Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Gastrointestinal stromal tumors (GISTs) are the most common soft tissue tumors of the gastrointestinal (GI) tract, with a worldwide annual incidence of about 15 cases per million. Although there was historic controversy over the distinction between GIST, smooth muscle tumors, and nerve sheath tumors, by the late 1990s, it was established that GISTs exhibit differentiation of interstitial cells of Cajal and represent a distinct diagnostic entity. We now know that most GISTs harbor mutually exclusive mutations in KIT (80%) or platelet-derived growth factor receptor-α (PDGFRA) (∼7%). The remaining 10% to 15% of so-called “wild-type GISTs” are a heterogeneous group of tumors that includes succinate dehydrogenase (SDH)–deficient GIST (accounting for 8% of GISTs), as well as neurofibromatosis-associated and BRAF -mutant GIST ( Table 7.1 ). Correct subclassification is critical for determining the prognosis and appropriate treatment.
| Mutation | Prevalence (%) |
|---|---|
| KIT | 80 |
| Exon 9 | 8 |
| Exon 11 | 70 |
| Exon 13 | 1 |
| Exon 17 | 1 |
| PDGFRA | 7 |
| Exon 12 | 1.5 |
| Exon 14 | 0.5 |
| Exon 18 | 5 |
| SDH Deficiency | 8 |
| SDHA inactivating mutation | ~3 |
| SDHB , SDHC , SDHD inactivating mutations | ~2 |
| SDHC promoter hypermethylation | ~3 |
| NF1 Biallelic Inactivation | 2–3 |
| BRAF V600E | <1 |
a NF , Neurofibromatosis; PDGFRA , platelet derived growth factor receptor alpha; SDH , succinate dehydrogenase (subunits A-C).
In the United States, GIST has an estimated annual incidence of 3300 to 6000 cases per year and most commonly occurs in middle-aged to older adults (median age, 60 years). Clinically detected tumors in the GI tract most commonly occur in the stomach (60%) and small intestine (35%), with most remaining cases accounted for by colorectal tumors; GISTs of the esophagus and appendix are rare. GIST can also present as omental, peritoneal, and retroperitoneal tumors, although these are typically large tumors that very likely originally originated in the GI tract but may have detached from their point of origin. GIST exhibits a wide range of behavior, from benign “micro-GISTs,” present in approximately 30% of the population based on autopsy series, to aggressive sarcomas. Prognosis depends in part on tumor location; for example, small intestinal GISTs are about twice as likely as gastric GISTs to behave in a clinically aggressive fashion.
Clinical presentation depends on tumor size and location. Large masses can cause abdominal pain, anemia, or GI bleeding. Micro-GISTs are found incidentally during surgery and may often be sent for frozen section analysis. GIST can also present as a component of multiple hereditary syndromes, including Carney-Stratakis syndrome, Carney triad, and neurofibromatosis. Carney-Stratakis syndrome (the dyad of gastric GIST and paraganglioma) is a familial syndrome caused by mutations in SDHx genes; these mutations account for about 65% of SDH-deficient GISTs. Patients with SDHA germline mutations mostly present with GISTs as young to middle-aged adults, whereas patients with other SDH germline mutations present in childhood and adolescence. Carney triad (gastric GIST, paraganglioma, and pulmonary chondroma) is a nonfamilial syndrome associated with SDHC promoter hypermethylation and not germline mutations. Neurofibromatosis type 1 (NF1) is the most common autosomal dominant genetic disorder and is associated with development of neurofibromas, ganglioneuromas, and malignant peripheral nerve sheath tumors (MPNSTs), among other tumor types. NF1 also predisposes patients to GISTs that are predominantly found in the small intestine and can be multiple in the same patient. These tumors are mostly low risk and are associated with interstitial cell hyperplasia. Last, there are rare instances of familial germline mutations in KIT and PDGFRA ; patients with these mutations develop GISTs with nearly 100% penetrance, in a background of interstitial cell hyperplasia. Patients with KIT germline mutations also develop urticaria pigmentosa and cutaneous hyperpigmentation.
Radiologic features are not specific, but most large mural gastric masses are GISTs. An example of a gastric GIST is shown in Fig. 7.1 .

Gastrointestinal stromal tumors vary in greatest dimension from smaller than 1 cm (micro-GISTs) to larger than 20 cm, and they can be submucosal, intramuscular, or subserosal, although most are centered in the muscularis propria. They can be solid or cystic with variable hemorrhage and necrosis, and there may be overlying mucosal ulceration. Generally, GISTs are well circumscribed. SDH-deficient GISTs have a characteristic multinodular or plexiform growth pattern. Fig. 7.2 shows a cross-section of a small intestinal GIST.

About 70% of gastric GISTs and nearly all of those in the small and large intestine exhibit spindle cell morphology, with short fascicles of bland cells with indistinct cell borders, palely eosinophilic cytoplasm, and uniform, tapered nuclei with evenly dispersed chromatin ( Figs. 7.3 and 7.4 ). Nuclear palisading is at least focally present in more than half of tumors. About 20% of GISTs have purely epithelioid morphology; these tumors nearly all occur in the stomach and are associated with PDGFRA mutations ( Fig. 7.5 ) or SDH deficiency ( Fig. 7.6 ). Epithelioid GISTs are composed of sheets and nests of cells with palely eosinophilic cytoplasm and uniform nuclei, often in a myxohyaline stroma ( Fig. 7.7 ; see also Figs. 7.5 and 7.6 ). The cell membranes are often better defined than spindle cell variants. SDH-deficient GIST has a characteristic multinodular or plexiform growth pattern and the cytomorphology is usually epithelioid (see Fig. 7.6 ). Gastric GISTs commonly have paranuclear vacuoles (see Figs. 7.3 and 7.7 ), and nearly half of small intestinal GISTs contain skeinoid fibers. Mitotic activity varies widely and is an important prognostic factor. Treatment effect can manifest as stromal hyalinization, decreased cellularity, or reduced mitotic activity. Rarely, tumors can develop heterologous rhabdomyosarcomatous differentiation after treatment, and both treated and treatment-naïve tumors can undergo dedifferentiation, in which there is a sharp transition from conventional morphology to high-grade pleomorphic regions that do not resemble GIST ( Fig. 7.8 ). Both rhabdomyosarcomatous differentiation and dedifferentiation are associated with imatinib resistance and a poor prognosis.






