Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Before attempting cannulation of the biliary tree for standard endoscopic retrograde cholangiopancreatography (ERCP), the appropriate equipment must be assembled (see Chapter 20 ). At baseline that equipment should include :
Side-viewing duodenoscope with biopsy channel of at least 3.2 mm to 4.2 mm
Guidewire , :
Hydrophilic or Hydrophilic tipped
Diameter: 0.018, 0.025, and 0.035 inches. The 0.035 inch is most commonly used, but the 0.025 size can be useful in smaller papillae and the 0.018 inch in the pancreatic duct. Although these have the advantage of being able to access narrower orifices, their smaller size makes them more pliable and more difficult to control.
Tip: angled or straight-tipped
Length:
Long-wire (420–480 cm) – This allows for universal exchange capabilities across all devices and brands and is also needed for complex rendezvous endoscopic ultrasound (EUS)–assisted cholangiopancreatography and single-operator cholangioscopy. It, however, does not allow for control by the endoscopist and instead relies heavily on excellent coordination with communication with a trained endoscopy assistant.
Short-wire system (184–270 cm) – Allows exclusive control of the guidewire by the endoscopist because it can be locked in place both at the level of the elevator and externally at the biopsy port to allow for easy exchange of devices over the wire.
Standard cannulation catheters (typically have two to three lumens to facilitate concurrent passage of a guidewire and contrast injection)
OR:
Sphincterotome – This includes an electrosurgical cutting wire at the distal end of the catheter. This is used primarily to perform a sphincterotomy. Additionally, applying tension on the cutting wire results in bowing of the sphincterotome which aligns its axis to facilitate cannulation.
Access (pre-cut) papillotomy catheters – These are used for precut sphincterotomies or biliary fistulotomies. The most commonly used of these catheters is the needle-knife (NK) , which has a retractable electrosurgical cutting wire.
There are two main techniques that can be used to cannulate the biliary system: traditional contrast-assisted biliary cannulation and guidewire-assisted biliary cannulation.
In traditional contrast-assisted biliary cannulation, the catheter is engaged with the papillary orifice and contrast is injected to delineate the trajectory and pathway of the bile duct. Afterwards, the catheter and/or a guidewire can be inserted directly into the bile duct. One of the main risks of this is inadvertent injection of contrast into the pancreatic duct, which increases the risk for post-ERCP pancreatitis (PEP; see Chapter 55 ).
This involves confirmed cannulation of the bile duct with a guidewire before the injection of any contrast. There are two methods to achieve this:
Touch guidewire technique: The tip of the catheter can be engaged with the biliary orifice and then the guidewire can be advanced into the common bile duct under fluoroscopic guidance. This technique may be associated with higher cannulation rates.
No-touch technique: Alternatively, the guidewire can be advanced one to two mm beyond the tip of the catheter and then advanced directly through the papillary orifice oriented towards the bile duct under fluoroscopy.
Guidewire-assisted biliary cannulation has been found to be superior to the contrast-assisted biliary cannulation technique because it results in higher rates of primary biliary cannulation, less need for precut sphincterotomy, and a lower risk of PEP.
Difficult biliary access is defined as the inability to achieve selective biliary cannulation by standard ERCP techniques within 10 minutes or five cannulation attempts or failure of access to the major papilla. Repeated attempts at biliary cannulation are independently associated with an increased risk of PEP. There are a few strategies that have been established to facilitate biliary cannulation in such instances.
