Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
In clinical practice, regular exercise is encouraged as an essential strategy in the prevention of incident coronary artery disease and heart failure. Regular, intense, and rigorous bouts of exercise, largely at a competitive level, have been associated with alterations in cardiac structure and function defined by the term “athlete’s heart.” Echocardiography plays a pivotal role in differentiating normal physiologic adaptations from occult pathologic disease. The implications of appropriate screening are even more critical in the assessment of individuals seeking preparticipation cardiac examinations. These examinations are performed with the goal of ensuring safe involvement in athletic events and prevention of sudden cardiac death (SCD), a rare but devastating consequence associated with underlying occult cardiomyopathies.
A constellation of findings related to chamber size and systolic and diastolic function can be used to help delineate adaptive versus pathologic remodeling patterns in the hearts of athletes. Understanding what constitutes appropriate patterns of structural changes from exercise and when to consider further testing has been studied, with consensus focused around three main phenotype profiles: predominance of hypertrophy, predominantly dilated with or without dysfunction, and right ventricular (RV) involvement.
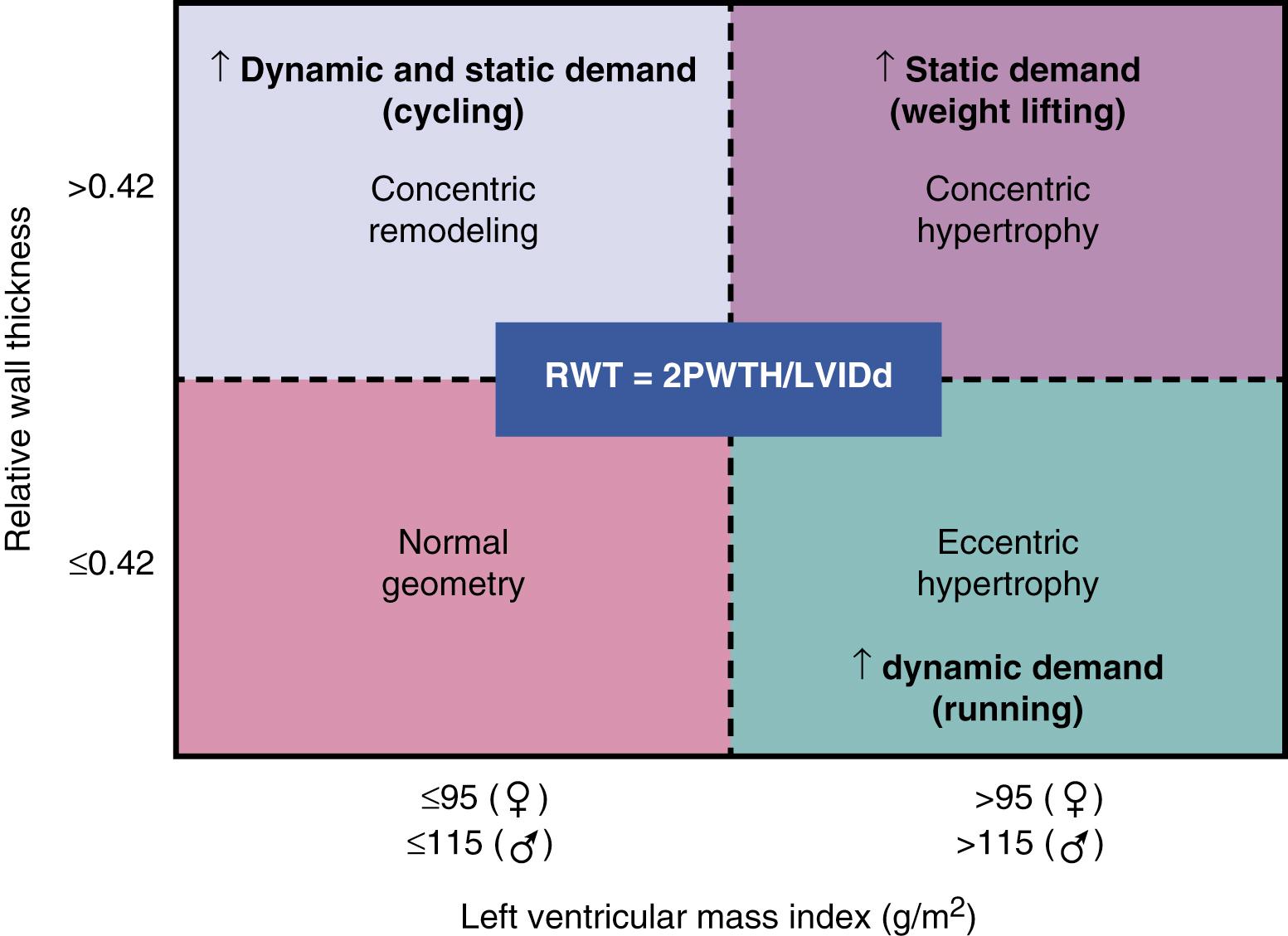
Basic exercise physiology dictates a direct relationship between the exercise intensity and oxygen consumption. This translates to a direct relationship between oxygen uptake and cardiac output (stroke volume × heart rate) in the healthy body, in which cardiac output may increase five to sixfold with exercise. The rise in cardiac output occurs because of a combination of increased sympathetic activation resulting in an increase in heart rate and an increase in preload. To meet the demands of the rigorous exercise regimen adopted by athletes, certain adaptations are made by the cardiovascular system that are based on the nature and intensity of exercise. Exercises that involve mostly static (isometric) components, characterized as short and intensive skeletal muscle contractions, are associated with a sudden rise in left ventricular (LV) afterload (pressure loading) during which the cardiac output must be maintained. Archetypal forms