Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Uterine mesenchymal tumors are neoplasms derived from or differentiating toward mesodermally derived tissues. Differentiation is typically toward normal constituents of the uterine corpus—endometrial stromal and myometrial smooth muscle cells. However, differentiation toward heterologous tissues—that is, mesenchymal tissue not normally present in the uterus (e.g., striated muscle, cartilage, or bone)—also may be seen.
Mixed epithelial and mesenchymal uterine tumors are composed of benign or malignant mesenchymal elements combined with benign or malignant epithelium. The classification of such mixed tumors depends on the morphologic assessment of both components.
The degree of risk for developing mesenchymal tumors is not uniform for all types of uterine mesenchymal neoplasia, and assessment of risk is further hindered by the rarity of most of these tumors. In fact, uterine sarcomas account for only 4% to 9% of uterine malignancies. Moreover, it is estimated that there are only 0.01 to 0.02 cases of uterine sarcoma/1000 women.
Despite their rarity, there have been recent insights into potential risk factors for certain subtypes of uterine mesenchymal neoplasia. Tamoxifen, an antiestrogenic drug widely prescribed for women at risk for breast cancer or used to treat women with breast cancer, is known to increase the risk for uterine carcinomas, presumably because of its agonistic effect at this site. The risk of uterine and endometrial cancer, of which most represent stage I adenocarcinoma, is almost doubled with tamoxifen use; however, this elevated risk appears to be only in women older than 50 years. Use of this drug may also increase a woman's risk for certain subtypes of uterine mesenchymal neoplasia, particularly adenosarcoma and carcinosarcoma. In some cases, there is a long latency period, so that long-term follow-up of all patients on tamoxifen is warranted. Follow-up ultrasound evaluation in patients with clinical symptoms may be useful in stratifying those women who need to undergo sampling of their endometrium. An abnormal ultrasound finding (i.e., thickened endometrial stripe, polyp, or uterine mass) in this group should prompt sampling. The risk for a uterine malignancy (both epithelial and mesenchymal types) associated with raloxifene, a drug currently prescribed to prevent osteoporosis and being tested in women with and at risk for breast cancer, is not clear.
In contrast to malignant mesenchymal tumors, the frequency of benign uterine leiomyomata is extremely high, with estimates of up to 85% of women of reproductive age and older. Consequently, leiomyomata are the most common uterine tumor, and this type is one of the most common tumors to affect women. Although their high frequency may complicate their epidemiologic analysis, more is known about risk factors for the development of uterine leiomyomata than for any other uterine mesenchymal tumor. As will be discussed in greater detail in subsequent sections, there is substantial evidence that genetic factors contribute to the development of leiomyomata. These factors are recognized at the familial and population levels. In particular, the risk of benign and possibly malignant uterine smooth muscle tumors are two- to threefold higher in black women.
Nongenetic (i.e., environmental and genetic by environmental) factors also have been implicated in the development or growth of uterine leiomyomata. The most frequently identified factor, smoking, appears to reduce the clinical frequency of leiomyomata in some but not all studies. Increased parity also is associated with a reduced risk of leiomyomata. Oral contraceptive use has been linked to leiomyomata in most studies. In particular, older age at menarche and early oral contraceptive use may be protective. In contrast, obesity is associated with larger or more symptomatic tumors. Environmental (perineal) exposure to talc increases the risk of leiomyomata by a factor of 2. Finally, increased dietary consumption of soy products, alcohol, and red meats and decreased consumption of green vegetables or foods rich in beta-carotenes have been implicated. Individually, these apparent risk factors for leiomyoma are a confusing hodge-podge. A potentially unifying explanation emphasizes the unopposed estrogen hypothesis invoked in endometrial carcinogenesis. Diet would tie into this hypothesis as a source of environmental estrogens. Another potential explanation is that gravidity alters the propensity for leiomyomata by structural, biochemical, and epigenetic remodeling of the uterus.
In general, most uterine mesenchymal tumors are intramural or intracavitary lesions. Clinical symptoms are related to their size; disruption of the uterine lining, with pelvic pain, pressure, and dysfunctional uterine bleeding, is the most common clinical manifestation. Although size is not a guarantee of a tumor being malignant, in general, most mesenchymal malignancies of the uterine corpus tend to be large (>10 cm) and may have already spread to involve contiguous structures at the time of presentation. Radiographic studies, particularly ultrasound and magnetic resonance imaging (MRI), may be useful in clinically assessing whether a mass is more likely to be benign or malignant. In addition to spread beyond the uterus, radiographic features suggestive of tumor necrosis may be present. Caveats include central infarction and degeneration of benign tumors, such as those that can be seen in large leiomyomata.
Currently, the World Health Organization (WHO) recognizes four main categories of endometrial stromal tumors: (1) endometrial stromal nodule; (2) endometrial stromal sarcoma (ESS), low grade; (3) ESS, high grade; and (4) undifferentiated uterine sarcoma. This categorization represents a significant change from the prior WHO categorization (2002), which had removed high-grade ESS from the lexicon and merits some discussion here to bring clarity to the current scheme.
