Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The contributions to this chapter by Dr. Jerrold R. Turner, Department of Pathology, Brigham and Women's Hospital, Boston, Massachusetts, in several previous editions of this book are gratefully acknowledged.
The gastrointestinal tract is a hollow tube consisting of the esophagus, stomach, small intestine, colon, rectum, and anus. Each region has complementary, highly integrated functions that regulate the intake, processing, and absorption of ingested nutrients and the disposal of waste products. The intestines are also where the immune system encounters a diverse array of antigens derived from food and endogenous gut microbes that must be tolerated as well as pathogens that may contaminate food and drink that must be eliminated. It should then come as no surprise that the gastrointestinal tract is commonly involved in a wide spectrum of infectious and inflammatory processes. In this chapter, we discuss diseases that affect each part of the gastrointestinal tract. Disorders that typically involve more than one region, such as Crohn disease, are considered according to the region they involve most frequently. We begin our discussion with diseases of the oral cavity, since that is where the journey of food begins.
Pathologic conditions of the oral cavity can be broadly divided into diseases affecting teeth and their support structures, oral mucosa, salivary glands, and the jaw. Discussed next are the more common conditions affecting these sites. Odontogenic cysts and tumors (benign and malignant), which are derived from the epithelial and/or mesenchymal tissues associated with tooth development, are also described briefly.
Bacteria in the oral cavity are directly or indirectly responsible for the most common disorders of the teeth and gums, caries, gingivitis, and periodontitis.
Dental caries result from focal demineralization of tooth structure (enamel and dentin) caused by acids generated during the fermentation of sugars by bacteria. Worldwide, caries is the main cause of tooth loss before 35 years of age. In the past, caries was pervasive in high-resource countries where there is ready access to foods containing large amounts of refined carbohydrates. However, the rate of caries has dropped markedly in countries such as the United States due to improved oral hygiene and fluoridation of drinking water. Fluoride is incorporated into the crystalline structure of enamel, forming fluoroapatite, which is resistant to degradation by bacterial acids. With the globalization of the world’s economy, processed foods are being increasingly consumed in lower-resource countries, and as a result the rate of caries is increasing in these regions of the world.
Inflammation involving the squamous mucosa, or gingiva, and associated soft tissues that surround teeth is called gingivitis. The most frequent cause is poor oral hygiene, which facilitates buildup of dental plaque and calculus between and on the surfaces of teeth. Dental plaque is a sticky biofilm composed of bacteria, salivary proteins, and desquamated epithelial cells. As it accumulates, plaque becomes mineralized to form calculus, or tartar. Subgingival plaque and associated bacteria lead to chronic gingivitis, marked by erythema, edema, and bleeding. Gingivitis may occur at any age but is most prevalent and severe in adolescence, where it is present in 40% to 60% of individuals, after which the incidence tapers off. Fortunately, gingivitis can be reversed, primarily by regular brushing and flossing of teeth and periodic cleaning, which reduces accumulation of plaque and calculus.
Periodontitis is an inflammatory process that affects the supporting structures of the teeth (periodontal ligaments), alveolar bone, and cementum. With progression, periodontitis may result in destruction of periodontal ligaments and alveolar bone and, eventually, tooth loss. Periodontitis is associated with poor oral hygiene that affects the composition of gingival bacteria. Facultative gram-positive organisms are associated with healthy teeth, whereas anaerobic and microaerophilic gram-negative bacteria colonize plaque within areas of active periodontitis. Bacterial species that are closely associated with periodontitis include Aggregatibacter (Actinobacillus) actinomycetemcomitans, Porphyromonas gingivalis, and Prevotella intermedia.
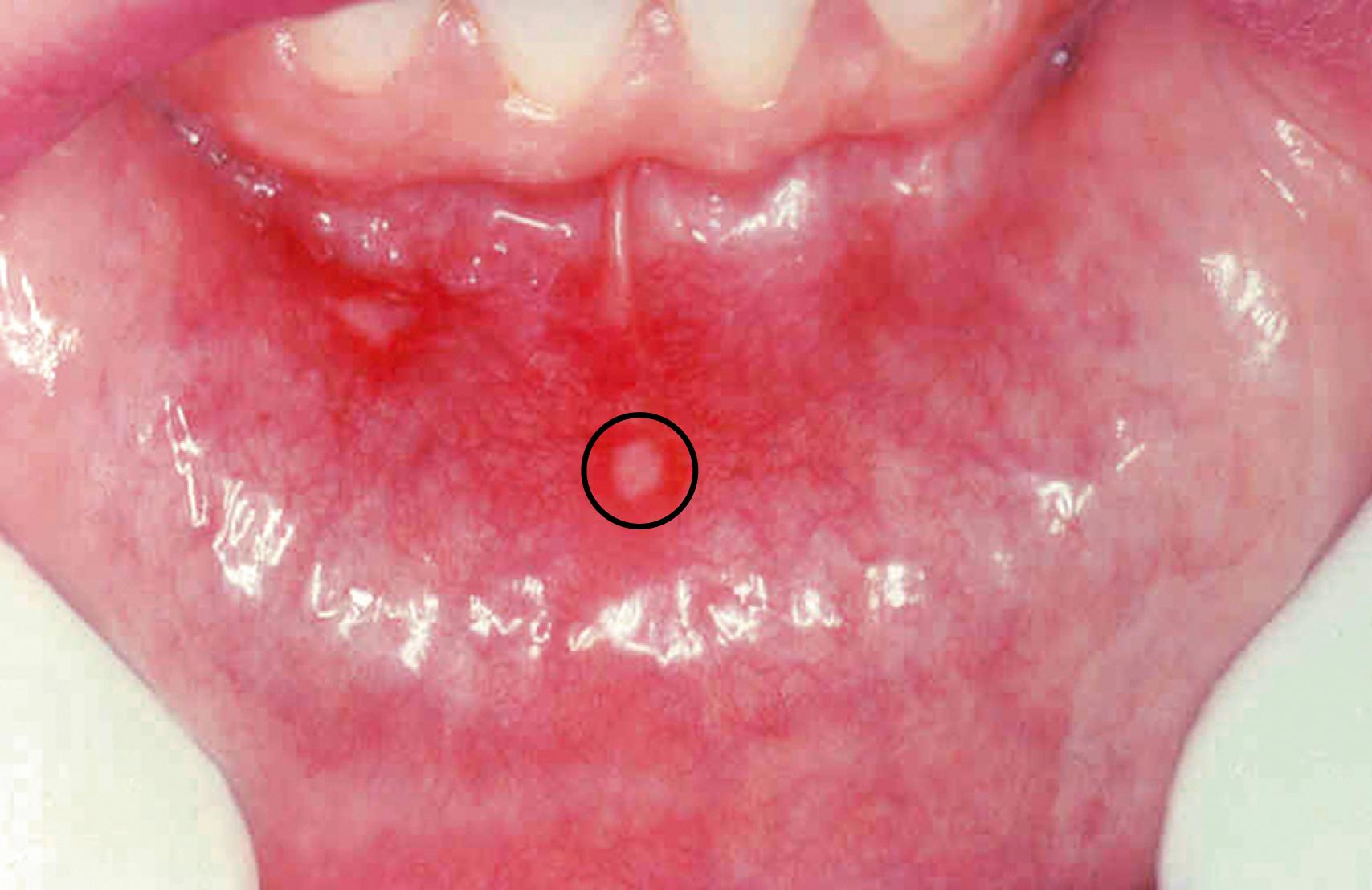
These common superficial mucosal ulcerations affect up to 40% of the population. They are more frequent in the first two decades of life, are extremely painful, and often recur. The cause is unknown; they tend to be familial and may be associated with celiac disease, inflammatory bowel disease, and Behçet disease, a rare vasculitic disorder. These ulcers can be solitary or multiple; typically, they are shallow and have a hyperemic base covered by a thin exudate that is rimmed by a narrow zone of erythema ( Fig. 13.1 ). In most cases they resolve spontaneously in 7 to 10 days.
Herpes simplex virus causes a self-limited primary infection that may reactivate if host resistance is compromised. Most orofacial herpetic infections are caused by herpes simplex virus type 1 (HSV-1), with the remainder being caused by HSV-2, a type that is more highly associated with genital herpes. Primary infections typically occur in children between 2 and 4 years of age and are often asymptomatic. However, in 10% to 20% of cases primary infection manifests as acute herpetic gingivostomatitis, with abrupt onset of vesicles and ulcerations throughout the oral cavity.
Most adults harbor latent HSV-1, which may reactivate, resulting in a so-called “cold sore” or recurrent herpetic stomatitis. Factors associated with HSV reactivation include trauma, allergies, exposure to ultraviolet light and extremes of temperature, upper respiratory tract infections, pregnancy, menstruation, and immunosuppression. Recurrent lesions usually occur at the site of primary inoculation or in adjacent mucosa innervated by the same axon and typically appear as groups of small (1- to 3-mm) vesicles. The lips (herpes labialis), nasal orifices, buccal mucosa, gingiva, and hard palate are the most common locations. Less often, the reactivated infection involves the cornea, producing pain, blurred vision, and photophobia. Lesions typically resolve spontaneously within 7 to 10 days, but patients who are immunocompromised and those with corneal involvement often require antiviral therapy. Morphologically, the lesions resemble those seen in esophageal herpes (see Fig. 13.8 ) and genital herpes ( Chapter 16 ). The infected cells become ballooned and have large eosinophilic intranuclear inclusions. Adjacent cells commonly fuse to form large multinucleated polykaryons.
Candidiasis is the most common fungal infection of the oral cavity. Candida albicans is a normal component of the oral flora and only produces disease under unusual circumstances. Predisposing factors include:
Immunosuppression
The presence of certain strains of C. albicans
A permissive oral microbial flora (microbiota)
Broad-spectrum antibiotics that alter the normal microbiota may promote oral candidiasis, which most commonly appears as a pseudomembranous form known as thrush. Thrush is characterized by a superficial, curdlike, gray to white inflammatory membrane composed of matted organisms enmeshed in a fibrinosuppurative exudate that can be readily scraped off to reveal an underlying erythematous base. In individuals who are mildly immunosuppressed or debilitated (e.g., patients with diabetes), the infection usually remains superficial, but spread to deep sites may be seen with more severe immunosuppression, such as in organ or hematopoietic stem cell transplant recipients and patients with neutropenia, chemotherapy-induced immunosuppression, or AIDS.
Fibromas are submucosal nodular fibrous tissue masses that form when chronic irritation (trauma from teeth or dentures) results in reactive connective tissue hyperplasia ( Fig. 13.2 A). They occur most often on the buccal mucosa along the bite line. Treatment consists of surgical excision and removal of the source of irritation.
