Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
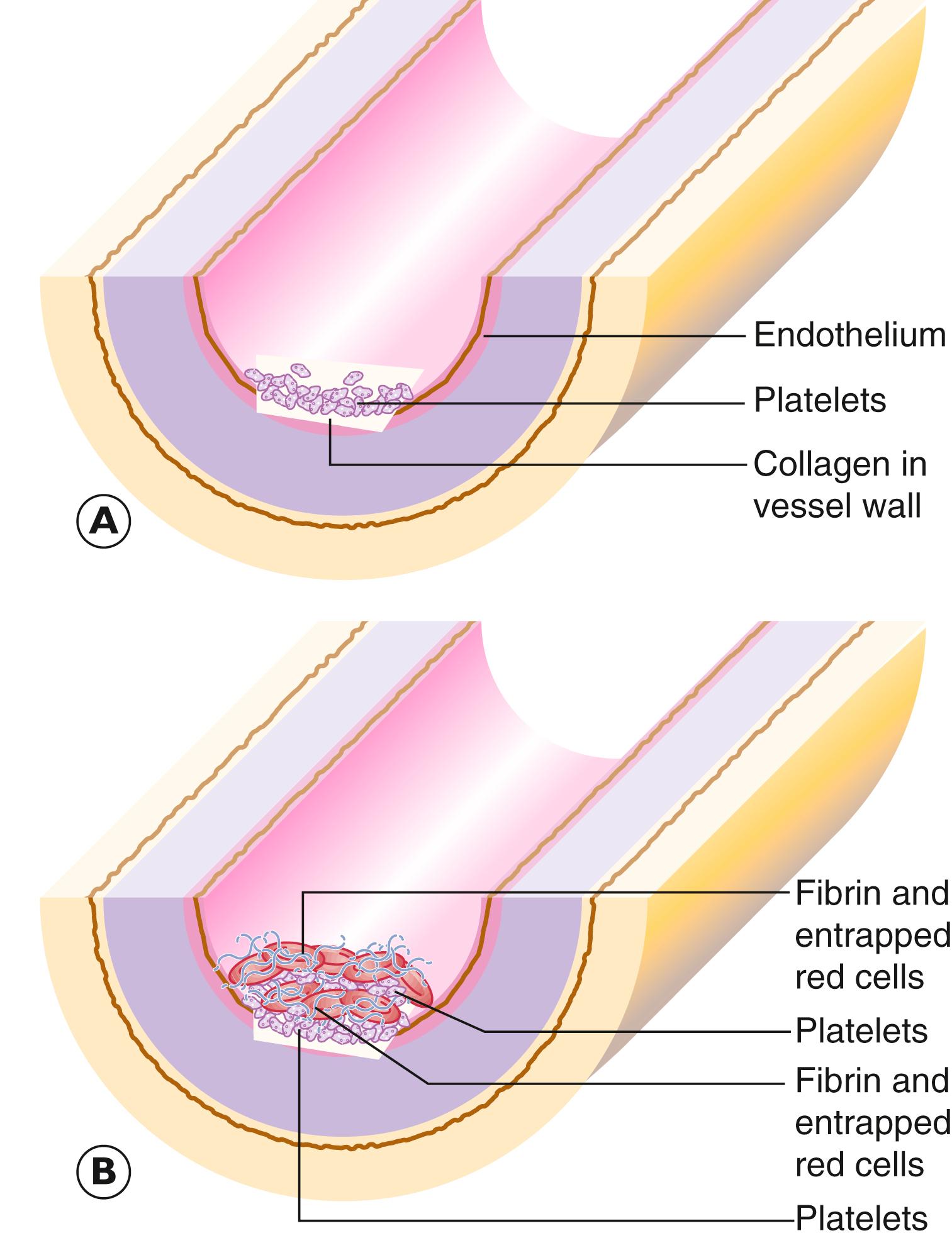
The vascular system normally contains fluid blood. However, it is often necessary for blood to coagulate to prevent bleeding following injury to a vessel wall. To control this process of haemostasis , there are interacting systems that either promote or inhibit the process of blood coagulation. These systems are normally well regulated. Under certain pathological circumstances these dynamics may be disrupted, leading either to haemorrhage or thrombosis . Thrombosis is a dynamic process in which a thrombus , composed of a solid, tangled mass of fibrin, platelets, erythrocytes and other blood constituent cells, forms within the lumen of a blood vessel in response to disruption of the normal blood flow. Thrombosis can be differentiated from coagulation in so far as coagulation occurs within static blood, often at the site of an injury. Thrombosis is a dynamic process and can occur more frequently in certain diseased states.