KIT (also known as CD117) is a receptor tyrosine kinase expressed in interstitial cells of Cajal, mast cells, melanocytes, a variety of epithelia, fetal endothelial cells, and a subset of CD34-positive hematopoietic stem cells. Oncogenic KIT mutations were discovered in the late 1990s to be present in the majority of GISTs, along with KIT overexpression. DOG1 (discovered on GIST1), a chloride channel normally expressed in the interstitial cells of Cajal, was also found to be expressed in GIST (hence the name “discovered on GIST 1”). Overall, KIT is positive in 95% of GISTs ( Fig. 7.9 ), and DOG1 is also positive in 95% ( Fig. 7.10 ). Fewer than 3% of tumors are negative for both markers; these tend to be epithelioid PDGFRA -mutant GISTs, and PDGFRA immunohistochemistry (IHC) is strongly and diffusely positive in essentially 100% of these tumors (see Fig. 7.5 ). PDGFRA is also weakly expressed in stromal cells, presenting a potential pitfall; only strong and diffuse staining should be used to support the diagnosis of GIST. CD34 is positive in about 70% of GISTs, although its diagnostic utility is now limited, given the superior sensitivity and specificity of KIT, DOG1, and PDGFRA IHC. Caldesmon is positive in 70% of GISTs, S-100 in 1% to 2% (especially duodenal GISTs), and desmin in 1% to 2% (but up to 10% in epithelioid PDGFRA -mutant GISTs; see Fig. 7.5D ). Keratin positivity is rare.
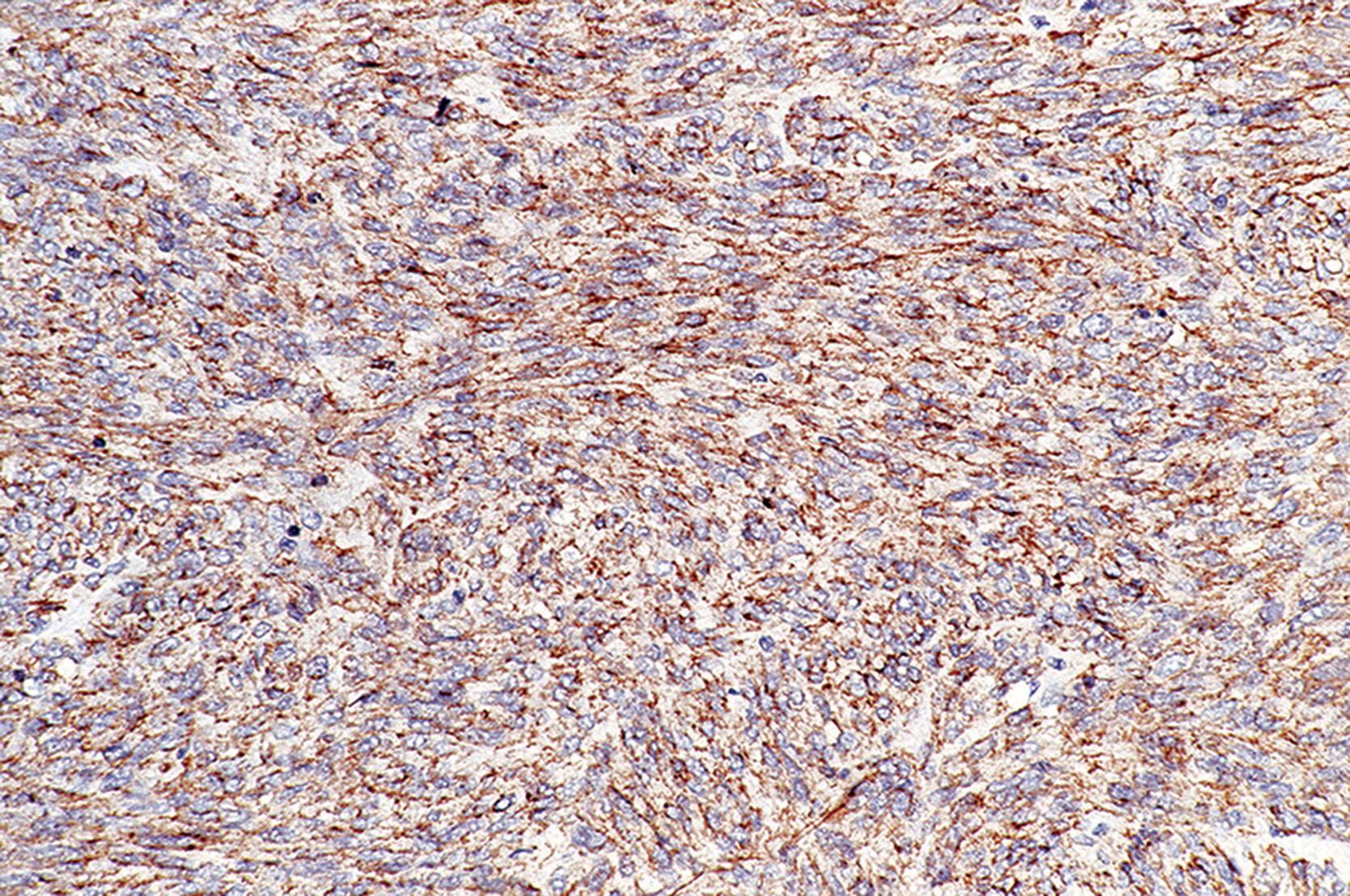

Immunohistochemistry is also useful for diagnosing SDH-deficient GIST, which are generally positive for KIT and DOG1. The four SDH subunits (A–D) form a protein complex that is necessary to stabilize the SDHB protein. Therefore, loss of expression of any one of the subunits leads to loss of expression of SDHB. Thus, SDHB IHC can be used to screen for loss of expression of any subunit. SDHA expression is lost in about 35% of tumors, an event that is reliably detectable using SDHA IHC and that generally indicates underlying SDHA mutations. Of the 65% of SDH-deficient GISTs that express SDHA, about 30% have mutations in SDHB , SDHC , or SDHD , and the remaining 35% have SDHC promoter hypermethylation.
