Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Venous thromboembolism (VTE), which includes both deep venous thrombosis (DVT) and pulmonary embolism (PE), is very common. Although the etiology of acute venous thrombosis (VT) is defined by Virchow triad of hypercoagulability, stasis, and endothelial injury, it is clearly a multifactorial process influenced by many other conditions. The acute thrombotic process must be managed appropriately to relieve edema and pain, to prevent death from PE, and to avoid reoccurrence and the long-term sequelae of postthrombotic syndrome (PTS), which is estimated to occur in 30% of patients older than 8 years, although it may be higher, especially with iliofemoral DVT. In addition to compression and ambulation, there are a number of pharmacologic treatment options: unfractionated heparin (UFH), low-molecular-weight heparin (LMWH), and vitamin K antagonists (VKAs), such as warfarin (Coumadin). Many new and alternative agents are becoming available and may require less rigorous monitoring. Length of anticoagulation typically ranges from 3 months for a first-time event, although certain conditions may warrant lifelong therapy. In other cases, surgical placement of inferior vena cava (IVC) filters may be indicated to prevent further propagation of PE. The present chapter is divided into sections encompassing discussions on all of these topics, including pathophysiology of VT, epidemiology of thrombosis, biomarkers, risk factors, anticoagulants (including the new agents), length of anticoagulation for VTE treatment, thrombolysis and vein wall abnormalities after DVT, diagnosis and treatment of superficial thrombophlebitis, and a discussion about inferior vena caval interruption.
VT is governed by the three principles of the Virchow triad: hypercoagulability, stasis, and endothelial injury. Activation of coagulation is critical in the pathogenesis of DVT. Although the hemostatic system is continuously active, thrombus formation is ordinarily confined to sites of local injury by a precise balance between activators and inhibitors of coagulation and fibrinolysis. A prothrombotic state may result either from imbalances in the regulatory and inhibitory systems or from activation exceeding antithrombotic capacity. Regardless of etiology, most venous thrombi originate in areas of static, low blood flow, often behind valve pockets. Furthermore, many risk factors for acute DVT are associated with immobilization and slow venous flow, and these mechanisms have been advanced to explain the role of stasis in thrombogenesis. For example, in comparison to pulsatile flow, static streamline flow is associated with significant hypoxia at the depths of the venous valve cusps and may induce endothelial injury. Under basal conditions, the endothelium provides a vasodilatory and local fibrinolytic environment via plasminogen activators, such as tissue plasminogen activator and urokinase plasminogen activator. In this setting, coagulation, platelet adhesion, platelet activation, inflammation, and leukocyte activation are inhibited. After a disturbance of the endothelium, a prothrombotic and proinflammatory state occurs and is defined by vasoconstriction, activated platelets, and upregulation of cellular mediators to recruit inflammatory cells to the site of endothelial damage. Indeed, experimental evidence suggests that platelets may be involved with early VT.
Individually, stasis, endothelial injury, and activated coagulation factors may be insufficient to provoke thrombosis. However, when localized in regions of stasis and incited by endothelial disruption, the coagulation cascade allows activated factors to rapidly intensify the thrombotic stimulus, leading to platelet aggregation, coagulation factor localization, and fibrin formation. Thus thrombosis appears to be a multifactorial phenomenon, with convergence of several pathologic factors often required to produce a thrombotic event. A good example of this clinically is the postoperative patient who suffers an infectious complication.
The pathophysiology of VT is a cyclic event, amplified by the associated inflammatory process. With inflammation, there is an increase in tissue factor, platelet reactivity, fibrinogen, phosphatidylserine expression on membrane surfaces, and PAI-1 (inhibiting fibrinolysis) and a decrease in thrombomodulin (and thus a decrease in the activity of protein C). Cell adhesion molecules, such as selectins, allow leukocyte transmigration and are intimately involved in this process. Venous stasis and ischemia result in upregulation of P-selectin, and this localizes microparticles, which are prothrombotic, to the area of evolving thrombosis. The role of the inflammatory cell adhesion molecule P-selectin in thrombosis is critical. Data suggest that elevated levels of soluble P-selectin combined with a clinical examination favoring DVT has a positive predictive value for diagnosing DVT far exceeding that of D dimer. More in-depth reviews highlight this interesting area for research.
