Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
A wide variety of conditions can cause vesicles, pustules, bullae, erosions, and ulcerations during the newborn period and infancy. Accurate and prompt diagnosis is important because some of the underlying disorders represent potentially life-threatening infections; conversely, many are benign and self-limited. Therefore, it is essential to develop a systematic approach to the evaluation and treatment of newborns and infants with these types of skin lesions. An algorithm outlining initial and subsequent investigations is presented in Fig. 34.1 . This chapter highlights many causes of vesiculopustular and erosive eruptions during the first year of life, and more exhaustive lists are provided in Tables 34.1 and 34.2 . The majority of infectious etiologies are also covered in Chapter 74, Chapter 75, Chapter 76, Chapter 77, Chapter 78, Chapter 79, Chapter 80, Chapter 81, Chapter 82 .
| DIFFERENTIAL DIAGNOSIS OF VESICULOPUSTULAR DISEASES | |||||
|---|---|---|---|---|---|
| Disease | Usual age of onset | Skin: morphology | Skin: distribution | Diagnostic studies (skin) | Comments |
| Infectious diseases (more common conditions are in bold ) | |||||
| Staphylococcal diseases: pyoderma and bullous impetigo (see Ch. 74 ) ( Fig. 34.3A ) | Few days to weeks or older | Pustules, bullae, occasionally vesicles; superficial erosions with collarettes of scale, crusts; furuncles | Favors diaper and periumbilical areas | Gram stain: Gram-positive cocci in clusters; bacterial culture | May occur in outbreaks |
| Group A streptococcal infection (see Ch. 74 ) | Few days to weeks or older | Isolated pustules, honey-colored crusts; moist, foul-smelling, fiery red intertriginous erythema | Any body region; may affect moist umbilical stump or skin folds | Gram stain: Gram-positive cocci in chains; rapid antigen test; bacterial culture | Occasionally cellulitis, meningitis, pneumonia |
| Group B streptococcal infection | Birth to first few days | Vesicles, bullae, erosions, pustules, honey-colored crusts | Any body region | Gram stain: Gram-positive cocci in chains; bacterial culture | Pneumonia, bacteremia, meningitis |
| Listeria monocytogenes infection (see Ch. 74 ) | Birth to first few hours | Hemorrhagic pustules and petechiae | Generalized, especially trunk and extremities | Gram stain: Gram-positive rods; bacterial culture | Sepsis; respiratory distress; history of maternal fever and premature labor |
| Haemophilus influenzae infection (see Ch. 74 ) | Birth to first few days | Vesicles, crusted papules | Any body region | Gram stain: Gram-negative bacilli; bacterial culture | Bacteremia, meningitis |
| Pseudomonas aeruginosa infection (see Ch. 74 ) | Days to weeks or older | Erythema, pustules, hemorrhagic bullae, necrotic ulcerations | Any region, especially anogenital area | Gram stain: Gram-negative rods; bacterial culture | Prematurity, low birth weight, immunodeficiency |
| Congenital candidiasis ( Figs 34.3B & 34.7 ) | Birth to first few days | Erythema, small papules and pustules, fine scaling; “burn-like” if extremely premature (see Table 34.2 ) | Any region; palms/soles often involved; nails may be affected | KOH: budding yeast; fungal culture; placental/umbilical cord lesions may be present | Prematurity, foreign body in cervix/uterus are risk factors; ascending in utero infection |
| Neonatal candidiasis | 1–2 weeks of age | Beefy red patches with scale; satellite pustules and papules | Diaper area, other intertriginous zones, face | KOH: budding yeast, pseudohyphae; fungal culture | Usually otherwise healthy; acquisition during delivery or postnatally; oral thrush |
| Aspergillus infection (see Ch. 77 ) | Few days to weeks | Pustules (often clustered) rapidly evolve to necrotic ulcers | Any region, especially macerated skin in the diaper area and under adhesive tape/occlusive dressings | Skin biopsy: septate hyphae; tissue fungal culture | Extreme prematurity |
| Neonatal HSV infection (see Ch. 80 ) ( Fig. 34.14 ) | Birth to 2 weeks, usually after 5 days | Vesicles, pustules, crusts, erosions | Any region, especially scalp, torso; may involve mucosa | Tzanck preparation, PCR, DFA or immunoperoxidase slide test, viral culture | Signs of sepsis; irritability, lethargy; must exclude herpes meningitis/encephalitis |
| Intrauterine HSV infection | Birth | Vesicles, pustules, widespread erosions, scars, areas of absent skin | Any body region | Tzanck preparation, PCR, DFA or immunoperoxidase slide test, viral culture | Low birth weight, microcephaly, chorioretinitis |
| Neonatal varicella (see Ch. 80 ) | Birth to 2 weeks | Vesicles on an erythematous base | Generalized distribution | Tzanck preparation, PCR, DFA, viral culture | Maternal primary varicella infection 7 days before to 2 days after delivery |
| Herpes zoster (see Ch. 80 ) | Typically 2 weeks or older | Vesicles on an erythematous base | Dermatomal pattern | Tzanck preparation, PCR, DFA, viral culture | Maternal primary varicella during pregnancy or up to a few days after delivery, or neonatal varicella |
| Scabies (see Ch. 84 ) | Typically 3–4 weeks or older | Papules, nodules, crusted areas, vesicles, pustules, burrows | Any region; especially axillae, groin, palms/soles, and wrists | Mineral oil preparation; dermoscopy | Family members may have pruritus and similar lesions |
| Common transient conditions | |||||
| Erythema toxicum neonatorum ( Fig. 34.2 ) | Typically 24–48 hours but can be birth to 2 weeks | Erythematous macules, papules, pustules > vesicles, wheals | Any region, except almost always spares palms/soles | Clinical; Wright's stain: eosinophils | Term infants >2500 g |
| Transient neonatal pustular melanosis ( Fig. 34.4 ) | Birth | Pustules without erythema; collarettes of scale; hyperpigmented macules | Any region; most often forehead, neck, lower back, shins; may affect palms/soles | Clinical; Wright's stain: neutrophils, occasional eosinophils, cellular debris | Term infants; more common in infants of African descent |
| Miliaria crystallina (see Ch. 39 ) ( Fig. 34.5A ) | Birth to early infancy | Fragile vesicles without erythema | Forehead, upper trunk, arms most common | Clinical | Sometimes history of overheating or fever |
| Miliaria rubra (see Ch. 39 ) ( Fig. 34.5B ) | Typically ≥1 week | Erythematous papules with superimposed pustules | Forehead, neck, upper trunk; occluded areas most common | Clinical; Wright's stain: variable inflammatory cells but not prominent eosinophils | Sometimes history of overheating or fever |