In the “double-wire technique,” a guidewire is deliberately placed in the pancreatic duct while the biliary cannulation device is preloaded with a second guidewire to reattempt biliary cannulation. Theoretically, the pancreatic guidewire assists by straightening the common channel and common bile duct (CBD), thus facilitating biliary cannulation. Furthermore, the direction of the pancreatic wire exiting the papilla on endoscopy may provide anatomic cues regarding the optimal axis for biliary cannulation. Although this technique has been associated with high rates of cannulation in difficult situations, it has not been conclusively shown to be superior to standard cannulation and may be associated with an increased risk of PEP. , Placement of a pancreatic duct stent facilitates biliary duct cannulation and also reduces the risk of PEP by ensuring pancreatic duct drainage and by minimizing further inadvertent entry into the pancreatic duct on repeated attempts at biliary cannulation. ,
Access “precut” sphincterotomy refers to the technique of incising the papilla before obtaining biliary access. Precutting can be a useful technique to achieve selective bile duct cannulation when standard approaches fail. The needle-knife (NK) catheter is typically used for this procedure. The NK catheters have a retractable bare electrosurgical cutting wire that extends from the tip of the catheter. The exposed needle can then be inserted into the papillary orifice, and the cut is directed upward in the axis of the bile duct, generally in the 11 to 12 o’clock position. Another variation of this technique involves using the NK to begin an incision above the ampullary orifice to directly access the CBD by creating a biliary fistula (“fistulotomy”).
Precut sphincterotomy has been associated with biliary cannulation rates exceeding 90%. , Nevertheless, precutting is not without risk because the incision is performed without the guidance of a wire within the duct. Earlier experience with precut sphincterotomy demonstrated an increased risk of complications such as PEP, bleeding, and perforation. , Nevertheless, given that precut sphincterotomy has traditionally been used as a second option or last resort during difficult cannulation, it has been suggested that precut sphincterotomy may be a surrogate marker of difficult cannulation and not an independent predictor of PEP. , More recent data have indicated no overall increased risk of complications when compared with persistent attempts at cannulation and may even be associated with a lower risk of PEP compared with repeated attempts at cannulation. , , , , Some centers have modified this technique and perform a “shallow” precut sphincterotomy, which uses only three mm of the NK, which has demonstrated overall lower complication rates. These centers have even advocated for this to be used as the first-line approach for all ERCPs.
Overall, there is good evidence to recommend early precut sphincterotomy in cases of challenging biliary cannulation but not enough to recommend this universally as the first-line approach for all ERCPs. It is important to emphasize that this approach should be performed by an experienced biliary endoscopist familiar with the nuances and technical aspects of this approach.
Transpancreatic precut (transeptal) sphincterotomy (TPS) for biliary access was first described by Goff and is thus sometimes known as the Goff technique. In this technique, after selective cannulation of the pancreatic duct, precut sphincterotomy is performed by cutting the septum between the pancreatic and bile duct with the standard sphincterotome directed cephalad toward the bile duct. Since its introduction, several studies have demonstrated high rates of bile duct cannulation without increased complication rates compared with other precut techniques used for difficult biliary cannulation, when used in expert hands. , To reduce the risk of PEP after TPS, it is further recommended that a pancreatic stent be left in situ.
The decision on the type of precut technique used can be determined based on papilla morphology (e.g., for protuberant papilla where NK fistulotomy may be preferential) or based on the presence of inadvertent pancreatic duct cannulation (where transpancreatic precut sphincterotomy may be preferred). It is again crucial that any of these precut techniques be performed by an expert endoscopist.
ERCP in patients with surgically altered anatomy can be technically difficult. There are two main challenges that need to be overcome to successfully complete the procedure. The first challenge is to reach the papilla or bilioenteric anastomosis in altered luminal anatomy. The second challenge is to be able to cannulate and perform the intended intervention from an altered position with the available endoscopes and accessories.
In Bilroth II anatomy, the distal stomach is resected and an end-to-side gastrojejunostomy has been created. From the gastrojejunal anastomosis, an afferent limb leads toward the proximal duodenum, whereas the efferent limb leads to the distal small bowel. In the Braun variation, there is additionally a side-to-side jejuno-jejunostomy between the afferent and efferent limbs. ERCP is performed by intubating the afferent limb and cannulating the papilla from a caudal angle. Although the afferent limb may be short, identification of the limb and navigating through the sharp angulation of this limb can be challenging with the conventional side-viewing duodenoscope, and even more so with the Braun variation. An alternative is to perform the entire procedure with a forward-viewing gastroscope or pediatric colonoscope. This can be further aided by placing a transparent cap on the tip of the endoscope, which facilitates navigation through the tortuous afferent limb and stabilizes the scope position for selective biliary cannulation. Other options for afferent limb intubation and biliary cannulation include use of balloon-assisted enteroscopy, spiral enteroscopy, or use of an anterior oblique-viewing endoscope. , There are limitations, however, to using these endoscopes because they lack elevators, which aid with biliary cannulation, and there are less ERCP-directed accessories available.