The incidence of recurrent, fatal, and nonfatal VTE has been estimated to be 300,000 to 600,000 cases annually in the United States. VTE has been estimated to occur with an incidence of approximately 1 per 1000 adult patients annually. This figure is supported by the 35-year population-based Rochester Epidemiology Project database of Olmstead County, Minnesota, which demonstrated an overall average age- and sex-adjusted annual incidence of VTE of 122 per 1,000,000 person-years. This landmark study also demonstrated higher age-adjusted rates in men compared with women (134 vs. 115 per 100,000 patients, respectively). First-time, or incident, VTE cases are estimated to occur in approximately 250,000 individuals annually in the United States. However, there is a difference in ethnic groups diagnosed with VTE. African Americans demonstrate greater than a 25% increased incidence in first-time VTE compared with whites (103 vs. 141 per 100,000 patients). Conversely, the incidence of first-time VTE in both Hispanic and Asian or Pacific Islanders is significantly lower than in whites (62 vs. 104 per 100,000 patients; 29 vs. 104 per 100,000 patients, respectively). Nevertheless, the issue of VTE is not isolated to the United States. Recent estimates across the European Union have totaled nearly 700,000 cases of DVT, 435,000 cases of PE, and 543,000 fatalities.
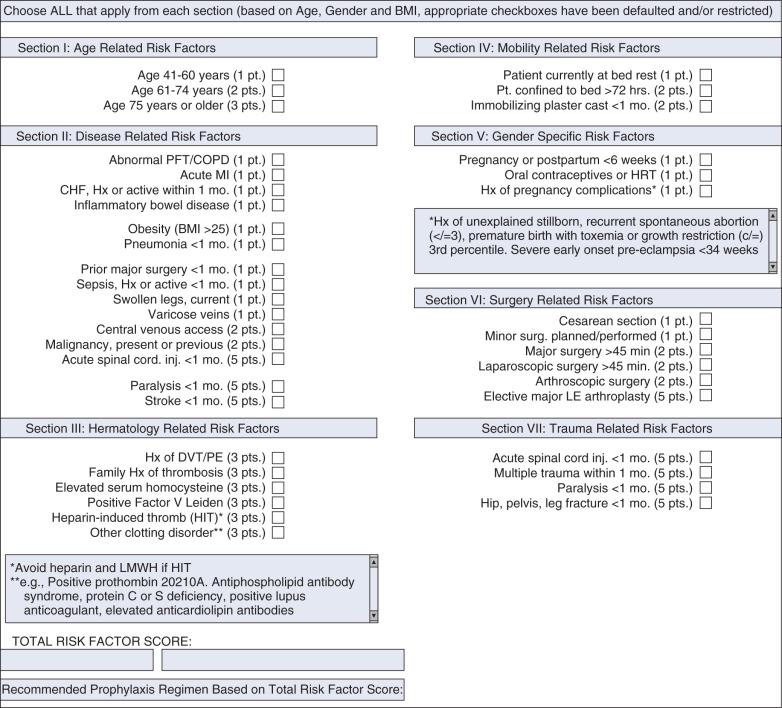
Although estimated incidences of VTE in both the community and hospital have been well documented, individuals at risk for VTE have been less well studied. Substantial differences have been noted in the distribution of risk factors between inpatients and outpatients. Extrapolating hospital inpatient data on the estimated 38 million patients discharged in 2003, 31% were considered to be at risk for VTE secondary to either major surgery or a medical illness. Although the incidence of VTE varies with the population studied, use of thromboprophylaxis, the intensity of screening, and the diagnostic test used, specific risk factors have been significantly associated with increasing VTE. Patients at highest risk are exemplified in the critically ill and trauma patients in whom bleeding risks prohibit thromboprophylaxis, and immobility contribute up to a greater than 80% DVT rate in specific populations. Risk stratification for VTE based on a number of individualized patient-specific clinical attributes was initially proposed by Caprini. In a large, single-center validation study, a modified Caprini risk assessment tool was applied to patients undergoing general surgery, associating higher scores with an increased incidence of VTE. Currently, electronic order entry to mandate a modified Caprini risk assessment tool is recommended for all hospitalized patients ( Fig. 49.1 ). Recognition of patients at high risk for VTE is a critical part in its prevention. In the next section, risk factors for VTE will be discussed in detail.