The 2002 classification reflected terminology first proposed in 1966 by Norris and Taylor in their seminal paper on endometrial stromal neoplasms, although the definitions of these categories have changed. The main difference in the prior classification centered on the definition of low-grade ESS and its separation from what was originally termed high-grade endometrial stromal sarcoma. As originally proposed in 1966, separation into low- and high-grade categories was solely based on mitotic activity—if a tumor that morphologically resembled endometrial stroma had fewer than 10 mitoses/10 high-power fields (hpf), it was considered low grade, whereas a tumor with more than 10 mitoses/hpf was considered high grade. However, since this original publication, the clinical relevance of separating those examples of ESS with morphology similar to normal proliferative phase stroma into low- and high-grade categories based on mitotic activity has not been confirmed in subsequent studies, most notably by Evans and Chang et al. Moreover, the category of high-grade ESS had represented a heterogenous group of tumors, including those that resembled endometrial stroma and those that were poorly differentiated, being composed of larger cells with a greater degree of nuclear anaplasia akin to the mesenchymal component of carcinosarcoma. Therefore, in the 2002 WHO classification scheme, there remained two categories of endometrial sarcoma—low grade and undifferentiated—based on differences in tumor morphology rather than mitotic activity. A low-grade ESS is a tumor composed of cells that morphologically resemble non-neoplastic proliferative phase endometrial stroma that infiltrates the surrounding myometrium in a characteristic finger-like permeative fashion and typically invades lymphatic or vascular spaces. In contrast, an undifferentiated endometrial sarcoma is a poorly differentiated sarcoma composed of cells that do not resemble proliferative phase endometrial stroma, which usually shows destructive infiltration of the myometrium. Despite this attempt at a neat separation into diagnostic categories, it is now clear that there is a subset of ESSs that are histologically higher grade, with distinctive uniform morphology, immunophenotype, and genetic alterations. This is now recognized as high-grade ESS, as will be discussed below.
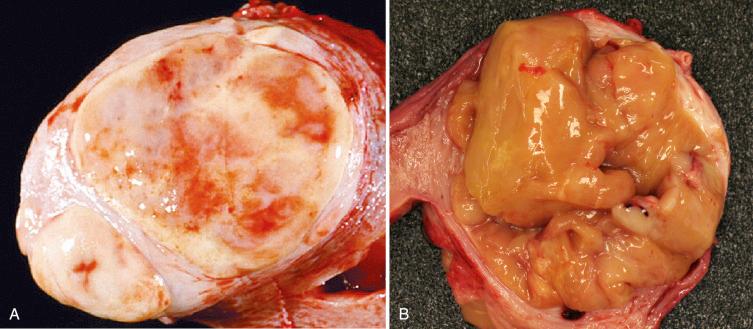
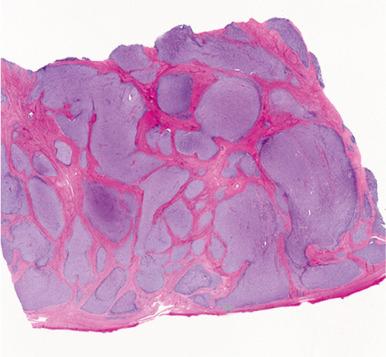
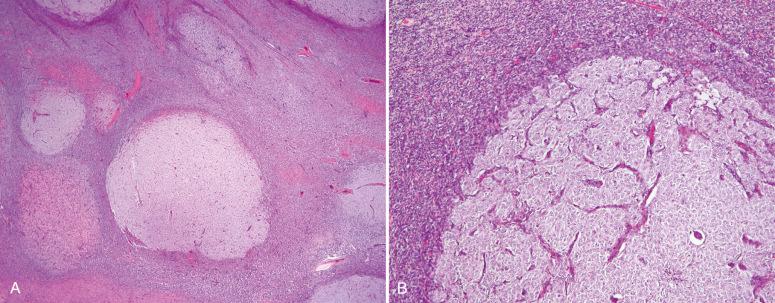
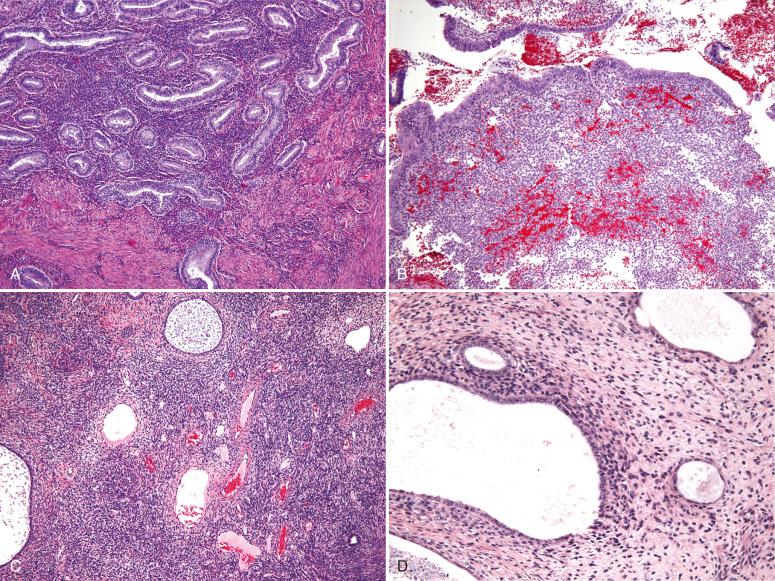
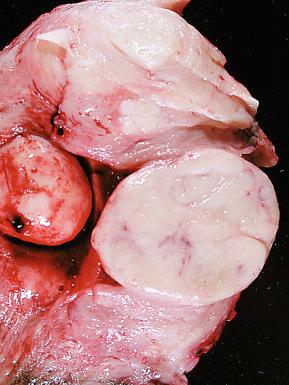
Endometrial stromal nodules may occur at any age but are typically encountered in women in their fifth and sixth decades. They may be located intramurally, with no connection to the endometrial surface (raising interesting possibilities concerning histogenesis and a shared developmental origin of uterine stroma and smooth muscle), may be located submucosally at the endomyometrial junction, or may project into the endometrial cavity as a polypoid mass. On gross examination, they are characteristically well circumscribed and may be mistaken for a leiomyoma; however, endometrial stromal nodules are usually softer in consistency, tend more commonly to be yellow, and lack the characteristic whorled bulging appearance of a typical leiomyoma ( Fig. 20.1 ). Hemorrhage and cystic degeneration may be seen (see Fig. 20.1B ). Endometrial stromal nodules can vary in size but are usually less than 10 cm, although larger tumors have been described.