of static exercise include wrestling, throwing events, martial arts, and weight lifting, with adaptive changes manifesting as concentric left ventricular hypertrophy (LVH) without an ostensible increase in LV size. By comparison, dynamic (also called isotonic or endurance) exercise consists of prolonged bouts of repetitive cycles of relaxation and contraction of skeletal muscle, typified by activities such as swimming, running, and rowing. These prolonged periods of exercise are associated with increased oxygen demands of the skeletal muscle and volume loading of the LV, the needs of which are met by augmentation of cardiac output. The long-term effects of this type of exercise include LV dilatation to accommodate larger stroke volumes, and eccentric LVH in addition to more rapid diastolic filling and increased systolic contractility. An algorithmic approach to this dichotomous remodeling response has been termed the “Morganroth hypothesis,” but it has faced criticism for failing to capture the effects of combined static and dynamic activity that cause combined pressure and volume loading on the LV. It is more realistic to recognize that the cumulative proportions of each types of exercise will exhibit a range of expected remodeling patterns for the individual athlete ( Fig. 64.1 ).
Although LV dilatation and hypertrophy are prototypical effects of prolonged static and dynamic exercise, there is a spectrum of features associated with the syndrome of athlete’s heart. These include an LV wall thickness that is increased but rarely exceeds 12 mm with relative symmetric involvement of the LV segments. LV dilatation may occur, with LV end-diastolic dimensions typically less than 60 mm. Degree of LV end diastolic volume increase is likely gender dependent, with three-dimensional echocardiography demonstrating greater volumes in male athletes. Preservation of systolic function is a hallmark of athlete’s heart, reflecting the efficiency of a highly trained cardiovascular system. Occasionally in elite athletes, lower normal systolic function has been reported, with evidence of adequate augmentation with exercise testing. Additional methods for evaluation of systolic function with speckle tracking have demonstrated normal LV strain values even in athletes with LVH; thus, it stands out as an additional method for differentiating normal variants from pathology.