The “gold standard” for diagnosis of DVT remains duplex ultrasound. However, there has been research directed toward the development of a simple, reliable, and accurate serum biomarker for the presence of DVT. Given that ultrasound can be both operator dependent and, in some institutions, unavailable at off-hours, a serum biomarker that speeds and simplifies diagnosis would be of great benefit to patients; moreover, the ability to obviate the need for ultrasound would represent a significant cost savings to the health care system.
Currently, the most well-validated and widely used serum biomarker is D dimer, a fibrin-degradation product. D dimer is evaluated by a blood test using one of a variety of enzyme-linked immunosorbent assays (ELISAs). Pretest probability of VTE is determined based on a clinical score, most commonly the Wells score. VTE can be excluded safely in patients in whom there is a low pretest probability and a negative D dimer. When combined with Wells score, D dimer performs excellently in a number of patient populations, including outpatients, emergency department patients, pregnant patients, and cancer patients. Concerns remain regarding the specificity of the test, which can be elevated in numerous other states, such as postoperatively and in postpartum individuals; these states also represent risk factors for VTE. D dimer appears to be less effective for diagnosis in certain patient populations, including the elderly and orthopedic surgery patients. Age adjustment of cutoff values may increase the diagnostic performance of the test in elderly individuals.
Soluble P-selectin, a cell adhesion and signaling molecule released with endothelial injury, has been evaluated as a biomarker for DVT. Elevated P-selectin is associated with increased risk of VTE in cancer patients. Vandy and colleagues demonstrated good rule-in performance for soluble P-selectin, with a positive predictive value of 91%—significantly more accurate at ruling in than D dimer with a positive predictive value of 69%. Although not yet widely available clinically, the literature suggests that a combination of Wells score, D dimer, and P-selectin may provide the greatest diagnostic performance by alleviating the low specificity of D dimer alone.
A higher incidence of VTE has consistently been associated with advanced age. In a community-based study of phlebographically documented DVT, the yearly incidence of DVT increased progressively from almost 0 in childhood to 7.65 cases per 1000 in men and 8.22 cases per 1000 in women by age 80. Similarly, in a more recent study reevaluating the Framingham Heart Cohort, age was significantly associated with VTE, with a hazard ratio per increase in 10 years of age of 1.69 (95% confidence interval [CI] 1.48 to 1.92).
Immobilization promotes venous stasis and is a major risk factor for VTE. The prevalence of lower extremity DVT in autopsy studies parallels the duration of bed rest, with DVT occurring in 15% of patients dying after 0 to 7 days of bed rest as opposed to 79% to 94% of those dying after 2 to 12 weeks. Preoperative immobilization is associated with postoperative DVT, contributing a twofold higher risk. Patients with extremity paresis have a threefold higher risk for DVT and PE independent of hospital confinement, and with no significant differences in platelet counts, fibrinogen levels, von Willebrand factor (vWF), platelet factor-4, or beta thromboglobulin levels between patients with or without DVT—signifying immobilization is the major contributing factor.
Despite the questionable importance of prolonged travel as a risk factor for VTE, in 2001 the World Health Organization acknowledged an association between air travel and VTE. Lapostolle and coworkers observed that over an 86-month period, 56 of 135.3 million airline passengers had severe PE. The frequency of PE in those who traveled more than 5000 km was 150-fold higher than those who traveled less than 5000 km. Paganin and associates observed a high incidence of VTE in patients with risk factors for DVT who traveled long distances. These investigators concluded that low mobility during flight was a modifiable risk factor for PE and that those with additional risk factors should increase their mobility. Most recent estimates stipulate a risk of DVT of 1/600 for flights longer than 4 hours and 1/500 for flights longer than 12 hours.