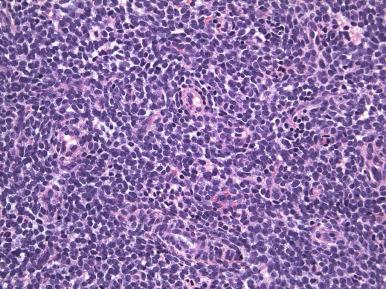
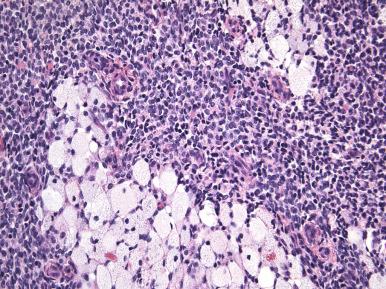
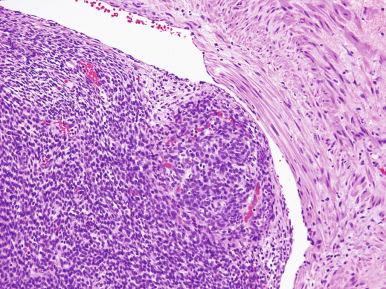
Histologically, an endometrial stromal nodule is characterized by sharp circumscription between the neoplastic cells and surrounding endometrium or myometrium, with a pushing interface ( Fig. 20.2 ). Sometimes, however, there may be some irregularities in the border in the form of small lobulated or finger-like extensions into surrounding myometrium. By definition, no lymphatic or vascular invasion is present, and these foci should be less than three in number and less than 3 mm from the main mass. Tumors with more than three foci or with extension more than 3 mm from the main mass are considered as low-grade ESS with limited infiltration (see below). The neoplastic cells of endometrial stromal nodules resemble proliferative phase endometrial stroma, being composed of cells with uniform round to ovoid nuclei that have scant to moderate amounts of eosinophilic to amphophilic cytoplasm ( Fig. 20.3 ). These cells appear to whorl around the prominent vascular component, which resembles the spiral arterioles of non-neoplastic endometrium; foamy stromal cells may be conspicuous ( Fig. 20.4 ). The vessels are typically evenly spaced and uniform in caliber throughout the neoplasm; however, on occasion, larger, thick-walled vessels may be present, although this is usually only a focal finding in a minority of cases.

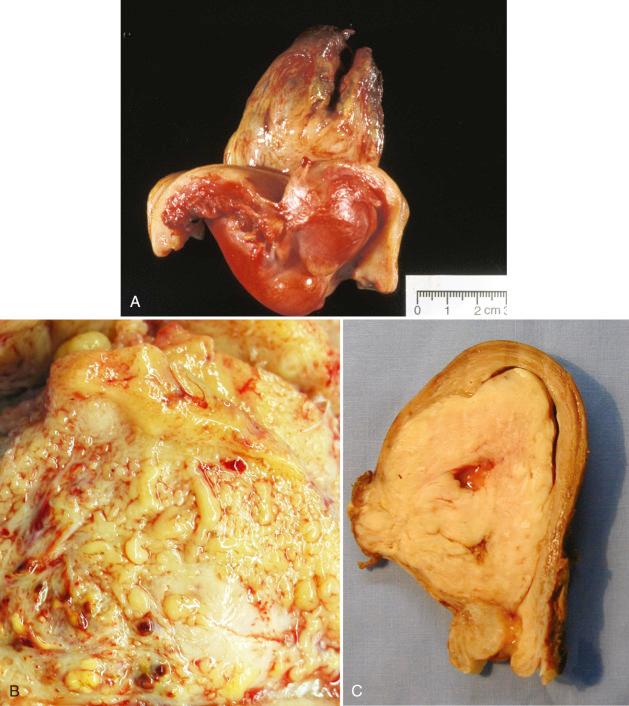
Women with low-grade ESS are typically perimenopausal but tend to be younger than those who present with other types of uterine sarcoma. They usually present with abnormal uterine bleeding or symptoms related to a uterine mass or extrauterine spread. The gross appearance can be variable, although a common feature is its tan to yellow color and soft consistency. Tumors may present as the following: (1) intramyometrial nodular masses; (2) an intracavitary polypoid mass; (3) diffuse myometrial infiltration, with expansion of the uterine wall; or (4) any combination of these patterns ( Fig. 20.5 ). Involvement of parametrial vessels may impart a wormlike appearance on gross examination.