When using a duodenoscope, the papilla is usually visible en face upon reaching the second portion of the duodenum. From this position, the papilla appears rotated by 180 degrees, and as such biliary cannulation generally proceeds toward the five o’clock position instead of the 11 o’clock position used for standard ERCP. Hence standard straight cannulas may be preferable for selective bile duct cannulation compared with the upward-curved papillotomes. Alternative devices that have been explored to facilitate bile duct access in these patients include the Bilroth II sphincterotome with a downward “reversed bow” curved wire, rotatable sphincterotomes, pull-type sphincterotomes, or triple-lumen needle-knives. In expert hands these have success rates of over 90%. , ,
Despite the inherent challenges of Bilroth II anatomy, the success rates for ERCP are similar amongst the different techniques used. In a large single-center series of 713 patients, the success rate for afferent limb intubation and biliary or pancreatic duct cannulation using the duodenoscope was 87% and 94%, respectively. Similarly, ERCP success rates with overtube-assisted enteroscopy (single-balloon [SBE], double- balloon [DBE], spiral enteroscopy) exceeded 90% in patients with Bilroth II anatomy. , A systematic review comparing the different techniques has reported rates of access and selective biliary cannulation exceeding 95% for both side-viewing and forward-viewing endoscopy.
In additional to the potential risks associated with conventional ERCP, Bilroth II anatomy increases the risk of adverse events, including perforations at the gastrojejunal anastomosis or within the afferent limb itself. The risk of this has been noted to be marginally higher with side-viewing endoscopy. ,
At this time, some society guidelines recommend the side-viewing endoscope as a first option with forward-viewing endoscopes (gastroscope, pediatric colonoscope, and balloon enteroscope) as the second choice in cases of failure. Given that there are pros and cons to both side-viewing conventional endoscopes and forward-viewing scopes, the endoscopist should be familiar with multiple techniques and be prepared to change strategies on a case-by-case basis, depending on the intraprocedural findings.
Roux-en-Y reconstruction has been used in bariatric gastric bypass, gastric resections, pancreaticoduodenectomy (Whipple procedure), resection of biliary malignancies, reconstruction of benign biliary strictures, and in some cases of liver transplant (see Chapters 42 , 117A , and 119A ).
Briefly, in patients with Roux-en-Y anatomy, the jejunum is divided into two segments close to the ligament of Treitz. The distal segment of this is anastomosed to the stomach or gastric remnant and forms the “Roux” limb. A jejunojejunal anastomosis is then formed by anastomosing the proximal segment further down on the distal segment. Proximally, this connects to the duodenum and the biliary system and forms the “biliopancreatic” limb.
Before attempting ERCP, a comprehensive review of the patient’s operative reports, imaging, and prior procedures are necessary, and a discussion with their surgeon would be ideal to develop a strategic approach. Emphasis should be placed on the type of resection and anastomosis, length of both limbs, and whether there is a native papilla or other type of bilioenteric anastomosis.
Roux-en-Y anatomy poses a major challenge for ERCP, given the length of bowel that must be traversed to reach the papilla or bilioenteric anastomosis. This is often prohibitive for duodenoscopes, and again the altered orientation makes cannulation difficult. To improve success rates, various combinations and adaptations of forward-viewing and side-viewing scopes have been employed.