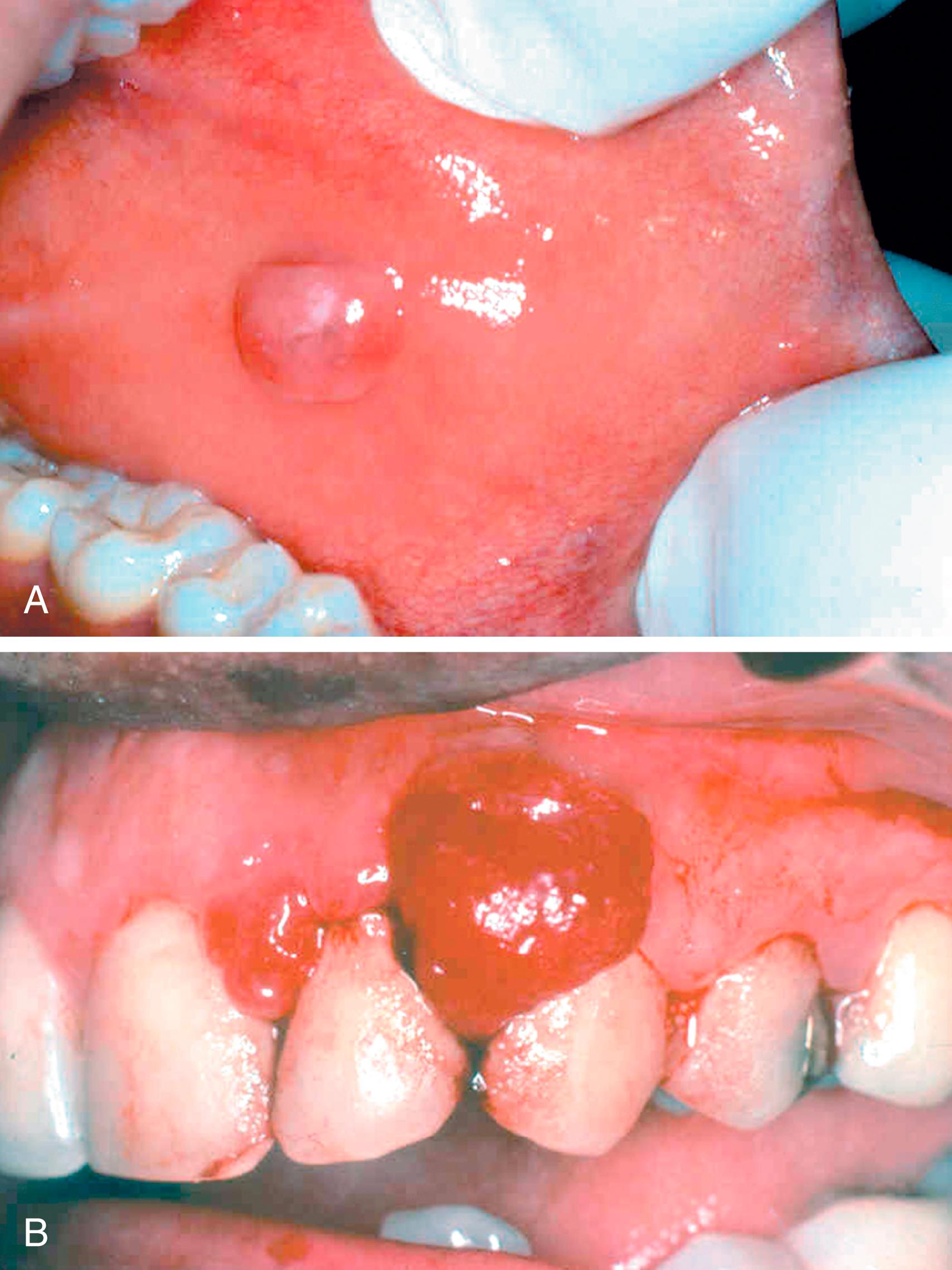
Pyogenic granuloma ( Fig. 13.2 B) is an inflammatory lesion typically found on the gingiva of children, young adults, and pregnant women ( Chapter 8 ). The lesions are highly vascular, giving them a red to purple color, and often ulcerated. In some cases, their growth may be so rapid as to suggest a malignant neoplasm. However, histologic examination demonstrates a proliferation of capillaries similar to those seen in granulation tissue. Pyogenic granulomas may regress or undergo fibrosis, sometimes associated with calcification. Complete surgical excision is the definitive treatment.
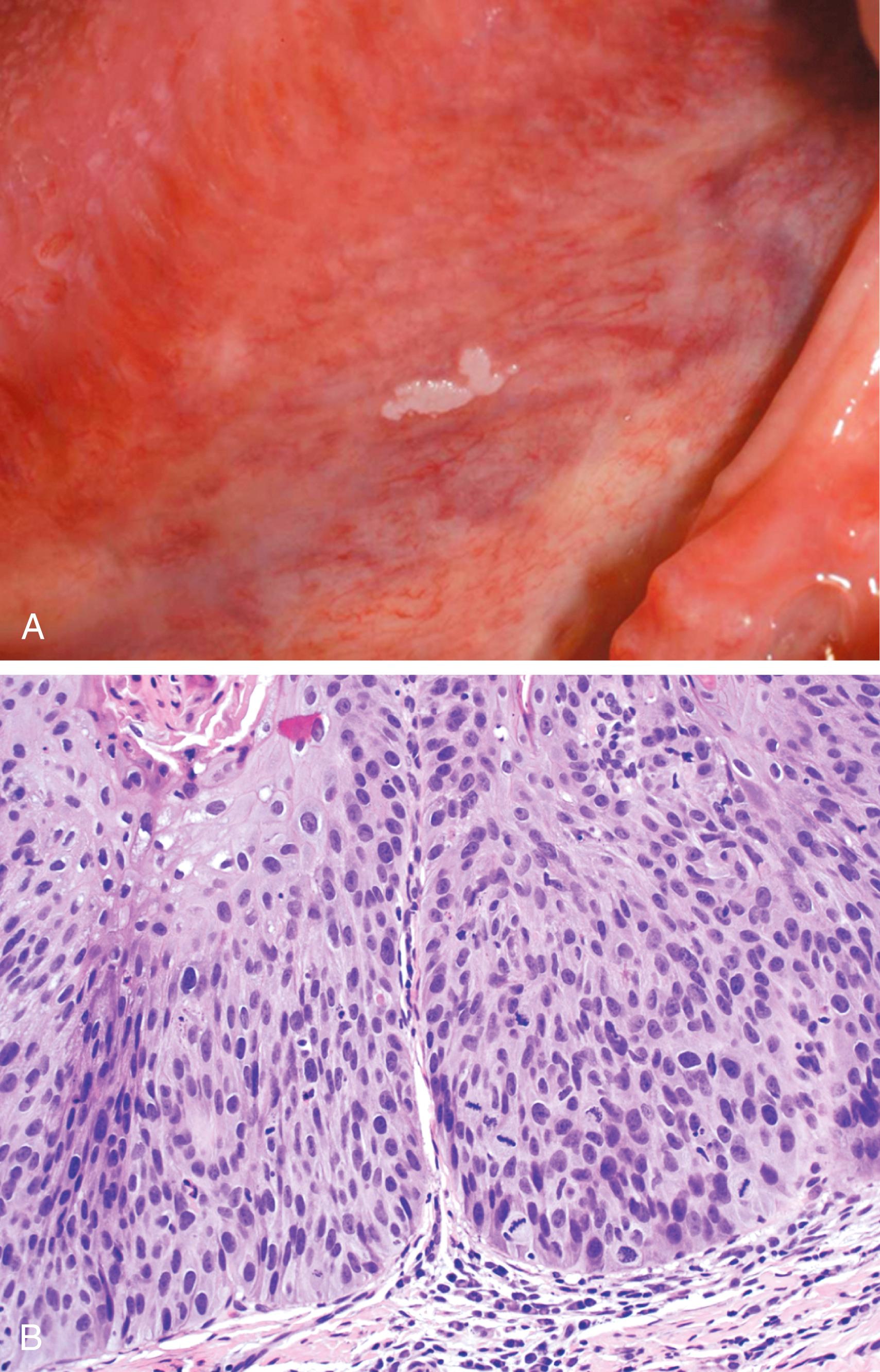
Leukoplakia and erythroplakia are squamous lesions of the oropharynx that may be precursors of squamous cell carcinoma. The terms are reserved for lesions that arise in the absence of any known cause; accordingly, lesions caused by chronic irritation or entities such as lichen planus and candidiasis are not leukoplakia. Approximately 3% of the world’s population has leukoplakic lesions, of which 5% to 25% are dysplastic and at risk for progression to squamous cell carcinoma. Thus, until proven otherwise by histologic evaluation, leukoplakia is considered precancerous. A related, less common entity, erythroplakia, is associated with a much greater risk for malignant transformation than leukoplakia. Leukoplakia and erythroplakia are most common in adults between 40 and 70 years of age and have a 2 : 1 male predominance. Although the etiology is multifactorial, the most important risk factor is tobacco use, including smokeless tobacco.
Leukoplakia typically appears as a well-demarcated gray-white plaque ( Fig. 13.3 A). On histologic examination leukoplakia and erythroplakia show a spectrum of epithelial changes ranging from a hyperplastic, hyperkeratotic but orderly squamous epithelium to lesions with marked dysplasia, sometimes associated with carcinoma in situ ( Fig. 13.3 B). Dysplastic lesions are often associated with an underlying inflammatory cell infiltrate consisting of lymphocytes and macrophages. The most severe dysplastic changes are associated with erythroplakia, which undergoes malignant transformation in more than 50% of cases. Grossly, erythroplakia appears as a red, velvety, sometimes eroded, flat or slightly depressed lesion.
Approximately 95% of cancers of the oral cavity are squamous cell carcinomas, with the remainder largely consisting of adenocarcinomas of salivary glands. Oral squamous cell carcinoma is an aggressive epithelial malignancy. The overall incidence of oral squamous cell carcinoma has risen since the 1980s, despite a decline in tobacco use, due to an increase in human papillomavirus–associated cancers, which are now believed to outnumber those related to alcohol and tobacco in the United States and Europe.
Squamous cancers of the oropharynx arise through two distinct pathogenic pathways, one involving exposure to carcinogens and the other to infection with high-risk variants of human papilloma virus (HPV). Carcinogen exposure in the United States and Europe mainly stems from chronic use of alcohol and/or tobacco (both smoked and chewed), while in India and Southeast Asia, chewing of betel nuts and leaves (paan) are predisposing factors. DNA sequencing of tobacco-associated cancers has revealed a mutational signature consistent with exposure to carcinogens. The most commonly mutated genes include TP53, genes that regulate proliferation (e.g., RAS ), and genes that regulate squamous differentiation (e.g., NOTCH ). The elevated risk for development of additional primary tumors in these patients has led to the concept of “field cancerization”; this hypothesis suggests that multiple neoplastic clones develop independently as a result of years of chronic mucosal exposure to carcinogens.
By contrast, HPV-related tumors tend to occur in the tonsillar crypts and harbor oncogenic “high-risk” HPV subtypes, particularly HPV-16. Exposure to HPV occurs through orogenital sex. As was discussed in Chapter 6 , high-risk subtypes of HPV express the viral oncoproteins E6 and E7, which inhibit the critical tumor suppressors p53 and RB, respectively. HPV-associated tumors carry far fewer mutations than those associated with tobacco exposure and often overexpress p16, a cyclin-dependent kinase inhibitor.
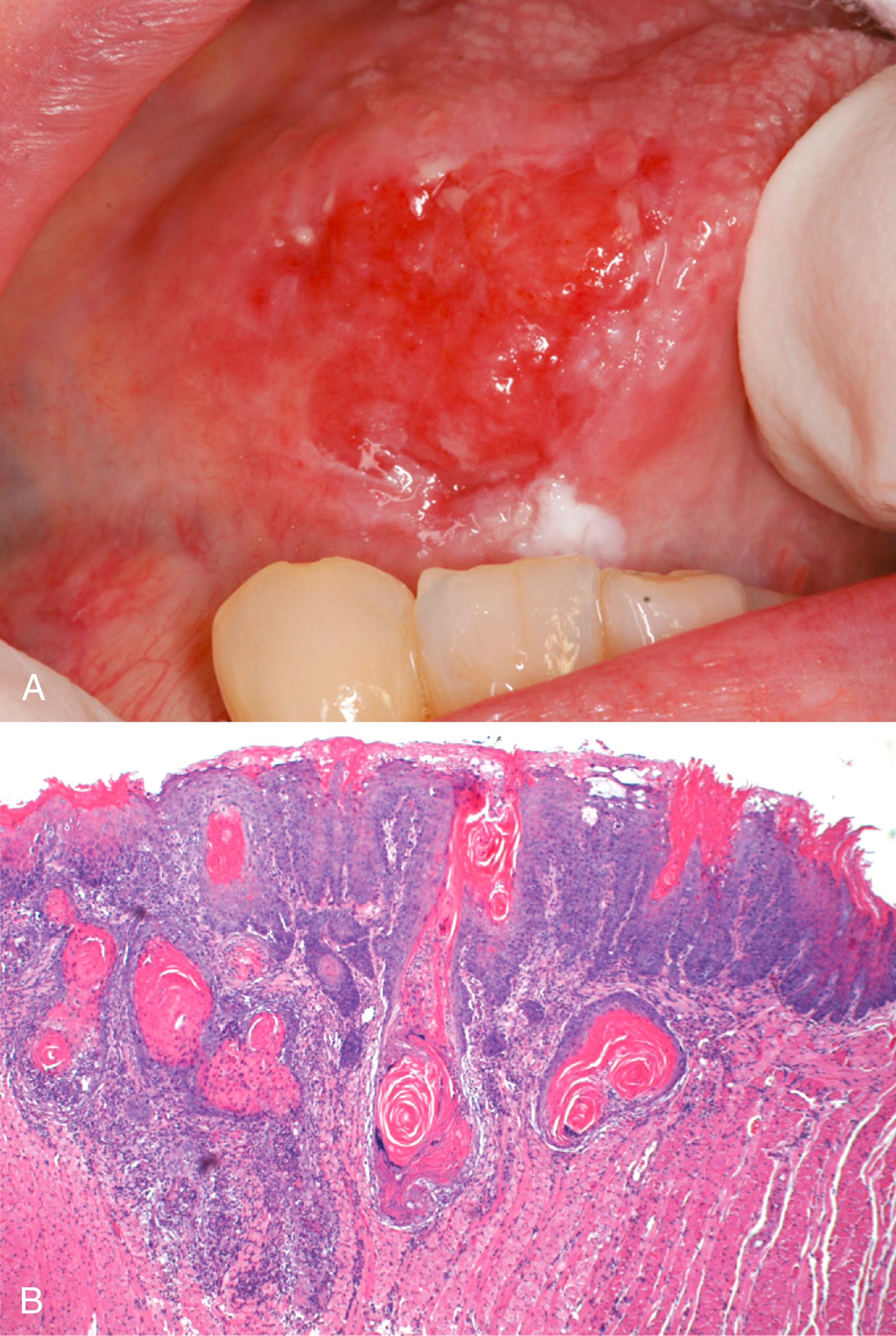
Squamous cell carcinoma may arise anywhere in the oral cavity. The most common locations for carcinogen-associated cancers are the ventral surface of the tongue, floor of the mouth, lower lip, soft palate, and gingiva ( Fig. 13.4 A). In early stages, these cancers may appear as raised, firm pearly plaques or irregular, roughened, or verrucous mucosal thickenings. The surrounding mucosa may show leukoplakia or erythroplakia. By contrast, HPV-associated cancers arise in the tonsil or the back of the tongue. As these lesions enlarge, they typically form ulcerated and protruding masses that have irregular and indurated or rolled borders. Histologic patterns range from well-differentiated keratinizing neoplasms ( Fig. 13.4 B) to anaplastic, sometimes sarcomatoid, tumors. However, the degree of histologic differentiation, as determined by the relative degree of keratinization, does not correlate with biologic behavior. Typically, oral squamous cell carcinoma infiltrates locally before it metastasizes. The cervical lymph nodes are the most common sites of regional metastasis; frequent sites of distant metastases include the mediastinal lymph nodes, lungs, and liver.
