Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Intrathecal drug delivery for the treatment of pain is an outgrowth of pioneering research into spinal cord mechanisms and receptors that modulate nociceptive signal transmission to and within the central nervous system. Since the 1970s, researchers have identified the peptide substance P in small dorsal root ganglion cells and observed that opioids would produce analgesia. These findings, as well as the observation that substance P release evoked from C fibers is blocked by morphine, marked the beginning of the current trend toward focusing on the selective regulation of spinal afferent processing. Since then, there has been much research in this area, leading to the widespread use of intrathecal therapy to treat chronic intractable pain. Although this therapy has proven extremely beneficial to many patients, it is also associated with high costs, high maintenance, and risks requiring careful patient selection. The first section focuses on the patient selection, technologies used for chronic intrathecal drug delivery, system implantation, and troubleshooting. The second section reviews the medications used in intrathecal drug delivery systems.
##
There are essentially three methods of intraspinal drug delivery: (1) externalized systems, (2) partially externalized systems, and (3) totally implanted systems. The selection of the technique will depend on the goal of therapy, ranging from short term intrathecal trialing to long term therapy to treat chronic pain. Each technique has associated risks and costs, which must be considered when selecting the technique.
An externalized intrathecal catheter can be used for trialing intrathecal drugs or for short term therapeutic infusions. Such a system is best for infusions lasting hours to days but has been successfully used for longer term delivery in terminally ill patients lasting a few weeks. The use of an externalized catheter raises the concern for a local infection at the catheter exit site or that the catheter may serve as a conduit for either epidural or intrathecal spread of infection. Using a strict aseptic technique for catheter insertion can minimize the risk of insertion site infection, and the placement of a chlorhexidine impregnated catheter patch at the insertion site further reduces this risk ( Fig. 72.1 ). There is no evidence that systemic antibiotics prevent these infections. In a terminally ill patient with a very short life expectancy, the risk-benefit ratio may favor the use of an externalized catheter for intrathecal drug delivery. Some practitioners tunnel these catheters under the skin for some distance to provide stability and reduce the likelihood of inadvertent catheter withdrawal. Currently, the United States Food and Drug Administration (FDA) has not approved any externalized intrathecal catheters, and it is common to use epidural or lumbar drain catheters for this purpose.
Partially externalized systems are those in which the intrathecal catheter is tunneled and connected to a subcutaneous port. A needle then accesses the port percutaneously, and medication is delivered with tubing connected to an external pump. Compared to an externalized system, this permits greater patient freedom of movement and reduces the risk of inadvertent catheter removal. Partially externalized systems are intended for single bolus injections. There continues to be a risk of colonization and infection of the port pocket with prolonged or repeated needle access. These systems have limited application for long term, continuous intrathecal drug delivery.
Totally implanted systems are the most common and have the catheter and pump completely internalized by surgical implantation. These are used for long term infusions and afford the patient much greater freedom of movement. A totally implanted system is more expensive and requires a more invasive surgical procedure than an externalized or partially externalized system. Although it is generally recommended that implanted pumps be reserved for patients with a life expectancy of greater than six months, , this therapy may improve quality of life in patients with life expectancy as low as two to three months. However, patients with a life expectancy of fewer than six months should be carefully selected.
There are two main types of intrathecal infusion systems, fixed-rate and variable-rate programmable pumps. All implanted pumps have two percutaneous access ports. One port is for refilling the drug reservoir, and there is a separate intrathecal catheter access port for bolus injection through, or aspiration from, the catheter. The infusion systems are generally implanted in the lower abdominal subcutaneous fat and connected to a catheter, which is tunneled subcutaneously around the abdominal wall into the intrathecal space. , However, the pumps may also be implanted subcutaneously in the lower back in selected patients (see the discussion that follows). The septum puncture life of the percutaneous refill port is very high: 500 punctures for the most commonly used pump, the Medronic Synchromed II pump. It would be unlikely to reach this number of punctures during the life of the pump (5.5 years for the Synchromed II).
