Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Access video content for this chapter online at Elsevier eBooks+ ![]()
Our first liposuction was undertaken in 1987, but it was not until 1993 that a more detailed treatment protocol was established for arm lymphedema. Liposuction for leg lymphedema was established 5 years later. Initially the “dry” technique was used. The introduction of use of a tourniquet and tumescence has made liposuction a safe procedure, and there is no longer any need of blood transfusions. Liposuction is the only method to completely reduce a chronic non-pitting lymphedema. The result is maintained with compression garments.
Typical indications:
All primary and secondary lymphedema with an excess volume of 10%.
Minimal pitting: 4–5 mm in arms and 5–6 mm in legs.
No active cancer
No effect of conservative treatment
No wounds
No age limit
Liposuction in the only method to completely reduce a non-pitting chronic lymphedema where the excess volume is dominated by adipose tissue. The incidence of postmastectomy arm lymphedema varies between 13% and 52%, depending in part on whether axillary lymph nodes have been removed and postoperative radiation has been given. The sentinel node technique had decreased the incidence of postoperative lymphedema to an estimated incidence of approximately 6%–8%. Immediate lymphatic reconstruction shows an incidence of 9.1% at short-term follow-up. For legs, primary lymphedema is a relatively rare condition, affecting 1 in 100,000 people, while secondary lymphedema is more common. One in 1000 people in the US will experience secondary lymphedema.
The outcome of the surgical procedure as well as of the radiation of the tissue often results in destruction of lymphatic vessels. When this is combined with the removal of lymph nodes and tissue scarring, the lymphatic vessels that remain are likely to be unable to remove the load of lymph. The remaining lymph collectors become dilated and overloaded due to an increase in endolymphatic pressure, and their valves become incompetent, preventing the lymphatics from performing their function. This failure spreads distally until even the most peripheral lymph vessels, draining into the affected system, also become dilated. The smooth muscle cells in the vessel walls become fibrotic leading to narrowing or occlusion of the lymphatics, which further affects the lymph transport.
In a parallel process, the cells of the mononuclear phagocytic system of the mesenchymal tissues begin to lose their capability to remove the protein that accumulates. The accumulated interstitial proteins, as osmotically active molecules, attract fluid to the area. This accumulation of protein and fluid is usually a transitory phase, lasting between 1 and 3 weeks.
In the latent phase, there may still be no clinical signs initially of any discernible lymphedema. The latent phase normally varies from about 4 months to 10 years. At the end of the latent phase, pitting of the edematous extremity on pressure can be observed. This can be objectively measured by plethysmography and by decreased tissue compressibility using a tissue tonometer.
The enlargement of the extremity leads to discomfort and complaints in the form of heaviness, weakness, pain, tension, and a sensory deficit of the limb, as well as anxiety, psychological morbidity, maladjustment and social isolation, and increasing hardness of the limb. Adipose tissue deposition starts already within the first year after lymphedema onset. In time, there is also an increase in the adipose tissue content of the swollen arm. The author has observed this clinically since 1987, when the first lymphedema patient was operated on. This phenomenon led to further research as presented in this chapter.
There are various possible explanations for the adipose tissue hypertrophy. There is a physiological imbalance of blood flow and lymphatic drainage, resulting in the impaired clearance of lipids and their uptake by macrophages. There is increasing support, however, for the view that the fat cell is not simply a container of fat, but it behaves like an endocrine organ and a cytokine-activated cell, and chronic inflammation plays a role here. The same pathophysiology goes for primary and secondary leg lymphedema.
For more detailed information about investigational advances and the relationship between slow lymph flow and adiposity, as well as that between structural changes in the lymphatic system and adiposity, see studies published by Harvey et al . and Schneider et al .
The findings of increased adipose tissue in intestinal segments in patients with Crohn disease, known as “fat wrapping”, have clearly shown that inflammation plays an important role.
Tonometry can distinguish if a lymphedematous arm is harder or softer than a contralateral arm (if unaffected). If a lower tissue tonicity value is recorded in the edematous arm, it indicates that there is accumulated lymph fluid in the tissue, and these patients are candidates for conservative treatment methods. In contrast, patients with a harder arm, compared with the healthy one, have an adipose tissue excess that can successfully be removed by liposuction.
In Graves ophthalmopathy, a major problem is an increase in the intra-orbital adipose tissue volume leading to exophthalmos. Adipocyte-related immediate early genes (IEGs) are overexpressed in active ophthalmopathy and cysteine-rich, angiogenic inducer 61 (CYR61) may have a role in both orbital inflammation and adipogenesis and serve as a marker of disease activity.