A second component that supports the presence of athlete’s heart is normal (or supranormal) diastolic function. This is caused by the ability of the highly trained and compliant LV to maintain efficient stroke volume and filling pressures even at elevated heart rates. Echocardiographic manifestations include normal mitral inflow patterns with normal E-wave deceleration time and elevated mitral e′ velocities, reflecting enhanced LV compliance and suction in early diastole. Some studies suggest tissue doppler e′ may migrate toward the normal range with increasing age in athletes. Additional supportive findings include normal pulmonary vein flow patterns, and E/e′ values typically below 8, reflecting normal left atrium (LA) pressures in this population.
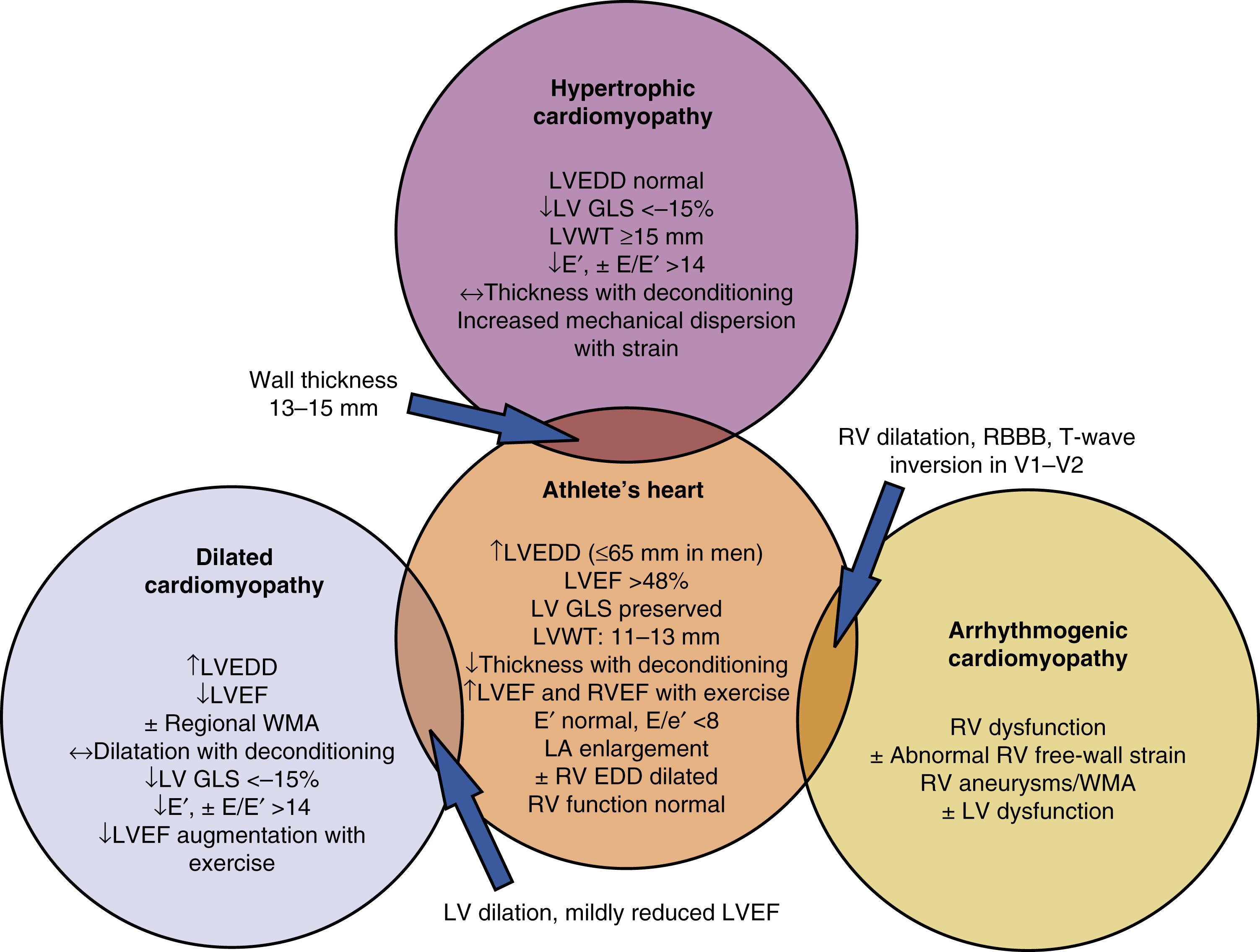
As outlined earlier, wall thickness and LV mass are often increased in athletes, but significant LVH (defined by most studies as LV wall thickness >12 mm) is prevalent in only 1% to 4% of endurance athletes. Demographics may play a role in the prevalence of LVH because female athletes or those younger than 16 years of age have been shown to mount lower degrees of LVH in response to exercise, whereas African American athletes have a higher prevalence and magnitude of LVH. Although isometric exercise would, in theory, generate more pronounced degrees of LVH, this has not borne out consistently in observational studies. Most often, the increased LV wall thickness seen in athletes involves the LV walls in a relatively symmetric fashion, with segment thickness variation of 2 mm or less and ratio of interventricular septal to posterior wall thickness of less than 1.5:1. Additionally, the degree of LVH is accompanied by proportionate increases in LV end-diastolic volumes, accounting for the increased pressure and volume-loading demands of the LV. Furthermore, as outlined earlier, adaptive LVH from athlete’s heart typically demonstrates normal diastolic function. Findings of LV wall thickness exceeding 16 mm should be considered pathologic, particularly when accompanied by normal LV cavity dimensions, reduced mitral annular e′ or elevated E/e′ ratio, electrocardiographic (ECG) evidence of pathologic q waves or T-wave inversions, family history of cardiac disease, and any evidence of LV outflow tract obstruction; these factors all suggest the presence of hypertrophic cardiomyopathy (HCM). Cases in which the LV wall thickness falls between 13 and 16 mm