Despite advances in treatment of oral squamous cell carcinoma, the overall survival at 5 years is only 50%, in large part because it is often diagnosed at an advanced stage. Multiple primary tumors may be present at initial diagnosis but more often are detected later, in an estimated 3% to 7% of patients per year. Therefore, surveillance and early detection of new premalignant lesions are critical for the long-term survival of patients diagnosed with oral squamous cell carcinoma.
Advanced stages of disease are treated with a combination of surgery, radiation, and chemotherapy. Tumors that arise in the setting of carcinogen exposure are responsive to immune checkpoint inhibitors, presumably because they harbor a high burden of tumor neoantigens. The prognosis for patients with HPV-positive tumors is better than for those with HPV-negative tumors, possibly because HPV-positive tumors are less genetically heterogeneous. The HPV vaccine is protective against cervical cancer and if widely adopted should also sharply reduce the frequency of HPV-associated oral squamous cell carcinoma.
There are three major salivary glands—parotid, submandibular, and sublingual—and numerous minor salivary glands distributed throughout the oral mucosa. Inflammatory or neoplastic disease may develop within any of these.
Xerostomia is defined as a dry mouth resulting from a decrease in the production of saliva. Its incidence varies among populations but has been reported in more than 20% of individuals older than 70 years of age. It is a major feature of the autoimmune disorder Sjögren syndrome, in which it is usually accompanied by dry eyes ( Chapter 5 ). A lack of salivary secretions is also a major complication of radiation therapy. However, xerostomia is most frequently observed as a side effect of many commonly used medications, including anticholinergic, antidepressant/antipsychotic, diuretic, antihypertensive, sedative, muscle relaxant, analgesic, and antihistaminic agents. Nontherapeutic drugs such as methamphetamine, cocaine, and cannabis can also cause xerostomia. On examination, the oral cavity may reveal only dry mucosa and/or atrophy of the papillae of the tongue, with fissuring and ulcerations, or, in Sjögren syndrome, concomitant inflammatory enlargement of the salivary glands. Complications of xerostomia include increased rates of dental caries and candidiasis, as well as difficulty in swallowing and speaking.
Inflammation of the salivary gland, referred to as sialadenitis, may be induced by trauma, viral or bacterial infection, or autoimmune disease. The most common viral sialadenitis is mumps, a paramyxovirus infection that predominantly involves the parotid glands. Mumps produces interstitial inflammation marked by a mononuclear inflammatory infiltrate. While mumps is usually a self-limited benign condition in children, in adults the virus may cause pancreatitis or orchitis; the latter sometimes results in sterility.
Mucocele is the most common inflammatory lesion of salivary glands. It results from blockage or rupture of a salivary gland duct, with consequent leakage of saliva into the surrounding connective tissue stroma. Mucocele occurs most often in toddlers and young and old adults and typically manifests as a fluctuant swelling of the lower lip that may change in size, particularly in association with meals ( Fig. 13.5 A ). Histologic examination demonstrates a cyst-like space lined by granulation tissue or fibrous connective tissue and filled with mucin and inflammatory cells, particularly macrophages ( Fig. 13.5 B). Complete excision of the lesion and the minor salivary gland is the definitive treatment.
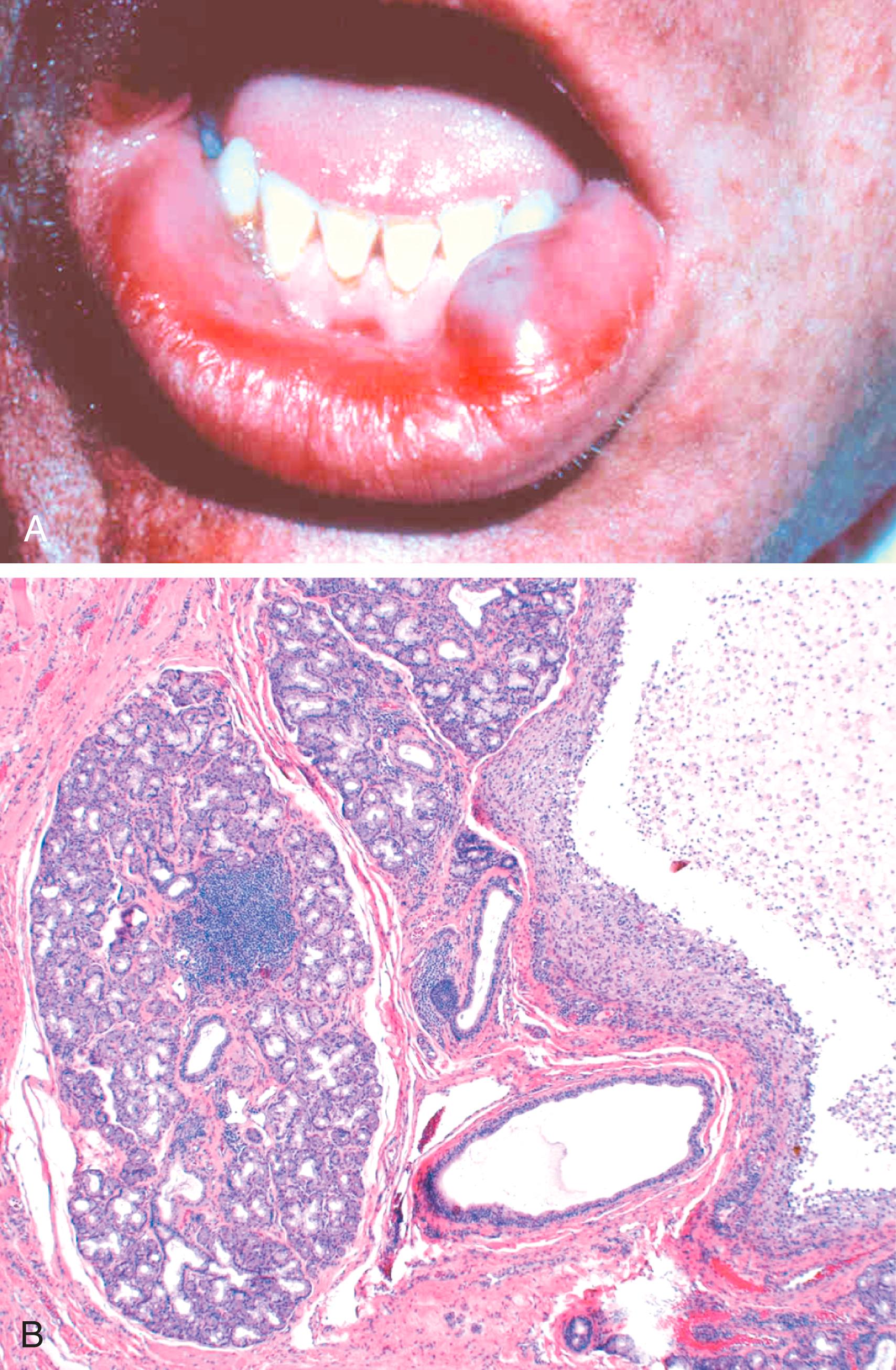
Bacterial sialadenitis is a common infection that most often involves the major salivary glands, particularly the submandibular glands. Duct obstruction by stones (sialolithiasis) is a common antecedent to infection; it may also be induced by impacted food debris or by edema secondary to injury. Dehydration and decreased secretory function may also predispose to formation of stones and consequent bacterial invasion. The most frequent pathogens are Staphylococcus aureus and Streptococcus viridans. Bacterial sialoadenitis may produce nonspecific interstitial inflammation of the affected glands or, when caused by staphylococci or other pyogens, may lead to suppuration and abscess formation.
Autoimmune sialadenitis, better known as Sjögren syndrome, is discussed in Chapter 5 .
Despite their relatively simple morphology, the salivary glands give rise to at least 30 histologically distinct tumors ( Table 13.1 ), a small number of which account for more than 90% of cases. Salivary gland tumors are relatively uncommon, representing less than 2% of human tumors. Approximately 65% to 80% arise within the parotid, 10% in the submandibular gland, and the remainder in the minor salivary glands, including the sublingual glands. Approximately 15% to 30% of tumors in the parotid glands are malignant. By contrast, approximately 40% of submandibular, 50% of minor salivary gland, and 70% to 90% of sublingual tumors are cancerous. Thus, the likelihood that a salivary gland tumor is malignant seems to be inversely proportional to the size of the gland.
| Benign | Malignant |
|---|---|
| Pleomorphic adenoma (50%) | Mucoepidermoid carcinoma (15%) |
| Warthin tumor (5%) | Acinic cell carcinoma (6%) |
| Oncocytoma (2%) | Adenocarcinoma NOS (6%) |
| Cystadenoma (2%) | Adenoid cystic carcinoma (4%) |
| Basal cell adenoma (2%) | Malignant mixed tumor (3%) |
Salivary gland tumors usually occur in adults. Parotid gland neoplasms cause swelling in front of and below the ear. Benign tumors may be present for months to several years before coming to clinical attention, while cancers more often come to attention promptly because of their more rapid growth. However, the only reliable way to differentiate benign from malignant lesions is through histopathologic evaluation.
Pleomorphic adenoma is a benign tumor that consists of a mixture of ductal (epithelial) and myoepithelial cells and often exhibits both epithelial and mesenchymal differentiation. Pleomorphic adenomas represent about 60% of parotid gland tumors, are less common in the submandibular glands, and are relatively rare in the minor salivary glands.
Pleomorphic adenomas typically manifest as rounded, well-demarcated masses of several centimeters in greatest dimension. Although they are encapsulated, in some locations (particularly the palate), the capsule is not fully developed, and expansile growth produces protrusions into the surrounding tissues. The cut surface is gray-white and typically contains myxoid and blue translucent chondroid areas. Their most striking histologic feature is their characteristic heterogeneity. Epithelial elements are dispersed throughout the tumor stroma, which may contain variable mixtures of myxoid, hyaline, chondroid (cartilaginous), and even osseous tissue. In some pleomorphic adenomas, the epithelial elements predominate; in others, they are present only in widely dispersed foci. This histologic diversity has given rise to the alternative, albeit less preferred, name mixed tumor. Epithelial elements resembling ductal or myoepithelial cells are arranged in ducts, acini, irregular tubules, strands, or sheets. These are typically dispersed within a mesenchyme-like background of loose myxoid tissue containing islands of cartilage and, rarely, foci of bone ( Fig. 13.6 ). Sometimes the epithelial cells form well-developed ducts lined by cuboidal to columnar cells with an underlying layer of deeply chromatic, small myoepithelial cells. In other instances, there may be strands or sheets of myoepithelial cells. Islands of well-differentiated squamous epithelium may also be present. In most cases, no epithelial dysplasia or mitotic activity is evident. No difference in biologic behavior has been observed between the tumors composed largely of epithelial elements and those composed largely of mesenchymal elements.