Preoperative investigation with volume-rendered computer tomography (VRCT) images showed a significant preoperative increase of adipose tissue in the swollen arm, the excess volume consisting of 81% (range 68%–96%) fat.
Analyses with dual X-ray absorptiometry (DXA) that was compared to plethysmography in 18 women with arm lymphedema following a mastectomy showed a significant increase of adipose tissue, 73% (range, 43%–111%), in the non-pitting swollen arm before surgery. The increase in adipose tissue develops when the lymphedema appears or soon thereafter. Complete reduction was achieved after one year with an excess fat volume reduction of 139%. Excess lean volume (muscle) was reduced by 54% due to the reduced heaviness of the arm.
Adipogenesis in response to lymphatic fluid stasis is associated with a marked mononuclear cell inflammatory response.
Lymphatic fluid stasis potently upregulates the expression of fat differentiation markers both spatially and temporally.
The underlying pathophysiology of lymphedema drives adipose-derived stem cells toward adipogenic differentiation.
Consecutive analyses of the content of the aspirate removed under bloodless conditions using a tourniquet showed a very high content of adipose tissue in 105 women with postmastectomy arm lymphedema (mean 94%, range 58%–100%).
A new staging of lymphedema is suggested because current lymphedema staging systems are inadequate since local patterns of fluid and fat accumulation and of the overall tissue composition are not taken into account.
Lymphedema adipose cells are larger than in controls.
A common misunderstanding among clinicians is that the swelling of a lymphedematous extremity, whether it is primary or secondary, is due purely to the accumulation of lymph and/or fibrosis. Lymph can be removed by the use of noninvasive conservative regimens such as complex decongestive therapy (CDT) and controlled compression therapy (CCT). These therapies work well when the excess swelling consists of accumulated lymph but do not work when the excess volume is dominated by adipose tissue (see Fig. 3.5.4 ). The same may go for microsurgical procedures using lymphovenous shunts, lymph vessel transplantation, and lymph node transfer.
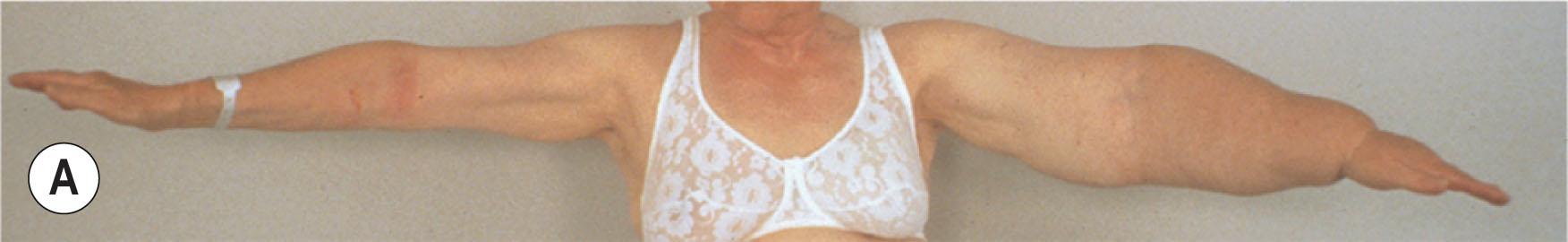
Today, chronic non-pitting arm lymphedema of more than 4 L in excess can be effectively removed by use of liposuction without any further reduction in lymph transport. For arm lymphedema, long-term outcome up to 21 years has not shown any recurrence ( Fig. 3.5.1 ).