| Neonatal cephalic pustulosis (neonatal “acne”) ( Fig. 34.6 ) | ~5 days to 3 weeks | Papules and pustules on erythematous base | Cheeks, forehead, chin, eyelids; less commonly neck, upper chest, scalp | Clinical; Giemsa stain: yeast forms, neutrophils | Otherwise well |
| Uncommon and rare non-infectious diseases | |||||
| Acropustulosis of infancy ( Fig. 34.10 ) | Typically 3–6 months, occasionally birth to weeks | Vesicles and pustules | Hands and feet; occasionally scalp, trunk | Clinical; assess for scabies infestation; skin biopsy: intraepidermal vesicle/pustule with neutrophils and occasionally eosinophils | Severe pruritus; lesions recur in crops; subset with prior scabies |
| Eosinophilic pustular folliculitis of infancy ( Fig. 34.11 ) | Birth to 14 months, mean 6 months | Papules and pustules | Scalp > face > trunk, extremities | Skin biopsy: dense mixed infiltrate with eosinophils, often but not invariably centered on hair follicles | Pruritus; lesions recur in crops; often peripheral eosinophilia; neonatal eosinophilic pustulosis variant favors the face in premature boys |
| Congenital and neonatal Langerhans cell histiocytosis (see Ch. 91 ) ( Fig. 34.12 ) | Birth to weeks | Vesicles, crusts, papules, nodules, petechiae | Any body region, especially flexural sites, palms/soles, scalp | Skin biopsy: S100 + /CD1a + histiocytes with reniform nuclei, focal invasion of epidermis | Occasional mucosal or extracutaneous involvement; pure cutaneous form often resolves spontaneously, but later cutaneous and systemic relapses possible |
| Incontinentia pigmenti (see Ch. 62 ) ( Fig. 34.13 ) | Birth to weeks | Vesicles, hyperkeratotic papules along the lines of Blaschko | Vesicular lesions most common on the extremities | Skin biopsy: eosinophilic spongiosis with necrotic keratinocytes; genetic analysis ( IKBKG / NEMO ) | Ocular, CNS, and dental involvement common but often not evident at birth; X-linked dominant, patients usually female |
| Autosomal dominant hyper-IgE syndrome (see Ch. 60 ) | Birth to weeks | Single and grouped vesicles or papulopustules | Face, scalp, upper trunk, axillae, diaper area | Skin biopsy: intraepidermal vesicle with eosinophils, eosinophilic folliculitis; genetic analysis ( STAT3 ) | Eosinophilia with variably elevated IgE levels; abscesses, pneumonias, and pneumatoceles often develop after neonatal period |
| Vesiculopustular eruption of transient myeloproliferative disorder in Down syndrome | Days to weeks | Vesicles and pustules | Face > trunk, extremities; sites of adhesive dressings, minor trauma | Intraepidermal spongiotic vesiculopustules, infiltrate containing immature myeloid cells | Trisomy 21 or mosaicism for trisomy 21; severe leukocytosis with immature myeloid cells; increased risk of myeloid leukemia |
| Erosive pustular dermatosis of the scalp | Weeks to months | Pustules, erythema with scale-crust, erosions; alopecia and scarring | “Halo scalp ring” pattern, vertex of scalp | Clinical; skin biopsy: alopecia, scarring, mixed dermal infiltrate | Prolonged labor and delivery; necrotic caput succedaneum at birth |
| Neonatal Behçet disease (see Ch. 26 ) | First week | Vesiculopustular, purpuric and necrotic skin lesions; oral and genital ulcers | Lesions favor hands and feet as well as oral and genital mucosae | Clinical | Maternal history of Behçet disease; diarrhea, vasculitis |
| Pustular psoriasis, including deficiency of interleukin-36 receptor antagonist (DITRA; AR) and CARD14-associated pustular psoriasis (CAMPS; AD) (see Chs 8 and 45 ) | Weeks to months or older | Pustules and pustular lakes within areas of erythema | Often generalized with erythroderma; pustules in any body region, especially the palms/soles | Skin biopsy: spongiform pustules and microabscesses within the epidermis, parakeratosis, dilated dermal capillaries; consider genetic analysis ( IL36RN , CARD14 ) | Occasional fever; often resistant to therapy |
| Deficiency of interleukin-1 receptor antagonist (DIRA; see Ch. 45 ) | Birth to weeks | Pustules within areas of erythema | Often generalized with erythroderma; oral lesions | Skin biopsy: neutrophilic microabscesses within acanthotic epidermis, parakeratosis, dilated dermal capillaries; genetic analysis ( IL1RN ) | Sterile osteolytic or hyperplastic bone lesions, neonatal distress; dramatic response to IL-1 antagonists |
| Perforating neutrophilic and granulomatous dermatitis associated with immunodeficiency | Birth to weeks | Papules evolve into vesicles, pustules, crusts, and ulcers | Varies: face, extremities, perineum | Skin biopsy: granulomas, neutrophilic infiltrate, transepidermal elimination of degenerated collagen and debris through hair follicles | Primary immunodeficiencies, including APLAID (autoinflammation and phospholipase C γ2-associated antibody deficiency and immune dysregulation) |
| DIFFERENTIAL DIAGNOSIS OF BULLAE, EROSIONS AND ULCERATIONS | |||||
|---|---|---|---|---|---|
| Disease | Usual age | Skin: morphology | Skin: distribution | Diagnostic studies (skin) | Comments |
| Infectious diseases | |||||
| Staphylococcal scalded skin syndrome (see Ch. 74 ) ( Fig. 34.17 ) | Few days to weeks; rarely congenital | Erythematous patches, fragile bullae, superficial erosions, peeling in sheets | Generalized with periorificial and intertriginous accentuation | Biopsy: epidermal separation at granular cell layer; culture positive only at primary site(s) of infection (toxin-mediated) | Irritability, temperature instability; high risk of secondary sepsis, fluid/electrolyte abnormalities |
| Group B streptococcal infection | (see Table 34.1 ) | ||||
| Pseudomonas aeruginosa infection | (see Table 34.1 ) | ||||
| Congenital syphilis (see Ch. 82 ) | Birth to first few days | Bullae or erosions; erythema and desquamation | Any region, especially perioral, palms/soles | Darkfield examination of serous exudates, DFA; serologic studies (treponemal and non-treponemal) | Snuffles, hepatosplenomegaly, periostitis with pseudoparalysis |
| “Invasive fungal dermatitis” due to candidiasis > other fungi (see text and below) in very-low-birth-weight premature newborns ( Fig. 34.7 ) | Birth to 2 weeks | “Burn-like” erythema with desquamation and erosions; intertriginous maceration | Any region | KOH: budding yeast; fungal culture; placental/umbilical cord lesions may be present | Risk factors: birth weight <1000 g, vaginal delivery, systemic corticosteroid administration, prolonged hyperglycemia; high risk of disseminated systemic infection |
| Aspergillus infection | (see Table 34.1 ) | ||||