The first intrathecal pump, manufactured by Shiley Infusaid, became commercially available in 1982. The Infusaid and other early pumps were non-programmable fixed-rate infusion systems. These pumps were simple devices without batteries, in which pressurized gas pushes the drug from a reservoir through a valve and into the catheter. The kinetic energy of the pressurized gas is increased every time a new drug is injected into the reservoir. The chief advantage of these pumps is the simplicity of design and lack of batteries. Unfortunately, they cannot vary the dose delivered without changing the concentration of the drug in the reservoir. The last of these pumps, the Codman 3000 ( Fig. 72.2 ), was discontinued in 2018. Fixed infusion rate pumps have not been used with any frequency in the United States for many years. However, some of these pumps may still be implanted and functional in patients since there are no batteries; thus they never require replacing because of battery failure. The Codman pump was available in different sizes and various flow rates. The flow rate is controlled by a flow restrictor. Flow rates may vary for different drugs, drug combinations, and especially with high drug concentrations, based on differences in viscosity. In addition, changes in body temperature and atmospheric pressure will change flow rate by affecting the gas pressure within the system.

Programmable pumps allow the clinician to vary infusion rates to increase or decrease the dose without changing the concentration of the drug or drugs in the pump. The original programmable pump was the Medtronic Synchromed, first introduced in 1991. This device was upgraded to the Medtronic Synchromed II ( Fig. 72.3 ), which allows patient-controlled intrathecal bolus dosing via a remote-control device. The Synchromed II uses a peristaltic roller system ( Fig. 72.4 ) to move the drug from the reservoir to the implanted intrathecal catheter. The Synchromed II is available in 20 mL and 40 mL reservoir sizes and is FDA approved for baclofen, morphine, and ziconotide.

The other programable intrathecal pump on the market is the Flowonix Prometra II implantable pump. Similar to the older non-programmable pumps, the original Prometra and newer Prometra II use a pressurized gas chamber as the driving force. Unlike those older fixed-rate infusion pumps, a programmable flow-metering valve allows for a variable drug delivery rate. The Prometra pump’s low energy requirement is its main advantage. Since the batteries are used to control the electronics only, not to pump the drug, the battery has a ten-year life span before the need to surgically replace the pump. The Prometra is available in a 20 mL reservoir and is FDA approved for morphine. The issues with variable flow resulting from viscosity, temperature, and residual reservoir volume have been addressed by the advanced flow-metering valve.
Both the Synchromed II and Prometra pumps are MRI conditional. However, the conditions for MRI management are different between the two. , As discussed above, the Synchromed pump is driven by a peristaltic pump that can be programmed to rotate at different rates to adjust drug daily dose. When in the MRI, the peristaltic rotor will suspend in the magnet and stop the flow of the drug. When removed from the magnet, the rotator will restart the drug flow as programmed. The MRI might result in a permanent pump stall if the rotor does not resume functioning. The risk of a permanent pump stall increases as the pump moves closer to a 90-degree position in the magnet. This may occur in patients with the pump flipped 90 degrees to normal placement in the abdominal wall pump pocket. Pumps in the traditional position within the abdominal wall are closer to the 0-degree position with a lower risk.
The Prometra pumps have a flow-metering valve that will move into the fully open position while in the MRI. Since the pump reservoir is under positive pressure, this would result in immediate release of the entire pump reservoir contents resulting in overdose. Therefore in the original Prometra pump, it is critical to remove all drugs from the reservoir prior to an MRI. The Prometra II pump has a safety mechanism that if the flow-metering valve abruptly opens, a secondary valve closes, thus stopping the flow. The pump will not restart until this secondary valve is reset. Despite the secondary valve’s safety measure, the manufacturer’s recommendations for MRI compatibility with the Prometra II pump are to remove the contents of the drug reservoir prior to an MRI.