Push enteroscopy (using a forward-viewing enteroscope or push colonoscope) can be an alternative when the biliopancreatic limb cannot be reached with the duodenoscope. , Deep enteroscopy platforms, including SBE, DBE, and spiral enteroscopy, were developed to allow access to the distal small bowel and have quoted success rates of reaching the papilla of up to 86%. , , Although the longer enteroscopes may facilitate navigation through the surgically altered anatomy compared with the duodenoscope, this advantage comes with several limitations. First, the lack of a side-viewing perspective and an elevator can potentially make cannulation more difficult. Second, there are limited accessories specifically designed to use with the longer endoscopes to perform diagnostic and therapeutic interventions. The use of larger-diameter biliary stents can be limited by the size of the working channel of the endoscope, and even smaller-caliber accessories may be difficult to advance through the channel when the longer endoscope is torqued or looped in the surgically altered bowel. Lastly, these procedures can be long (median ranging from one to three hours), with the increased risk of prolonged general anesthesia. Success rates of device-assisted enteroscopy range from 70% to 86% across multiple studies, with the main limiting factor being the enteroscopy, as cannulation rates are upwards of 85% once the papilla or bilioenteric anastomosis is reached.
Transoral ERCP in patients with Roux-en-Y gastric bypass (RYGB) can be challenging given the relatively longer Roux and biliopancreatic limbs that must be traversed. Hence, alternative access routes through the remnant stomach directly to the native papilla have been explored.
Percutaneous Transgastric ERCP – This involves creation of a gastrostomy to the excluded stomach and can be done via open surgery or via a percutaneous gastrostomy placed by interventional radiology, but is most commonly performed laparoscopically. In laparoscopic-assisted transgastric ERCP, a 15-mm trocar is inserted into the excluded stomach is used to pull it adjacent to the abdominal wall, and is secured with a purse-string suture. The duodenoscope can then be inserted through the trocar into the excluded stomach and advanced in an anterograde manner to the papilla. If subsequent ERCPs are anticipated, a gastrostomy tube may be inserted to maintain and allow for maturity of the gastrostomy tract over two to four weeks. This technique has quoted success rates of up to 100% and low rates of severe adverse events, even in lower volume community hospitals. It has the advantage of facilitating use of standard side-viewing duodenoscopes and ERCP accessories, which improves success rates of cannulation and interventions when compared with enteroscopy-assisted ERCP. , Additionally, laparoscopy allows for diagnosis and management of internal hernias, adhesions, and cholecystectomy if indicated in the same setting (see Chapter 24 ). Coordination between gastroenterology and surgical teams and maintenance of a sterile field are the main challenges with this approach.