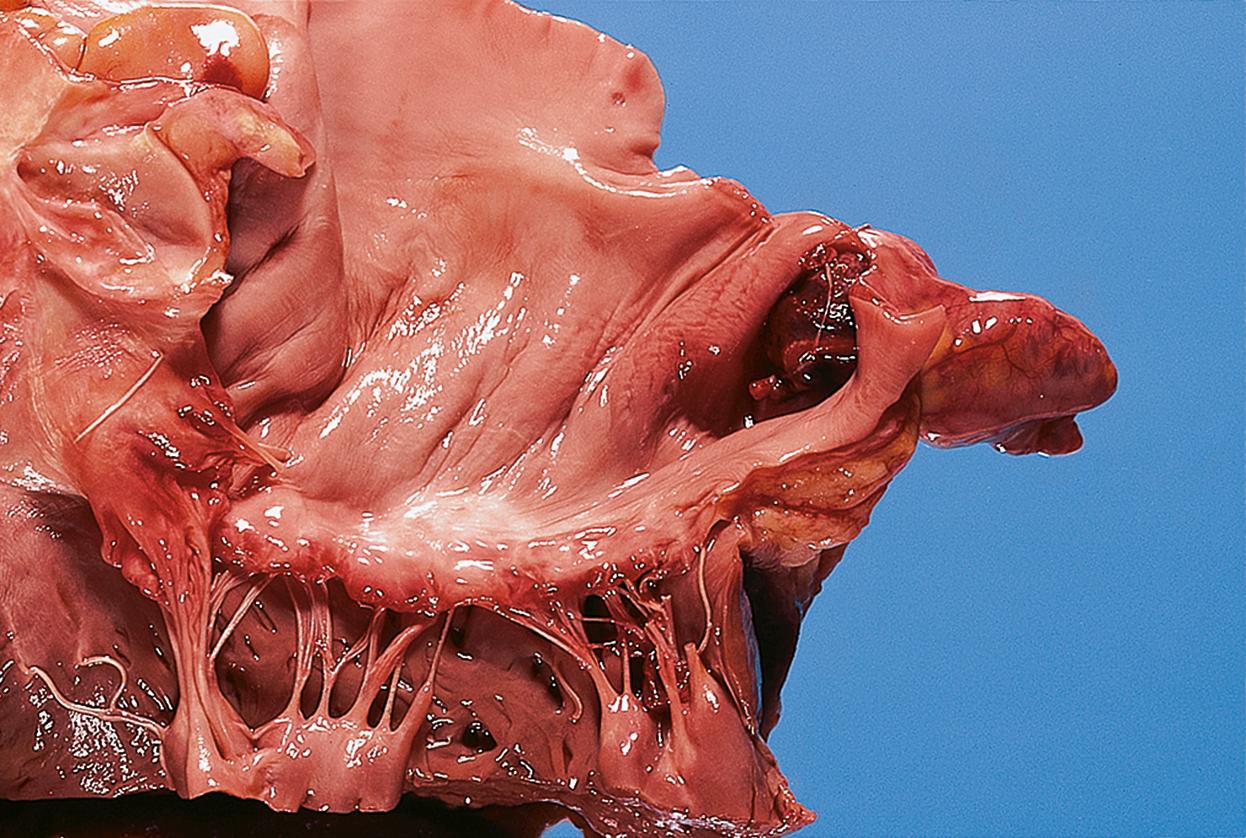
Thrombosis may occur in any part of the circulation, but most commonly occur in large veins ( E-Fig. 9.1 G ), large arteries, in the heart chambers ( E-Fig. 9.2 G ) and on heart valves ( Fig 11.16, Fig 11.17 ). Three major factors, alone or in combination, predispose to thrombosis. These are often referred to as Virchow’s triad :
Damage to the endothelium is the main cause of arterial and intracardiac thrombosis: This damage may be either through physical endothelial disruption or as a consequence of endothelial cell dysfunction. Hence hypertension, turbulent blood flow and bacterial endotoxins may contribute to endothelial dysfunction and thrombus formation.