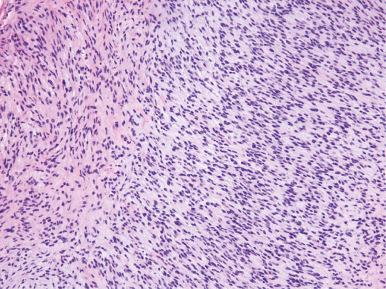
Histologically, the neoplastic cells of low-grade ESS are morphologically indistinguishable from those of an endometrial stromal nodule; they are composed of small, bland-appearing cells resembling proliferative phase endometrial stroma. The mitotic rate is typically low (usually <5 mitoses/10 hpf), although it can be variable and sometimes brisk; necrosis is uncommon. A diagnosis of sarcoma is based on the presence of extensive myometrial infiltration ( Fig. 20.6 ) or invasion of lymphatic or vascular spaces ( Fig. 20.7 ). Characteristically, the neoplastic cells invade as rounded to irregularly shaped nests of varying size, which have been described as a finger-like permeation of the surrounding myometrium.
Endometrial stromal nodules and low-grade ESS, can occasionally exhibit a diverse range of differentiation including sex cord–like, epithelial (e.g., endometrioid type glands), smooth muscle, and, rarely, skeletal muscle differentiation. A fibroblastic appearance due to extensive stromal hyalinization secondary to increased collagenous matrix production may sometimes occur, making recognition as a stromal neoplasm difficult ( Fig. 20.8 ). However, in general, other areas of morphologically typical stromal neoplasia are usually present. In addition, the characteristic vascular component and arrangement of the tumor cells around it are maintained. Other less common variants include features such as epithelioid differentiation, tumor cells with clear or granular cytoplasm, bizarre cells, pseudopapillary growth, and adipose tissue.
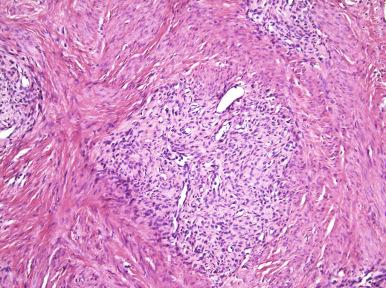
Endometrial stromal neoplasms with smooth muscle differentiation have been the focus of a limited number of studies, partly because of underrecognition and their uncommon occurrence. If more than 30% of the tumor is comprised of smooth muscle, they are designated as mixed endometrial stromal–smooth muscle tumors. Histologic features of smooth muscle differentiation include the following: (1) typical smooth muscle morphology reminiscent of that seen in leiomyomata; (2) nodules with central prominent hyalinization (the so-called starburst pattern) ( Fig. 20.9 ); and (3) irregular islands that can be discrete or merge imperceptibly with the areas characteristic of stromal differentiation ( Figs. 20.10 and 20.11 ). These tumors are of endometrial stromal derivation (see later, “ Molecular Genetics ”); therefore, determination of benign versus malignant should be made using the criteria for endometrial stromal tumors (e.g., presence or absence of myometrial invasion, lymphatic or vascular invasion).
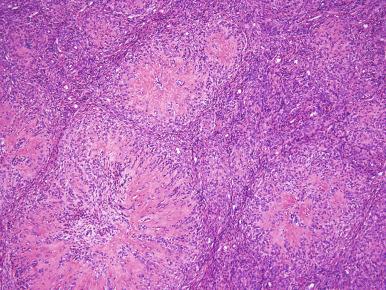
Endometrial stromal nodules and low-grade ESS may exhibit morphologic features of ovarian sex cord stromal tumors. These elements may be present focally, most often in the form of interanastomosing trabeculae ( Fig. 20.12 ), cords and, less commonly, tubules. They may also be the predominant pattern in which the stromal element is less conspicuous or absent; in this scenario, the term uterine tumor resembling ovarian sex cord tumor (UTROSCT) has been used. UTROSCTs show striking morphologic overlap with ovarian sex cord tumors mimicking granulosa cell, Sertoli cell, and even retiform Sertoli cell tumors ( Fig. 20.13 ). Whether these tumors represent endometrial stromal neoplasms has been a subject of debate, although molecular evidence has suggested that they may not be of endometrial stromal derivation (see later, “ Molecular Genetics ”). In addition to the morphologic resemblance, the sex cord–like elements are often positive for markers of sex cord differentiation, such as inhibin and O-13/CD99, suggesting that these foci represent true sex cord differentiation akin to that seen in the ovary.
The presence of sex cord–like differentiation in an endometrial stromal nodule and low-grade ESS does not appear to have an impact on biologic behavior. Histopathologic criteria for the distinction between a stromal nodule and stromal sarcoma, as outlined previously, are applied, regardless of the presence of sex cord–like elements. Less is known concerning the natural biologic behavior of tumors that show exclusive sex cord–like differentiation (UTROSCTs). Although evidence has suggested that they are benign—they are generally well-circumscribed, yellow, gray, or tan masses, and there have been no reported incidences of metastasis —the outcome in patients treated conservatively is not known, because all patients typically have undergone hysterectomy. We have also seen rare examples of UTROSCT with worrisome histologic features, including necrosis and brisk mitotic rate; in such cases, the biologic behavior should not be presumed to be benign but rather best considered uncertain.
Divergent differentiation down epithelial lines also may be occasionally seen in endometrial stromal nodule or low-grade ESS, usually as well-formed endometrioid glands ( Fig. 20.14 ). Although divergent differentiation is the most likely explanation for their presence, some examples may represent entrapped non-neoplastic endometrial glands. In general, this type of differentiation is focal, and the main differential diagnosis is the distinction from adenomyosis, particularly adenomyosis with sparse glands. The latter is usually encountered as an incidental finding in postmenopausal women and can be distinguished from low-grade ESS primarily based on the atrophic appearance of the stroma and the absence of a grossly identifiable mass.