Approximately 23% to 26% of patients with acute DVT have a previous history of DVT, and histologic studies confirm that acute thrombi are often associated with fibrous remnants of previous thrombi in the same or nearby veins. Depending on sex and age, population-based studies have demonstrated that recurrent thromboembolism develops in 1 of every 11 to 50 persons with a previous episode of thromboembolism, with the risk for recurrent thromboembolism being higher in patients with idiopathic DVT.
The evidence supporting obesity as a risk factor for DVT has been controversial. In postmenopausal women, a body mass index of greater than 25 to 30 kg/m 2 has been associated with significantly increased risk. Some investigators have reported obesity to be associated with a twofold greater risk for postoperative DVT, but multifactorial analysis by others has not shown obesity to constitute an independent risk. However, more recently a review of the Framingham Heart cohort revealed in multivariate analysis a body mass index [BMI] ≥ 30 afforded a hazard ratio of 1.88 (95% CI 1.44 to 2.45) for VTE. Similarly, in other studies, obesity has been found to be a risk factor for recurrent DVT.
Approximately 20% of all first-time VTE events are associated with malignancy. Either DVT or PE will develop in an estimated 1 in 200 individuals with malignancy, a fourfold higher risk than in those without malignancy. Considering all-cause in-hospital mortality for cancer patients, one in seven will die of PE. Correlation with location of the tumor reveals that the highest rates of VTE are associated with pancreatic malignancies, followed by kidney, ovary, lung, and stomach malignancies. Venous compression secondary to tumor growth, cancer-associated thrombocytosis, immobility, indwelling central lines, and chemotherapy or radiation therapy are all risk factors that increase the likelihood of developing VTE. Furthermore, as many as 90% of patients with cancer have abnormal coagulation parameters, including increased levels of coagulation factors, elevated fibrinogen or fibrin degradation products, thrombocytosis, and elevated levels of circulating prothrombotic microparticles.
All components of the Virchow triad may be present in surgical patients: perioperative immobilization, transient changes in coagulation and fibrinolysis, and the potential for gross venous injury. In addition, surgery constitutes a spectrum of risk that is influenced by patient age, coexistent thrombotic risk factors, type of procedure, extent of surgical trauma, length of the procedure, and duration of postoperative immobilization. Approximately half of postoperative lower extremity thrombi detected by 125 I-labeled fibrinogen scanning develop in the operating room, and the remainder occur over the next 3 to 5 days. However, the risk for development of DVT does not end uniformly at hospital discharge. In one study, 51% of the thromboembolic events that occurred in patients undergoing gynecologic surgical procedures occurred after initial discharge. One of the most at risk groups for VTE are those patients undergoing bariatric surgery, in which VTE was identified in 1% to 6% of patients, with a mean time to diagnosis of 24 days post operation. Consequently, many bariatric surgery programs have begun prophylactic LMWH administration for the first month following the procedure.
Despite improvements in trauma care and thromboembolism prophylaxis, DVT remains a significant source of morbidity and mortality in injured patients. In modern venographic series the incidence of DVT in the trauma patient is as high as 58%. Recent trauma was the second most common risk factor for thromboembolism in the Olmsted County study by Heit and associates and was associated with a nearly 13-fold increase in risk. As with postoperative DVT, several pathophysiologic elements may be responsible for the high incidence of DVT in trauma patients. Immobilization by skeletal fixation, paralysis, and critical illness are obviously associated with venous stasis, whereas mechanical injury is important after direct venous trauma and central venous cannulation. Less appreciated is the hypercoagulable state after depletion of coagulation inhibitors and components of the fibrinolytic system. Finally, more recently, acute trauma has also been linked to neutrophil activation with the formation of intravascular neutrophil extracellular traps or “NETs” that have a clear link with activation of the coagulation cascade and VTE.
Resistance to activated protein C was initially described by Dahlbach and associates in 1993. Subsequent studies from the Leiden University Hospital in the Netherlands revealed a mutation in the gene for factor V conferring resistant to cleavage by activated protein C. The factor V Leiden mutation has an autosomal dominant mode of inheritance and is at least tenfold more common than other inheritable defects. The relative risk for first-time DVT is increased sevenfold in those who are heterozygous for the factor V Leiden mutation. However, the relative risk is increased eightyfold in individuals who are homozygous for the mutation.