As an alternative to this multidisciplinary approach, a technique using percutaneous- assisted transprosthetic endoscopic therapy has been described. , In this technique, an enteroscope is advanced transorally into the excluded stomach, followed by the creation of a percutaneous endoscopic gastrostomy. The gastrostomy tract is then dilated to allow for the placement of a fully covered esophageal self-expanding metal stent (SEMS). A duodenoscope can then be advanced through the stent to perform antegrade ERCP. This has just been described in a single-center case series, however, and has not been widely adopted.
EUS-Directed Transgastric ERCP (EDGE) – This is another option for patients with RYGB (see Chapter 22 ). It involves the creation of a fistula between the gastric pouch or proximal Roux limb and the excluded stomach under EUS guidance, using a lumen-apposing metal stent (LAMS). A duodenoscope is then inserted orally and advanced through this fistula to the excluded stomach, and the ERCP is completed in the standard manner. There is a risk of acute stent migration when attempting ERCP in the same session that the LAMS is placed, which can lead to a free perforation of both the gastric pouch/roux limb or the excluded stomach. Because of this risk, some centers perform a “staged EDGE” whereby the ERCP is performed in a separate session after placement of the LAMS. , However, if a single-stage EDGE is desired, such as in cases of acute cholangitis, some investigators have reported success with anchoring the LAMS in place using either an over-the-scope clip (OTSC) or with endoscopic suturing. , This procedure has technical and clinical success rates ranging from 91% to 100%, with main adverse events being related to bleeding, perforation, and stent dislodgement. , There is also concern for weight regain because of the creation of the gastro-gastric fistula, although weight loss has actually been more common in the short term. , The fistula often closes spontaneously after removal of the LAMS or can be closed by endosuturing or by using an OVESCO if necessary , . The EDGE procedure has higher clinical and technical success when compared with enteroscopy-assisted ERCP but does have more adverse events. When compared with the laparoscopic transgastric approach, the EDGE procedure has similar success and adverse event rates but has shortened procedure times and shorter hospital stays. ,