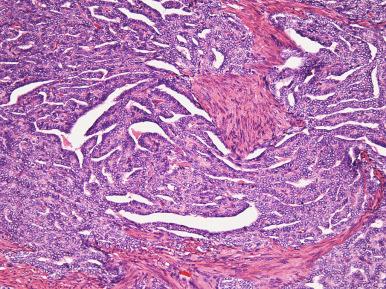
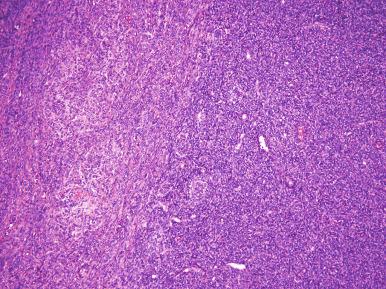
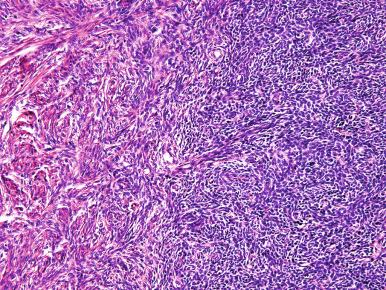
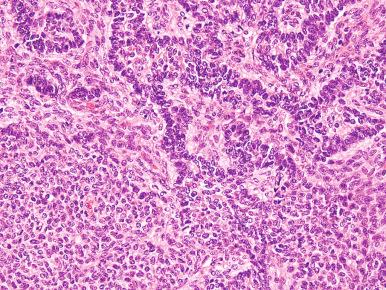
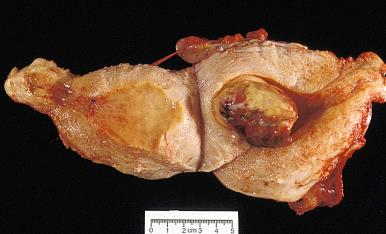
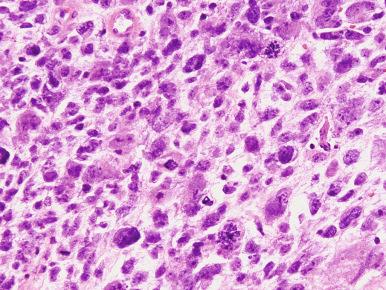
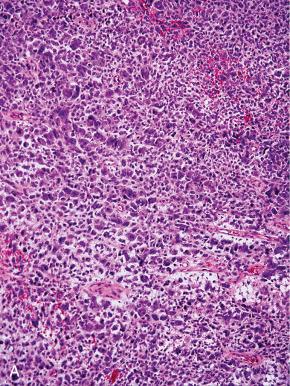
High-grade ESS is a recently described subtype of endometrial stromal neoplasia that is recognized by its distinctive morphologic appearance, immunohistochemical profile, and molecular genetic abnormality. These uncommon tumors occur in patients over a wide age range (mean, 50 years). Most patients present with abnormal uterine bleeding and/or symptoms related to extrauterine extension because these are often high-stage tumors. On gross examination, they typically form intracavitary polypoid masses, often with hemorrhage and necrosis ( Fig. 20.15A ), are poorly circumscribed, and show extensive permeation of the uterine wall, often with extension to the serosa (see Fig. 20.15B ). Histologically, the tumor shows extensive finger-like permeation of the myometrium and lymphovascular invasion ( Fig. 20.16 ); however, destructive infiltration with splaying of individual muscle fibers is also commonly seen ( Fig. 20.17 ). The tumor typically contains morphologically low- and high-grade areas, which can sometimes be appreciated on low-power examination as hypercellular and hypocellular areas ( Fig. 20.18A ); these areas can be juxtaposed with transition from a low- to high-grade area or intermingled (see Fig. 20.18B ). The morphologically low-grade areas can have a fibroblastic or myxoid appearance, similar to the fibrous and myxoid variants of low-grade ESS, and are composed of cells with round to oval nuclei and scant cytoplasm ( Fig. 20.19 ). The morphologically high-grade areas typically show nested and corded growth and are composed of a uniform population of histologically higher grade epithelioid tumor cells, with scant to moderate amounts of eosinophilic cytoplasm and round to oval vesicular nuclei with irregular nuclear contours and nucleoli, but without nuclear pleomorphism notable at low-power magnification (4–10× objective; Fig. 20.20 ). As a point of reference, the nuclei are typically four to six times larger than a lymphocyte. There is a prominent delicate arborizing capillary network in which small concentric arterioles are only present occasionally. Mitotic activity is brisk, typically more than 10 mitoses/10 hpf.