| Zygomycosis, trichosporonosis (see Ch. 77 ) | Days to weeks | Generalized peeling and skin breakdown or cellulitis evolving into necrotic ulcer | Any body region | Skin biopsy and tissue fungal culture | Extreme prematurity |
| Intrauterine HSV infection | (see Table 34.1 ) | ||||
| Congenital (fetal) varicella syndrome (see Ch. 80 ) | Birth | Erosions, ulcers, scarring, aplasia cutis congenita | Any region, especially extremities; may have dermatomal pattern | Variably positive Tzanck preparation, PCR, DFA, viral culture | Maternal varicella in the first or early second trimester; limb atrophy, CNS/eye abnormalities |
| Conditions with exogenous causes | |||||
| Sucking blisters ( Fig. 34.8 ) | Birth | Flaccid bulla or linear erosion; most often solitary | Radial forearm, wrist, hand, fingers, occasionally foot | Clinical | Due to sucking on affected areas in utero |
| Irritant contact dermatitis | First few days to weeks or older | Glazed erythema, vesiculation, punched-out erosions | Diaper area, especially on convex surfaces | Usually clinical | Severe form referred to as Jacquet erosive diaper dermatitis |
| Perinatal trauma/iatrogenic injury | Birth or neonatal period | Erosions, ulcerations, scars, atrophic lesions | Depends on cause of trauma | Usually clinical | Perinatal history of fetal scalp electrode monitor, prolonged labor and/or vacuum or forceps delivery, history of amniocentesis, etc. |
| Perinatal gangrene of the buttock | Days | Sudden-onset erythema and cyanosis, then gangrenous ulcers | Buttocks | Clinical | Umbilical artery catheterization in some cases |
| Associated with birthmarks and related conditions | |||||
| Erosions overlying large congenital melanocytic nevi (see Ch. 112 ) | Birth to first few days | Erosions, ulcerations | Superimposed on the congenital melanocytic nevus, most often on the back | Clinical; skin biopsy to exclude melanoma if persistent or other unusual features | Neurocutaneous melanosis present in subset of patients |
| Infantile hemangioma (see Ch. 103 ) | Birth to first few weeks or months | Ulceration, often with a bright red border | Any site, but diaper area and lip most common | Clinical | Cause often not clinically apparent until hemangioma begins to proliferate |
| Aplasia cutis congenita (see Ch. 64 ) ( Fig. 34.9 ) | Birth | “Bullous” form: round/oval, sharply demarcated with overlying membrane; other types with raw, full-thickness ulcer; may occasionally present as scar | Scalp (especially vertex) or face most common; other sites depending on etiology | Usually clinical; imaging studies to evaluate underlying bone, CNS | Possible associations: CNS defects, sagittal sinus thrombosis, trisomy 13, limb-reduction abnormalities |
| Linear porokeratosis, porokeratotic eccrine ostial and dermal duct nevus (PEODDN) (see Chs 62 , 109 , & 111) | Birth or weeks to years later | Occasionally eroded at birth; usually erythema and keratotic border or (in PEODDN) spines | Often extremities, but any site possible | Skin biopsy: cornoid lamella (may not be evident in newborn period) | Risk of squamous cell carcinoma; mosaic GJB2 mutations in PEODDN |
| Uncommon and rare non-infectious diseases | |||||
| Mastocytosis (see Ch. 118 ; Fig. 34.18 ) | Birth to weeks or months | Localized/discrete lesions : papules, plaques or nodules with intermittent superimposed wheals, bullae, erosions; often hyperpigmented Diffuse form : blistering and erosions superimposed on infiltrated skin with “peau d’orange” appearance |
Any body region | Positive Darier sign; skin biopsy: increased mast cells in dermis | Variably present: flushing, irritability, diarrhea, abdominal pain |
| Epidermolysis bullosa (see Ch. 32 ) | Birth to first few days or older | Mechanically induced blisters and erosions; depending on type: mucosal erosions, aplasia cutis congenita of anterior legs, scarring, milia, nail dystrophy | Widespread or limited, depending on type; most often extremities, especially hands/feet | Biopsy of induced blister for immunofluorescence antigen mapping ± electron microscopy; genetic analysis | Difficulty feeding, failure to thrive; occasionally corneal, respiratory tract or gastrointestinal (e.g. pyloric atresia) involvement; anemia |
| Kindler syndrome (see Ch. 32 ) | Birth to weeks or older | Trauma-induced blisters and erosions; progressive poikiloderma | Bullae most common in acral sites; poikiloderma is widespread with skip areas | Skin biopsy: immunostaining with anti-kindlin-1 antibody; genetic analysis ( FERMT1 ) | Photosensitivity; gingivitis, colitis, mucosal stenoses (e.g. esophageal, urethral); acquired syndactyly (toes, proximal fingers); AR inheritance |
| Epidermolytic ichthyosis (see Ch. 57 ; Fig. 34.16 ) | Birth | Erythroderma with blisters and erosions | Generalized | Skin biopsy: epidermolytic hyperkeratosis; genetic analysis ( KRT1 , KRT10 ) | Risk of sepsis, fluid and electrolyte imbalances; AD inheritance |
| Maternal autoimmune bullous disease | Birth | Depends on type of maternal disease: tense or flaccid bullae, erosions | Variable; often widespread | Skin biopsy with DIF usually diagnostic | Maternal history of blistering disease, but occasionally inactive during pregnancy |
| Bullous pemphigoid (see Ch. 30 ) | Usually 2 months of age or older | Tense bullae | Favor hands and feet but may be generalized | Skin biopsy: subepidermal bullae with eosinophils; DIF: linear pattern IgG at DEJ | |
| Linear IgA bullous dermatosis (see Ch. 31 ) | Rarely at birth, usually later infancy or childhood | Tense blisters often form rosette or sausage shapes | Widespread but often concentrated in the girdle area; usually spares mucosa | Skin biopsy: subepidermal bullae with neutrophils; DIF: linear pattern IgA at DEJ | |
| Neonatal (congenital) lupus (see Ch. 41 ) | Birth (~20%) to weeks or months | Congenital erosions/ulcers, crusting, rarely bullae as well as classic annular erythematous plaques; atrophy, scarring, background of livedo reticularis | Face favored, especially the periorbital area, but can be more widespread | Skin biopsy: epidermal atrophy, vacuolar interface dermatitis, variable mucin | Mother and infant with antibodies to SSA/Ro, SSB/La, and/or U1RNP; variable heart block, cardiomyopathy, hepatobiliary disease, cytopenias |
| Toxic epidermal necrolysis (TEN; see Ch. 20 ) or TEN-like GVHD (see Chs 52 , 60 ) | Usually 6 weeks of age or older, except for intrauterine GVHD | Tender erythema, bullae, epidermal sloughing/erosions | Often widespread, evolving rapidly over hours to days; mucosal involvement | Skin biopsy: subepidermal blister with epidermal necrosis (usually full thickness) | Typically in setting of Gram-negative sepsis or due to maternofetal or transfusion-associated GVHD in infants with severe combined immunodeficiency |