EUS-Directed Transenteric ERCP (EDEE) – This technique has been recently described and involves the creation of an enteroenteric anastomosis to facilitate ERCP in non–RYGB surgical anatomy (e.g., Whipple, hepaticojejunostomy, Bilroth II, and duodenal switch). The pancreaticobiliary limb is first identified either by enteroscopy or via direct EUS puncture from the stomach, duodenum, or jejunum and filled with a solution of contrast, saline, and methylene blue. A target is then identified where the distance between the two luminal walls is less than 1 cm and there are no intervening vascular structures on Doppler. EDEE is then performed using either a 15 mm or 20 mm LAMS. A standard duodenoscope can then be passed through the LAMS to facilitate completion of the ERCP. In this study, 22% of ERCPs were done in the same session as the LAMS placement but did not demonstrate any increased risk of adverse events compared with those that were done in a separate session. Additionally, despite no stent fixation being done for any of the procedures, there were no occurrences of stent migration. This procedure has technical and clinical success rates of up to 100% and 94.4%, respectively, with an adverse event rate of 5.6%. This procedure provides an additional option to access the pancreaticobiliary region in patients with complicated surgically anatomy in institutions where this expertise is available.
In summary, selection of the appropriate technique for biliary access in patients with surgically altered anatomy should be individualized and would likely involve a combination of methods and endoscopic tools based on patient factors and operator’s expertise.
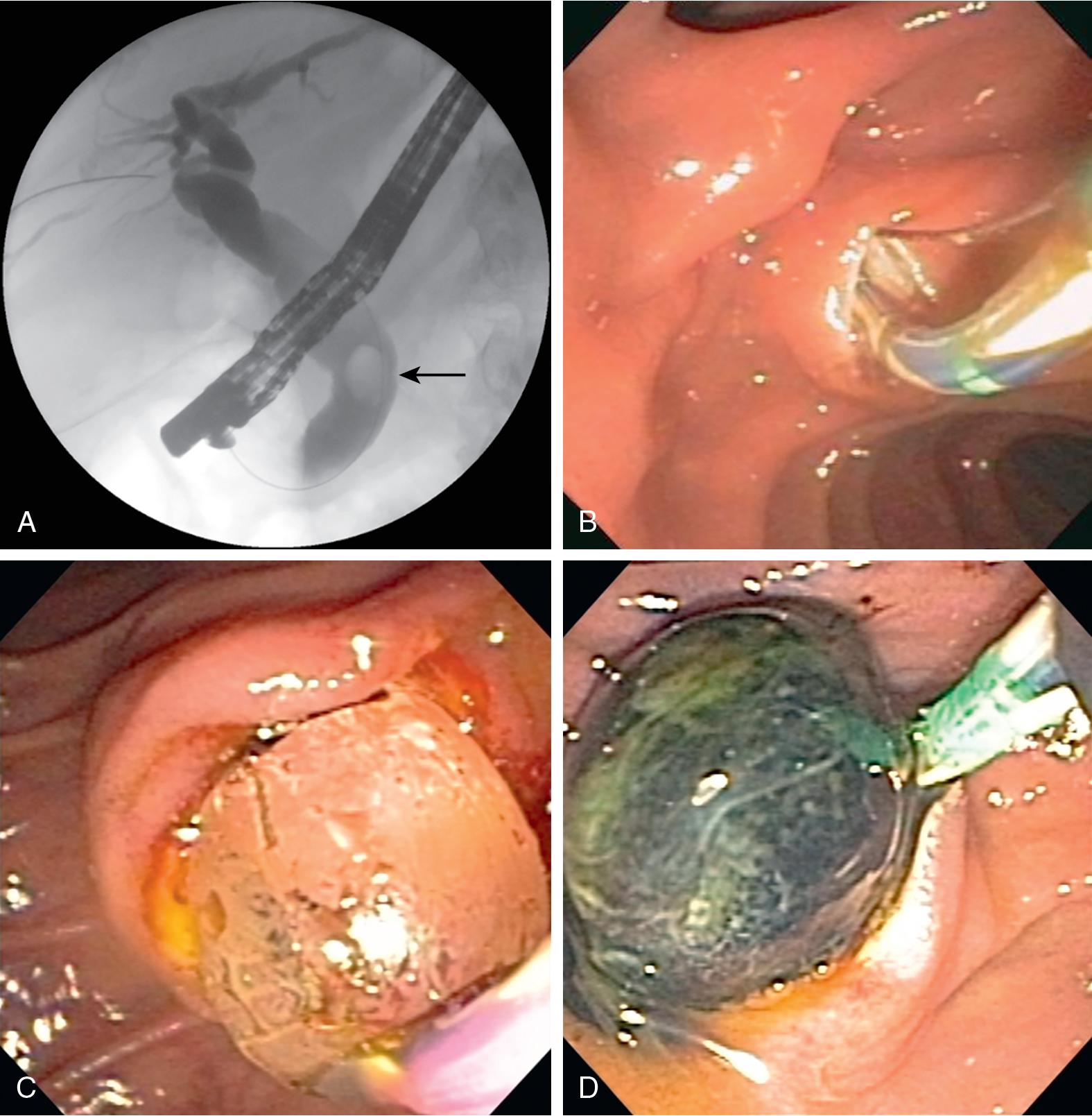
ERCP success rates for biliary and pancreatic duct decompression can be anywhere from 76% to 98% depending on operator expertise, alterations in anatomy, and the etiology of biliary obstruction (see Chapter 20 ). Alternative methods of biliary decompression have traditionally included percutaneous transhepatic drainage or surgery (see Chapters 20 , 31 , 42 , and 52 ). With the advancement of curvilinear-array echoendoscopes and peripheral devices, EUS-guided biliary drainage (EUS-BD) has become increasingly reported either via an intrahepatic (hepaticogastrostomy) or extrahepatic (choledocho-duodenostomy) approach ( Fig 30.1 A). This approach has been found to have better clinical success outcomes and less adverse events when compared with percutaneous drainage.
EUS-BD has conventionally been used as a second-line therapy when ERCP is unsuccessful. , Nevertheless, there are increasing reports, including two randomized controlled trials (RCTs), of EUS-BD being used as the primary procedure for biliary decompression with comparable success rates and decreased adverse event rates compared with ERCP. , In cases of malignant biliary obstruction, these techniques have a pooled technical and clinical success rate of 95% and 97%, respectively, and adverse event rates (mainly biliary peritonitis and cholangitis) of 19%. At this time, given inter-institution variations in access to EUS, it cannot be universally recommended as the initial procedure for biliary decompression.
There are three main EUS-guided techniques: rendezvous, anterograde stenting, and direct transluminal drainage, which are described in detail in the following sections.