Intrathecal drug delivery systems (IDDSs) are reserved for treating severe and refractory pain in two types of patients: those with cancer pain and those with non-cancer pain who have failed all conservative therapies. The cost and invasive nature of IDDSs necessitate a careful evaluation to determine whether each individual patient is appropriate and likely to achieve benefit. A thorough assessment includes an evaluation of pain-generating pathologic conditions, medical comorbidities, psychosocial issues, patient compliance history, economic and healthcare coverage, and anatomic and technical considerations.
In advanced stages of cancer, the prevalence of pain exceeds 60%. One-third of those patients report moderate to severe pain. Comprehensive medical management (CMM) can significantly reduce pain and manage the adverse effects of opioids and other analgesics. Despite aggressive therapy, some patients continue to endure severe pain. Others are unable to tolerate systemic opioids because of toxicity. A 2002 multi-center randomized trial compared CMM alone versus CMM plus an IDDS. They found that cancer patients receiving CMM plus an IDDS had reduced pain, fewer drug toxicities, and prolonged survival than controls. Patients with significant toxicity or with refractory pain that has been inadequately controlled by the systemic administration of opioids and other analgesics are potential candidates for an IDDS ( Table 72.1 ). Intrathecal therapy may be especially valuable in patients with neuropathic pain from tumor invasion of neural plexuses, unstable pathologic fractures, painful impending spinal cord compression, and visceral tumors with or without autonomic dysfunction that results in gut dysmotility.
| Study | Number of Subjects | Type/Duration | Details | Efficacy |
|---|---|---|---|---|
| Smith et al. 2002 |
200 | RCT/four weeks | Advanced cancer pain; CMM (99) vs. IDDS (101) | IT plus CMM improved pain and reduced opioid adverse effects |
| Staats et al. 2004 |
111 | RCT/11 days | Refractory pain because of cancer or AIDS; ziconotide (71) vs. placebo (40) | IT ziconotide significant improvement in pain scores vs. placebo |
| Rauck et al. 2003 |
119 | Prospective Observational; up to 16 months | Refractory cancer pain; addition of IT morphine | Significant reduction in pain intensity and systematic opioid use |
| Mercadante et al. 2007 | 55 | Prospective Observational; up to six months | Advanced cancer pain; addition of IT morphine + levobupivacaine | Improved pain, decreased oral opioid, decreased opioid adverse effects |
| Dupoiron et al. 2012 | 77 | Prospective Observational; 90 days | Refractory cancer pain; ziconotide + morphine, ropivacaine, and/or clonidine | Improvement in numeric rating scale pain scores |
| Brogan et al. 2015 |
58 | Prospective Observational; 14–82 days |
Refractory cancer pain; addition of IT analgesics | Decrease in numeric rating scale pain scores; decreased percentage with severe pain |
In patients with a life expectancy of less than two to three months, a percutaneous catheter and external pump can be used. These patients may be discharged from the hospital with hospice care or home-health services for the care of the catheter and infusion if appropriate services are locally available.