| Intrauterine epidermal necrosis | Birth | Widespread erosions and ulcerations without vesicles or pustules | Generalized, spares mucous membranes | Skin biopsy: epidermal necrosis and calcification of pilosebaceous units | Prematurity; brain infarcts, cardiomegaly, renal tubular necrosis; rapid mortality |
| Congenital erosive and vesicular dermatosis ( Fig. 34.15 ) | Birth | Erosions, vesicles, crusts, “scalded skin” | Generalized with relative sparing of face, palms/soles | Clinical diagnosis, often retrospective; skin biopsy (nonspecific): epidermal necrosis, neutrophils or mixed infiltrate; must exclude other etiologies (e.g. HSV) | Prematurity; heals with supple reticulated scarring; CNS/developmental abnormalities |
| Pyoderma gangrenosum (see Ch. 26 ) | Rare reports of congenital or neonatal onset | Sharply demarcated ulcerations with undermined borders | Any site, especially groin and buttocks | Clinical; exclusion of other etiologies; skin biopsy: infiltration of neutrophils without primary vasculitis or infection | Associations include inflammatory bowel disease, chronic recurrent multifocal osteomyelitis; consider immunodeficiency and autoinflammatory syndromes |
| Acrodermatitis enteropathica * (see Ch. 51 ) | Weeks to months | Sharply demarcated, eroded erythematous plaques with scale-crust; occasionally intact vesicles/bullae | Periorificial (i.e. around mouth, nose, eyes, anus, genitalia), neck folds, hands/feet | Low serum zinc and alkaline phosphatase levels | Irritability, diarrhea, failure to thrive; onset classically after weaning if breastfed; acquired zinc deficiency: low maternal breast milk zinc or prematurity; genetic form with AR inheritance ( SLC39A4 ) |
| Methylmalonic acidemia, other organic acidemias/aminoacidopathies * (see Fig. 51.13 ) | Days to weeks | Erythema with or without erosions | Periorificial or more widespread | Urine organic acid analysis; plasma isoleucine level (if restricted diet) | Lethargy, hypotonia, neutropenia, metabolic acidosis; in some cases, skin findings result from therapeutic restrictions of dietary amino acids |
| Restrictive dermopathy | Birth | Rigid tense skin with linear erosions, tears | Generalized; skin tears most common in flexural creases | Clinical; distinguish from Neu–Laxova syndrome (intrauterine growth retardation, microcephaly, abnormal brain development, edema, parchment-like or ichthyosiform skin); genetic analysis ( LMNA, ZMPSTE24 ) | Joint contractures, micrognathia, fixed facial expression, restrictive pulmonary disease; early mortality |
| Focal dermal hypoplasia (see Ch. 62 ) | Birth | Occasional blisters, but more often hypoplasia or aplasia of skin; linear and whorled pattern; also fat “herniation”, telangiectasias | Any body region | Clinical; skin biopsy: dermal hypoplasia with fat abutting epidermis; genetic analysis ( PORCN ) | Skeletal, eye and CNS abnormalities to varying degrees |
| Absent dermal ridges and congenital milia (Basan syndrome; see Ch. 64 ) | Birth | Bullae; absent dermal ridge patterns, multiple milia | Fingers, plantar surface of feet | Clinical; genetic analysis ( SMARCAD1 ) | AD inheritance |
| Porphyrias (see Ch. 49 ) | Days to months | Photosensitive erythema, blistering, erosions; scarring | Sun-exposed areas or more generalized if phototherapy for hyperbilirubinemia | Transient form: elevated plasma porphyrins; heritable forms: elevated urine, fecal and/or plasma porphyrins ± pink/fluorescent urine | Transient form usually related to hemolytic disease; rare heritable forms present with blistering in infancy, e.g. congenital erythropoietic porphyria |
| Neonatal purpura fulminans | Days | Initially purpura or cellulitis-like areas evolving to necrotic bullae or ulcers | Buttocks, extremities, trunk and scalp | Prolonged PT/PTT, low fibrinogen, elevated FDPs, low protein C or S levels | Related to sepsis or inherited protein C or S deficiency |
| Ankyloblepharon–ectodermal dysplasia–clefting (AEC) syndrome (see Ch. 63 ) | Birth | Erythroderma, peeling skin, superficial erosions | Generalized, with prominent scalp involvement | Clinical; genetic analysis ( TP63 ) | Associated findings include ankyloblepharon, ectodermal dysplasia, and cleft lip/palate |
* An acrodermatitis enteropathica-like eruption (periorificial or more widespread), often associated with edema and hypoalbuminemia, can occur in infants (typically 3–5 months of age) with cystic fibrosis as well as in those with organic acidemias/aminoacidopathies (see Ch. 51 ).
▪ Erythema toxicum ▪ Toxic erythema of the newborn
Vesiculopustular eruption characterized by an eosinophilic infiltrate
Affects nearly half of full-term neonates and resolves spontaneously over a few days
Often involves the trunk as well as the face, proximal limbs, and buttocks
Almost always spares the palms and soles
Small wheals, inflammatory papules, pustules, and/or vesicles surrounded by blotchy erythema
Erythema toxicum neonatorum (ETN) is a very common, benign condition. Originally described by Netlinger in 1472, it was known as “toxic erythema of the newborn” and was renamed erythema toxicum neonatorum by Leiner in 1912 . ETN occurs in approximately half of full-term neonates and only rarely in premature infants or those weighing less than 2500 g .
It has no gender predilection and is most commonly observed in Causcasian infants, although this may be related to the difficulty in perceiving erythema in infants with darker skin .
Rarely presenting at birth, ETN usually begins 24 to 48 hours after delivery, followed by a waxing and waning course over the next few days. Although the onset may be as late as 1–2 weeks of age, additional diagnoses need to be considered. Five distinct components may be present in various combinations: erythematous macules, wheals, and small pustules, vesicles and papules, usually measuring 1–2 mm. The characteristic central papule with a surrounding erythematous flare is reminiscent of a flea bite ( Fig. 34.2 ). Mechanical irritation may precipitate new lesions.
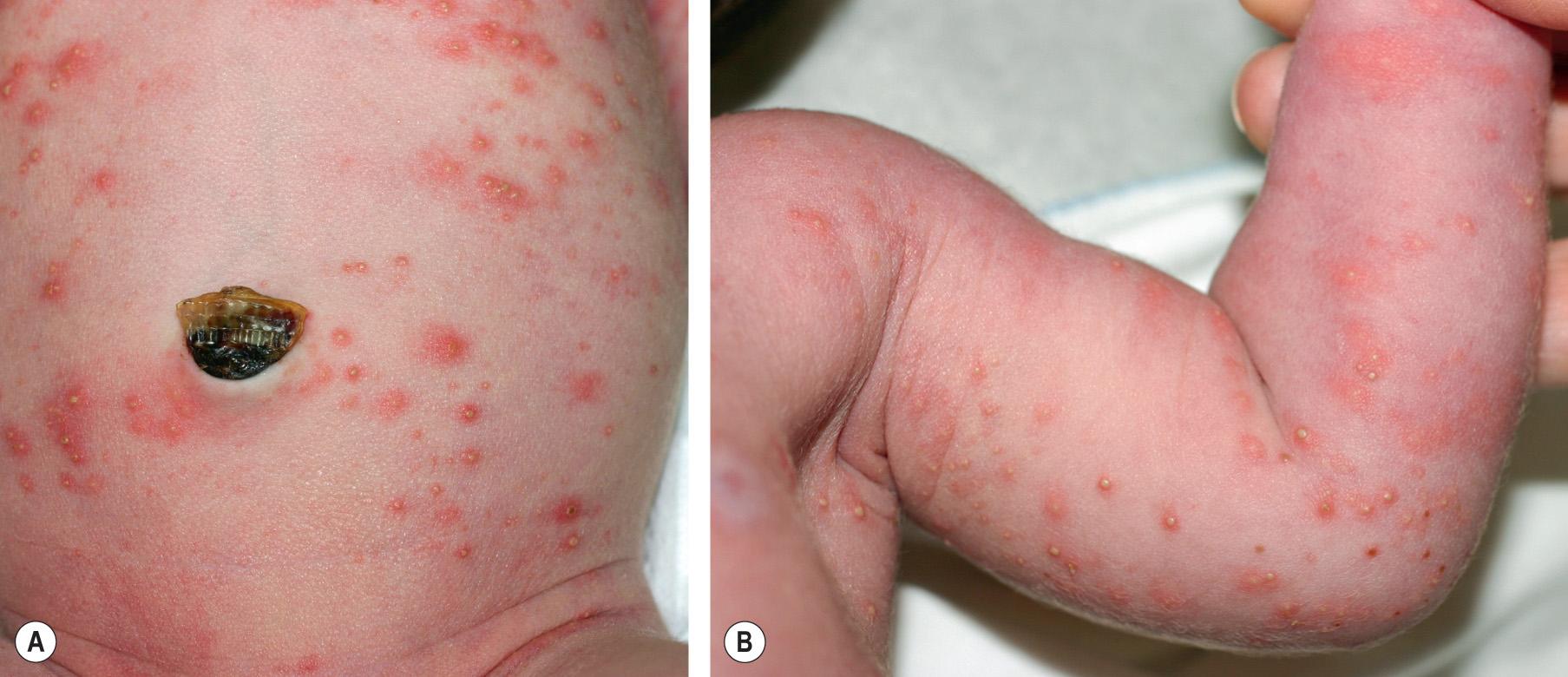
ETN most frequently affects the trunk but can also involve the face, buttocks, and proximal extremities; it almost always spares the palms and soles . Individual lesions rarely persist for more than a day, are asymptomatic, and require no therapy.