Disordered blood flow. Relative stasis is important in initiating thrombus in slow-flowing blood such as in veins. Turbulent blood flow predisposes to thrombus formation in arterial vessels and the heart through disruption of endothelial function and the creation of eddy currents in the blood with focal stasis.
Abnormal haemostatic properties of the blood (hypercoagulability). Hypercoagulable states may be either inherited or acquired. Of the inherited disorders, abnormalities in the factor V and prothrombin genes are commonest and may present with recurrent venous thrombosis. There are numerous acquired hypercoagulable states, such as those associated with changes in blood viscosity, for example in dehydration, major illness, disseminated carcinoma or in the post-operative state.
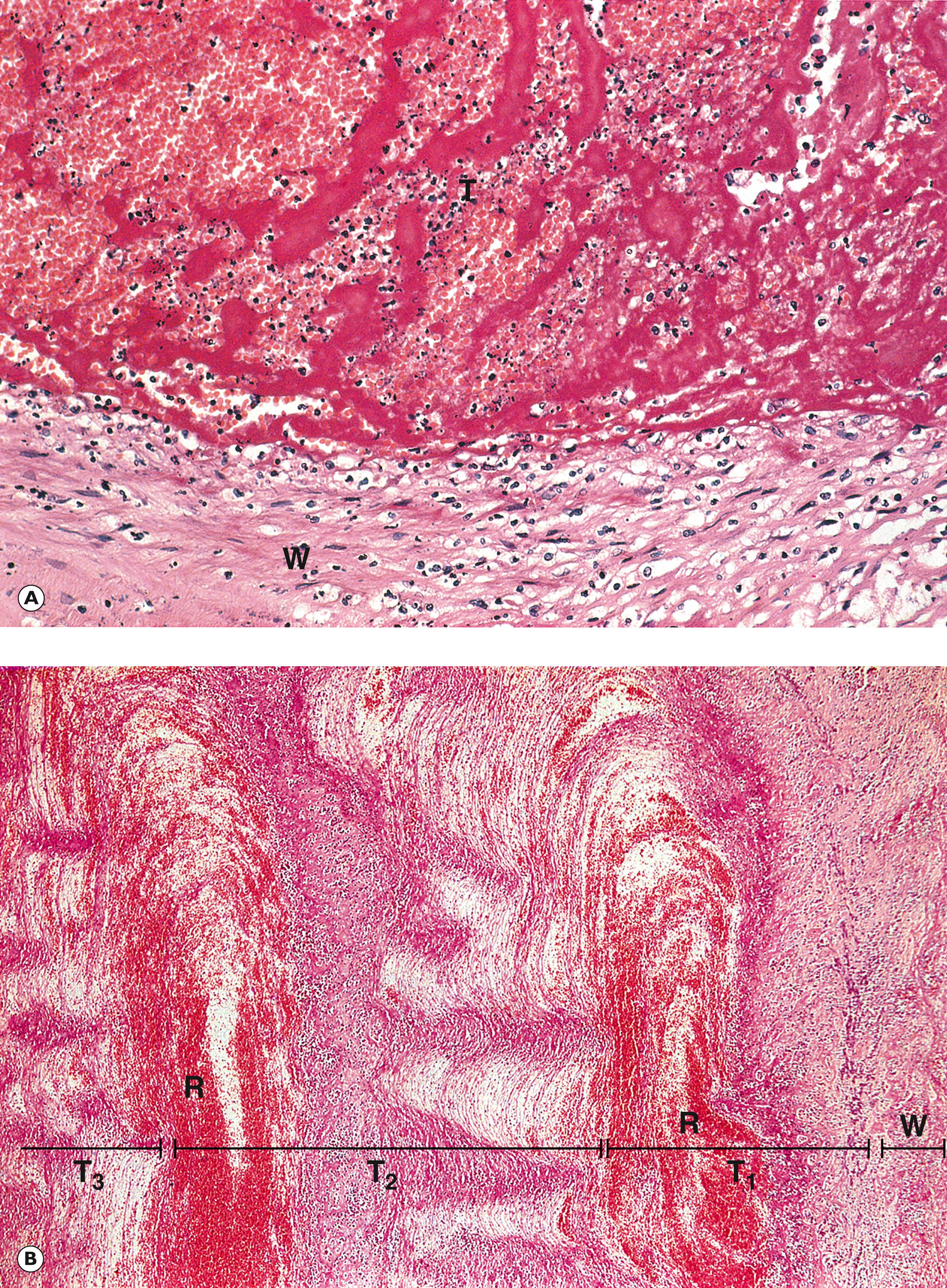