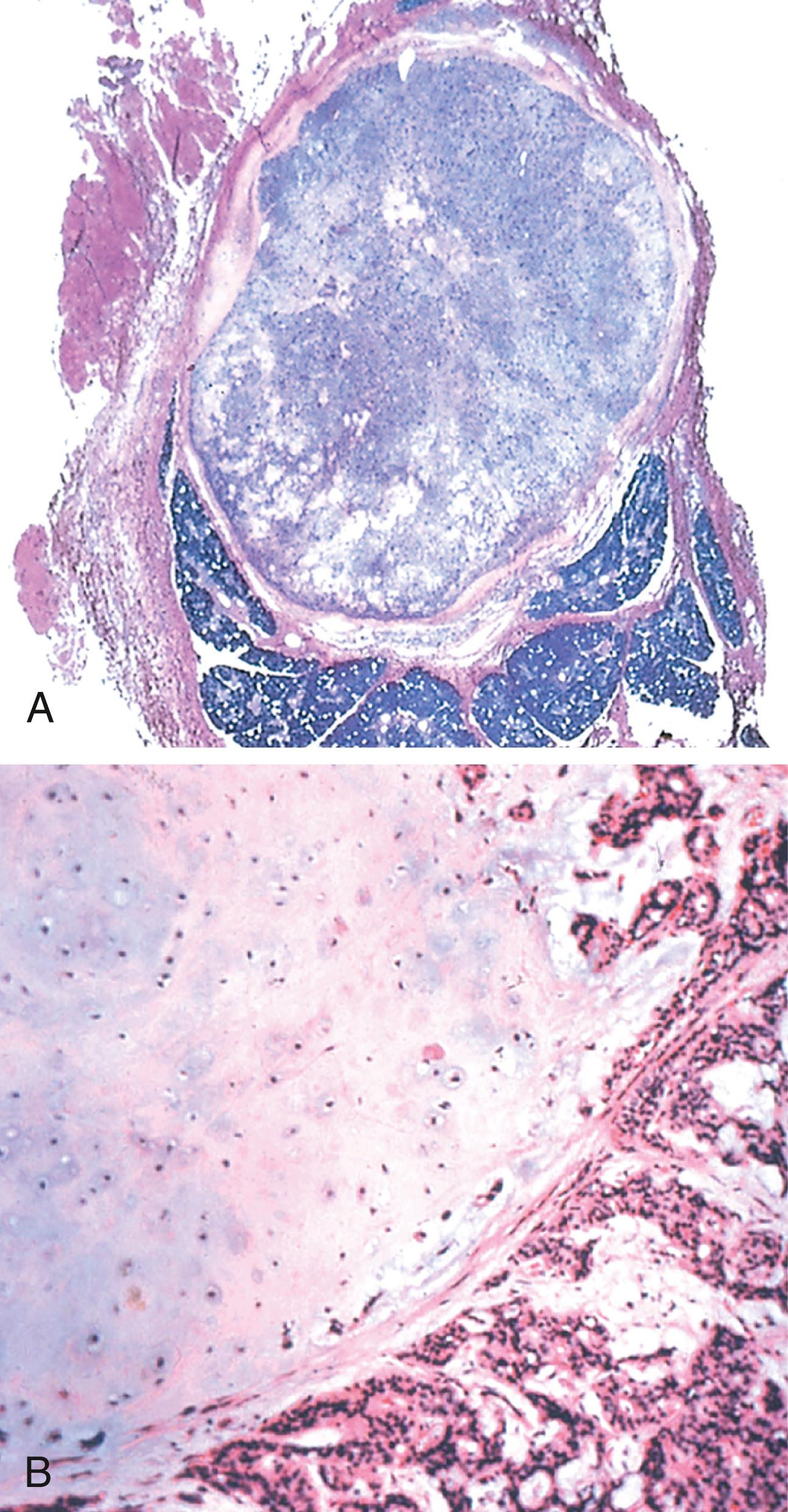
Pleomorphic adenoma presents as a slow-growing, painless, mobile, discrete mass. Recurrence occurs in 25% of cases after simple enucleation of the tumor and in 4% of cases after wider resection and stems from a failure to remove minute extensions of tumor into surrounding soft tissues.
Carcinoma arising in a pleomorphic adenoma is referred to variously as a carcinoma ex pleomorphic adenoma or malignant mixed tumor. The incidence of malignant transformation increases from 2% in tumors present for less than 5 years to almost 10% in those present for more than 15 years. These cancers usually take the form of an adenocarcinoma or undifferentiated carcinoma. Unfortunately, they are among the most aggressive malignant neoplasms of salivary glands, with mortality rates of 30% to 50% at 5 years.
Mucoepidermoid carcinoma is composed of variable mixtures of squamous cells, mucus-secreting cells, and intermediate cells. This neoplasm represents about 15% of all salivary gland tumors. While it mainly occurs in the parotids (60%–70%), it also accounts for a large fraction of salivary gland neoplasms in the other glands, particularly the minor salivary glands. Overall, mucoepidermoid carcinoma is the most common primary malignant tumor of the salivary glands.
Mucoepidermoid carcinomas may be up to 8 cm in diameter and, although apparently circumscribed, lack well-defined capsules and are often infiltrative. The cut surface is pale gray to white and frequently demonstrates small, mucinous cysts. On histologic examination, these tumors contain cords, sheets, or cysts lined by squamous, mucous, or intermediate cells ( eFig. 13.1 ). The latter is a hybrid cell type with squamous features and mucin-filled vacuoles, which are most easily detected with mucin stains. Cytologically, the tumor cells may be benign appearing or highly anaplastic and unmistakably malignant.

Clinical course and prognosis depend on histologic grade. Low-grade tumors may invade locally and recur in about 15% of cases but metastasize only rarely and afford a 5-year survival rate of over 90%. By contrast, high-grade neoplasms and, to a lesser extent, intermediate-grade tumors are invasive and difficult to excise. As a result, they recur in 25% to 30% of cases, and about 30% metastasize to distant sites. The 5-year survival rate is only 50%.
Odontogenic cysts are derived from remnants of odontogenic epithelium present within the jaws. In contrast to the rest of the skeleton, epithelium-lined cysts are quite common in the jaws and may be either inflammatory, developmental, or neoplastic in origin. Only the most common lesions are considered here.
Dentigerous cysts originate around the crown of an unerupted tooth and are thought to result from a developmental defect involving of the dental follicle (primordial tissue that makes the enamel surface of teeth). They are lined by a thin, stratified squamous epithelium and are typically associated with a dense chronic inflammatory infiltrate in the surrounding connective tissue. Complete removal is curative.
Keratocystic odontogenic tumor is a neoplasm that occurs most frequently in individuals between 10 and 40 years of age, predominantly in males and typically in the posterior mandible. Previously considered to be a developmental aberration, it is now known to have clonal chromosomal aberrations and often harbors loss-of-function mutations in tumor suppressor genes, including PTCH1 . As would be expected, individuals with nevoid basal cell carcinoma syndrome, which is caused by germline mutations in PTCH1 , are at increased risk for keratocystic odontogenic tumor. On histologic examination, the cyst lining consists of a thin layer of keratinized squamous epithelium with a prominent basal cell layer. It is locally aggressive and has a high recurrence rate. Effective treatment requires complete surgical removal.
The periapical cyst has an inflammatory etiology. This common lesion occurs at the tooth apex as a result of long-standing pulpitis, which may be caused by advanced caries or trauma. Necrosis of the pulpal tissue may traverse the length of the root and exit the apex of the tooth into the surrounding alveolar bone, leading to a periapical abscess. Over time, granulation tissue (with or without an epithelial lining) may develop. Periapical inflammatory lesions persist due to bacterial infection or necrotic tissue in the area. Successful treatment consists of complete removal of the offending material followed by restoration or extraction of the tooth.
Odontogenic tumors are a complex group of lesions with diverse histologic appearances and clinical behaviors. Most are true neoplasms, either benign or malignant. They are derived from odontogenic epithelium, ectomesenchyme, or both. The two most common and clinically significant tumors are ameloblastoma and odontoma.
Ameloblastoma arises from odontogenic epithelium. It is typically cystic and slow growing and, despite being locally invasive, has an indolent course. The cysts are lined by palisading columnar epithelium that sometimes undergoes squamous differentiation and that overlies a loose stroma with stellate cells ( eFig. 13.2 ). Odontoma , the most common type of odontogenic tumor, arises from epithelium but shows extensive deposition of enamel and dentin. It is cured by local excision.
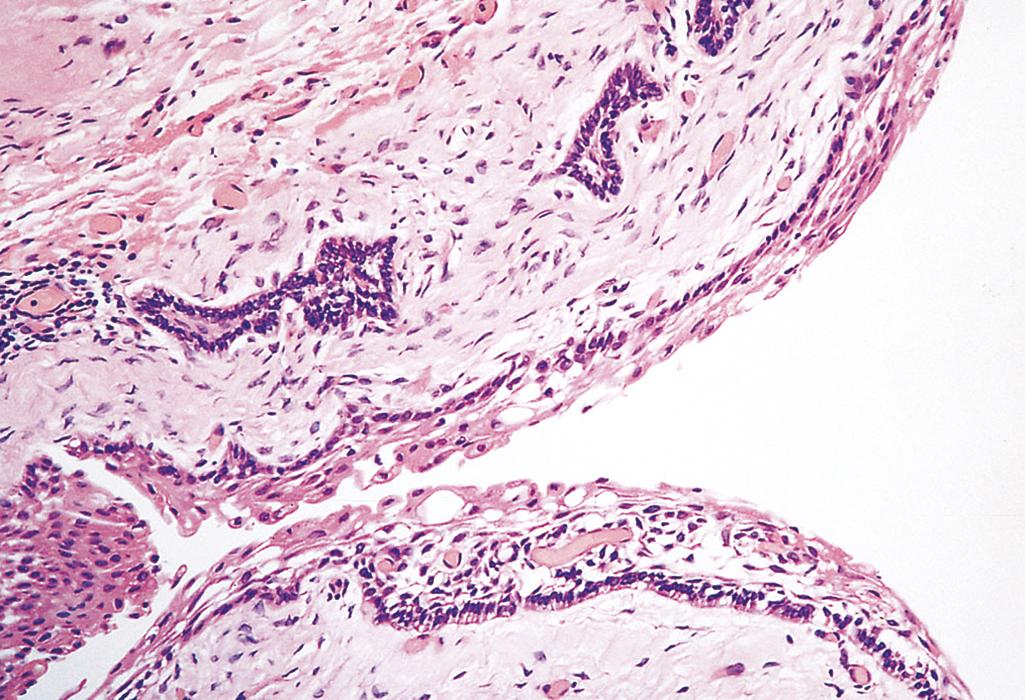
The esophagus develops from the cranial portion of the foregut. It is a hollow, highly distensible muscular tube that extends from the epiglottis to the gastroesophageal junction, located just above the diaphragm. Acquired diseases of the esophagus range from lethal cancers to the persistent “heartburn” of gastroesophageal reflux, which may be chronic and incapacitating or merely an occasional annoyance.