The prothrombin G20210A mutation, first described by Poort and associates in 1996, was demonstrated in 28 families with a documented history of VTE. The mutation is characterized by a transition from guanine to adenine at the 20210 nucleotide on the prothrombin gene, and the mechanism of increased risk of thrombogenesis is unknown. The risk for VTE in the presence of the prothrombin G20210A mutation is threefold higher in heterozygotes than in wild type, and the presence of homozygosity further increases this risk. Although prothrombin G20210A is rare in individuals of Asian or African descent, additional investigations have shown that in those with spontaneous DVT, the incidence of the mutation may range from 7% to 16%.
Protein C is a vitamin K–dependent serine protease that inhibits the coagulation system by inactivating factors Va and VIIIa. In a study of 10,000 healthy blood donors the observed prevalence of inherited protein C deficiency was 1.45 per 1000. In a study of 2132 patients with VTE, 3.2% were found to have protein C deficiency. It is estimated that individuals who are heterozygous for protein C deficiency have a sevenfold higher risk for the development of VTE than the general population.
Protein S is a vitamin K–dependent cofactor for the protein C–mediated inactivation of factors Va and VIIIa. Inheritance of protein S deficiency is autosomal dominant, and it is more common than protein C deficiency. The incidence of protein S–associated VTE has been reported to be 5% to 7%, with the prevalence in the general population being 0.13%. It is estimated that individuals who are heterozygous for protein S deficiency have an 8.5-fold higher risk for the development of VTE than the general population.
Antithrombin inhibits thrombin as well as factors Xa, IXa, XIa, and XIIa. Antithrombin deficiency is inherited in an autosomal dominant fashion. The majority of patients are heterozygous because homozygote patients usually die in utero. The incidence of VTE associated with antithrombin deficiency has been reported to be 0.5% to 3%, with a prevalence in the general population of 0.2%. However, it is estimated that individuals who are heterozygous for antithrombin deficiency have a twentyfold higher risk for the development of VTE than in the general population.
The incidence of VTE in the pregnant population is fivefold to tenfold greater than in nonpregnant, age-matched controls. VTE accounts for approximately 10% of all maternal deaths. The risk for VTE is highest in the postpartum period, with greatest risk occurring at 3 to 6 weeks post partum and in those patients whom underwent cesarean section. DVT in pregnancy has been attributed to impaired venous outflow secondary to uterine compression, and up to 97% of reported thromboses have been isolated to the left leg. Furthermore, pregnancy is associated with a transient hypercoagulable state because of increases in levels of fibrinogen; vWF; and factors II, VII, VIII, and X.
Case-control and population-based studies have established the use of oral contraceptives as an independent risk factor for the development of DVT. Most studies have reported odds ratios of 3.8 to 11.0 for idiopathic thrombosis, with an unweighted summary relative risk of 2.9 in 18 controlled studies. Approximately one-quarter of idiopathic thromboembolic events in women of childbearing age have been attributed to oral contraceptives. The risk of hospital admission for a thromboembolic event, including cerebral thrombosis, has been estimated to be 0.4 to 0.6 per 1000 for oral contraceptive users versus 0.05 to 0.06 for nonusers. Estrogenic compounds also increase the risk for VTE when used for suppression of lactation, in the treatment of carcinoma of the prostate, and as postmenopausal replacement therapy. Several studies have now reported a twofold to fourfold higher risk in women taking hormone replacement therapy. This increased risk is greatest during the first year of treatment.
The use of peripherally inserted central catheters (PICCs) and centrally inserted central catheters (CICCs) for hemodynamic monitoring, infusion, antibiosis, and supplementation of alimentation have been associated with an increased risk of DVT. This is particularly true for upper-extremity thrombus, with as many as 65% being related to central venous cannulation. Although the incidence of symptomatic thrombosis may be low, studies using objective surveillance have reported thrombosis to occur at a mean incidence of 28% after subclavian cannulation. This risk also extends to femoral venous catheters, where ipsilateral thrombosis develops in 12% of patients undergoing placement of large-bore catheters for trauma resuscitation. In a single institution study of 64,000 admissions, central venous catheters of any type were associated with a 14-fold increased risk of upper-extremity DVT (UEDVT), with PICC lines having a fourfold increased risk of UEDVT compared with CICC lines.