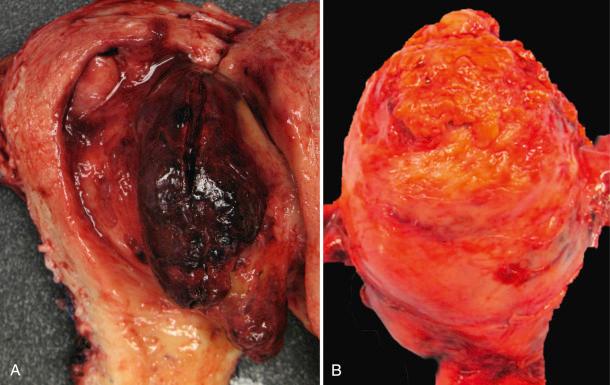
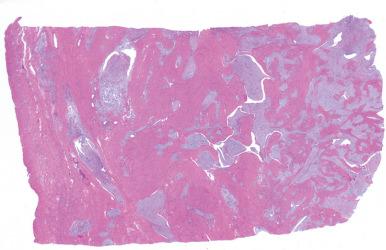
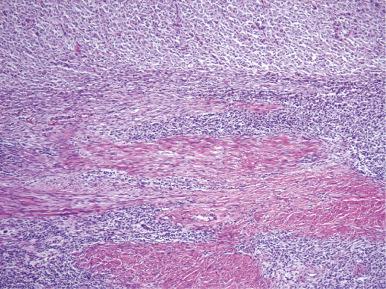
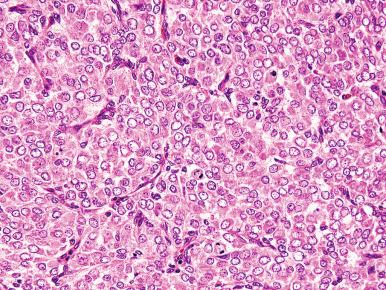
Usually, an undifferentiated endometrial sarcoma presents as one or more tan-yellow to gray, fleshy, intracavitary polypoid masses ( Fig. 20.21 ). Hemorrhage and necrosis are common. Histologically, the neoplastic cells show marked cellular atypia and numerous mitoses, including atypical forms, without evidence of differentiation toward endometrial stroma both cytomorphologically ( Fig. 20.22 ) and by pattern of growth within the myometrium. In general, an undifferentiated endometrial sarcoma has a diffuse and destructive infiltrative pattern as opposed to the characteristic wormlike pattern and propensity for intravascular extension characteristic of low-grade ESS. Because the tumor cell morphology of undifferentiated endometrial sarcoma is often likened to the mesenchymal component of a carcinosarcoma, exclusion of this latter entity should always be considered. In fact, the diagnosis of undifferentiated endometrial sarcoma should only be considered following exclusion of a poorly differentiated carcinoma, leiomyosarcoma, and carcinosarcoma, all of which may be morphologically similar in appearance ( Fig. 20.23 ). Extensive sampling and immunohistochemical staining (e.g., epithelial and smooth muscle markers) may be required. Undifferentiated uterine sarcomas have been subdivided into pleomorphic and uniform types. Many tumors in the latter category are now known to represent a high-grade ESS, as described above.
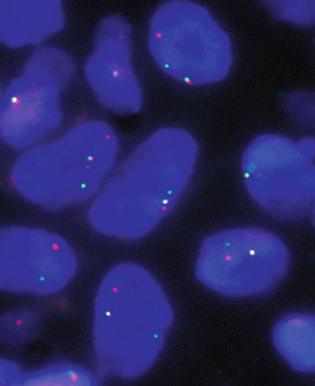
Chromosomal rearrangements involving chromosomes 6, 7, and 17 are the most frequent cytogenetic abnormalities that have been reported. The most common rearrangement is a reciprocal, balanced translocation between chromosomes 7 and 17, the t(7;17)(p15;q21). Two previously unknown genes, termed JAZF-1 and SUZ12 ( JJAZ-1) , were identified at the chromosomal sites of breakage in 7p15 and 17q21, respectively. In both stromal nodules and low-grade ESS, chimeric JAZF-1 – SUZ12 ( JJAZ-1) mRNA transcripts can be detected by reverse transcription polymerase chain reaction (PCR) in most cases. Moreover, fluorescence in situ hybridization (FISH) analysis can be performed on formalin-fixed, paraffin-embedded tissue. In this technique, two fluorescently labeled human genomic probes that flank the chromosome 7 breakpoint in the t(7;17)(p15;q21) are used to test for the presence of the translocation. In a tumor that contains the translocation, the fluorescent-labeled probes are split apart, yielding separate red and green signals ( Fig. 20.24 ). A yellow signal is produced by the close apposition of the two probes, marking the normal copy of chromosome 7.
The second most frequent translocation is the t(6;7)(p21;p15), in which there is fusion of JAZF-1 with PHF-1 . The PHF1 gene at 6p21 can also fuse with EPC1 at 10p11 or MEAF6 at 1p34. Interestingly, ESSs showing sex cord–like differentiation often have a PHF1 genetic rearrangement. Two additional translocations have been described in ESSs, a t(X;22) (p11;q13) and t(X;17) (p11.2;q21.33) associated with a ZC3H7B- BCOR fusion and MBTD1-CXorf67 fusion, respectively. Although endometrial stromal tumors are genetically heterogeneous, the different genes involved in low-grade ESSs are functionally related ( PHF1, SUZ12, EPC1, MBTD1 ), being members of the polycomb gene family. Of interest, ZC3H7B-BCOR , MEAF6-PHF1 , and EPC1-PHF1 fusions were also identified in ossifying fibromyxoid tumors and JAZ1-PFH1 in an ossifying sarcoma of the heart. Rarely, ESSs can show MDM2 amplification by FISH as well as MDM2 expression by immunohistochemistry, a potential pitfall, particularly in tumors occurring in locations more common to liposarcoma (e.g., peritoneum, retroperitoneum).
Endometrial stromal nodules and low-grade ESSs may show evidence of the t(7;17) translocation by FISH, suggesting that the stromal nodule may be a precursor lesion for low-grade ESS and that the t(7;17) is an early genetic abnormality in the development of low-grade ESS. In addition, FISH of mixed endometrial stromal–smooth muscle tumors has shown evidence for the translocation in the endometrial stromal and smooth muscle components, supporting the concept that these tumors are of endometrial stromal derivation.