For the patient with chronic non-cancer pain, a comprehensive evaluation to establish the cause of the pain should be performed as part of the decision process regarding whether an intrathecal pump is appropriate. The evaluation should include a detailed history, physical examination, radiologic studies, and assessment of prior therapies and their outcomes. Many diagnoses and painful conditions may be amenable to intrathecal drug delivery in certain cases. These include neuropathic syndromes, such as spinal cord injury, diabetic peripheral neuropathy, complex regional pain syndrome, postherpetic neuralgia, and phantom limb pain. Additionally, mixed nociceptive and neuropathic conditions, such as post-laminectomy pain syndrome or chronic pancreatitis, and nociceptive pain conditions, such as chronic vertebral compression fractures and severe spinal degeneration, may be appropriate for an IDDS in some circumstances after thorough patient selection and discussion. , ,
Implanted drug delivery therapy should be reserved for those patients who are refractory from more conservative treatment. A stepwise approach starting with the least invasive and costly therapies is recommended. , This may begin with exercise programs, relaxation techniques, and over-the-counter analgesics. Subsequent steps include adjuvant analgesics, muscle relaxants, physical therapy, and psychological therapies. More invasive therapies include neuraxial somatic and sympathetic blocks and spinal cord stimulation. The top tier of this stepwise treatment pyramid is reserved for the most invasive therapies, including intrathecal drug delivery and neuro-destructive procedures, such as cordotomy, myelotomy, and dorsal rhizotomy. , There are few controlled trials of intrathecal therapy for chronic non-cancer pain. However, observational studies suggest such therapy can provide pain relief in certain patients who have failed to achieve relief with more conservative treatments ( Table 72.2 ). , Determining the efficacy of long-term intrathecal drug therapy remains one of the greatest challenges in chronic non-cancer pain management. One of the reasons for the lack of randomized controlled trials is that the high cost and invasiveness of such treatment pose ethical challenges in designing a double-blind, placebo-controlled study. Therefore one can only rely on the retrospective reviews of patients receiving this therapy and attempt to make conclusions on the efficacy of this treatment. Many studies have attempted to determine the efficacy of long-term intrathecal morphine therapy based on the classification of the pain. However, a review of these articles reveals large discrepancies in pain classification between studies. Because pain is often multifactorial (e.g. failed back syndrome versus HIV neuropathy versus complex regional pain syndrome), it is difficult to determine what pain syndromes are the most responsive to this therapy and have long-term benefits.
| Study | Number of Subjects | Type/Duration | Details | Efficacy |
|---|---|---|---|---|
| Winkelmuller et al. 1996 | 120 | Retrospective; six months to 5.7 years follow up | Mixed neuropathic/nociceptive pain; morphine, bupivacaine, hydromorphone | 74% reported improvement in pain, averaging a 58% reduction |
| Anderson et al. 1999 |
30 | Retrospective | Failed back surgery; rotation from morphine to hydromorphone because of adverse effects or poor pain control | 37% had improved pain control; the majority with decreased adverse effects (nausea, pruritis, edema) |
| Kumar et al. 2001 |
25 trialed 16 implanted | Prospective, single-center, non-randomized | Non-malignant pain; morphine; follow up 13–49 months | Trial 64% with >50% pain reduction; implant avg reduction 57.5% at final follow up |
| Rainov et al. 2001 |
30 trialed, 26 implanted | Prospective, single-center, non-randomized | Failed back surgery syndrome; polydrug infusion of morphine + clonidine, bupivacaine, or midazolam; mean follow up 27 months | Average pain intensity 8/10 to 3/10, remained in the range of 3–5/10 throughout follow up |
| Deer et al. 2004 |
166 trialed, 136 implanted | Prospective, non-randomized, multi-center registry | Mechanical neuropathic, mixed low back/leg pain. Trialing and implant for non-malignant pain; follow up 12 months | 93% trial success; 82% implanted; 48% reduction in the low back; 32% for leg pain at 12 months |
| Rauck et al. 2006 |
220 | Randomized, double-blind, placebo controlled | Ziconotide slow titration over three weeks; 112 ziconotide, 108 placebo | Statistical significance for ziconotide over three weeks; higher adverse effects |
The aging, chronic pain patient may experience some of the same age-related comorbidities as any individual. Many of these comorbid conditions can complicate the implantation and use of an intrathecal pump. These medical issues and their potential interaction with intrathecal therapy must be considered during the patient selection process.
Diabetes mellitus affects over 8% of the United States population and nearly 27% of those over 65 years. Patients with diabetes experience poor wound healing and suffer from an increased incidence of surgical site infections. – Implantable devices pose a special risk of surgical wound infections. With implantable intrathecal pumps, wound infections have been found to be the most common device-related complication. Patients with diabetes, especially if blood glucose control is poor, should be counseled on the increased risk, and implanters should check a hemoglobin A1C prior to surgical implant and consider postponing surgery for an A1C >8.