Gastrointestinal stromal tumors are not classified as benign or malignant tumors; instead, a risk stratification scheme is used, with prognosis determined by using site, size, and mitotic index ( Table 7.2 ). Mitoses should be counted in a 5-mm 2 area; on most modern microscopes, 5 mm 2 is close to 20 hpf. When GISTs behave in a clinically aggressive fashion, they most commonly metastasize to the abdominal cavity and liver. Metastasis can occur more than 10 years after surgical resection, underscoring the need for long-term follow-up.
| Tumor Parameter | Risk of Progressive Disease b (%) | ||||
|---|---|---|---|---|---|
| Mitotic Index | Size (cm) | Stomach | Duodenum | Jejunum/ileum | Rectum |
| ≤5 per 5 mm 2 | ≤2 | None (0%) | None (0%) | None (0%) | None (0%) |
| >2–≤5 | Very low (1.9%) | Low (8.3%) | Low (4.3%) | Low (8.5%) | |
| >5–≤10 | Low (3.6%) | Insufficient data | Moderate (24%) | Insufficient data | |
| >10 | Moderate (10%) | High (34%) | High (52%) | High (57%) | |
| >5 per 5 mm 2 | ≤2 | None | Insufficient data | High | High (54%) |
| >2–≤5 | Moderate (16%) | High (50%) | High (73%) | High (52%) | |
| >5–≤10 | High (55%) | Insufficient data | High (85%) | Insufficient data | |
| >10 | High (86%) | High (86%) | High (90%) | High (71%) | |
a Does not apply to succinate dehydrogenase–deficient gastrointestinal stromal tumors.
Several studies have established relationships between specific GIST genotypes and their corresponding clinicopathologic phenotypes. The protein structures of KIT and PDGFRA receptors are similar, and activating a mutation in either one of these two genes leads to activation of similar signal transduction pathways and these mutations are thought to be initiating events in GIST tumorigenesis.
Eighty percent of GISTs have KIT mutations, and 7% have PDGFRA mutations. KIT mutations occur in exon 11 (overall accounting for 70% of GISTs), exon 9 (8%), exon 13 (1%), and exon 17 (1%) (see Table 7.1 ). Exon 11 (juxtamembrane domain) mutant GISTs occur in the stomach and small intestine, and they have spindle cell or mixed morphology. Exon 11 mutations include in-frame deletions (60%–70%), missense point mutations (20%–30%), and internal tandem duplications; whereas exon 11 point-mutant GISTs have a better prognosis than other GISTs, tumors with exon 11 deletions behave more aggressively. Recent clinical trials demonstrated that patients with exon 11 deletions benefit from an extended course of adjuvant imatinib therapy.
Exon 9 (dimerization motif domain) mutations occur in tumors typically located in the small and large intestines; these tumors have spindle cell or mixed morphology, and they require higher doses of imatinib to achieve treatment response. Exon 13 and exon 17 mutations together account for 2% of GISTs; these tumors mostly occur in the small intestine, have spindle cell morphology, and have inconsistent responses to imatinib.
About 8% of GISTs harbor mutations to PDGFRA ; as in KIT -mutant GISTs, these occur in different exons corresponding to different protein domains. PDGFRA mutations occur in exon 18 (accounting for 7% of GISTs overall), exon 12 (1.5%), and exon 14 (0.5%). PDGFRA -mutant GISTs predominantly occur in the stomach, and essentially all have purely epithelioid morphology. A particular missense mutation (D842V) accounts for the majority of exon 18 mutations and confers imatinib resistance; recently, the Food and Drug Administration (FDA)-approved avapritinib to treat patients with these GISTs. Exon 12– and exon 14–mutant GISTs have variable responses to imatinib.
PDGFRA mutations, KIT mutations, and biallelic NF1 inactivation are thought to be initiating events in GIST tumorigenesis; all three subtypes undergo the same additional chromosomal deletions as part of an apparent stepwise progression toward higher- risk tumor biology. Given that they share the same progression, it is perhaps not surprising that the prognosis of these three GIST subtypes can be determined using the same risk stratification system (see Table 7.2 ).