Clinical series have reported VTE to complicate inflammatory bowel disease (IBD) in 3.9% of all cases. Specifically, in one series, with 2284 admitted patients with IBD, 3.4% of Crohn disease (CD) patients developed VTE (2.4% DVT, 1.5% PE), whereas 4.7% of ulcerative colitis (UC) patients developed VTE (3.2% DVT, 2.4% PE). Overall, IBD patients have an approximately threefold increased risk of VTE compared with the general population. Patients with CD have incidence rates of 31.4 and 10.3 per 10,000 person-years for DVT and PE, respectively. Meanwhile, UC patients are reported as having incidence rates of 30.0 and 19.8 per 10,000 person-years for DVT and PE, respectively. Such thrombosis events occur more frequently in patients with active disease and may affect unusual sites such as the cerebral veins. IBD has been linked to intravascular neutrophil activation and subsequent release of NETs, which may explain this systemic prothrombotic diathesis.
In patients with systemic lupus erythematosus (SLE), the incidence rates of DVT, PE, and VTE are estimated at 3.33, 2.58, and 5.32 per 1000 person-years, respectively. In comparison to normal controls, patients with SLE have adjusted hazard ratios for DVT, PE, and VTE of 4.46, 3.04, and 3.55, respectively. Among patients with SLE, those with lupus anticoagulant have a sixfold higher risk for VTE, whereas those with anticardiolipin antibodies have a twofold greater risk. As earlier, SLE has also been linked to systemic neutrophil activation and NET formation, which offers explanation for the increased rates of VTE.
Therapy for DVT is undertaken with the goals of reducing the risk of thrombus extension, thrombus recurrence, and ultimately reducing the risk of PE. Immediate systemic anticoagulation should be administered for proximal DVT (tibial veins and above) because the risk of extension or recurrent venous thromboembolism is significantly increased if anticoagulation is not therapeutic within the first 24 hours. In cases of suspected PE, this may necessitate heparinization before completion of diagnostic testing. Confirmation of DVT with duplex imaging is rapid and, as a result, usually precedes anticoagulation.
Historically, initial systemic anticoagulation has been undertaken with UFH with a loading dose of 80 U/kg and infusion rate adjusted to achieve activated partial thromboplastin time to 2 to 2.5 times normal. However, because of decreased rates of thrombotic events, decreased bleeding complications, decreased mortality (in cancer patients), and improved pharmacokinetic profile, LMWH is preferred over UFH for the initial treatment of acute DVT in most cases. LMWHs are dosed in a weight-based fashion and rarely require monitoring, except in circumstances such as renal failure, pregnancy, and morbid obesity. LMWH can be given subcutaneously once or twice daily with equal efficacy on an outpatient basis. Following initial anticoagulation with UFH or LMWH, most patients are switched to an oral agent for either short-term (3, 6, or 12 months) or indefinite anticoagulation.
There are many LMWHs in clinical use; enoxaparin, dalteparin, and tinzaparin are approved for use in the United States. The class of LMWHs are defined most notably as being a more specific inhibitor to factor Xa than factor IIa, and the average molecular weight of the compound is 30% less than UFH. Clinical trial data have nearly unanimously revealed that LMWHs are as effective if not more effective than UFH, with their safety profile at least equivalent.
A limitation to LMWH use is in those patients with renal dysfunction (creatinine clearance less than 30 mL/min). In these cases, effects are not predictable and safe dosing is difficult because LMWHs have the majority of their elimination by the kidneys. In addition, in an obese patient or another patient whose dosing appears complicated, anti-Xa activity can be measured to titrate therapeutic effect. The half-life of LMWH is longer and as such may not be ideal for a high-risk patient with bleeding concerns, but in most other patient groups the LMWHs are viable and in many cases the current preferred method of anticoagulation for VTE.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here