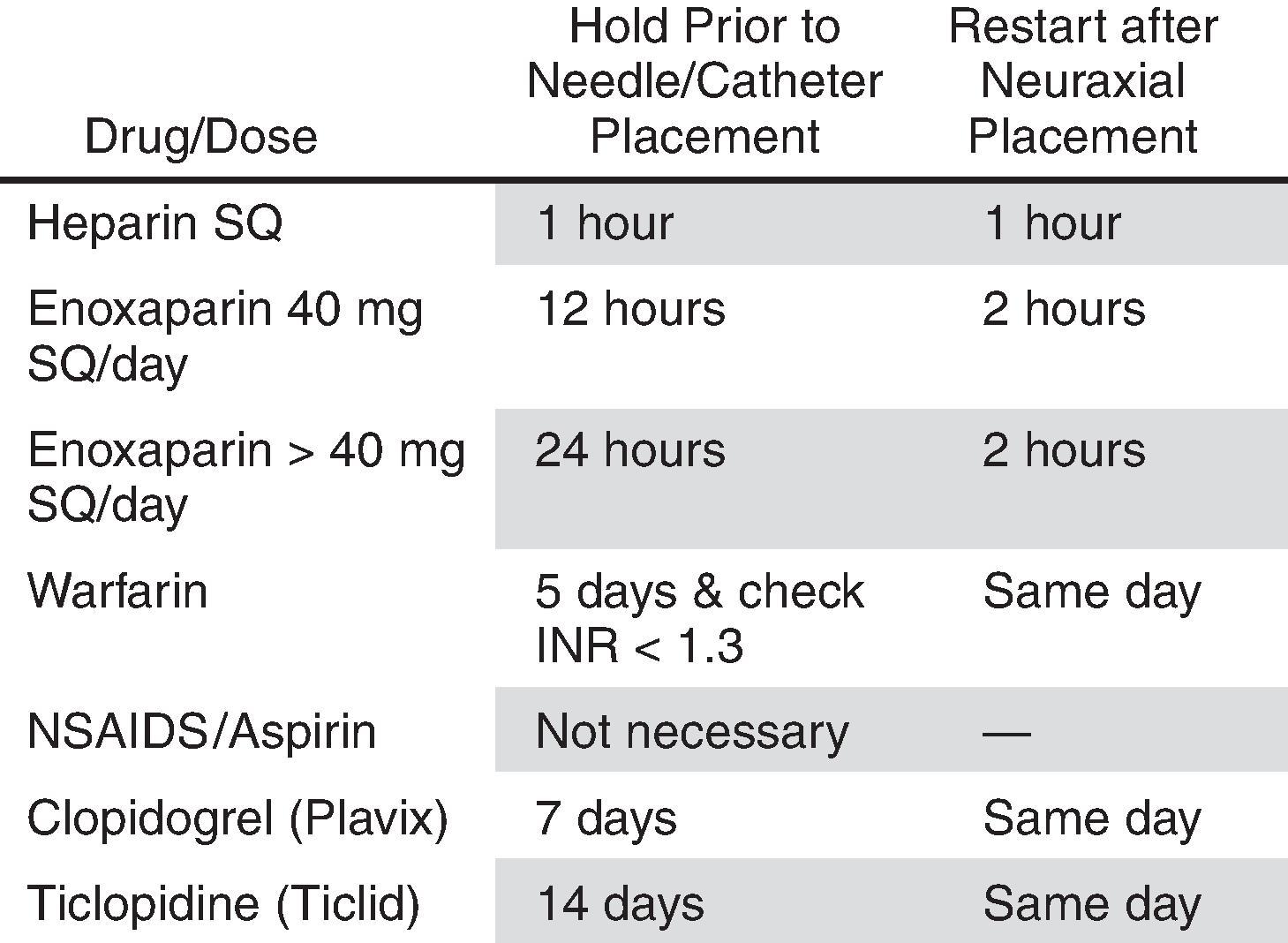
Patients on anticoagulant therapy are at increased risk of epidural hematoma formation after placement or removal of epidural or spinal catheters. , Additionally, multiple new anticoagulant agents have made the management of anticoagulation for invasive pain procedures more of a challenge. Many of these agents carry specific warnings related to spinal injection procedures but may not provide clear guidance regarding perioperative or peri-procedural management. A group of chronic pain societies, including the American Society of Regional Anesthesia and Pain Medicine, the European Society of Regional Anaesthesia and Pain Therapy, American Academy of Pain Medicine, the International Neuromodulation Society, the North American Neuromodulation Society, and the World Institute of Pain, have published guidelines regarding the management of drugs that affect coagulation in the setting of invasive pain procedures ( Box 72.1 ). They divide pain procedures into three risk categories: low, intermediate, and high. The implantation of intrathecal pumps is considered by this guideline to be a high-risk procedure. Scattered case reports of anticoagulant therapy restarted after implantation of intrathecal pumps offered no clear evidence for or against the safe use of intrathecal pumps in these patients.
| Drug/Dose | Hold Prior to Pump Implantation | Restart After Implant |
|---|---|---|
| Heparin SQ (BID or TID) | 24 h | 6–8 h |
| Enoxaparin 40 mg SQ/day | 12 h | 12–24 h |
| Enoxaparin >40 mg SQ/day | 24 h | 12–24 h |
| Coumadin | Five days & check INR<1.3 | 6 h |
| NSAIDS | Five half lives | 24 h |
| Aspirin | Six days primary prophylaxis; shared decision-making secondary prophylaxis | 24 h |
| Clopidogrel (Plavix) | Seven days | 12–24 h |
| Prasugrel (Effient) | Seven to ten days | 24 h |
| Ticagrelor (Brilinta) | Five days | 24 h |
| Dabigatran (Pradaxa) | Four days | 24 h |
| Rivaroxaban (Xarelto) | Three days | 24 h |
| Apixaban (Eliquis) | Three days | 24 h |
