphorylated sugars, and triose phosphate. Its gram-negative cell wall contains an abundant 135-kDa, immunodominant, tetragonally arranged, S-layer surface protein; lipopolysaccharide; and peptidoglycan. Differential expression of genes occurs with its infection of mammalian or arthropod host cells. It possesses an extracellular dormant form that remains infectious in louse feces for months or more. Technical barriers to the genetic manipulation of R. prowazekii have limited extensive study of its virulence mechanisms, but development of insertional mutants, site-directed knockouts, and maintenance of inserted plasmids offer tools for more extensive study. To date, only phospholipase D and phospholipase A have been identified as virulence factors of R. prowazekii.
Typhus affected the outcomes of wars from the 1500s until the end of the 19th century. During the Russian campaign of 1812, typhus was responsible for the deaths of many of the 700,000 troops of Napoleon. During World War I, the Bolshevik revolution, and its aftermath, an estimated 30 million cases of typhus occurred in the Soviet Union alone, with 3 million deaths. During World War II, epidemics of typhus occurred in eastern Europe, North Africa, concentration camps, and southern Italy, where dichlorodiphenyltrichloroethane (DDT) was used against lice to abort an epidemic in Naples in 1944.
R. prowazekii is transmitted between patients by the human body louse (Pediculus humanus corporis) , which is strictly adapted to humans, lives in the clothes, and takes a blood meal five times daily. Lice become infected while feeding on the blood of rickettsemic patients. R. prowazekii enters the louse gut epithelial cells and replicates by means of binary fission until the massively infected cells burst and are released into the louse feces 5 to 7 days after ingestion. Lice are not adapted to an elevated body temperature and leave febrile patients for a new host. Rickettsia -laden louse feces are deposited on the skin and clothes and are introduced into the new host by being scratched into the louse-bitten skin or being rubbed into mucous membranes such as the conjunctiva; they are also transmitted through inhalation.
Persons who recover from typhus fever remain latently infected and are susceptible to reactivation of the infection and rickettsemia, which can infect body lice and ignite an epidemic under circumstances of crowding, extreme poverty, cold climate, and poor hygiene, which can lead to a high prevalence of louse infestation ( Fig. 189.1 ). The louse population can expand rapidly (11% daily). Lice are currently prevalent in poor countries and found on homeless persons in developed countries. In France, 35% of members of the homeless population have lice. Risk factors for reactivation of latent infection in humans have not been determined, but waning immunity, poor nutrition, alcoholism, and stress have been hypothesized as potentially important.
Typhus is endemic in the Peruvian Andes, Burundi, and Rwanda. The most recently documented outbreak occurred in a youth rehabilitation center, where the recorded attack and case-fatality rates were 10% and 3.5%, respectively. Cases have been diagnosed in Russia, in patients from Morocco, Algeria, and Senegal, and in a homeless person in France. Given the geographic distribution of latent typhus infection from known historic epidemics, the neglect with which this disease has been handled, and the limited availability of laboratory diagnostic methods, the current epidemiology of louse-borne typhus, particularly in marginal populations of mountainous areas in Asia, South and Central America, and Africa, is unknown.
An extrahuman reservoir of R. prowazekii exists in a large portion of southern flying squirrels (Glaucomys volans), which are distributed from Florida to Maine and westward to Minnesota and eastern Texas. In stark contrast with the louse-human cycle, in which 15% of humans die and 100% of lice develop rickettsial destruction of the intestine with extravasation of the red blood meal into the hemocele and death, R. prowazekii causes subclinical rickettsemia in flying squirrels and no ill effects on the squirrel's own species of lice and fleas. Infections are apparently transmitted to humans by flying squirrel fleas or mucous membrane or inhalation exposure to the feces of the flea or louse. These illnesses occur mainly during winter, when flying squirrels enter buildings. Clusters of cases have occurred in association with flying squirrel–infested attics and walls of dwellings. A case of recrudescent typhus has been described in a patient 11 years after R. prowazekii infection related to close contact with flying squirrels. The role of ticks in transmitting R. prowazekii is currently unknown, although this source would appear to be a potential explanation for a case of R. prowazekii originating in southern Texas.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here