constitute a clinical challenge termed the “gray zone,” and diagnosis may be best adjudicated by the use of additional modalities. One option is the use of speckle-tracking echocardiography (STE), with limited studies indicating that athlete’s heart is associated with preserved LV global longitudinal strain (GLS). Mechanical dispersion (MD) by STE, defined as the segmental time to maximal shortening, is an additional echocardiographic parameter with promise in differentiating pathologic hypertrophy as seen with HCM from athlete’s heart. Increasingly, MD reflects increased or heterogenic variation in systolic thickening, which may occur in regions of abnormal hypertrophy. Another option for supplemental imaging comes in the form of cardiac magnetic resonance imaging (CMRI), which has the ability to detect subclinical fibrosis and asymmetric hypertrophy patterns, findings that both support a diagnosis of HCM. Ultimately, consideration of the global clinical picture, which includes resting ECG, family history, and diastolic function, are essential in contextualizing echo findings. If there is clinical equipoise regarding the true diagnosis, detraining can be implemented ( Fig. 64.2 ).
LV noncompaction is an increasingly reported phenotype of inherited cardiomyopathy that can also display overlap with hypertrabeculation of the LV noted in young asymptomatic athletes. This finding is distinct from the increased LV wall thickness associated with athlete’s heart and characterized as a preponderance of noncompacted myocardium with increased LV trabeculation and recesses, with some studies reporting it to be present in 20% of athletes. One very small case series suggests that increased trabeculation can occur as a response to exercise training, but data in this area are extremely sparse. Pathologic LV noncompaction cardiomyopathy is diagnosed by ratios between noncompacted and compacted myocardium of more than 2.0 at end-systole on echocardiography and more than 2.3 at end-diastole on CMRI, but data have shown these criteria are present in 10% of asymptomatic athletes and thus may not be specific. In cases in which LV noncompaction cardiomyopathy is of concern, the presence of family history, ventricular arrhythmias, LV dysfunction or diastolic dysfunction, and abnormal CMRI imaging should prompt concern for true pathology. Detraining has an unknown impact on trabeculation burden.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here