SDH deficiency is seen in about 8% of GISTs and is mutually exclusive with PDGFRA and KIT mutations and NF1 inactivation. SDH-deficient GIST represents the majority of pediatric GIST but also occurs in a significant subset of adults. SDH-deficient GISTs are almost always gastric and show an epithelioid morphology with a unique plexiform architectural growth pattern (see Fig. 7.6 ). SDHA, SDHB, SDHC, and SDHD proteins form a multimeric complex in the inner mitochondrial membrane that is critical for cellular metabolism. Whereas SDHA mutations are present in about 35% of SDH-deficient GISTs, about 30% have mutations in SDHB , SDHC , or SDHD . The remaining 35% of SDH-deficient GISTs have SDHC promoter hypermethylation, which silences expression of SDHC, leading to SDH deficiency. SDHB, SDHC , or SDHD mutations are usually present in the germline, and these patients also develop paragangliomas in the familial Carney-Stratakis syndrome, as discussed earlier.
The prognosis of SDH-deficient GIST is different from the other described genetic subtypes, so the risk stratification system developed for KIT - and PDGFRA -mutant tumors does not apply. Instead, SDH-deficient GISTs frequently give rise to slowly growing lymph node metastases that progress over the course of several years to decades. SDH-deficient GISTs do not respond to imatinib therapy, and as of now, there is no targeted therapy to treat patients with these tumors.
BRAF -mutant GISTs are rare. Tumors harboring BRAF V600E occur in the small intestine and have spindle cell morphology. Although they do not respond to imatinib, they have been shown to respond to dabrafenib, a BRAF inhibitor.
The differential diagnosis for spindle cell GIST includes leiomyoma, leiomyosarcoma, schwannoma, and desmoid fibromatosis. Leiomyoma and leiomyosarcoma have more brightly eosinophilic cytoplasm, and they exhibit distinct cell borders in contrast to the indistinct cell borders of GIST. Leiomyoma and leiomyosarcoma also have blunt-ended nuclei, unlike the tapered nuclei of GIST. IHC is useful to resolve the differential diagnosis: leiomyoma and leiomyosarcoma commonly have diffuse desmin expression, which would be unusual in GIST, and they are negative for KIT and DOG1.
Gastric schwannoma typically has a peripheral cuff of lymphocytes and neural cytomorphology with buckled nuclei and mild atypia. The cells of schwannoma show diffuse S-100 expression and are negative for DOG1 and KIT.
Desmoid fibromatosis is more infiltrative than GIST and has longer fascicles. IHC demonstrates nuclear β-catenin positivity in 70% to 80% of desmoid fibromatoses, which is negative in GIST. Desmoid fibromatosis does not express KIT or DOG1.
The differential diagnosis for epithelioid GIST includes neuroendocrine tumors and gastric adenocarcinoma. Neuroendocrine tumors usually have smaller cells and darker nuclei with speckled chromatin. IHC for synaptophysin, chromogranin, KIT, and DOG1 readily distinguishes the two. Gastric adenocarcinoma exhibits significant nuclear pleomorphism and hyperchromasia, in contrast to the uniform nuclei and evenly dispersed chromatin of epithelioid GIST.
Surgical excision is the treatment for patients with resectable GIST. Narrow margins suffice because tumors tend not to recur at the surgical site; the exception is SDH-deficient GISTs, likely because of their multinodular, infiltrative growth pattern. For most KIT -mutant GISTs and some PDGFRA -mutant GISTs, imatinib is the first-line therapy in the setting of metastases or locally advanced disease and as adjuvant therapy in GISTs that stratify into intermediate- or high-risk groups (see Table 7.2 ). Neoadjuvant imatinib is also used in some cases to reduce the size of locally advanced tumors prior to surgery. Imatinib resistance eventually develops in all treated GISTs, usually by means of secondary KIT mutations. Sunitinib and regorafenib are FDA-approved second- and third-line therapies, respectively, that are used in the setting of imatinib resistance. As mentioned earlier, avapritinib was recently FDA approved for first-line treatment of patients with PDGFRA exon 18–mutant GIST, which is resistant to imatinib, sunitinib, and regorafenib.
Gastrointestinal stromal tumor (GIST) is the most common mesenchymal tumor in the gastrointestinal (GI) tract and exhibits differentiation of the interstitial cells of Cajal
3300 to 6000 cases per year in the United States
Most commonly occurs in the stomach (60%) and small intestine (35%)
Tumor mortality depends in part on primary site: intestinal GISTs are about twice as likely to behave aggressively compared with gastric GISTs.