A thrombus has a defined architecture and consistency, which reflects the manner and stages of its formation and the nature of blood flow in the vicinity. For example, thrombus formed in an artery is usually dense and composed mainly of aggregated platelets and fibrin ( Fig. 9.2 ) .
R red cells T thrombus W arterial wall
Once thrombus is formed there are several possible outcomes:
Propagation: The thrombus itself is thrombogenic and may attract more platelets and fibrin to the site, resulting in it enlarging and growing along the vessel, ultimately leading to vessel occlusion and/or embolisation if left untreated.
Embolisation: In addition to the triad of factors contributing to thrombus generation, Virchow coined the term embolus for ‘fragments from the end of a softening thrombus that are carried along by the current of the blood and driven into remote vessels’ . This term is now applied not only to fragments of thrombus (thromboemboli) , but to any material forming in, or entering, the bloodstream and passing with the circulation to lodge in a more distal vessel.
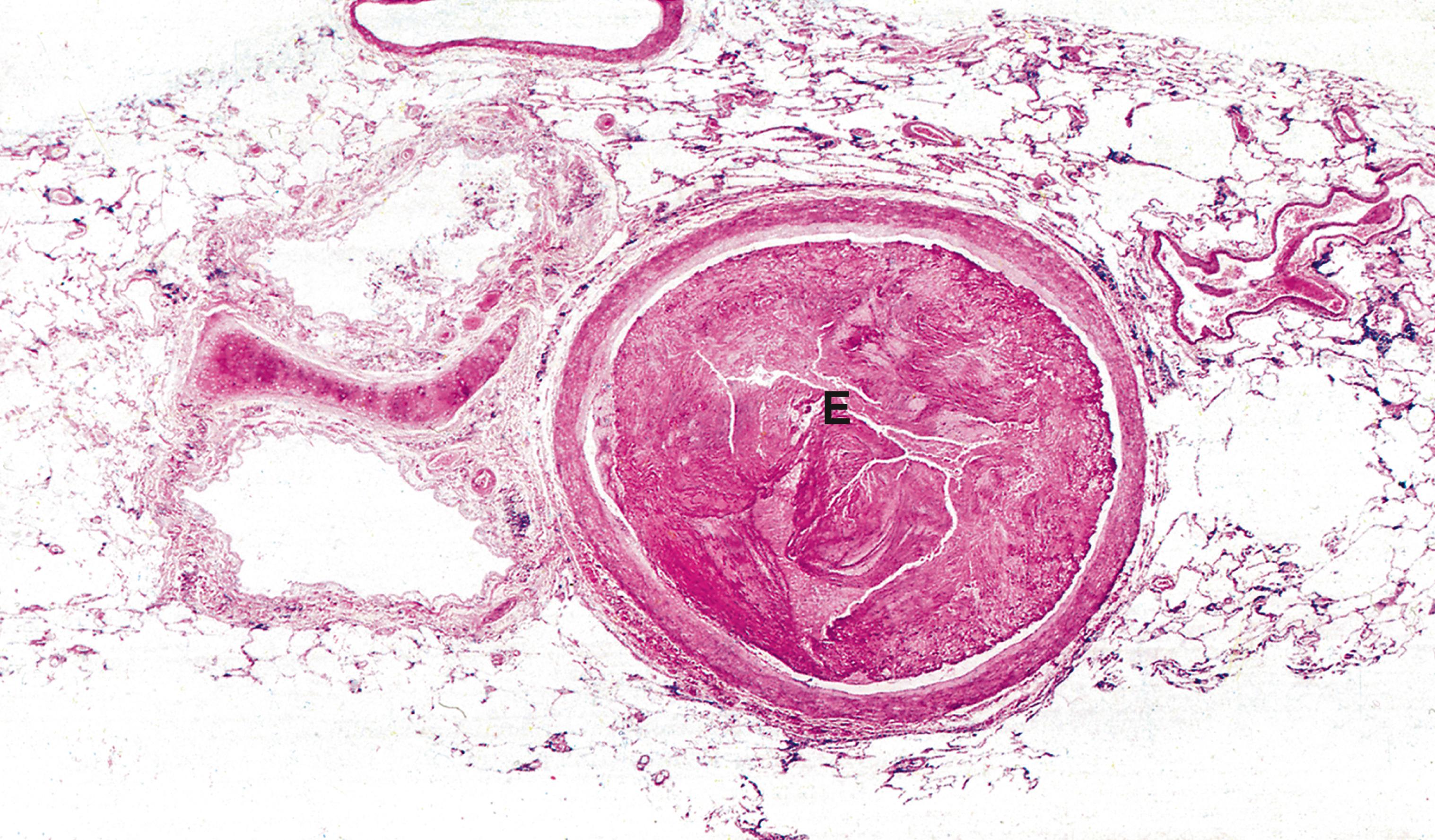
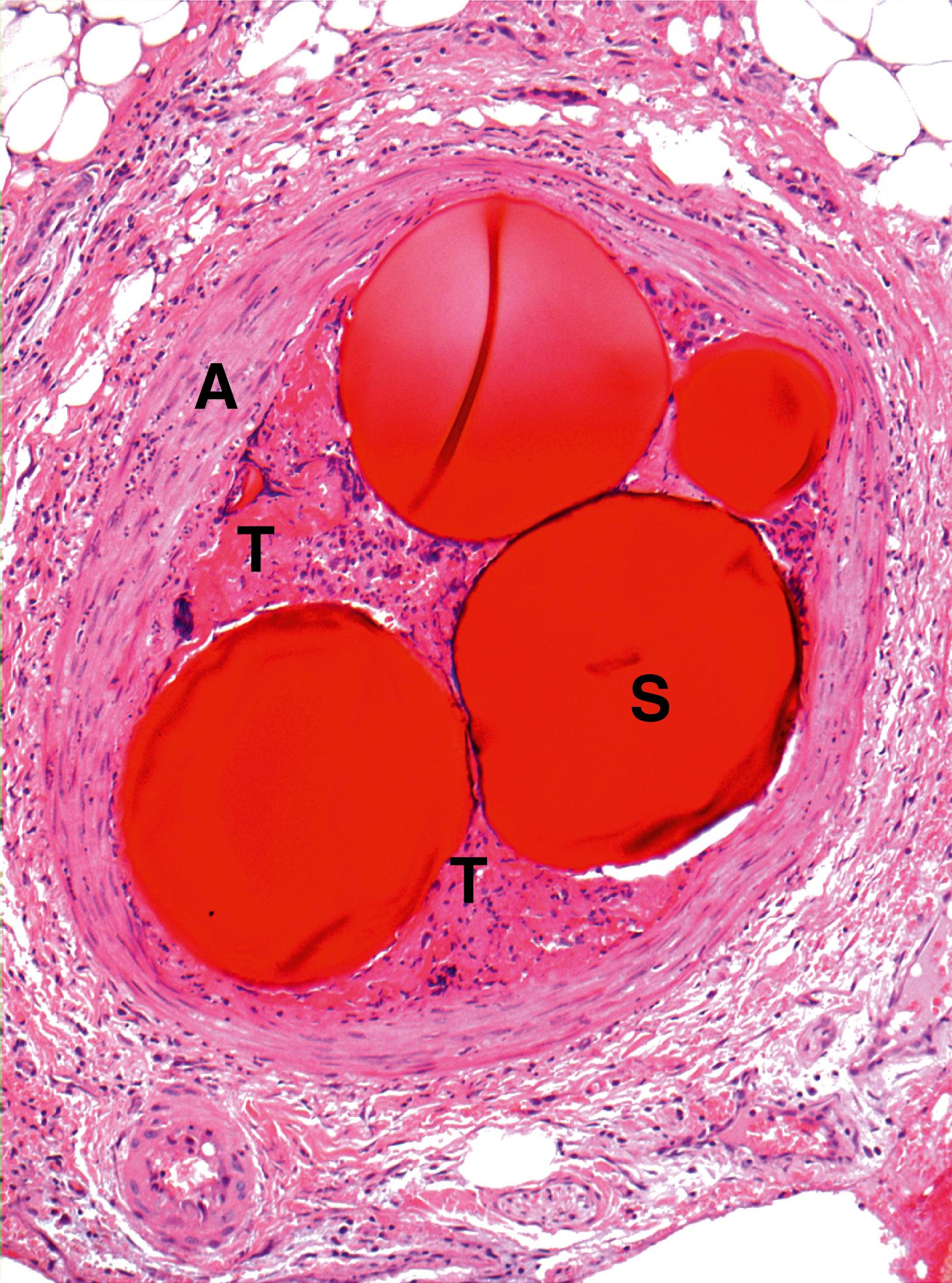
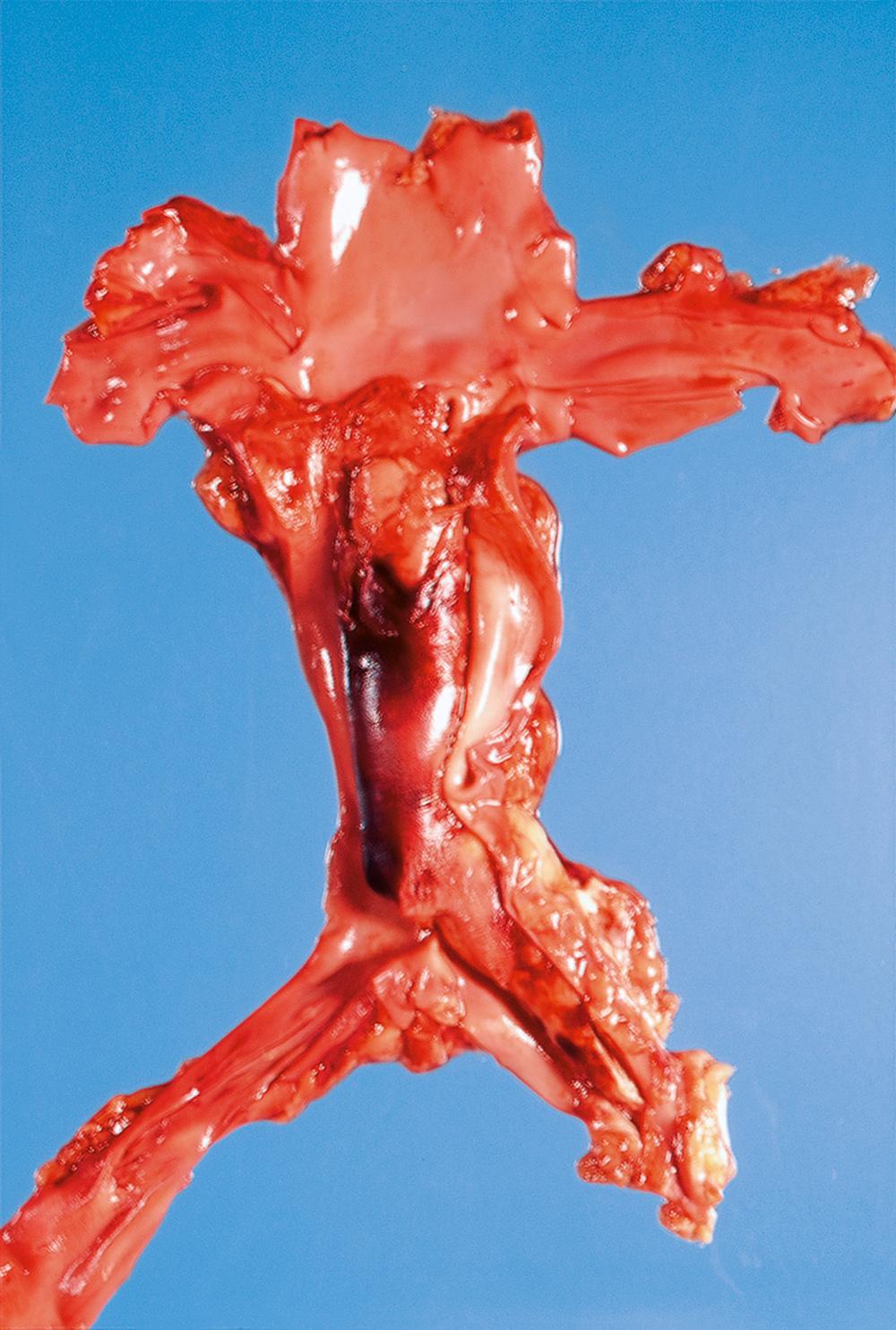
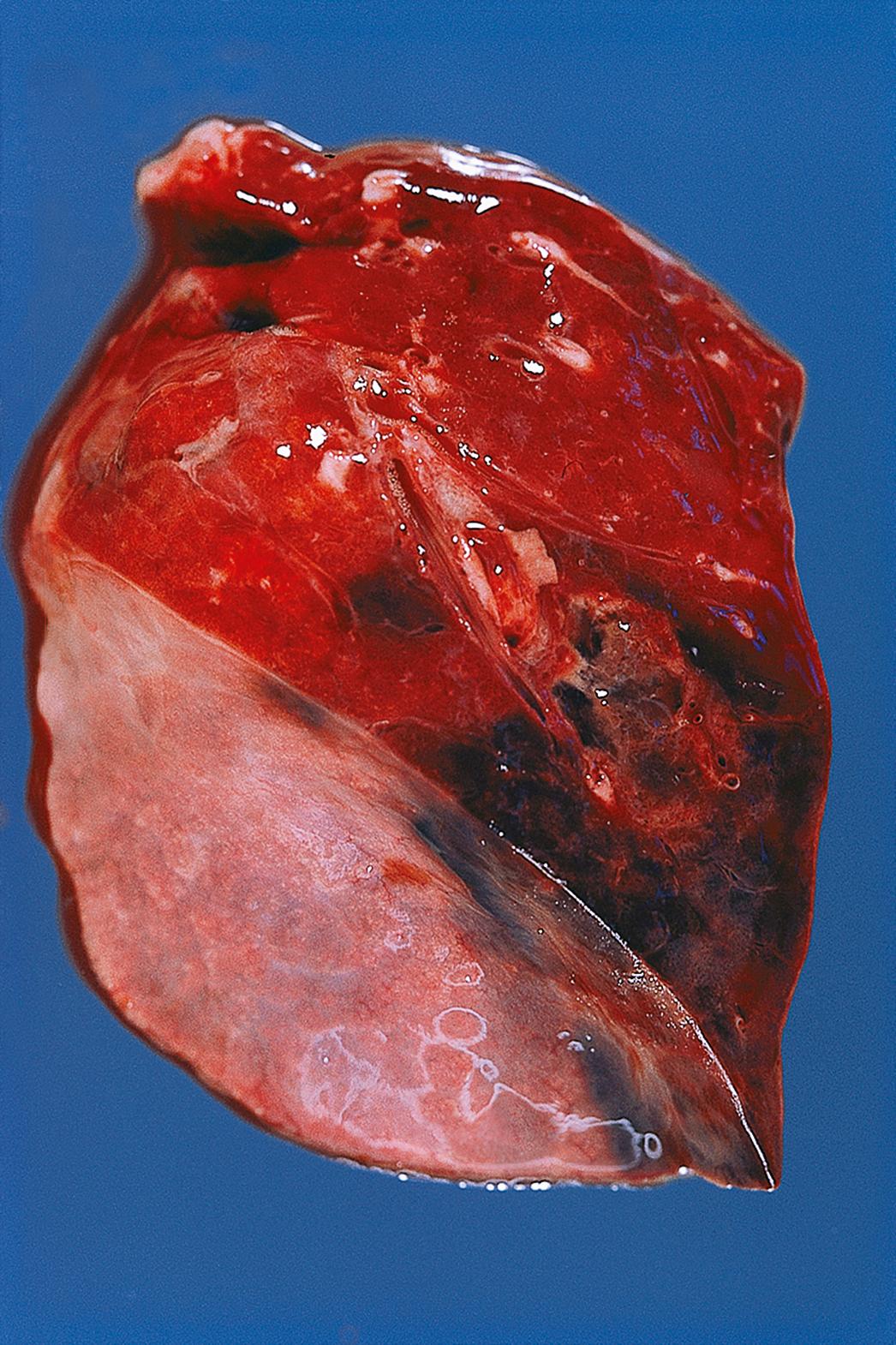
Emboli are most commonly caused by detachment of all or part of a thrombus from its site of formation. Venous thromboembolism is of clinical importance as, for example, following deep venous thrombosis in the leg, the thrombus may propagate as far as the common iliac vein or even the inferior vena cava. Part of such a huge thrombus may readily become detached and pass via the right side of the heart to lodge in the pulmonary arterial tree as a pulmonary embolus , which is often fatal ( Fig. 9.4 ) and ( E-Fig. 9.3 G ).