This tumor has a characteristic genetic abnormality—t(10;17)(q22;p13)—associated with a YWHAE-FAM22 (NUTM2AB) fusion. FISH analysis on formalin-fixed, paraffin-embedded tissue for the YWHAE-FAM22 rearrangement has become more widely available; however, in most cases, confirmatory FISH does not need to be performed if the morphology and immunophenotype are characteristic (see below).
These tumors have been thought to represent endometrial stromal neoplasms that show entirely sex cord differentiation. However, they do not have the JAZF1-SUZ12(JJAZ1) fusion, which is the most common aberration seen in low-grade endometrial stromal neoplasms. Thus, it is now believed that these may not represent an endometrial stromal neoplasm.
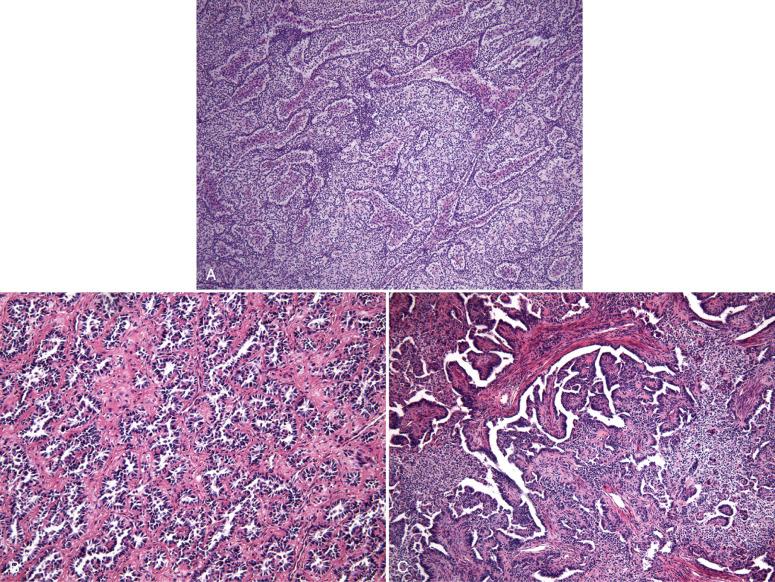
Recognition of a morphologically low-grade endometrial stromal neoplasm in a curettage can sometimes be difficult because it must be distinguished from potential mimics, including endometrial basalis, aglandular functionalis, endometrial polyp, and adenosarcoma. Multiple fragments of aglandular cellular stroma containing spiral arteriole-like vessels with an expansile sheetlike growth are characteristic of a stromal neoplasm. In one study, curettage of a stromal neoplasm typically produces fragments of aglandular stroma measuring more than 5 cm ( Fig. 20.25 ). Distinction from fragments of basalis is made by the presence of an orderly component of glands and lack of the rich vasculature so characteristic of stromal neoplasia ( Fig. 20.26A ). Strips of aglandular functionalis, usually associated with submucosal leiomyomata, tend to be less cellular and show features of compression or reactive surface changes (see Fig. 20.26B ). Fragments of stroma-rich endometrial polyps usually exhibit other features of polyps, including large, thick-walled vessels and abnormal glandular architecture (see Fig. 20.26C ). An adenosarcoma also may exhibit a cellular stroma but typically has glandular cuffing, albeit sometimes subtle (see Fig. 20.26D ). Appreciation of a more spindled atypical stroma without a rich vascular network facilitates this distinction.
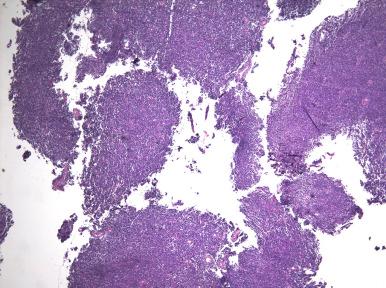
Once a stromal neoplasm is suspected based on histopathologic findings in the endometrial sampling, the next step in interpretation is determination of whether the tumor is benign or malignant. Unfortunately, this distinction is based on whether or not there is myometrial or lymphovascular invasion, two criteria that are difficult to interpret in biopsy or curettage material. One could raise the possibility of sarcoma if some fragments of tissue contain myometrium infiltrated by stromal tumor; however, prudent clinicopathologic correlation is critical. In general, the diagnosis of an endometrial stromal neoplasm, with a comment on one's inability to distinguish between a stromal nodule and low-grade endometrial sarcoma in the submitted material, will be the most likely course of action.
Endometrial stromal nodules are considered to be benign, nonrecurring neoplasms. Most women with stromal nodules have been treated by hysterectomy, due not only to their incidental discovery in hysterectomy specimens but also to the difficulty in distinguishing between stromal nodules and stromal sarcoma in biopsy or curettage material (discussed previously). In some cases, depending on the location and size of the tumor mass, preservation of fertility may be possible with partial uterine resection, which would include the mass and a rim of myometrium to assess for invasion. Preoperative imaging as to the feasibility of such an approach would be mandatory to assess for circumscription of the mass.
Only a few women with stromal nodules have been treated conservatively (local excision), and none of the tumors have recurred. In a case of an endometrial stromal nodule treated by local excision, an 8-year follow-up has been benign. Occasionally, some endometrial stromal nodules exhibit more than a 3-mm extension into the surrounding myometrium but lack the typical overt myometrial permeation seen in an ESS. The term endometrial stromal tumors with limited infiltration has been proposed for such tumors. Difficulty in predicting biologic behavior for these tumors is compounded by the fact that few cases have been described, all have been treated by hysterectomy, and long-term follow-up is not known. In practice, we recommend diagnosing these as ESS with limited infiltration or as endometrial stromal neoplasm with limited infiltration, with a comment suggesting that the tumor may pursue a benign clinical course but clinical follow-up is nevertheless recommended.