Tumor size and mitotic count also influence clinical behavior
No sex predilection
May be overrepresented in African Americans
Most commonly occurs in adults, with a median age at presentation of 60 years
Pediatric tumors are typically succinate dehydrogenase deficient
Clinical presentation depends on primary site but usually includes abdominal pain, symptoms of GI bleeding such as anemia or melena or obstruction
Small tumors are often detected incidentally
Tumors are not classified as benign or malignant; instead, they are risk stratified based on anatomic site, tumor size, and mitotic activity (see Tables 7.2 )
Prognosis and treatment are determined based on the driving mutation and risk category
Food and Drug Administration-approved treatment includes imatinib, sunitinib, and regorafenib (nonspecific tyrosine kinase inhibitors) and, more recently, avapritinib (used for PDGFRA exon 18-mutant GIST)
Mural mass, sometimes with protrusion into the gastrointestinal lumen
Fibrous to fleshy cut surface, with variable hemorrhage and/or cystic degeneration
Spindled or epithelioid cells with uniform nuclei and relatively limited atypia
Nuclear palisading common
Gastric gastrointestinal stromal tumors (GISTs) commonly have paranuclear vacuoles
About half of small intestinal GISTs have skeinoid fibers
Positive: DOG1 (discovered on GIST1; 95%), KIT (95%), CD34 (70
Variable: smooth muscle actin (30%), S-100 (5%)
Platelet-derived growth factor receptor-α ( PDGFRA )–mutant GISTs: positive for PDGFRA (nearly 100%), DOG1 (95%), KIT (85%), desmin (≤10%)
Determines optimal treatment
KIT mutations (80%) in exon 11 (70%), exon 9 (8%), exon 13 (1%), exon 17 (1%), or exon 8 (rare)
PDGFRA mutations (7%) in exon 18 (5%), exon 12 (1.5%), or exon 14 (0.5%)
Succinate dehydrogenase (SDH) deficiency (8%): SDHA mutations (35%); SDHB , SDHC , or SDHD mutations (30%); SDHC promoter hypermethylation (35%); no KIT or PDGFRA mutations
Biallelic NF1 inactivation (2%–3%); no KIT or PDGFRA mutations
BRAF V600E mutation (<1%)
Spindle cell GIST: leiomyoma or leiomyosarcoma (desmin positive, KIT/DOG1 negative); schwannoma or malignant peripheral nerve sheath tumor (S-100 positive, KIT negative); desmoid fibromatosis (70%–80% nuclear β-catenin positivity; 95% negative for KIT)
Epithelioid GIST: neuroendocrine tumor (synaptophysin and chromogranin positive, KIT and DOG1 negative), gastric adenocarcinoma (keratin positive, more atypical or pleomorphic)
Smooth muscle tumors of the GI tract consist of leiomyoma and the much rarer leiomyosarcoma. Colonic leiomyomas classically arise in association with the muscularis mucosae and present as distal polyps on screening colonoscopy, usually in men in their seventh decade. Esophageal leiomyomas often present as intramural masses; patients have a median age of 35 years, there is a male predominance (∼70%), and the clinical course is indolent. In the esophagus, the vast majority of spindle cell tumors are leiomyomas. Smooth muscle tumors of the stomach and small intestine are rare; GIST is much more common at these locations. Leiomyosarcoma of the GI tract is very rare and generally follows an aggressive course. Leiomyosarcoma of the colon shows a slight male predominance and presents in adults. Leiomyosarcoma is exceptional in the stomach. Leiomyomatosis has been described in the small intestine and colon.
The radiologic features are not specific and are not distinguishable from those of GIST.
Esophageal leiomyomas form whorled white to pale yellow masses similar to uterine leiomyomas. Leiomyomas arising in the muscularis mucosae of the colorectum present as mucosal polyps, usually smaller than 1 cm. Leiomyosarcomas of the colorectum tend to be intraluminal bulging exophytic and polypoid masses, which may show necrosis and hemorrhage.
Smooth muscle tumors of the GI tract have the same features of smooth muscle differentiation seen at other anatomic sites, namely perpendicularly oriented fascicles of spindle cells with distinct cell borders, brightly eosinophilic cytoplasm, longitudinal striations, and blunt-ended or cigar-shaped nuclei ( Figs. 7.11 to 7.15 ). Leiomyomas are bland and resemble normal smooth muscle. They lack atypia and pleomorphism and generally show no or minimal mitotic activity. Leiomyosarcoma is characterized by conspicuous mitotic activity, cytologic atypia, and necrosis.





Smooth muscle tumors show expression of smooth muscle actin (SMA), desmin, and caldesmon, but expression of the latter two markers may be variable in extent, especially in leiomyosarcoma. KIT, CD34, and S-100 protein are generally not expressed. Approximately 40% of leiomyosarcomas show variable keratin expression, which may be extensive occasionally and make distinction from sarcomatoid carcinoma difficult. Esophageal leiomyomas often contain numerous mast cells that are KIT positive, which may result in an erroneous diagnosis of GIST.
The differential diagnosis is primarily with GIST and schwannoma, and the majority of lesions are readily separable by IHC. Sarcomatoid carcinoma may mimic leiomyosarcoma because both are composed of fascicles of atypical spindle cells, which may express keratins. Identification of a more epithelioid or conventional carcinomatous component allows for recognition of sarcomatoid carcinoma. In addition, the latter does not show desmin expression.
The prognosis of leiomyomas is excellent in all sites because they are benign. In general, GI tract leiomyosarcomas have a poor prognosis, and about 75% of patients die of disease.
Benign or malignant neoplasms with smooth muscle differentiation
Leiomyoma is far more common than leiomyosarcoma, and leiomyoma is the most common spindle cell tumor of the esophagus.
Small polypoid leiomyomas arise in association with the muscularis mucosae of the colorectum
Leiomyomas are benign; in contrast, 75% of patients with leiomyosarcomas of the gastrointestinal tract die of disease
Occur in adults (average, 60 years); slight male predominance
Whereas smooth muscle tumors of the esophagus present as an intramural mass, smooth muscle tumors of the colon present as polypoid lesions
Smooth muscle tumors of the small intestine can present as either intramural masses or polypoid lesions
Poor prognosis for leiomyosarcomas; leiomyomas are benign.
All lesions except small polypoid leiomyomas of the colorectum are treated surgically when possible; there is no targeted chemotherapy as there is for gastrointestinal stromal tumor
Esophageal leiomyomas are whorled, whitish masses
Leiomyosarcomas are large, exophytic, mural-based masses, with variable amounts of necrosis and hemorrhage
Fascicular spindle cell neoplasm; tumor cells have brightly eosinophilic cytoplasm, distinct cell borders, and blunt-ended cigar-shaped nuclei
Leiomyomas are bland, resemble normal smooth muscle, and lack atypia and necrosis
Leiomyosarcomas show cytologic and nuclear pleomorphism and atypia, mitotic activity, and necrosis
Positive for desmin, smooth muscle actin, caldesmon
Negative for KIT, DOG1 and S-100 protein
Focal keratin expression is common in leiomyosarcoma
Usually negative for CD34
Gastrointestinal stromal tumor: cytoplasm is paler; may show skeinoid fibers or paranuclear vacuoles; KIT and DOG1 positive and desmin negative
Schwannoma: usually larger gastric lesions with a peripheral lymphoid cuff or smaller polypoid colonic lesions that entrap colonic crypts, different from the growth pattern of leiomyoma; tumor cells are positive for S-100
Sarcomatoid carcinoma: may be difficult to distinguish from leiomyosarcoma, given that both can show keratin expression; identification of an epithelioid or overtly carcinomatous component is particularly helpful and may require extensive sampling
Granular cell tumors are benign tumors that exhibit Schwannian differentiation. Although they can occur anywhere along the luminal GI tract, they have a striking predilection for the esophagus. Granular cell tumors usually occur in middle-aged adults and are more common in women and African Americans. Although they usually occur as unifocal tumors in the distal esophagus, they can also present as multiple sporadic esophageal tumors.