The diagnosis of ETN is usually made on clinical grounds. When necessary, the demonstration of numerous eosinophils via Wright stain of pustular contents can help to confirm the diagnosis. Peripheral eosinophilia is occasionally present. Histologically, pustules are intrafollicular, subcorneal, or intraepidermal and are composed of eosinophils and occasional neutrophils . An eosinophilic infiltrate may also be present in the upper dermis. The pathogenesis of ETN is unknown, although an inflammatory response to microbial colonization of the follicle in the first days of life has been postulated .
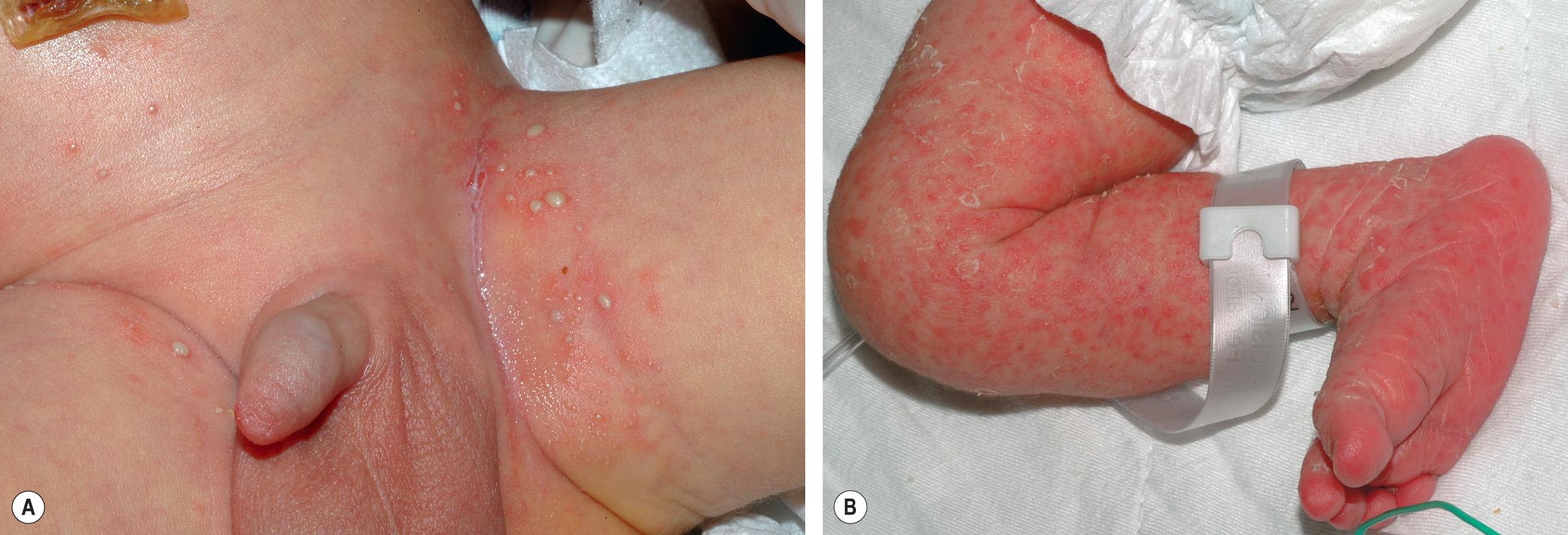
The differential diagnosis includes other pustular disorders of the newborn, some of which may have important medical sequelae (see Table 34.1 ). Transient neonatal pustular melanosis (TNPM) is primarily neutrophilic, present at birth, usually affects infants with darkly pigmented skin, and resolves as pigmented macules that last weeks to months. However, overlap between ETN and TNPM has been observed (see below). Infantile acropustulosis is an acral, rather than truncal, eruption that usually appears after the neonatal period. Eosinophilic pustular folliculitis of infancy favors the scalp and features lesions that often become crusted and recur in crops over a period of months to years. Herpes simplex viral (HSV) infection presents with discrete vesicular lesions with progression to hemorrhagic crusting. Staphylococcus aureus infections manifest as more well-developed superficial pustules ( Fig. 34.3A ) or bullae that rapidly rupture, resulting in an erythematous base with a surrounding collarette. Congenital candidiasis presents with multiple tiny pustules and desquamation, with skin scrapings showing budding yeast and pseudohyphae on potassium hydroxide (KOH) examination ( Fig. 34.3B ). Both staphylococcal and candidal infections result in more persistent lesions that do not undergo the rapid spontaneous resolution characteristic of ETN. Miliaria rubra (prickly heat) is usually concentrated on the forehead and upper torso, with somewhat longer-lived lesions that lack a prominent erythematous flare or eosinophilic infiltrate .
▪ Transient neonatal pustulosis ▪ Lentigines neonatorum
Develops in ~5% of newborns with darkly pigmented skin
Prominent superficial pustules without erythema that rupture, leaving a collarette of scale and hyperpigmentation that may last several months
Sterile subcorneal neutrophilic infiltrate
Favors the forehead, chin, neck, back and shins but may be widespread
Originally described in 1961 as “lentigines neonatorum” , the full spectrum of TNPM was not recognized until 15 years later . It occurs most commonly in black infants and is infrequently seen in Caucasians. Lesions are virtually always present at birth and they have three different morphologies that can coexist or occur sequentially. At delivery, superficial, 2–10 mm vesiculopustules ( Fig. 34.4A ) are present at sites such as the chin, forehead, nape of the neck, lower back, buttocks and shins, and, less often, the remainder of the face, trunk, palms, and soles. These lesions may go undetected, as the fragile intra- and subcorneal pustules can be subtle or wiped away during the first bath. The second phase features slightly hyperpigmented macules with fine collarettes of scale at sites of ruptured and resolving pustules ( Fig. 34.4B ). Finally, residual brown macules may persist for several months. The name “lentigines” is a misnomer, as these lesions result from postinflammatory hyperpigmentation. Sometimes only the brown macules are noted at birth, suggesting that a previous pustular phase occurred in utero . Less commonly, infants with TNPM may present at a few weeks of age with extensive pigmented macules but no history of preceding pustules.