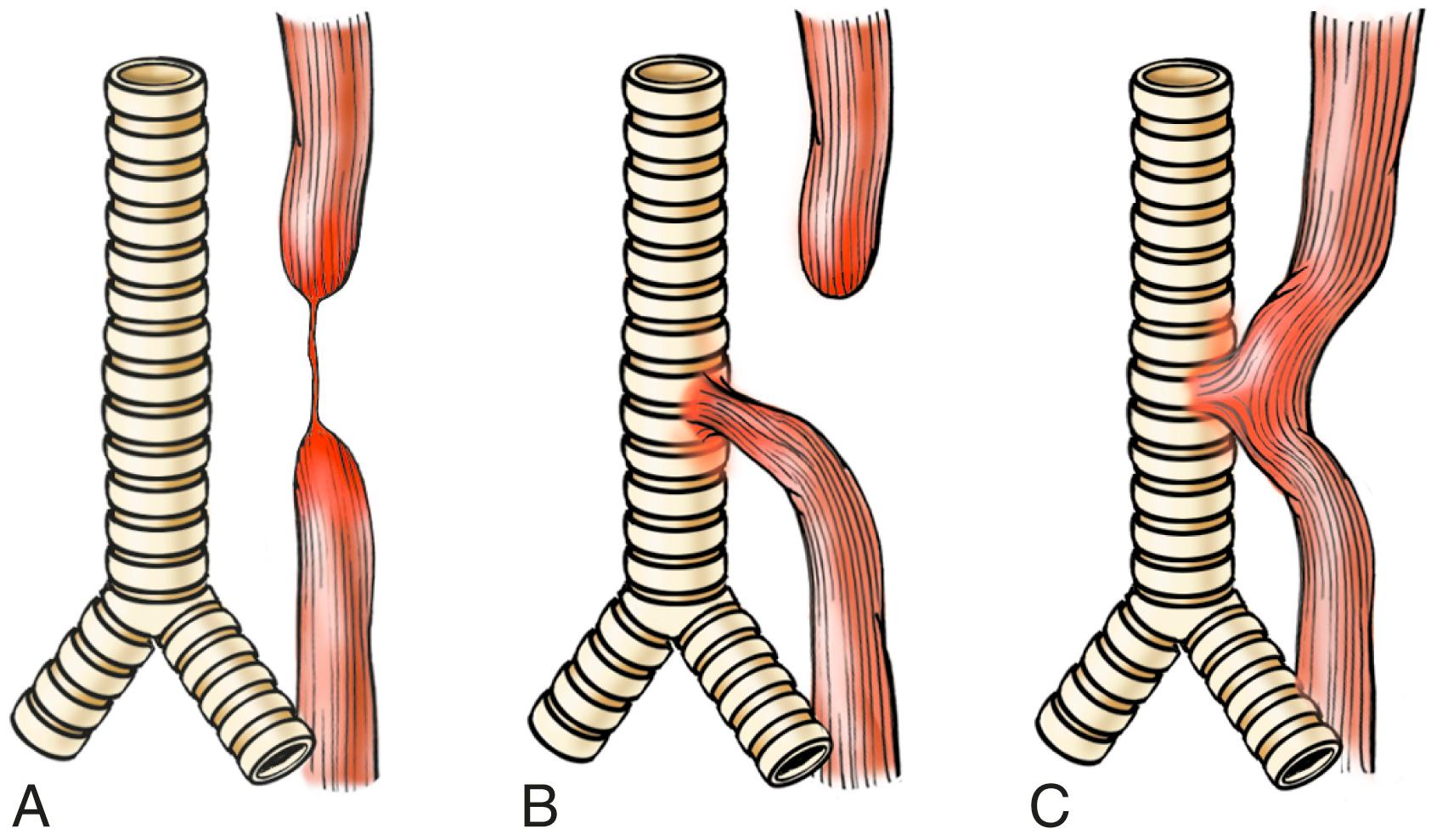
Atresia, fistulas, and duplications may occur in any part of the gastrointestinal tract, including the esophagus ( eFig. 13.3 ). When they involve the esophagus, they are discovered shortly after birth, usually because of regurgitation during feeding, warranting prompt surgical repair. Absence, or agenesis, of the esophagus is extremely rare; atresia, in which a thin, noncanalized cord replaces a segment of esophagus, is more common. It occurs most frequently at or near the tracheal bifurcation and is usually associated with a fistula connecting the upper or lower esophageal pouches to a bronchus or the trachea. This abnormal connection may result in aspiration, suffocation, pneumonia, or severe fluid and electrolyte imbalances.
Esophageal stenosis may be congenital or (more commonly) acquired. When acquired, the narrowing is generally caused by fibrous thickening of the submucosa and atrophy of the muscularis propria due to inflammation and scarring secondary to chronic gastroesophageal reflux, systemic sclerosis, irradiation, ingestion of caustic agents, or other forms of severe injury. Stenosis-associated dysphagia is usually progressive: difficulty eating solids typically occurs long before problems with swallowing liquids appear.
Efficient delivery of food and fluids to the stomach requires coordinated waves of peristaltic contractions. Esophageal dysmotility interferes with this process and can take several forms, all characterized by dyscoordinated contraction or spasm of the muscularis. Because it increases esophageal wall stress, spasm also can cause small diverticula to form. Esophageal dysmotility can be separated into several forms depending on the nature of the contractile abnormalities.
Achalasia is characterized by the triad of incomplete lower esophageal sphincter (LES) relaxation, increased LES tone, and esophageal aperistalsis. Primary achalasia is caused by degeneration of distal esophageal inhibitory neurons and is, by definition, idiopathic. Loss of neural innervation due to damage within the esophagus, the extraesophageal vagus nerve, or the dorsal motor nucleus of the vagus may lead to secondary achalasia. This occurs in Chagas disease, in which Trypanosoma cruzi infection causes destruction of the myenteric plexus, failure of LES relaxation, and esophageal dilatation. As mentioned in Chapter 9 , T.cruzi is an important cause of myocarditis in South and Central America. Achalasia-like disease may also be caused by diabetic autonomic neuropathy, infiltrative disorders such as malignancy, amyloidosis and sarcoidosis, and lesions of dorsal motor nuclei, which may be due to polio or surgical ablation.
Ectopic tissues (developmental rests) are common in the gastrointestinal tract. The most frequent site of ectopic gastric mucosa is the upper third of the esophagus, where it is referred to as an inlet patch. Although such tissue is generally asymptomatic, acid released by ectopic gastric mucosa may lead to dysphagia, esophagitis, Barrett esophagus, or, rarely, adenocarcinoma. Gastric heterotopia, patches of ectopic gastric mucosa in the small bowel (e.g., within a Meckel diverticulum , a remnant of the omphalomesenteric duct that is present in approximately 2% of individuals) or the colon, may present with occult blood loss due to local injury caused by acid secretion.
Before returning to the heart, venous blood from the gastrointestinal tract is delivered to the liver via the portal vein. This circulatory pattern is responsible for the first-pass effect, in which drugs and other materials absorbed in the intestines are processed by the liver before entering the systemic circulation. Diseases that impede portal blood flow cause portal hypertension and may lead to the development of esophageal varices, an important cause of massive, life-threatening bleeding.
One of the few sites where the splanchnic and systemic venous circulations communicate is the esophagus. Portal hypertension induces development of collateral channels that allow portal blood to shunt into the caval system. However, the increased blood flow enlarges and dilates subepithelial and submucosal venous plexi within the distal esophagus. These vessels, termed varices, develop in 50% of patients with cirrhosis, most commonly in association with alcohol-related liver disease. Worldwide, hepatic schistosomiasis is the second most common cause of varices. A more detailed consideration of portal hypertension is provided in Chapter 14 .
Varices are often asymptomatic, but their rupture can lead to massive hematemesis and death and constitutes a medical emergency. Despite intervention, as many as 20% of patients die from the first bleeding episode, either as a direct consequence of hemorrhage or due to hepatic coma triggered by hypovolemic shock and metabolic disturbances. Among those who survive, additional episodes of hemorrhage, each potentially fatal, occur in as many as 60% of cases.
The most common esophageal lacerations are Mallory-Weiss tears, which are often induced by severe retching or vomiting. Normally, a reflex relaxation of the gastroesophageal musculature precedes the antiperistaltic contractile wave associated with vomiting. This relaxation may fail during prolonged vomiting and, as a result, the refluxing gastric contents may cause the esophageal wall to stretch and tear. Patients usually present with hematemesis.
The roughly linear lacerations of Mallory-Weiss syndrome are longitudinally oriented and usually cross the gastroesophageal junction ( Fig. 13.8 A). These superficial tears generally heal quickly without intervention. By contrast, transmural esophageal tears (Boerhaave syndrome) result in severe mediastinitis and usually require prompt surgical intervention.
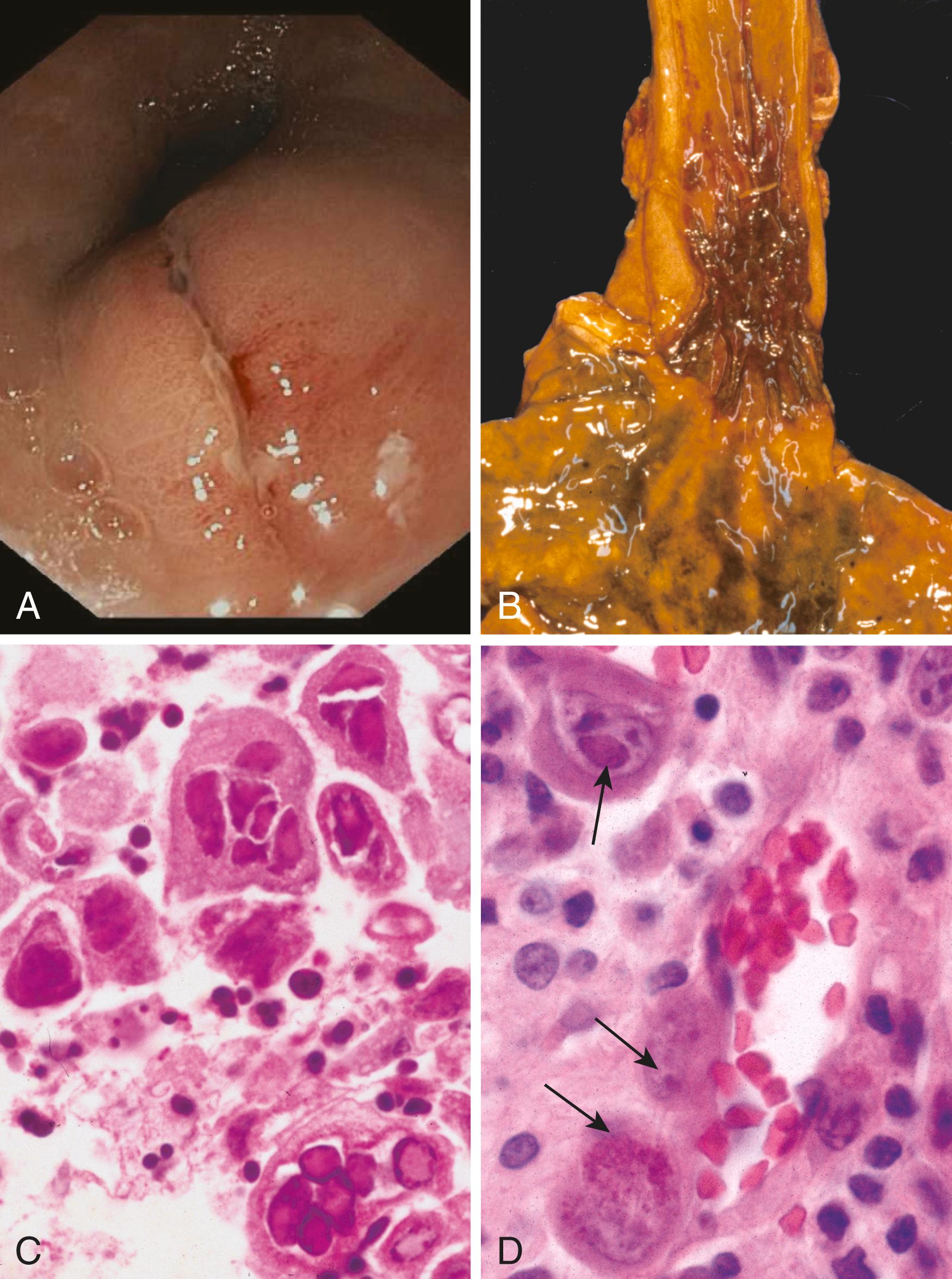
The stratified squamous mucosa of the esophagus may be damaged by a variety of irritants including alcohol, corrosive acids or alkalis, excessively hot fluids, and heavy smoking. Medicinal pills, most commonly doxycycline and bisphosphonates, may adhere to the esophageal lining and dissolve in the esophagus rather than passing immediately into the stomach, resulting in pill-induced esophagitis . Esophagitis due to chemical injury generally causes only self-limited pain, particularly odynophagia (pain with swallowing).
Hemorrhage, stricture, or perforation may occur in severe cases. Iatrogenic esophageal injury may be caused by cytotoxic chemotherapy, radiation therapy, or graft-versus-host disease. The morphologic changes are nonspecific, consisting of ulceration and acute inflammation. Irradiation also causes blood vessel damage, adding an element of ischemic injury.
Infectious esophagitis may occur in otherwise healthy individuals but is most frequent in those who are debilitated or immunocompromised. In these patients, esophageal infection by herpes simplex viruses, cytomegalovirus (CMV), or fungal organisms is common. Among fungi, Candida is the most common pathogen. The esophagus may also be involved in desquamative skin diseases, such as bullous pemphigoid and epidermolysis bullosa, and rarely by Crohn disease.
Infection by fungi or bacteria may be primary or complicate a preexisting ulcer. Nonpathogenic oral bacteria are frequently found in ulcer beds, while pathogenic organisms, which account for about 10% of infectious esophagitis cases, may invade the lamina propria and cause necrosis of the overlying mucosa. Candidiasis is characterized by adherent, grayish white pseudomembranes composed of densely matted fungal hyphae and inflammatory cells covering the esophageal mucosa, similar to that seen in the oral cavity.