Granular cell tumors are often found incidentally on endoscopy, and they appear as small polyps or plaques.
Most granular cell tumors are small (<1 cm) with a yellow-white cut surface and infiltrative peripheral growth.
Granular cell tumors are composed of sheets and nests of large polygonal cells with brightly eosinophilic, granular cytoplasm, and centrally located, small, hyperchromatic nuclei ( Figs. 7.16 and 7.17 ). There is frequently overlying squamous cell hyperplasia, a useful diagnostic clue. Occasionally, tumors can have spindle cell morphology, although the spindle cells still contain abundant granular cytoplasm. In conventional granular cell tumor, mitotic activity is low, and there is no necrosis.


Diffuse strong expression of S-100 protein and SOX10 is present in esophageal granular cell tumors ( Fig. 7.18 ). Granular cell tumors also overexpress transcription factor enhancer 3 (TFE3), but they do not harbor underlying TFE3 rearrangements.

Granular cell tumors were recently found to have recurrent loss of function mutations in ATP6AP1 and ATP6AP2 , which are endosomal pH regulatory proteins; loss of expression of these proteins was shown to be oncogenic and to lead to accumulation of intracytoplasmic granules.
The differential diagnosis is limited because the cytomorphology of granular cell tumor is unique. Nested examples can sometimes resemble alveolar soft part sarcoma, which also overexpresses TFE3; however, alveolar soft part sarcoma has significantly more cytologic atypia. Granular cell tumors with spindle cell morphology could be mistaken for schwannoma, but the latter does not contain abundant granular cytoplasm.
The vast majority of granular cell tumors are benign and do not recur even if incompletely excised. The rare malignant granular cell tumors tend to have worrisome features such as increased mitotic activity and necrosis, neither of which are features of conventional granular cell tumor.
Benign neoplasm with Schwannian differentiation composed of large polygonal cells with bright eosinophilic granular cytoplasm
Most commonly occur in the distal esophagus and rarely occur at other sites in the gastrointestinal tract
Wide age distribution, with a peak in middle age
More common in women than men
More common in African American patients
Small incidental polyps or plaques found during endoscopy
Mostly benign and do no recur even with positive margins
Rare malignant granular cell tumors have worrisome features such as high mitotic activity and necrosis
Subepithelial mass with a yellow cut surface and infiltrative borders
Sheets and nests of large polygonal cells with abundant eosinophilic granular cytoplasm; uncommonly, neoplastic cells can have spindled morphology
Overlying squamous cell hyperplasia may be present
Strong and diffuse expression of S-100 and SOX10
Alveolar soft part sarcoma: both overexpress transcription factor enhancer 3 (TFE3), but granular cell tumor lacks the TFE3 rearrangement of alveolar soft part sarcoma
Schwannoma: can resemble spindle cell granular cell tumor, and both diffusely expression S-100 and SOX10; however, schwannoma lacks abundant granular cytoplasm
Clear cell sarcoma-like tumor of the GI tract, also known as malignant GI neuroectodermal tumor (malignant GNET), is a rare tumor with distinctive pathologic and genetic features, different from conventional clear cell sarcoma of tendons and aponeuroses. These tumors have also been called “osteoclast-rich tumors of the GI tract with features resembling clear cell sarcoma of soft parts.” In contrast to conventional clear cell sarcoma of tendons and aponeuroses, these tumors usually show less nested growth pattern, contain osteoclast-like giant cells, and lack evidence of melanocytic differentiation ultrastructurally and immunohistochemically. They both, however, express S-100 and harbor the EWSR1-ATF1 or EWSR1-CREB1 fusion genes.
The most common site of involvement is the small intestine followed by the stomach and colon. Patients are usually young to middle-aged adults (mean age, 42 years; range, 17–77 years), and there is no apparent gender predilection. Presenting symptoms and signs include abdominal pain, intestinal obstruction, weight loss, anemia, and fever. Metastasis at the time of presentation is common.
Tumors grow as exophytic masses that protrude into the lumen of the intestine. Overlying mucosal ulceration is often present. Tumors may also grow as circumferential masses, mimicking carcinoma or lymphoma.
Tumors typically involve both submucosa and muscularis propria and range in size from 2 to 15 cm. Extension into mucosa may occur. Tumors are usually solid and firm, with a tan-white cut surface. Hemorrhage, necrosis, and cystic change may be present.
Malignant GNET is composed of sheets of round to epithelioid cells and in some cases ovoid to spindled cells ( Fig. 7.19 ). Tumor cells have small amounts of pale eosinophilic cytoplasm and round to oval nuclei with vesicular chromatin and small nucleoli ( Fig. 7.20 ). Mitotic activity is variable. The growth pattern is usually mixed, with focal areas of nested, pseudoglandular, pseudopapillary, microcystic, and fascicular architecture. Osteoclast-like giant cells are present in 50% of cases.