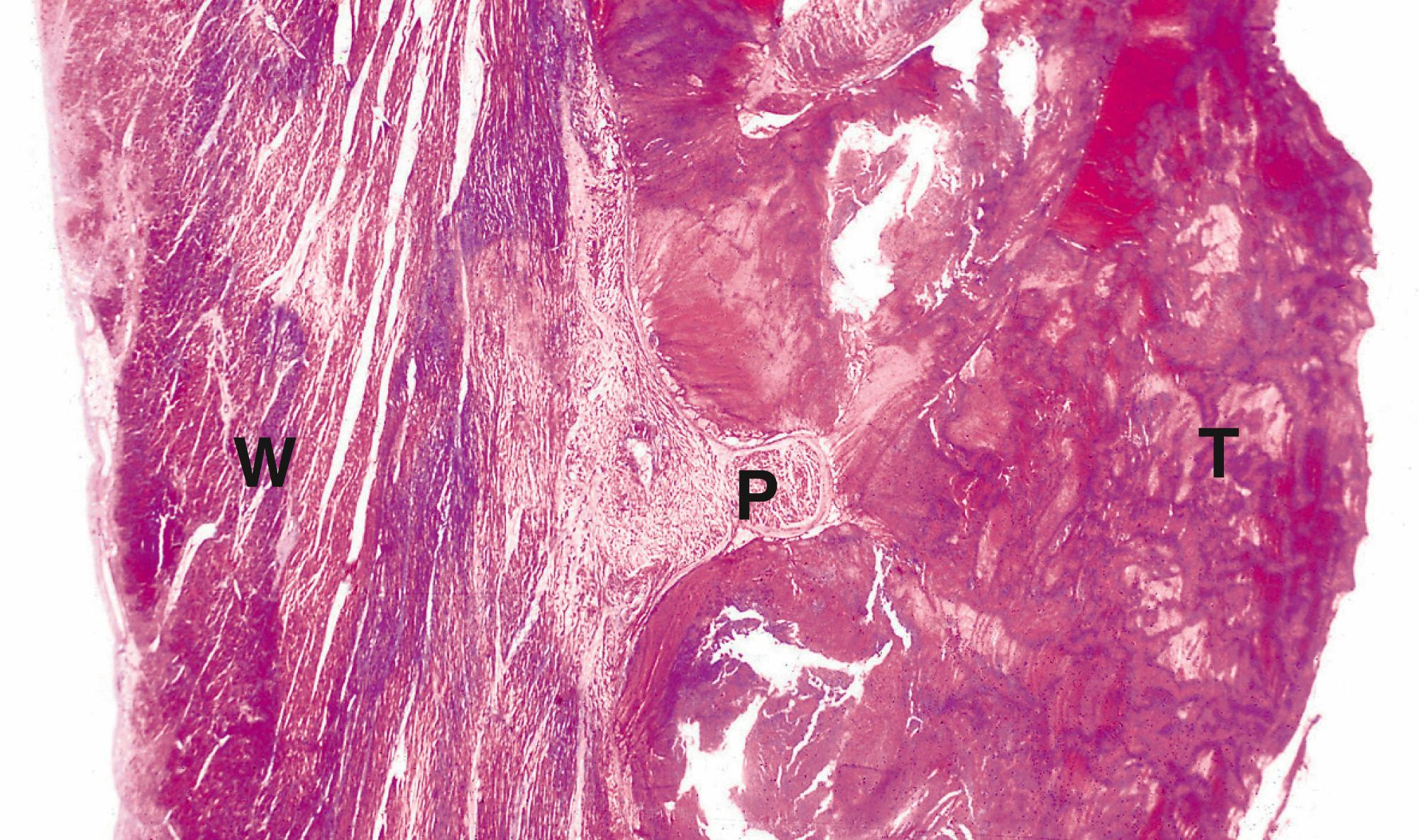
Systemic arterial thromboemboli most commonly arise from the heart or major arteries. In such cases, the thrombus often covers only part of the luminal wall as a plaque-like structure and is known as mural thrombus . Such thrombi may form on the ventricular endocardium following myocardial infarction ( Fig. 9.3 ). Emboli arising in the arterial system impact in peripheral arterial vessels where the most dramatic outcome is necrosis of the tissue supplied (infarction) ; this is described in detail in Ch. 10 .