The EUS rendezvous technique was first described by Mallery et al. in 2004 and involves EUS-guided wire placement into the bile duct in an antegrade fashion to facilitate subsequent retrograde biliary cannulation (see Chapter 20 ). The point of biliary duct entry (intrahepatic vs. extrahepatic) depends on accessibility and which route facilitates wire manipulation. Nevertheless, accessing the extrahepatic bile ducts from the second portion of the duodenum has been associated with a higher success rate. Whether it is through an intrahepatic or extrahepatic approach, a therapeutic linear echoendoscope is used to visualize the bile duct from the stomach or small intestine. Once an avascular plane has been identified using Doppler ultrasonography, an EUS needle is advanced into the bile duct (see Fig 30.1 B). Bile is aspirated, and contrast is injected to confirm position inside the bile system. A hydrophilic guidewire is then advanced antegrade through the EUS needle and into the bile duct and manipulated across the papilla. The needle is first exchanged out over the wire, followed by an exchange of the echoendoscope over the wire. The duodenoscope is then inserted transorally adjacent to the indwelling wire and advanced to the duodenum, with visualization of the wire traversing the papilla. The distal end of the indwelling guidewire can be grasped with forceps or snare and withdrawn through the accessory channel, and a cannulation catheter can be backloaded over the guidewire and re-advanced to the papilla. Alternatively, biliary cannulation can be accomplished in the standard retrograde fashion adjacent to the indwelling wire. Overall success and complication rates of the EUS-guided rendezvous technique are quoted as 80% to 86% and 10% to 15%, respectively. ,
This technique is useful in cases where conventional or even rendezvous ERCP cannot be performed because of inaccessibility of the papilla (e.g., because of proximal luminal obstruction or altered surgical anatomy, such as in RYGB). Similar to the previously detailed rendezvous technique, the bile duct is identified and accessed from the stomach or small intestine. The transmural tract is then dilated over the guidewire with either a balloon catheter or bougie to allow anterograde advancement of a stent into the bile system with subsequent deployment across the biliary obstruction and through the papilla. Success rates from various case series range from 57% to 100%, and complication rates range from 0% to 6%. ,
In cases where anterograde or rendezvous techniques for transpapillary drainage cannot be accomplished (e.g., impacted biliary calculi, papillary stenosis, and tumor infiltration), an EUS-guided transluminal approach can facilitate biliary decompression.