Strong diffuse expression of S-100 is present in all cases ( Fig. 7.21 ). SOX10 expression is also seen, reflecting the neuroectodermal differentiation of this tumor. In contrast, melanocytic markers HMB45 (human melanoma black), Melan-A, microphthalmia transcription factor (MiTF), and tyrosinase are negative. Tumor cells are also negative for KIT, DOG1, CD34, glial fibrillary acidic protein (GFAP), keratins, desmin, and SMA. Expression of synaptophysin and neuron-specific enolase (NSE) is seen in up to 50% of cases. Ultrastructural features of neural differentiation are seen by electron microscopy.

Malignant GNET is a translocation-associated tumor, with EWSR1-ATF1 and EWSR1-CREB1 fusion genes in approximately 90% of cases caused by t(12;22)(q13;q12) or t(2;22)(q34;q12), respectively. FISH for EWSR1 rearrangement is useful to exclude a diagnosis of malignant melanoma.
Conventional clear cell sarcoma typically arises in tendons or aponeuroses of the extremities of young adults and rarely involves the GI tract. Clear cell sarcoma is characterized by epithelioid or spindle-shaped tumor cells (or both) with clear or pale eosinophilic cytoplasm, in a nested or fascicular growth pattern with a delicate fibrovascular stroma. The nested growth pattern is more prominent than in malignant GNET. Melanin pigmentation is rarely seen, and osteoclast- like giant cells are not typical, in contrast to malignant GNET. In addition to S-100 protein expression, the tumor cells express secondary melanocytic markers such as HMB45, MART1, tyrosinase, and MiTF. Tumors usually harbor a t(12;22)(q13;q12), similar to malignant GNET.
Metastatic melanoma is common in the small intestine. Given the morphologic and immunohistochemical overlap, malignant GNET may easily be mistaken for metastatic melanoma and vice versa. As noted, malignant GNET lacks expression of secondary melanocytic markers, but this can also be the case in some metastatic melanomas. Clinical correlation to determine if there is a history of melanoma at other sites is essential, and demonstration of EWSR1 rearrangement can confirm the diagnosis of malignant GNET.
Monophasic synovial sarcoma rarely involves the GI tract, but when it does, it has a predilection for the stomach. The spindled tumor cells of synovial sarcoma typically have overlapping nuclei and less cytoplasm than those of malignant GNET and show a more uniform fascicular growth pattern without the heterogeneity of the former. By IHC, the tumor cells are negative for S-100 and instead show expression of keratin, epithelial membrane antigen (EMA), and TLE1. Identification of SS18 (SYT) rearrangement can help confirm the diagnosis in difficult cases.
Gastrointestinal stromal tumors showing mixed epithelioid and spindled cell morphology may mimic malignant GNET. However, pseudoglandular areas and osteoclast-like giant cells are not typical of GIST. IHC readily distinguishes the two tumor types; expression of KIT, DOG1, and CD34 is seen in virtually all GIST but not in malignant GNET. Focal S-100 expression is seen in a small subset of duodenal GIST.
Malignant GNET pursues an aggressive clinical course in most cases and has a poor prognosis. Metastasis to lymph nodes and liver is common. A study with clinical follow-up for 12 patients showed 50% died of disease with a mean survival period of 32 months.
Malignant neuroectodermal tumor
Extremely rare; usually arises in the small intestine
Malignant tumors with frequent metastases and death from disease
No gender predilection
Young to middle-aged adults
Pain, gastric outlet obstruction, bleeding, anemia, fever
Aggressive tumors with frequent metastasis
Intramural, 2- to 15-cm, solid masses, often with hemorrhage or necrosis
Sheets of epithelioid, round or spindled cells
Osteoclast-like giant cells in 50%
Mixed growth patterns: sheets-like, pseudoglandular, pseudopapillary, or nested
Positive for S-100
Negative for secondary melanocytic markers, KIT, DOG1 (discovered on GIST1), and CD34
EWSR1-ATF1 and EWSR1-CREB1 fusion genes in 90%
Gastrointestinal stromal tumor: especially myxoid or succinate dehydrogenase–deficient types: positive for KIT and DOG1
Metastatic melanoma: may be positive for secondary melanocytic markers; lacks EWSR1 rearrangement
Conventional clear cell sarcoma: rarely involves the gastrointestinal tract; more prominent nested growth pattern; also expresses HMB45 (human melanoma black), MART1, tyrosinase
Synovial sarcoma: more uniform fascicular growth pattern; lacks osteoclast-like giant cells; positive for EMA and TLE1; SS18 gene rearrangement
Inflammatory fibroid polyp is a benign tumor that most commonly arises in the gastric antrum or ileum and rarely in other intestinal locations. Patients usually present in their sixth decade, and there is a slight female predominance. Large gastric tumors can cause abdominal pain, gastric outlet obstruction, and GI bleeding, and large intestinal tumors commonly cause intussusception, often requiring emergent surgery. Smaller tumors can be found incidentally during endoscopy.
Endoscopically, there may be a small polypoid mass or a larger mass bulging into the lumen, with or without mucosal ulceration.
Gastric tumors are typically small (<3 cm) polypoid masses; small intestinal tumors tend to be larger (3 to >10 cm). They involve the lamina propria or submucosa and can extend into muscularis propria or serosa. The cut surface appears glistening and fleshy, consistent with the edematous stroma microscopically.
Tumors exhibit a diffuse growth pattern of stellate, ovoid or short spindled fibroblastic cells, most commonly in an edematous or myxoid stroma ( Fig. 7.22 ). Sometimes the stroma can appear more collagenous, and some tumors exhibit perivascular onion-skinning fibrosis. Inflammatory fibroid polyp is usually hypocellular, and neoplastic cells have at most minimal atypia. The lesion can resemble a non-neoplastic inflammatory lesion. Mitotic activity is low or absent. There are frequently admixed eosinophils, a useful diagnostic clue, as well as lymphocytes and histiocytes ( Fig. 7.23A ).