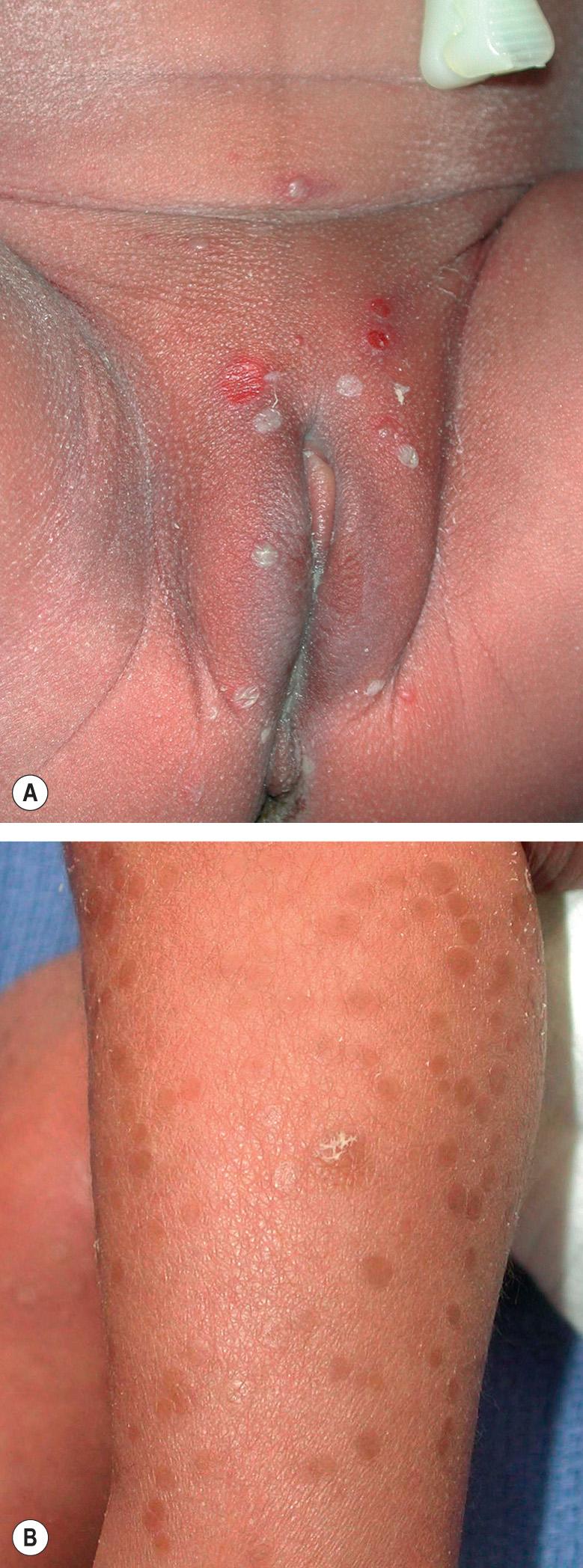
Histologic examination is rarely necessary to make the diagnosis, but when performed on early lesions, it demonstrates intra- or subcorneal pustules containing primarily neutrophils and occasionally some eosinophils . Pigmented macules show increased melanin within basal keratinocytes. The pathogenesis is unknown, and the differential diagnosis is similar to that discussed above for ETN. No treatment is necessary, and parents should be reassured of the benign and self-limited nature of the condition.
It has been postulated that TNPM is simply a variant of ETN, based on their overlapping clinical and histologic features as well as reports of the two conditions coexisting in the same patient. The term sterile transient neonatal pustulosis has been proposed to encompass the ETN–TNPM spectrum . However, most authors consider TNPM and ETN to represent separate entities.
▪ Heat rash ▪ Miliaria crystallina – “dew drops” ▪ Miliaria rubra – prickly heat
Three variants: miliaria crystallina, miliaria rubra and miliaria profunda (rare)
Due to obstruction of eccrine sweat ducts, often in association with overheating, over-swaddling, or a fever
Sites of predilection include the forehead, neck, upper trunk, and occluded areas
Spontaneous resolution with cooling
Prevention consists of avoidance of overheating and limiting excessive swaddling
Miliaria is a common condition that affects up to 15% of newborns, and it is seen more frequently in warm climates. Two types of miliaria (see Ch. 39 ) are typically observed in the newborn period. Miliaria crystallina , sometimes present at birth, is due to obstruction of the eccrine sweat duct as it courses through the stratum corneum. Sweat collects beneath the stratum corneum, causing clear, small, flaccid vesicles that are often likened to “dew drops” ( Fig. 34.5A ). Miliaria rubra , usually seen after the first week of life, is also caused by a blockage of the sweat ducts, but deeper within the spinous layer. The obstructed flow leads to leakage of fluid into the lower epidermis and upper dermis, resulting in an inflammatory response that produces small erythematous papules, papulovesicles, and pustules ( Fig. 34.5B ). Miliaria profunda , a variant caused by even deeper obstruction of the eccrine duct, is very rare in infants.