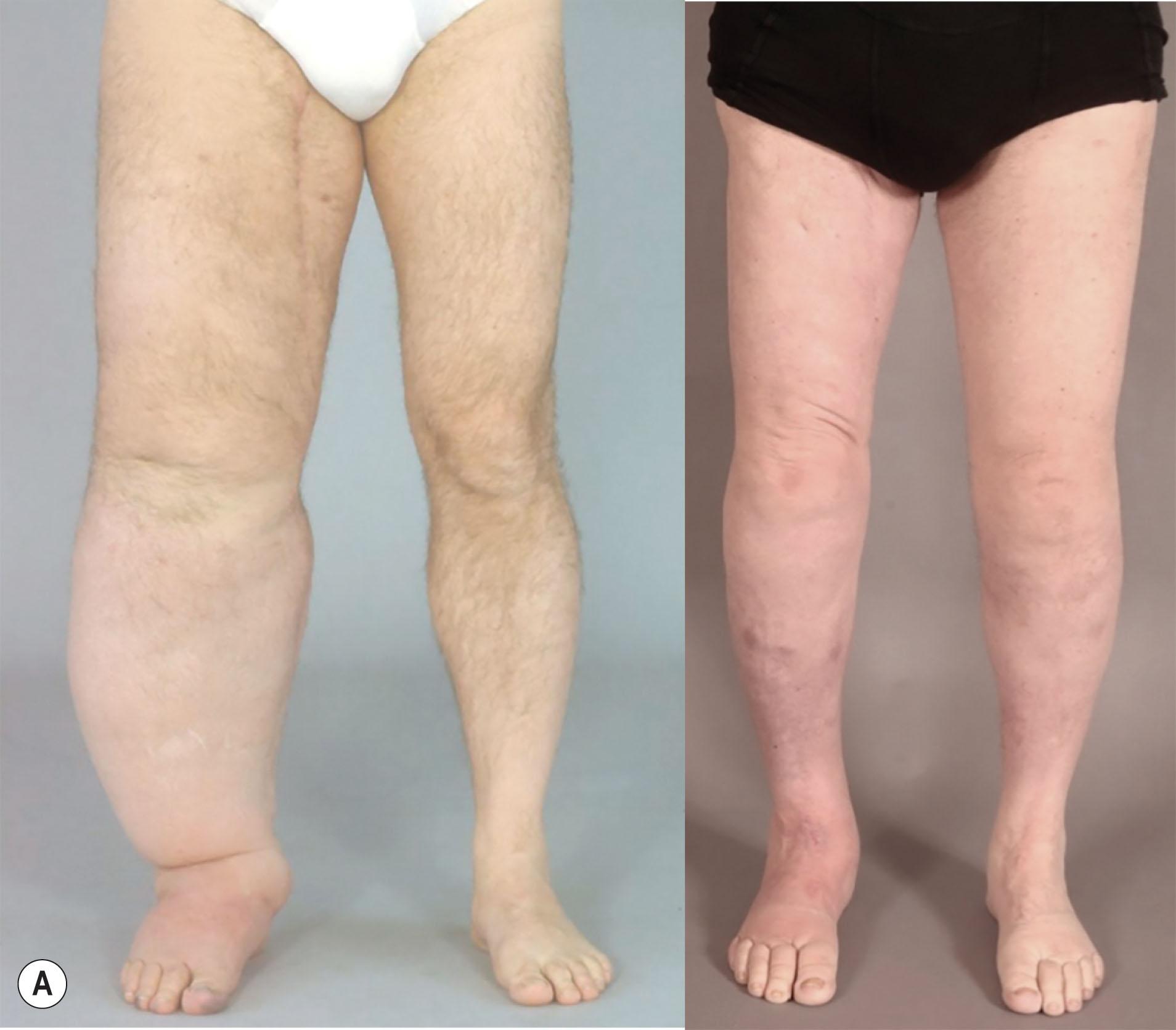
Regarding primary and secondary leg lymphedema, over 7 liters in excess volume can be completely reduced with long-lasting results up to 10 years ( Figs. 3.5.2 & 3.5.3 ).