Neoplastic cells are positive for CD34 and, in 20% of cases, SMA. They are virtually always negative for KIT, DOG1, desmin, and S-100, which can be useful to exclude histologic mimics. IHC for PDGFRA, when available, is helpful as it is diffusely positive in essentially all inflammatory fibroid polyps; because background stromal cells express PDGFRA at lower levels, it is important to only interpret strong diffuse positivity as a positive result ( Fig. 7.23B ).
Inflammatory fibroid polyp harbors mutations in PDGFRA , including some of the same mutations present in PDGFRA -mutant GIST. The exonic mutations correlate with tumor location, with mutations in exon 12 more commonly occurring in small intestinal tumors and exon 18 more commonly occurring in gastric tumors.
Myxoid GIST can appear similar to inflammatory fibroid polyp but is typically more densely cellular and lacks an eosinophil-rich inflammatory infiltrate. Many myxoid GISTs are PDGFRA mutant, such that PDGFRA IHC is not useful to resolve this differential diagnosis. However, in contrast to inflammatory fibroid polyp, the tumor cells of GIST are positive for KIT and DOG1.
Plexiform fibromyxoma arises in the gastric antrum, but in contrast to inflammatory fibroid polyp, it exhibits a multinodular or plexiform growth pattern and is predominantly located in the muscularis propria. Although plexiform fibromyxoma is also composed of bland spindle cells, it does not have the eosinophil-rich inflammatory infiltrate characteristic of inflammatory fibroid polyp. IHC for PDGFRA can be used to distinguish these entities.
Inflammatory myofibroblastic tumor (IMT) also contains an inflammatory infiltrate, but its neoplastic cells exhibit a fascicular growth pattern. IHC for PDGFRA and anaplastic lymphoma kinase (ALK) can discriminate between these tumor types.
Complete excision is curative. Very rarely, inflammatory fibroid polyp occurs in the inherited Devon polyposis syndrome.
Benign fibroblastic proliferation that commonly manifests as a gastric antral polyp or an intestinal mass causing intussusception
Rare tumor that typically arises in the lamina propria/submucosa of the gastric antrum, ileum, or, less commonly, other intestinal sites
Inflammatory fibroid polyps are benign and only rarely occur in a familial syndromic setting (Devon polyposis syndrome)
Slight female predominance
Wide age range, with peak in the sixth decade of life
Gastric tumors sometimes present with pain, gastric outlet obstruction, or bleeding
Small intestinal tumors can present with intussusception
Small tumors can be incidentally found during endoscopy
Benign and cured by simple excision
Wide size range (1–>10 cm); gastric tumors tend to be smaller than intestinal tumors
Uniform fleshy or glistening cut surface
Submucosal location, sometimes with infiltration into the muscularis propria
Hypocellular neoplasm with edematous or myxoid stroma rich in eosinophils
Bland stellate, ovoid, or short spindled neoplastic cells
Perivascular onion-skinning fibrosis is common
Positive for CD34, platelet-derived growth factor receptor-α (PDGFRA), and smooth muscle actin (20%)
Negative for S-100, KIT, DOG1 (discovered on GIST1), and desmin
Mutations in PDGFRA gene, with a different but somewhat overlapping exonic distribution compared with PDGFRA -mutant gastrointestinal stromal tumor (GIST)
GIST: especially myxoid or PDGFRA -mutant GIST, which is usually more cellular; positive for KIT and DOG1; both inflammatory fibroid polyp and PDGFRA -mutant GIST are positive for PDGFRA
Plexiform fibromyxoma: usually intramural rather than submucosal, has plexiform growth, lacks prominent inflammatory infiltrate
Inflammatory myofibroblastic tumor: more cellular, with fascicles of neoplastic myofibroblasts; negative for PDGFRA, 50% positive for anaplastic lymphoma kinase
Plexiform fibromyxoma is a benign tumor that virtually always arises in stomach, most often at the gastric antrum. Very rare cases arise in the small intestine. Patients are usually in their fifth decade, and there is no gender predilection. Presenting symptoms include abdominal pain, gastric outlet obstruction, and GI bleeding.
Endoscopically, there may be a bulging mass into the gastric lumen, with or without mucosal ulceration.
Tumors usually measure between 2 and 15 cm. They are typically intramural but may extend into mucosa or serosa. Tumors are lobulated and the cut surface variably red, tan, or white, with a gelatinous and glistening appearance because of the myxoid stroma.
Tumors arise within the muscularis propria and show a multinodular or plexiform growth pattern on low power examination, resulting in separation of smooth muscle bundles ( Fig. 7.24 ). The nodules are relatively hypocellular and contain bland oval-spindled tumor cells with at most minimal atypia, within a variably myxoid and collagenous stroma ( Fig. 7.25 ). There is typically a prominent capillary proliferation. The tumor cells have indistinct cytoplasm and small nuclei. Mitotic activity is low or absent. Extension into mucosa may result in mucosal ulceration. Subserosal or serosal tumor nodules may be present.


The immunoprofile is nonspecific: the spindled cells express SMA, either diffusely or focally, and are virtually always negative for KIT, DOG1, CD34, and S-100 protein. Focal expression of desmin or caldesmon has been reported in a small subset of tumors. IHC may be helpful to exclude histologic mimics.
A subset of tumors harbor MALAT1-GLI1 fusion, GLI1 polysomy, or PTCH1 loss, all of which are expected to activate the hedgehog pathway.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here