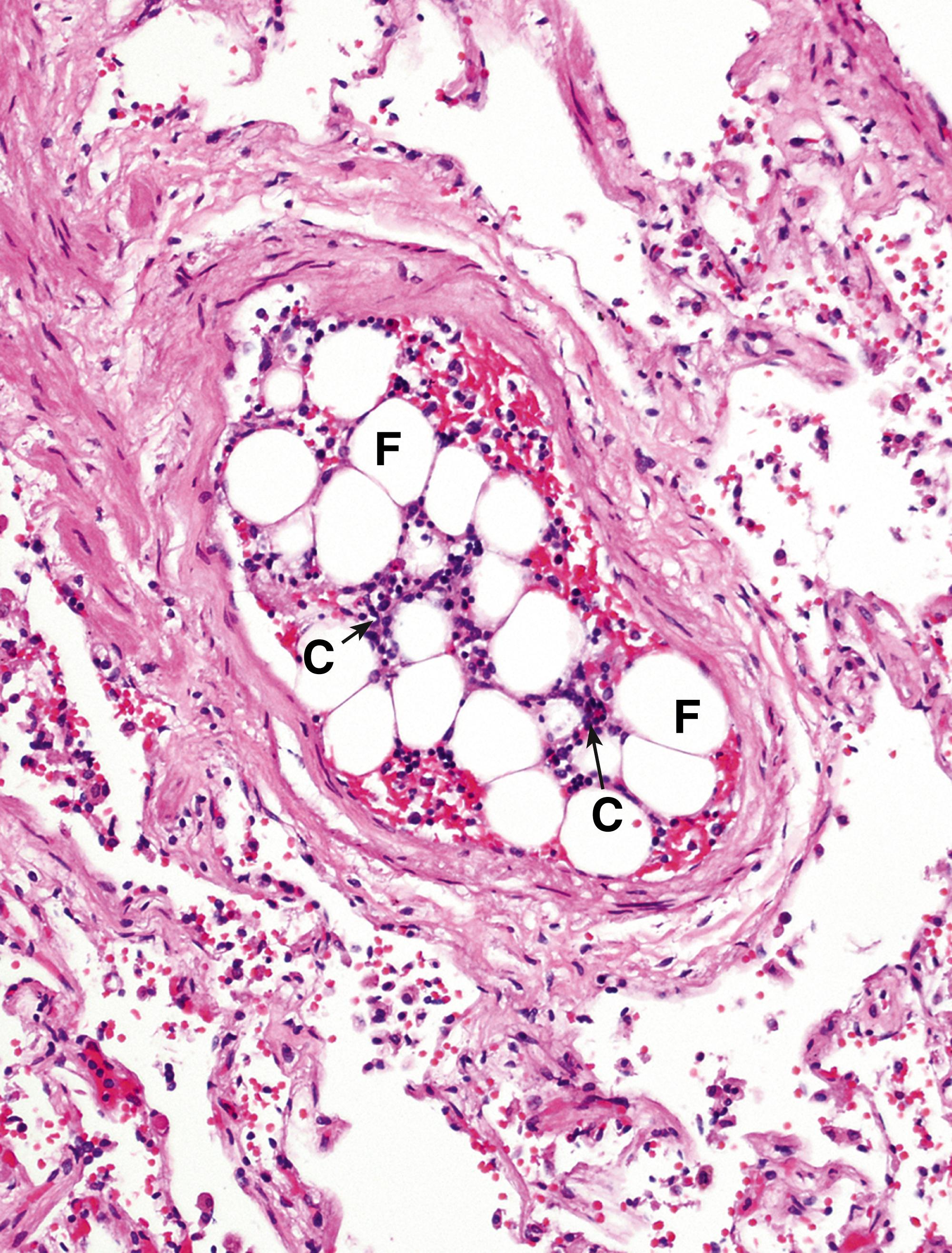
Whilst thrombotic emboli are the most common, emboli may also arise from other sources. These include atheromatous debris, tumour cells, bacterial vegetation from infected heart valves, fat and bone marrow after bone fracture ( Fig. 9.5 ), air entering the circulation and, rarely, amniotic fluid in cases of complicated pregnancy. Embolisation can also have a therapeutic role (see Fig. 9.6 ).
Resolution: This involves the normal physiological phenomenon of fibrinolysis as well as autolytic disintegration of the cellular elements of the thrombus. The end result is complete dissolution with restoration of blood flow. This process may be enhanced by the therapeutic administration of thrombolytic drugs, a principle of the acute management of stroke (see clinical box ‘Clinical management of thrombosis and embolism’).