Any active infection puts the patient at risk of a surgical site infection that may be superficial or deep into the pump pocket. While a superficial surgical site infection may be successfully treated medically, a pump pocket infection is virtually impossible to clear with antibiotic therapy and will require explanting the entire pump and catheter system. Any active or chronic bacterial or fungal infections, particularly with non-cancer pain patients, should be regarded as a contraindication to implantation.
More than 18 million Americans have obstructive sleep apnea (OSA). In addition to the risks of carbon dioxide retention and both systemic and pulmonary hypertension, the OSA patient has increased sensitivity to the respiratory depressant effects of opioids. There are no studies of intrathecal opioid use in OSA patients. However, case reports suggest that chronic use of oral opioids in those with OSA increases apnea duration and hypoxia severity. Chronic obstructive pulmonary disease (COPD) also results in carbon dioxide retention and increased risk of respiratory depression with the administration of opioids. A retrospective case-controlled study of postoperative COPD patients administered opioids found an increased risk of respiratory events (odds ratio [OR] 5.09; 95% confidence interval [CI]) than patients without chronic lung disease. Most of these events occurred within 24 h of surgery. The long-term risk of intrathecal opioid administration in patients with severe pulmonary disease is unknown. However, known risks of perioperative opioid administration in this population warrant careful monitoring during trialing as well as after implantation and initiation of therapy.
Additionally, patients who are chronic smokers are at increased risk of surgical site infections. The Polyanalgesic Consensus Conference (PACC) published several guidelines regarding intrathecal pump implantation and management. They recommend smoking cessation two months prior to elective intrathecal pump implant.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here