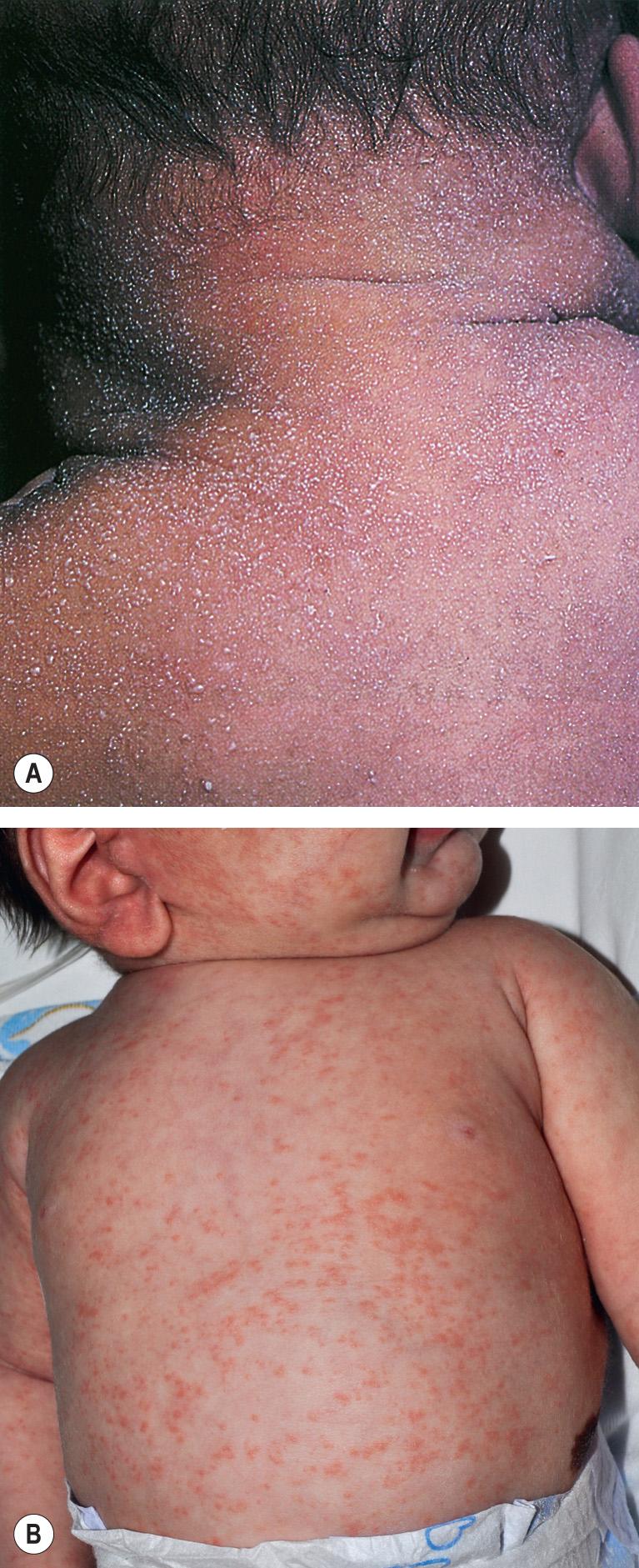
Both miliaria crystallina and miliaria rubra may be seen in relation to excessive warming inside an incubator, over-swaddling, fever, occlusive dressings, or a lack of air conditioning in warm climates. Lesions occur most commonly on the forehead, neck, and upper trunk as well as in areas of skin that have been occluded.
In miliaria crystallina, the characteristic histologic features are subcorneal or intracorneal vesicles that are centered on the acrosyringium with relatively little surrounding inflammation. In miliaria rubra, intraepidermal spongiosis and vesicles are seen along with a chronic inflammatory infiltrate in the dermis . An extracellular polysaccharide produced by Staphylococcus epidermidis present within the sweat duct may play a role , but this does not explain all cases, particularly congenital forms.
Miliaria resolves without treatment. Prevention is accomplished by avoidance of overheating and excessive swaddling when feasible.
▪ Neonatal acne
Onset usually within the first 2–3 weeks of life
Inflammatory non-comedonal pustular eruption, most commonly observed on the cheeks, and less often on the forehead, chin, scalp, and chest
Malassezia spp. have been implicated in its pathogenesis
Spontaneous involution over weeks to months
Historically referred to as “neonatal acne”, neonatal cephalic pustulosis lacks the comedones and more prolonged course that characterize infantile acne vulgaris ( Table 34.3 ; see Ch. 36 ). Neonatal cephalic pustulosis develops within the first 2–3 weeks of life and resolves spontaneously by 3 months of age. Discrete papulopustules overlying variable background erythema are most commonly seen on the cheeks but may also develop on the forehead ( Fig. 34.6 ), chin, eyelids, neck, upper chest, and scalp.
| NEONATAL CEPHALIC PUSTULOSIS VERSUS INFANTILE ACNE | ||
|---|---|---|
| Neonatal cephalic pustulosis | Infantile acne (see Ch. 36 ) | |
| Epidemiology | Common: >20–50% of term infants Male : female = 1 : 1 |
Uncommon Males > females |
| Proposed pathogenic factors | Inflammatory reaction to Malassezia spp. | Androgen production |
| Typical age of onset | ~5 days to 3 weeks | ~6 weeks to 1 year |
| Typical duration | Weeks to 3 months | 6 to 18 months |
| Morphology of lesions | Papules and pustules on an erythematous base | Open and closed comedones, papules, pustules, occasionally nodules |
| Distribution | Cheeks, forehead, eyelids, chin > neck, scalp, upper trunk | Primarily cheeks |
| Scarring | None | Occasionally |
| Treatment | Not usually necessary | More often necessary |
| Topical imidazole or hydrocortisone | Topical retinoids, benzoyl peroxide, antibiotics; occasionally oral antibiotics or retinoids | |
| Associated disease | Occasionally seborrheic dermatitis | Rarely an endocrine disorder, but need to consider when severe or other signs/symptoms (e.g. virilization, abnormal growth) Increased likelihood of severe adolescent acne |
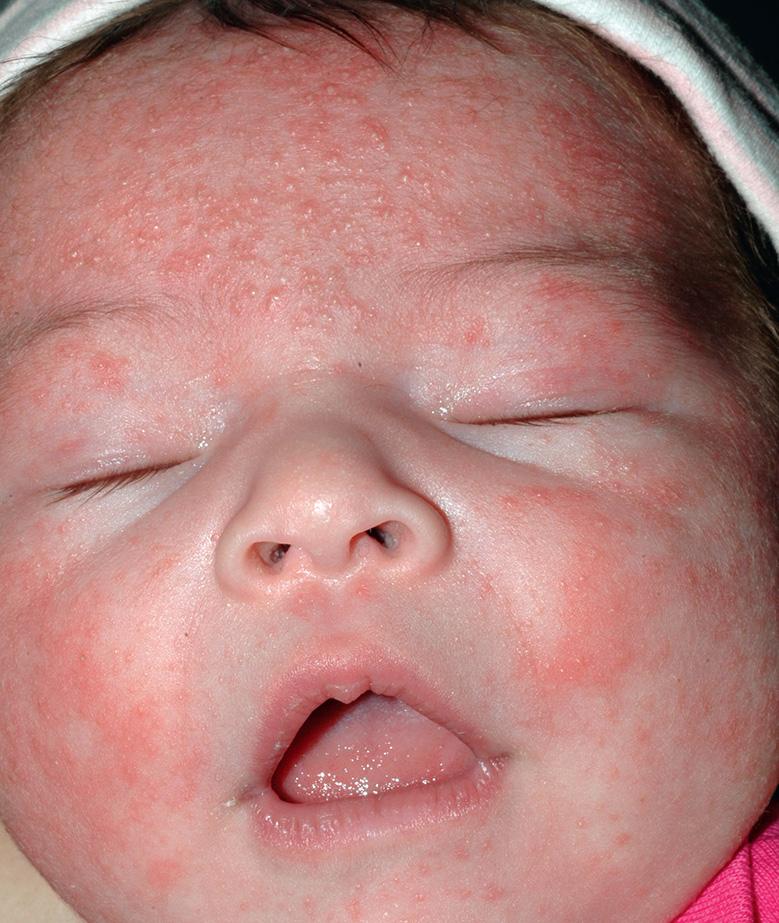
The results of some studies have suggested that an inflammatory response to Malassezia (Pityrosporum) spp. such as M. furfur, M. sympodialis , and M. globosa may be involved in the pathogenesis of neonatal cephalic pustulosis . However, other studies have found no correlation between colonization of neonatal skin with Malassezia spp. and the presence or severity of neonatal cephalic pustulosis . While these yeasts have been cultured from lesional skin in affected patients, they are part of the normal skin flora in humans, and thus their exact role in the condition remains uncertain.