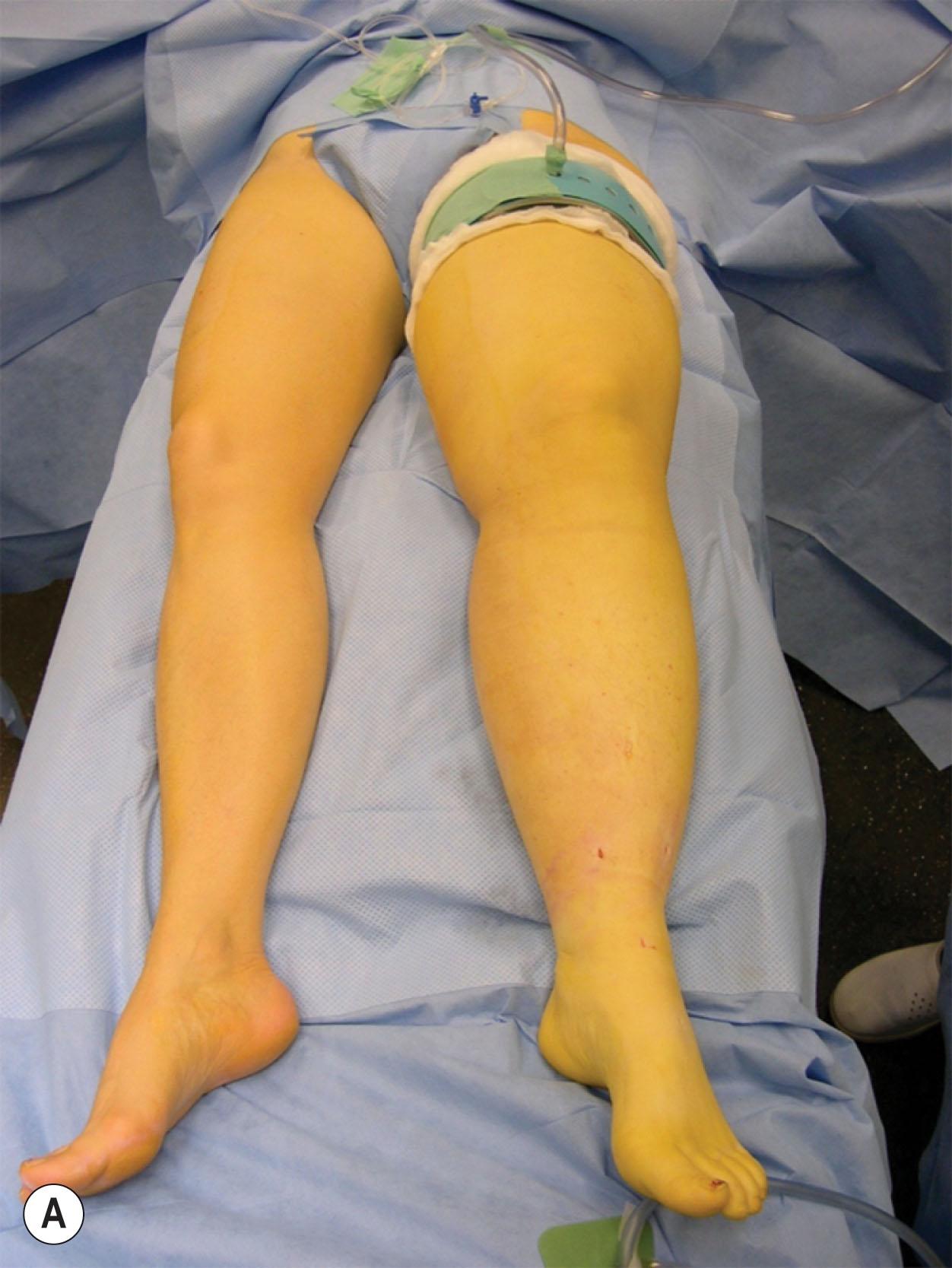
Made-to-measure compression garments (three leg-long with a panty Jobst Elvarex [Essity, Sweden] compression class 3 and two leg-long Jobst Elvarex compression class 2) are ordered 2 weeks before surgery based on measurements of the unaffected leg. One garment, to be put on the leg at the time of surgery, is sterilized and used for only 2 days since it loses some of its pressure by sterilization. The size of the garments is measured according to the size of the unaffected leg.
For the majority of patients, power-assisted liposuction (Lipomatic, Nutational Infrasonic Liposculpture, Euromi, Andrimont, Belgium) was performed to facilitate liposuction ( ![]() ). A tourniquet was placed as proximal as possible on the thigh. Then the leg was exsanguinated with Esmarch rubber rolls. The tourniquet was set to +100 mmHg above the systolic pressure, and the Esmarch rubber rolls were removed. Around 15–20 3- to 4-mm long incisions were made, and liposuction was performed using 15- and 25-cm-long cannulas with diameters of 3 and 4 mm, where the 4 mm was used more proximally. Initially, the foot was also treated, but since no fat could be aspirated, we ceased to treat this area. Circumferential liposuction was performed, step-by-step, from ankles to inguinal region, and as much of the hypertrophied fat was removed as possible using previously measured circumferences of the unaffected leg as a control ( Fig. 3.5.4 ).
). A tourniquet was placed as proximal as possible on the thigh. Then the leg was exsanguinated with Esmarch rubber rolls. The tourniquet was set to +100 mmHg above the systolic pressure, and the Esmarch rubber rolls were removed. Around 15–20 3- to 4-mm long incisions were made, and liposuction was performed using 15- and 25-cm-long cannulas with diameters of 3 and 4 mm, where the 4 mm was used more proximally. Initially, the foot was also treated, but since no fat could be aspirated, we ceased to treat this area. Circumferential liposuction was performed, step-by-step, from ankles to inguinal region, and as much of the hypertrophied fat was removed as possible using previously measured circumferences of the unaffected leg as a control ( Fig. 3.5.4 ).