The endoscopic appearance often provides a clue to the identity of the infectious agent in viral esophagitis. Herpes simplex virus (HSV) typically produces punched-out ulcers ( Fig. 13.8 B), and histopathologic analysis demonstrates nuclear viral inclusions within a rim of degenerating epithelial cells at the ulcer edge ( Fig. 13.8 C). By contrast, CMV causes shallower ulcerations. Biopsy of CMV lesions shows the characteristic nuclear and cytoplasmic inclusions within endothelial cells and stromal cells ( Fig. 13.8 D). Immunohistochemical staining for HSV and CMV antigens may be a useful diagnostic tool.
Reflux of gastric contents is the most frequent cause of esophagitis and the most common gastrointestinal ailment with which patients present in the outpatient setting in the United States. The associated clinical condition is termed gastroesophageal reflux disease (GERD). The stratified squamous epithelium of the esophagus is resistant to abrasion from foods but is sensitive to acid. Mucosal protection to acid is afforded by mucin and bicarbonate secreted from submucosal glands in the proximal and distal esophagus. More importantly, high lower-esophageal sphincter tone protects against reflux of acidic gastric contents, which are under positive pressure.
Reflux of gastric fluids is central to the development of mucosal injury in GERD. In some cases, duodenal bile reflux may exacerbate the damage. Conditions that decrease lower esophageal sphincter tone or increase abdominal pressure contribute to GERD and include alcohol and tobacco use, obesity, central nervous system depressants, pregnancy, hiatal hernia (discussed later), delayed gastric emptying, and increased gastric volume. In many cases, no definitive cause of reflux is identified.
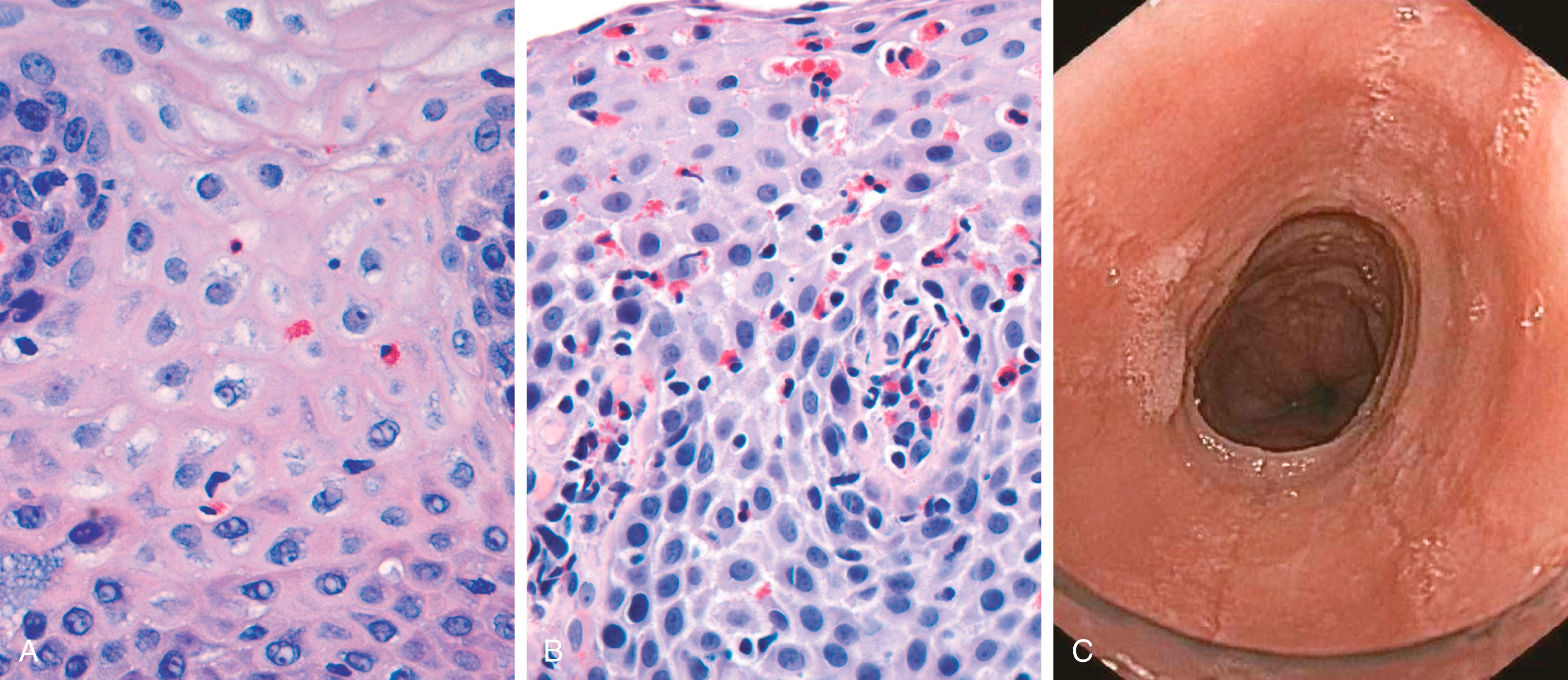
By endoscopy erythema may be the only alteration. In mild GERD the mucosal histology is often unremarkable. With more significant disease, eosinophils are recruited to the squamous mucosa, followed by neutrophils, which are usually associated with more severe injury ( Fig. 13.9 A ). Basal zone hyperplasia and elongation of lamina propria papillae may also be seen.
GERD is most common in those over 40 years of age. The most frequent symptoms are heartburn, dysphagia, and, less often, noticeable regurgitation of sour-tasting gastric contents. Rarely, chronic GERD is punctuated by attacks of severe chest pain that may be mistaken for heart disease. Treatment with proton pump inhibitors reduces gastric acidity and typically provides symptomatic relief. While the severity of symptoms is not closely related to the degree of histologic change, the latter tends to increase with disease duration. Complications include esophageal ulceration, hematemesis, melena, stricture development, and Barrett esophagus, a precursor lesion to esophageal carcinoma (see below).
Hiatal hernia is also associated with esophageal reflux. It is characterized by separation of the diaphragmatic crura and protrusion of the stomach into the thorax through the resulting gap. Congenital hiatal hernias are recognized in infants and children, but many are acquired in later life. They are symptomatic in fewer than 10% of adults. Because hiatal hernia may lead to LES incompetence, when present symptoms often resemble GERD.
Eosinophilic esophagitis is a chronic immunologic disorder characterized clinically by symptoms related to esophageal dysfunction and histologically by the presence of eosinophilic inflammation. Symptoms include food impaction and dysphagia in adults and feeding intolerance or GERD-like symptoms in children. The cardinal histologic feature is epithelial infiltration by large numbers of eosinophils, particularly superficially ( Fig. 13.9 B) and at sites far from the gastroesophageal junction. Their abundance can help to differentiate eosinophilic esophagitis from GERD, Crohn disease, and other causes of esophagitis. Endoscopically evident rings in the upper and mid portions of the esophagus ( Fig. 13.9 C) may also help to distinguish eosinophilic esophagitis from GERD. Patients with eosinophilic esophagitis are typically refractory to proton pump inhibitor treatment. Most patients are atopic, and many have atopic dermatitis, allergic rhinitis, asthma, or modest peripheral eosinophilia. Treatments include dietary restrictions to prevent exposure to food allergens (e.g., cow's milk, soy products) and corticosteroids.
Barrett esophagus is a complication of chronic GERD that is characterized by intestinal metaplasia of the esophageal mucosa and an increased risk for development of adenocarcinoma. The incidence of Barrett esophagus is rising: it is estimated to occur in as many as 10% of individuals with symptomatic GERD. Patients typically present between 40 and 60 years of age and males are affected more than females. Molecular studies indicate that Barrett epithelium shares many acquired driver mutations with adenocarcinoma, consistent with the view that Barrett esophagus is a precursor of cancer. In keeping with this, epithelial dysplasia, considered to be a precursor lesion, develops in 0.2% to 1% of individuals with Barrett esophagus each year; its incidence increases with duration of symptoms and increasing patient age. Although the vast majority of esophageal adenocarcinoma is associated with Barrett esophagus, most individuals with Barrett esophagus do not develop esophageal cancer.
Barrett esophagus is recognized endoscopically as tongues or patches of reddish, velvety mucosa extending upward from the gastroesophageal junction ( Fig. 13.10 A ). This metaplastic mucosa alternates with residual smooth, pale pink or gray squamous (esophageal) mucosa proximally and interfaces with light-brown columnar (gastric) mucosa distally ( Fig. 13.10 B and C). High-resolution endoscopes have increased the sensitivity of Barrett esophagus detection.
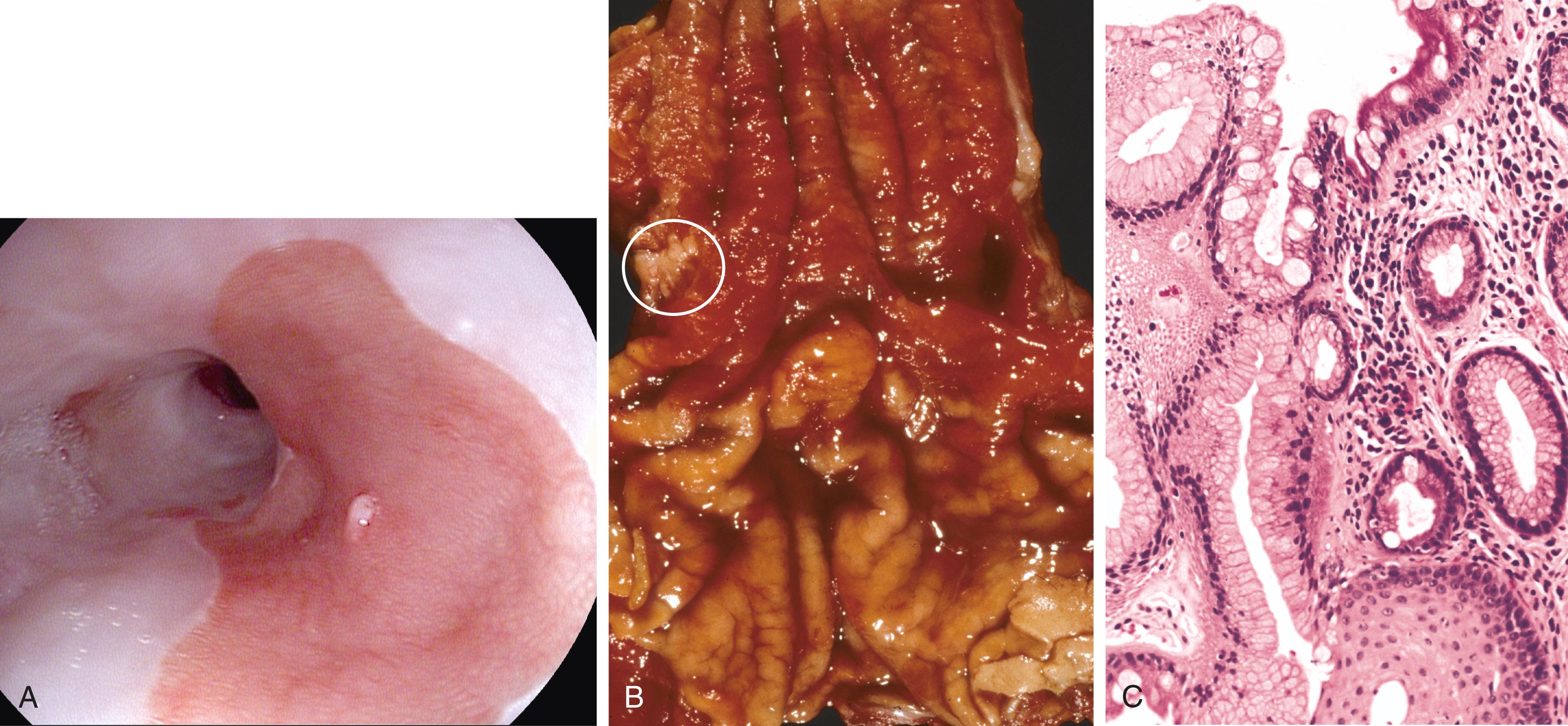
The defining feature of intestinal metaplasia is the presence of goblet cells, which have distinct mucin vacuoles that stain pale blue by H&E and impart the shape of a wine goblet to the remaining cytoplasm ( Fig. 13.10 C). Dysplasia is classified as low grade or high grade based on morphologic criteria.
Diagnosis of Barrett esophagus is usually prompted by GERD symptoms and requires endoscopy and biopsy. Most experts require both endoscopic evidence of abnormal mucosa more than 1 cm above the gastroesophageal junction and histologically documented intestinal metaplasia for diagnosis of Barrett esophagus. The optimal management is a matter of debate, but most clinicians recommend periodic surveillance endoscopy with biopsy to screen for dysplasia. Dysplasia is often treated and more advanced lesions, including high-grade dysplasia and intramucosal carcinoma, always require therapeutic intervention. Available modalities include surgical resection (esophagectomy) , radiofrequency ablation, and endoscopic mucosectomy.