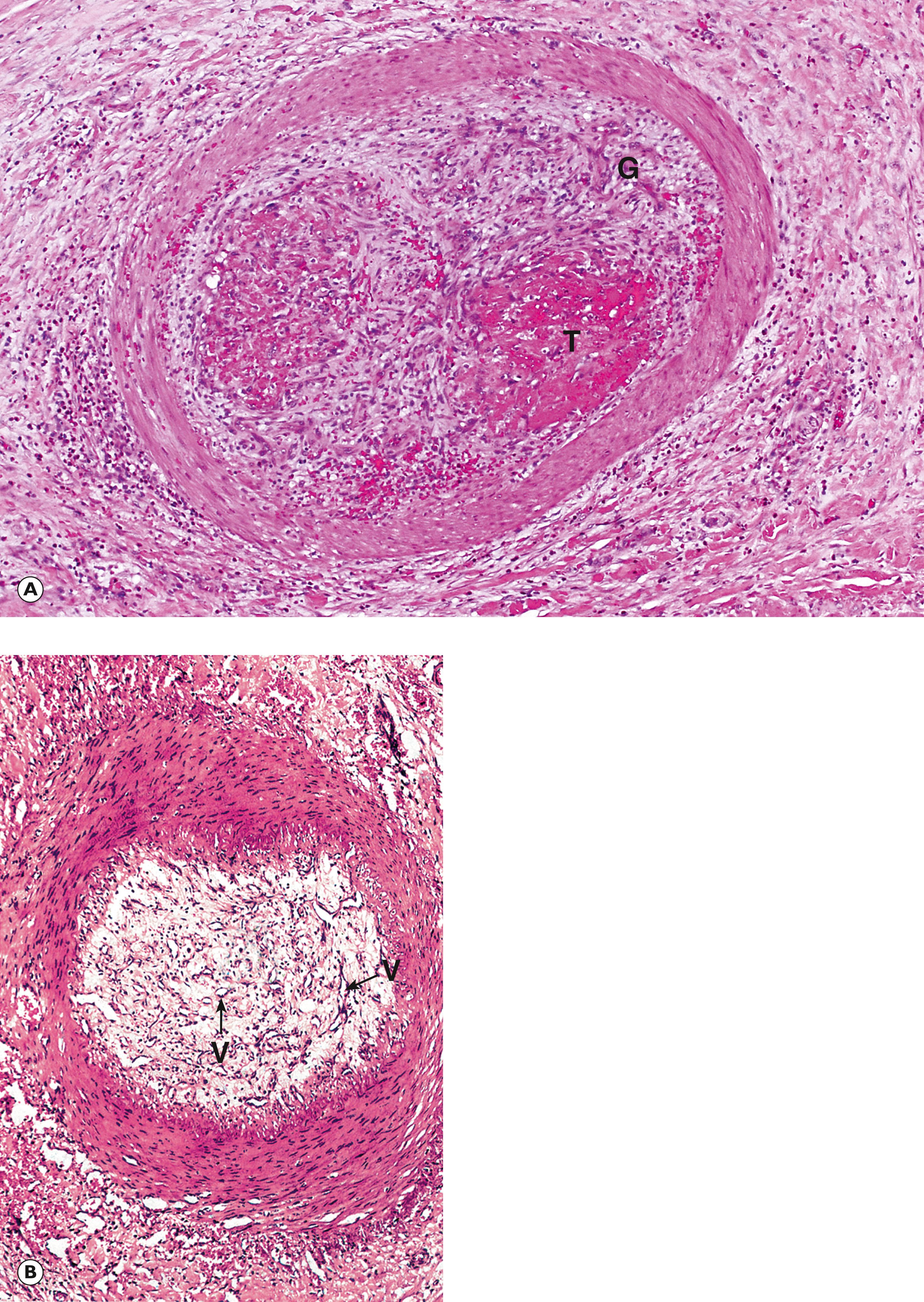
Organisation: Ingrowth of granulation tissue from the vessel wall and subsequent fibrous repair occur when fibrinolysis is ineffective in removing the thrombus. By an extraordinary mechanism, the organised thrombus may undergo recanalisation, a process whereby new vascular channels are formed to re-establish a patent lumen. The processes of organisation and recanalisation are illustrated in Fig. 9.7 .
E embolus P papillary muscle T thrombus W ventricular wall
To supplement the natural process of fibrinolysis and hasten dissolution of thrombus in clinical situations where early restoration of blood flow (reperfusion) to an organ may be indicated, a variety of pharmacological agents have been developed that aid breakdown (lysis) of the thrombus. Hence this treatment is known as thrombolysis and the drugs used are termed thrombolytics . These drugs act to increase the conversion of naturally occurring plasminogen to plasmin, which has powerful fibrinolytic properties. An example is recombinant tissue plasminogen activator (rt-PA).
Administration of these agents has transformed the acute management of stroke. The aim is to achieve reperfusion of the vascular bed as quickly as possible, to minimise the volume of tissue infarcted.
Embolectomy can help to treat ischaemic limbs and can prevent the need for amputation. In this process, either a surgeon or an interventional radiologist can use a catheter to remove an embolus from an artery within a limb, restoring blood flow to the ischaemic area.
Some patients take anticoagulant drugs such as warfarin, a coumarin-based medication, particularly if they have an irregular heart rhythm called atrial fibrillation. This has been shown to decrease the frequency of strokes caused by embolism of atrial thrombi into the cerebral arterial tree.
A artery C bone marrow cells F fat G granulation tissue S embolisation sphere T thrombus V blood vessels
| Feature | Definition | Figure |
|---|---|---|
| Thrombus | Aggregate of platelets, fibrin and blood cells formed in vessel lumen by the process of thrombosis | 9.1 9.2 9.3 |
| Virchow’s triad | Factors predisposing to thrombus formation namely damaged endothelium, abnormal blood flow and hypercoagulability | |
| Fate of thrombi | Propagation: enlargement with growth along vessel or occlusion of vessel | |
| Embolisation: see below | 9.4 | |
| Resolution: dissolution of the thrombus by fibrinolysis | ||
| Organisation: where fibrinolysis is unsuccessful, granulation tissue may form with subsequent fibrous repair and recanalisation | 9.7 | |
| Embolus | Fragment of thrombus or other material (e.g. bone marrow, air) passing with the circulation to lodge in a distal vessel | 9.5 9.6 |
Virchow’s triad states that the following three factors predispose to thrombosis: damage to endothelium, disordered blood flow and hypercoagulability. During thrombogenesis, activated platelets secrete which of the following substances?
Thromboxane A2
Thrombin
ATP
Histamine
Fibrin
Which ONE of the following does NOT pre-dispose to pulmonary thromboembolism?
Obesity
Factor V Leiden disorder
Oral contraceptive pill
Immobility
Haemophilia
Which of the following can be associated with an embolism?
Bone marrow
Atheroma
Tumour
Air
All of the above
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here