In the original variations of this procedure, the bile duct was accessed under EUS guidance, with placement of a guidewire similar to described above. The fistula tract was then dilated, and a stent was deployed over the guidewire (see Fig 30.1 C and D). This technique was initially performed with plastic stents. It was subsequently performed with SEMS and then LAMS, in an attempt to improve patency and reduce leakage and migration. More recently, with the introduction of electrocautery-enhanced LAMS (ECE-LAMS), this procedure can be reduced to a single step wherein the biliary duct is punctured with the ECE-LAMS system and the stent is deployed without the need for guidewire placement or fistula tract dilation. This procedure has been associated with technical and clinical success rates of 88% to 93% and 97% to 100%, respectively, with overall adverse event rates (mostly mild) of up to 36%.
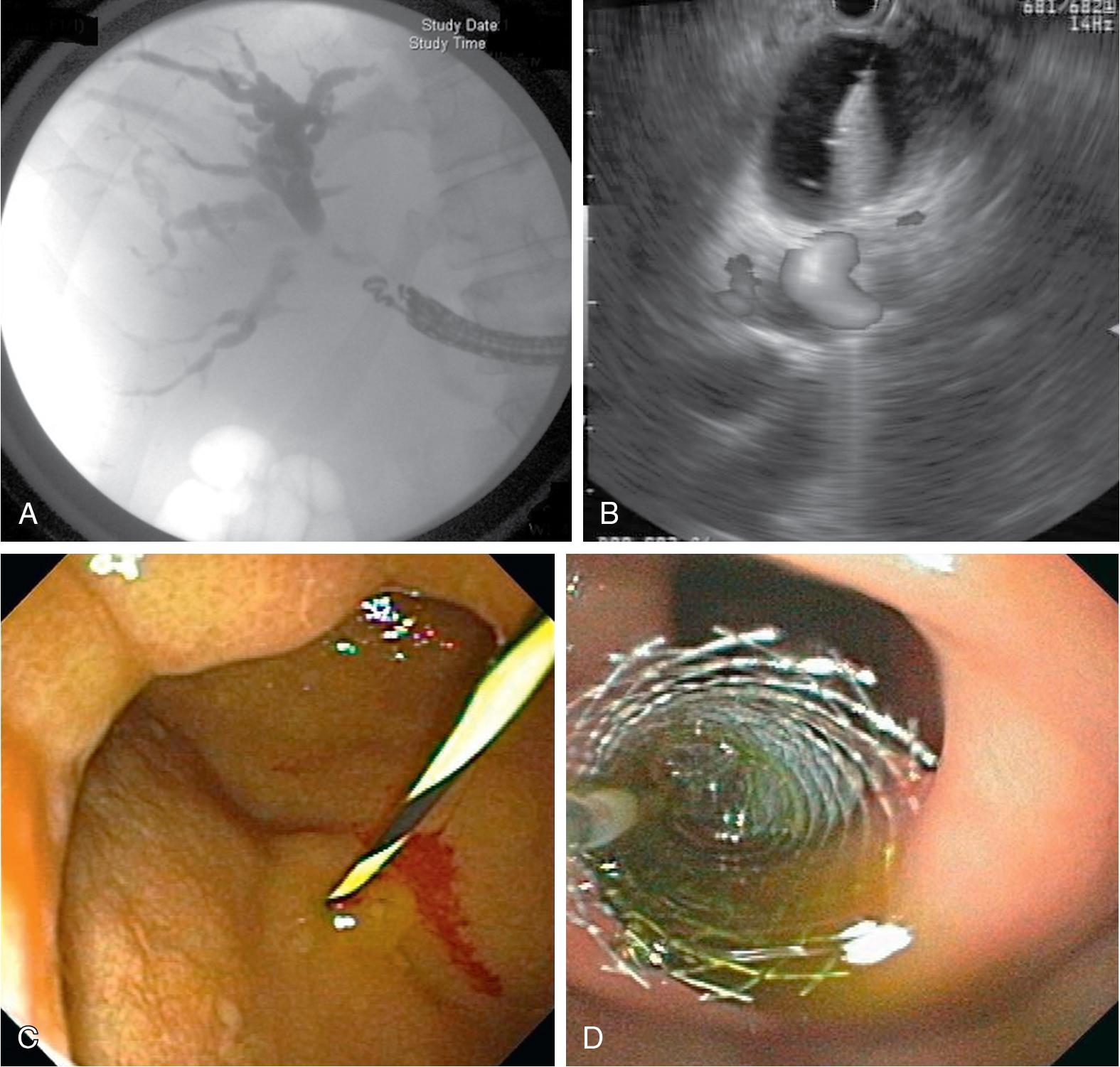
Complications of cholelithiasis accounts for over 300,000 hospital admissions annually at a cost of over four billion dollars. Choledocholithiasis is present in 10% to 15% of patients with symptomatic gallstones and can result in significant morbidity and mortality related to biliary obstruction, ascending cholangitis, and pancreatitis. Up to one-third of CBD stones may pass spontaneously, but for those that do not, ERCP with biliary sphincterotomy and stone extraction is considered the first-line management ( Fig 30.2A ; see Chapter 37 ).
Endoscopic sphincterotomy (EST) aims at opening the terminal part of the CBD by cutting the papilla and sphincter muscles. The basic technique of sphincterotomy has not changed significantly since its initial description. The standard sphincterotome, the Erlangen “pull-type” model, consists of a catheter containing an electrosurgical cutting wire exposed 20 to 25 mm near the tip of the sphincterotome. The leading tip distal to the wire, the “nose,” is five to ten mm in diameter. Once deep biliary cannulation has been achieved, the sphincterotome is retracted slowly, until one-fourth to one-half of the wire length is exposed outside the papilla. The sphincterotome is slightly bowed so the cutting wire is in contact with the roof of the papilla.
The incision is made by upward lifting of the sphincterotome with pressure against the papillary roof, but not excessively, to avoid a rapid large incision (“zipper”; see Fig 30.2 B). It is recommended that a current mode with alternating cutting and coagulating phases (e.g., Endocut) be used because this reduces the rates of uncontrolled cutting (“zipper”), PEP, and postsphincterotomy bleeding. , The size of the sphincterotomy varies on a case-by-case basis and can be limited by the length of the intraduodenal portion of the CBD. In general, the sphincterotomy should be of adequate size to allow the passage of the stone in the CBD. The size of sphincterotomy can be gauged by the ability to move the bowed sphincterotome across the opening, by passing an inflated balloon catheter through the site, and/or by eliminating the tapering or “pinch” of the intra-ampullary bile duct seen on fluoroscopy.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here