Adenocarcinoma and squamous cell carcinoma account for the majority of esophageal tumors. Worldwide, squamous cell carcinoma is more common, but adenocarcinoma is on the rise. Other rare tumors are not discussed here.
Esophageal adenocarcinoma typically arises in a background of Barrett esophagus and long-standing GERD. Risk for development of adenocarcinoma is greater in patients with documented dysplasia and in those who use tobacco, are obese, or who have had previous radiation therapy. In the United States, esophageal adenocarcinoma is seven times more common in men than in women and is more common in individuals of European descent. Incidence varies worldwide, with rates being highest in Western countries, including the United States, the United Kingdom, Canada, Australia, and the Netherlands, and lowest in Korea, Thailand, Japan, and Ecuador. In countries where esophageal adenocarcinoma is more common, the incidence has increased more rapidly than for almost any other cancer. As a result, esophageal adenocarcinoma, which represented less than 5% of esophageal cancers before 1970, now accounts for half of all esophageal cancers in some Western countries, including the United States. This increase is largely attributed to the increased incidence of GERD and associated Barrett esophagus.
Molecular studies suggest that the progression of Barrett esophagus to adenocarcinoma occurs over an extended period through the stepwise acquisition of genetic and epigenetic changes. This model is supported by the observation that epithelial clones identified in nondysplastic Barrett metaplasia persist and accumulate mutations during progression to dysplasia and invasive carcinoma. Chromosomal abnormalities and TP53 mutation are often present in the early stages of esophageal adenocarcinoma. Additional genetic changes and inflammation are thought to contribute to tumor progression.
Esophageal adenocarcinoma usually occurs in the distal third of the esophagus and may invade the adjacent gastric cardia ( Fig. 13.11 A ). Early lesions often appear as flat or raised patches in otherwise intact mucosa, while advanced tumors tend to form large exophytic masses, infiltrate diffusely, or ulcerate and invade deeply. On microscopic examination, Barrett esophagus is frequently present adjacent to the tumor. Tumors typically produce mucin and form glands ( Fig. 13.11 B).
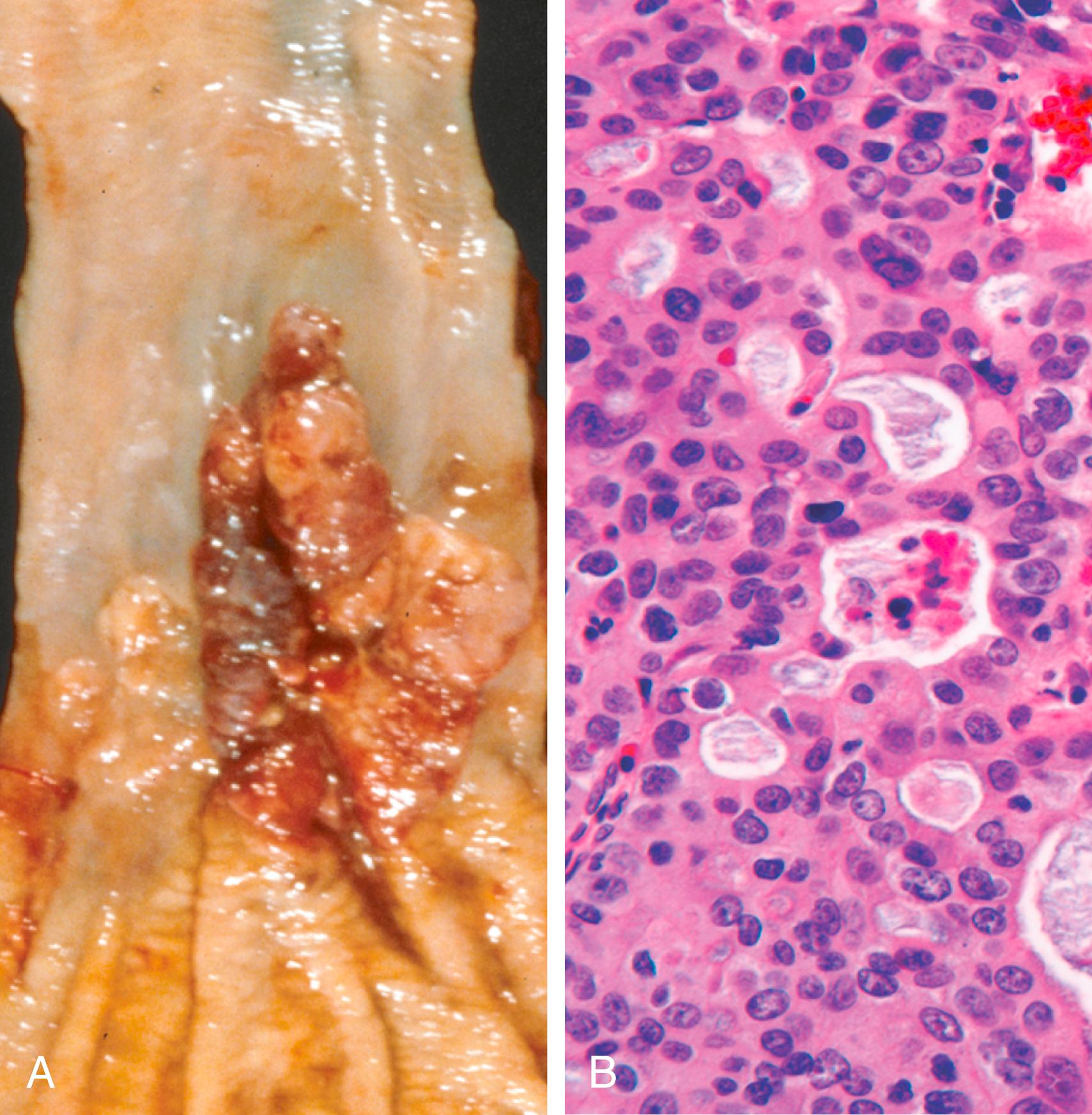
Patients most commonly present with pain or difficulty in swallowing, progressive weight loss, chest pain, or vomiting. By the time signs and symptoms appear, the tumor has usually invaded submucosal lymphatic vessels. As a result of the advanced stage at diagnosis, the overall 5-year survival rate is less than 25%. By contrast, 5-year survival is close to 80% in the few patients with adenocarcinoma limited to the mucosa or submucosa.
In the United States, esophageal squamous cell carcinoma typically occurs in adults older than 45 years of age and affects males four times more frequently than females. Risk factors include alcohol and tobacco use, caustic esophageal injury, achalasia, Plummer-Vinson syndrome (iron deficiency anemia, dysphagia, and esophageal webs), frequent consumption of very hot beverages, and previous radiation therapy to the mediastinum. The incidence of esophageal squamous cell carcinoma can vary by more than 100-fold between and within countries, being more common in rural and lower resource areas. The countries with highest incidences are Iran, central China, Hong Kong, Argentina, Brazil, and South Africa.
A majority of esophageal squamous cell carcinomas in Europe and the United States is associated with the use of alcohol and tobacco, the effects of which synergize to increase risk. However, esophageal squamous cell carcinoma is also common in some regions where alcohol and tobacco use are uncommon due to religious or societal norms. In these areas, nutritional deficiencies and exposure to polycyclic hydrocarbons, nitrosamines, and other mutagenic compounds, such as those found in fungus-contaminated foods, are suspected to be the risk factors. HPV infection has also been implicated in esophageal squamous cell carcinoma in high-risk regions. DNA sequencing of esophageal squamous cell carcinoma has identified frequent mutations in genes that regulate squamous differentiation (e.g., TP63 and NOTCH ) as well as genes encoding regulators of proliferation (e.g., cyclins, cyclin-dependent kinases, RB).
In contrast to the distal location of most adenocarcinomas, half of squamous cell carcinomas occur in the middle third of the esophagus ( Fig. 13.12 A ). Squamous cell carcinoma arises from in situ lesions that exhibit squamous dysplasia. Early lesions appear as small, grayish white plaquelike thickenings. Over months to years, they grow into tumor masses that may be polypoid and protrude into and obstruct the lumen. Other tumors are either ulcerated or diffusely infiltrative lesions that spread within the esophageal wall, where they can cause thickening, rigidity, and luminal narrowing. These cancers may invade surrounding structures including the respiratory tree, causing pneumonia; the aorta, often causing fatal hemorrhage; or the mediastinum and pericardium.
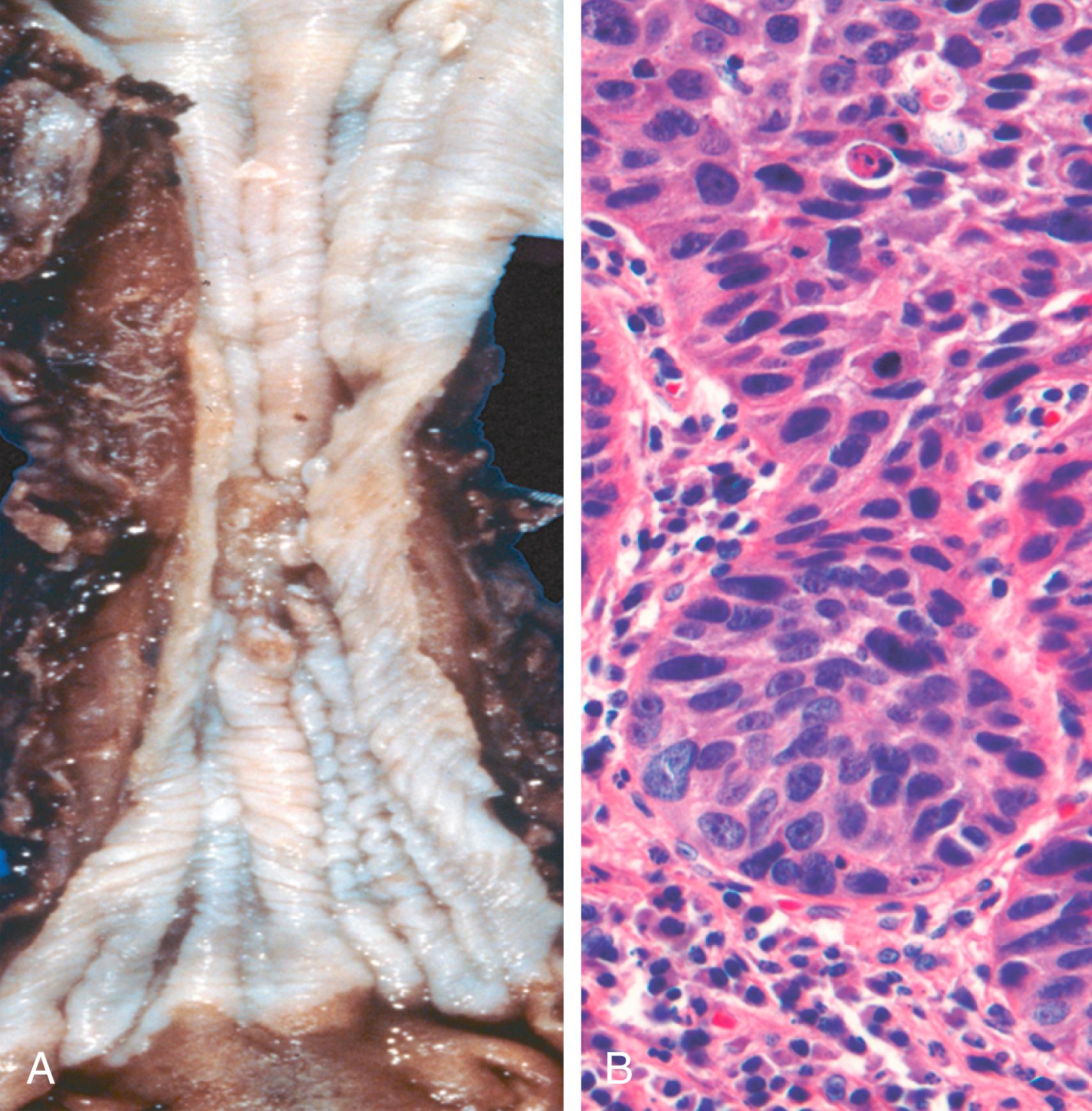
Most squamous cell carcinomas are moderately to well differentiated ( Fig. 13.12 B). Regardless of the histologic grade, symptomatic tumors have often invaded the esophageal wall at the time of diagnosis. The rich submucosal lymphatic network promotes circumferential and longitudinal spread, and intramural tumor nodules may be present several centimeters away from the main mass. The sites of lymph node metastases vary with tumor location: Cancers in the upper third of the esophagus tend to spread to cervical lymph nodes; those in the middle third most commonly metastasize to mediastinal, paratracheal, and tracheobronchial nodes; and those in the lower third spread to gastric and celiac nodes.