A Giemsa-stained smear of pustular contents may demonstrate yeast forms, neutrophils, and other inflammatory cells; culture of Malassezia spp. requires specialized media . The eruption can be treated with a topical imidazole or hydrocortisone, but this is not usually necessary .
Neonatal candidiasis is a common mucocutaneous infection acquired during delivery or postnatally, whereas congenital candidiasis is a relatively uncommon infection acquired in utero
Neonatal candidiasis presents after the first week of life and is characterized by pink–red patches with satellite papules and pustules that favor the diaper area, other intertriginous sites, and face; oral thrush is often present
When congenital candidiasis occurs in term neonates, it is characterized by a widespread vesiculopustular eruption, often involving the palms and soles
In very-low-birth-weight premature newborns, cutaneous candidiasis may present as “burn-like” erythema with desquamation and erosions
First-line therapy consists of topical anti-yeast medications (e.g. imidazoles), unless there is concern for systemic infection
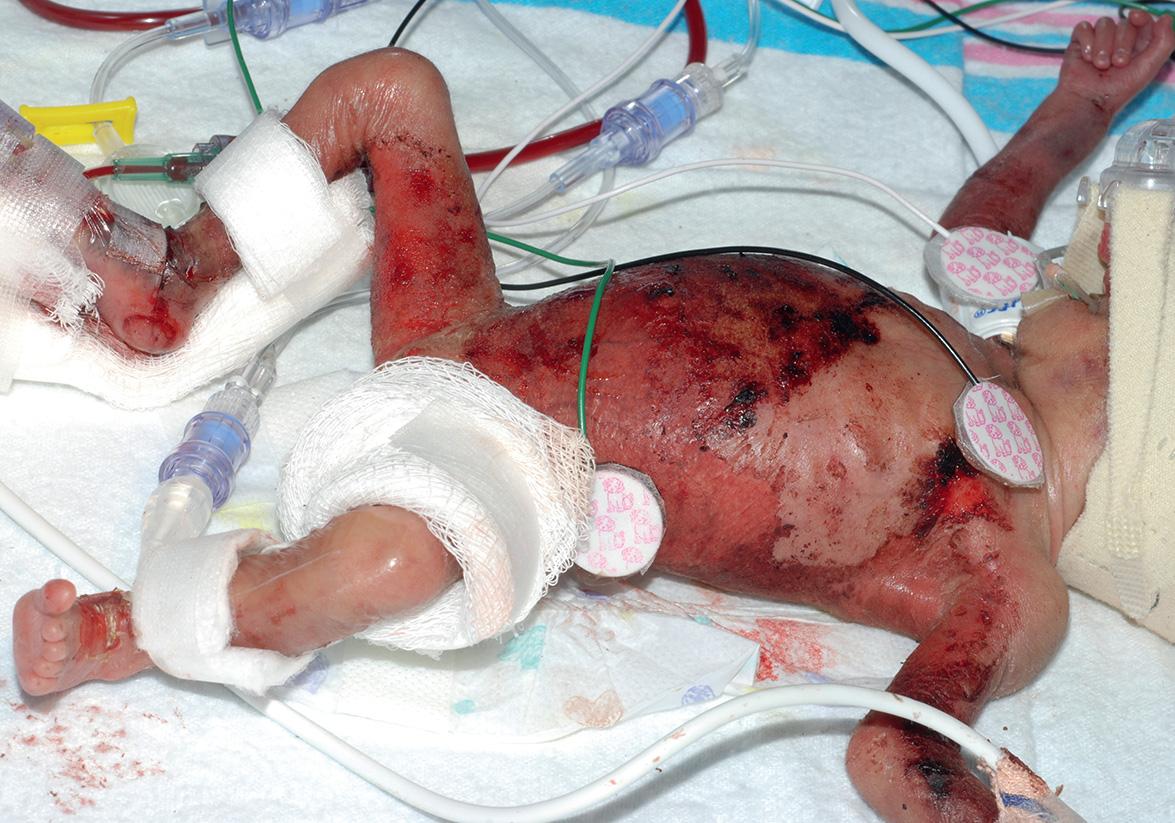
Neonatal candidiasis is common and acquired during delivery or postnatally, whereas congenital candidiasis is uncommon and acquired in utero . In addition, premature newborns with a very low birth weight (often <1000 g) who develop cutaneous candidiasis prenatally or during the first two weeks of life can develop an “ invasive fungal dermatitis ” with a high risk of systemic involvement.
Neonatal candidiasis presents after the first week of life and involves primarily the diaper area and oral mucosa . In some patients, other intertriginous areas and the face may be affected. Pink–red patches with satellite papules and pustules are the characteristic findings; pustules and scaling may also be present at the peripheral border.
In contrast, the relatively uncommon congenital candidiasis presents with a more widespread eruption, usually evident at birth but sometimes appearing as late as the 6 th day of life. Risk factors include a foreign body in the uterus or cervix (e.g. retained intrauterine device, cervical cerclage), premature delivery, and a maternal history of vaginal candidiasis . Skin lesions occur on the face, trunk, and extremities; the palms and soles are often involved, but the diaper area and oral mucosa are usually spared. Cutaneous findings vary and can include erythematous papules, vesiculopustules, diffuse erythema, and fine scaling . Typically, erythematous papules appear first, followed by pustules and desquamation (see Fig. 34.3B ). Nail dystrophy with yellowing and transverse ridging may be present and is occasionally the only manifestation.
“ Invasive fungal dermatitis ” in very-low-birth-weight newborns often presents as widespread “burn-like” erythema with desquamation and erosions ( Fig. 34.7 ). Maceration in intertriginous areas may also be present. Additional risk factors include vaginal delivery, systemic corticosteroid administration, and prolonged hyperglycemia. Although Candida albicans is the most frequent pathogen, infections with other Candida spp. and rarely Trichosporon or Aspergillus spp. can have similar clinical findings.
In neonates, the differential diagnosis includes other infections (e.g. staphylococcal, streptococcal, listerial, HSV) as well as miliaria, ETN, and TNPM (see Table 34.1 ). Budding yeast and pseudohyphae in a KOH preparation that includes scale and/or the pustule and its roof points to the diagnosis, which can then be confirmed by culture. In congenital candidiasis, characteristic yellow–white papules may be found on the umbilical cord.
For neonatal candidiasis, a topical anti-yeast medication (e.g. imidazole cream) is usually sufficient. Even if involvement is localized, premature neonates with very low birth weights (<1500 g) should be closely observed, with cultures of blood, urine, and CSF as indicated; treatment with parenteral antifungals should be considered, if there are signs of systemic infection .
Treatment of congenital candidiasis is dependent on the gestational age and weight of the infant. Premature infants (<27 weeks' gestational age and <1500 g) are at greater risk for the development of disseminated systemic candidiasis and therefore should be treated with parenteral antifungal agents after cultures have been obtained from the blood, urine, and CSF . Infants of a more advanced gestational age with no evidence of systemic infection can be treated with topical therapy as discussed for neonatal candidiasis. However, regardless of gestational age, systemic antifungal therapy should be considered for infants with congenital candidiasis who have respiratory distress, an elevated white blood cell count with a left shift, or other signs of systemic infection .
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here