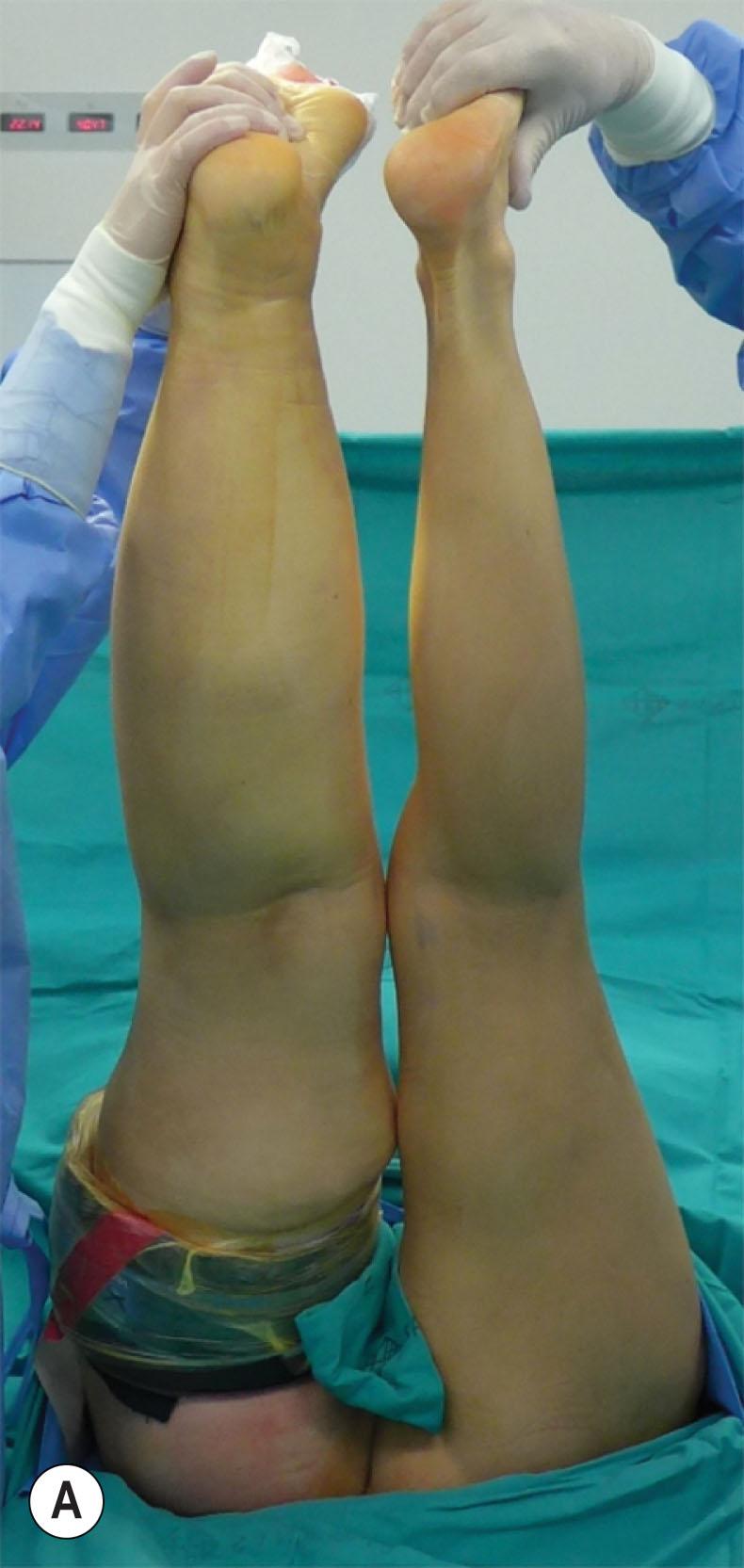
When the leg distal to the tourniquet had been treated, a sterilized custom-made leg-long compression garment with a panty was applied (Jobst Elvarex, compression class 3) to the leg to minimize bleeding and reduce postoperative edema. The tourniquet was then removed, and the most proximal part of the upper leg was treated using the tumescent technique, where 1000 mL saline mixed with 1 mg adrenaline and 40 mL lidocaine 2%, in total 2 liters, were injected. Normally the aspirate contains 90%–100% adipose tissue ( Fig. 3.5.5 ).

Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here