Manifestations of squamous cell carcinoma of the esophagus appear insidiously and include dysphagia, odynophagia (pain on swallowing), and obstruction. As with other forms of esophageal obstruction, patients may unconsciously adjust to the progressively increasing obstruction by altering their diet from solid to liquid foods. Extreme weight loss and debilitation may occur as consequences of impaired nutrition and tumor-associated cachexia. As with adenocarcinoma, hemorrhage and sepsis may accompany tumor ulceration. Occasionally, squamous cell carcinoma of the upper and mid esophagus presents with symptoms caused by aspiration of food via a tracheoesophageal fistula.
Although 5-year survival rates are 75% for patients with superficial esophageal carcinoma, they are much lower for patients with advanced tumors. Since most tumors are detected at an advanced stage, the overall 5-year survival rate is only approximately 10%.
Disorders of the stomach are a frequent source of disease, with inflammatory and neoplastic lesions being the most common. In the United States, symptoms related to gastric acidity account for nearly one-third of health care spending on gastrointestinal disease. In addition, despite a decreasing incidence in certain locales, including the United States, gastric cancer remains a leading cause of death worldwide.
The stomach is divided into four major anatomic regions: the cardia, fundus, body, and antrum. The cardia is lined mainly by mucin-secreting foveolar cells that form shallow glands. The antral glands are similar but also contain endocrine cells, such as G cells that release gastrin to stimulate luminal acid secretion by parietal cells within the gastric fundus and body. The well-developed glands of the body and fundus also contain chief cells, which produce and secrete digestive enzymes such as pepsin.
Gastritis results from mucosal injury. When neutrophils are present, the lesion is referred to as acute gastritis . When cell injury and regeneration are present but inflammatory cells are rare or absent, the term gastropathy is applied. Agents that cause gastropathy include nonsteroidal antiinflammatory drugs, alcohol, bile, and stress-induced injury. Acute mucosal erosion or ulceration, such as Curling ulcers or lesions following disruption of gastric blood flow, for example, in portal hypertension, can also cause gastropathy that typically progresses to gastritis. The term hypertrophic gastropathy is applied to a specific group of diseases exemplified by Ménétrier disease and Zollinger-Ellison syndrome (discussed later).
Both gastropathy and acute gastritis may be asymptomatic or cause variable degrees of epigastric pain, nausea, and vomiting. In more severe cases, there may be mucosal erosion, ulceration, hemorrhage, hematemesis, melena or, rarely, massive blood loss.
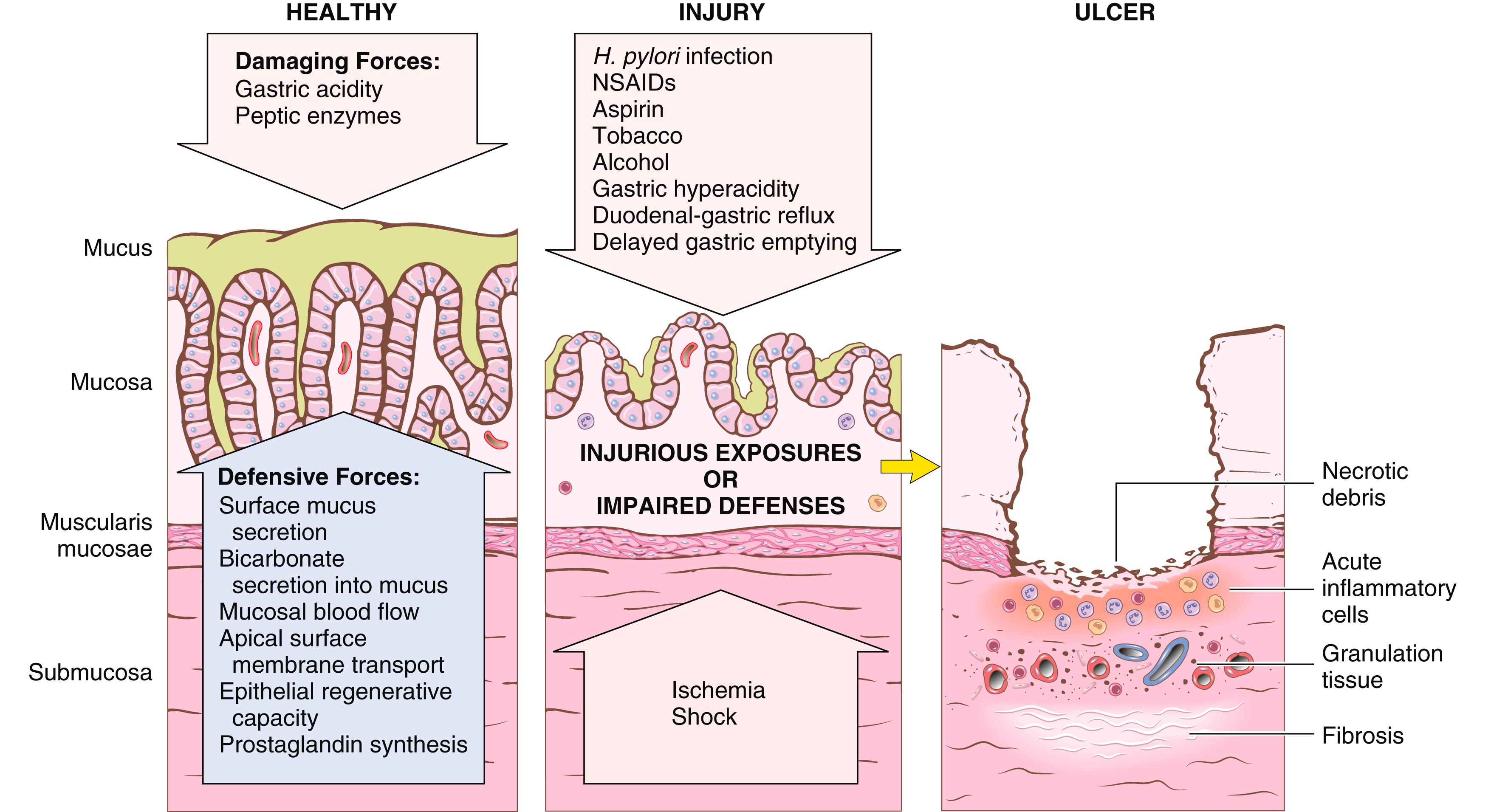
The gastric lumen normally has a pH close to 1—more than 1 million times more acidic than the blood. This harsh environment aids digestion but also has the potential to damage the mucosa. Several mechanisms have evolved to protect the gastric mucosa from “self-digestion” ( Fig. 13.13 ). Surface foveolar cells produce a thin layer of mucus that is resistant to acid and has a neutral pH due to secretion of bicarbonate ions by surface epithelial cells. This mucus layer also protects the mucosa from potential physical damage caused by food particles. In addition, any protons that diffuse back into the lamina propria are buffered by the rich blood supply of the gastric mucosa. Gastropathy, acute gastritis, and chronic gastritis may occur after disruption of these protective mechanisms. The main causes include:
Nonsteroidal antiinflammatory drugs (NSAIDs) inhibit cyclooxygenase (COX)-dependent synthesis of prostaglandins E 2 and I 2 , which protect the gastric lining by stimulating mucus and bicarbonate secretion, mucosal blood flow, and epithelial restitution.
The gastric injury that occurs in uremic patients and those infected with urease-secreting H. pylori may be due to inhibition of gastric bicarbonate transporters by ammonium ions.
Aging, which is associated with reduced mucin and bicarbonate secretion, has been suggested to explain the increased susceptibility of older adults to gastritis.
Hypoxemia and decreased oxygen delivery may account for an increased incidence of gastropathy and acute gastritis at high altitudes.
Ingestion of harsh chemicals, particularly acids or bases, either accidentally or in suicide attempts, leads to severe gastric mucosal damage as a result of direct injury to epithelial and stromal cells. Direct cellular damage also contributes to gastritis induced by excessive alcohol consumption, NSAID use, and radiation therapy. Agents that inhibit cell division, such as those used in cancer chemotherapy, may cause generalized mucosal damage due to insufficient epithelial renewal.
Histologically, gastropathy and mild acute gastritis may be difficult to recognize since the lamina propria shows only moderate edema and slight vascular congestion. The surface epithelium is intact, but hyperplasia of foveolar mucus cells is typically present. Neutrophils, lymphocytes, and plasma cells are not prominent.
The presence of neutrophils above the basement membrane in contact with epithelial cells is abnormal in all parts of the gastrointestinal tract and signifies active inflammation or, at this site, gastritis (rather than gastropathy). The term active inflammation is preferred over acute inflammation throughout the luminal gastrointestinal tract since neutrophils may be present in both acute and chronic disease states. With more severe mucosal damage, erosions and hemorrhage develop. Hemorrhage may manifest as dark punctae in an otherwise hyperemic mucosa. Concurrent presence of erosion and hemorrhage is termed acute erosive hemorrhagic gastritis.
Stress-related gastric injury occurs in patients with severe trauma, extensive burns, intracranial disease, major surgery, serious medical diseases, and other forms of severe physiologic stress. More than 75% of critically ill patients develop endoscopically visible gastric lesions during the first 3 days of their illness. In some cases, the ulcers are given specific names based on location and clinical associations. Examples are as follows:
Stress ulcers , which may occur in critically ill patients with shock, sepsis, or severe trauma
Curling ulcers , which may occur in the proximal duodenum of individuals who have suffered severe burns or trauma
Cushing ulcers , which may occur in the stomach, duodenum, or esophagus of patients with CNS injury, such as stroke; these have a high incidence of perforation
Stress-related gastric mucosal injury is most often due to ischemia caused by systemic hypotension or reduced gastric blood flow resulting from splanchnic vasoconstriction. The decrease in blood flow appears to reduce the secretion of bicarbonate while also diminishing buffering by blood, setting the stage for mucosal injury. In patients who are severely ill, systemic acidosis may also contribute to mucosal injury by lowering the intracellular pH of mucosal cells. CNS injury may lead to the formation of Cushing ulcers by stimulation of vagal nuclei, which transmit parasympathetic impulses that promote acid hypersecretion.
Stress-related gastric mucosal injury ranges from shallow erosions caused by superficial epithelial damage to deep lesions that penetrate the mucosa. Acute ulcers are round and typically less than 1 cm in diameter. The ulcer base is frequently stained brown to black by acid-digested extravasated red cells. Unlike peptic ulcers, which arise in the setting of chronic injury and are typically solitary lesions at the interface of the body and antrum, acute stress ulcers can be found anywhere in the stomach and are often multiple. They are sharply demarcated, with essentially normal adjacent mucosa, although there may be suffusion of blood into the mucosa and submucosa and some inflammatory reaction. The scarring and thickening of blood vessels that characterize chronic peptic ulcers are absent. Healing with complete reepithelialization occurs days or weeks after the injurious factors are removed.
Ulcers are associated with nausea, vomiting, melena, and coffee-ground hematemesis. Bleeding from superficial gastric erosions or ulcers sufficient to require transfusion develops in 1% to 4% of these patients. Other complications, including perforation, also may occur. Prophylaxis with proton pump inhibitors may blunt the impact of stress ulceration, but the most important determinant of outcome is the severity of the underlying condition.
The most common cause of chronic gastritis is infection with the bacillus Helicobacter pylori. Autoimmune gastritis, typically associated with gastric atrophy, is the most common cause in patients without H. pylori infection. Chronic NSAID use is a third important cause of gastritis in some populations, as discussed later. Less common causes include radiation injury and chronic bile reflux.
The signs and symptoms associated with chronic gastritis are typically less severe but more persistent than those of acute gastritis. Nausea and upper abdominal discomfort may occur, sometimes with vomiting, but hematemesis is uncommon.
The discovery of the association of H. pylori with peptic ulcer disease revolutionized the understanding of chronic gastritis. These spiral-shaped bacilli are present in gastric biopsy specimens from almost all patients with duodenal ulcers and a majority of those with gastric ulcers or chronic gastritis. Acute H. pylori infection is subclinical in most cases, and it is the subsequent chronic gastritis that ultimately brings the affected person to medical attention.
In the United States, H. pylori infection is associated with lower economic status, residence in areas with poor sanitation, and birth outside of the United States. Infection is typically acquired in childhood and may persist for life. Improved sanitation in many areas likely explains why H. pylori infection rates among younger individuals today are markedly lower than they were in similarly aged individuals 30 years ago. Worldwide, colonization rates vary from less than 10% to more than 80% as a function of age, geography, and social factors.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here