Patients with a low-grade ESS are treated by hysterectomy and bilateral salpingo-oophorectomy. Patients with tumors confined to the uterus (stage I) have an excellent prognosis, with a 5-year survival rate over 90%. Recurrence is not uncommon, ranging up to 25% in patients with stage I disease. Unfortunately, there are no histopathologic parameters to predict which patients with tumors confined to the uterus are at risk for recurrence. Distant metastases, principally involving the lung, may occasionally occur, often nearly a decade following the initial presentation. Following surgery, treatment options include local radiation therapy, which may reduce local failure. The impact of local radiotherapy on long-term survival, however, is not known. A low-grade ESS typically is positive for progesterone receptor. Therefore, hormonal therapy, particularly progestin therapy, is an option often considered in patients who present with advanced-stage disease or have recurrences.
Patients typically present with advanced-stage disease (stage III or IV ≫ stage I) and are treated with hysterectomy, bilateral salpingo-oophorectomy, and tumor debulking, if indicated. Patients frequently have recurrences, usually within a few years of initial surgery. Although experience is limited, adjuvant therapy may provide survival benefit. Hormonal therapy is likely ineffective because the morphologically high-grade component is typically negative for estrogen and progesterone receptor (see below).
Most of the studies of clinical outcomes with different treatment regimens do not clearly distinguish between undifferentiated uterine sarcoma and high-grade ESS. The latter category might encompass tumors that would be classified according to current WHO criteria as a low-grade ESS as well as undifferentiated uterine sarcoma. Undifferentiated uterine sarcoma is an aggressive neoplasm and treatment options, in addition to surgery, include consideration of radiation therapy for local control, as well as chemotherapy for systemic control.
Endometrial stromal tumors with smooth muscle differentiation or those that have a more fibrous or myxoid appearance may occasionally be confused with uterine smooth muscle tumors. Conversely, uterine smooth muscle tumors may mimic endometrial stromal tumors, particularly when the former is markedly cellular (e.g., highly cellular leiomyoma; see later, Figs. 20.75 and 20.76 ) or has prominent vascular invasion (e.g., intravenous [IV] leiomyomatosis; see later, Fig. 20.90 ) and IV leiomyosarcomatosis. In difficult cases, application of a panel of biomarkers may be helpful, provided that one is aware of potential pitfalls.
Endometrial stromal nodules and low-grade ESSs are usually only focally positive for smooth muscle actin and desmin; however, a subset of morphologically typical cases may show more extensive expression of these markers. In contrast, h-caldesmon is a more specific marker of smooth muscle differentiation than desmin and may be useful in this differential. Non-neoplastic and neoplastic endometrial stromal cells are typically negative for this marker, whereas non-neoplastic and neoplastic uterine smooth muscle is positive ( Fig. 20.27 ). In some cases of highly cellular leiomyoma, h-caldesmon may only show patchy or focal positivity (see later, Fig. 20.78 ).
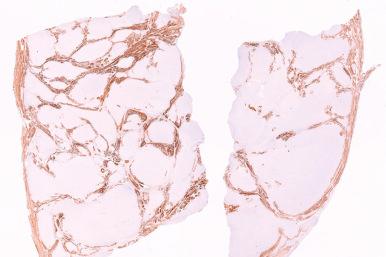
The role of CD-10 as a potential marker of endometrial stromal differentiation was proposed based on its unexpected expression in ESS. Subsequent analyses have shown that CD-10 is expressed in endometrial stromal cells of the cycling endometrium (less so in decidua), adenomyosis, endometriosis, and endometrial stromal tumors, both nodules and low-grade ESS ( Fig. 20.28 ). CD-10 is typically strongly and diffusely positive in non-neoplastic and neoplastic endometrial stroma; however, some endometrial stromal tumors may be negative for this marker. As a further caveat, smooth muscle tumors, particularly highly cellular leiomyomata and leiomyosarcoma, may be positive for CD-10 ( Fig. 20.29 ; see later, Fig. 20.79 ), but usually not to the degree seen in endometrial stromal tumors.
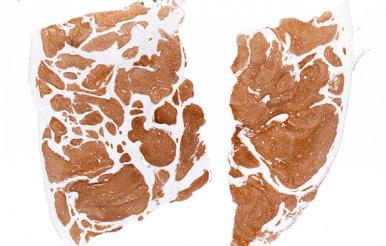
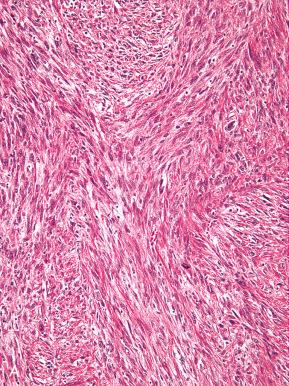
In general, the morphologic appearance of tumor cells and the growth pattern within myometrium can distinguish a low-grade ESS from leiomyosarcoma. In cases in which there is prominent lymphatic or vascular permeation by leiomyosarcoma, ESS may be considered. Although morphologic features such as the presence of a fascicular architecture, even in the intravascular component, can help facilitate its recognition as a malignant smooth muscle tumor, a panel of antibodies, including h-caldesmon, desmin, and CD-10, may be performed in difficult cases. Most low-grade ESSs will be CD-10–positive (diffusely), h-caldesmon–negative, and desmin variable (but usually focal); most leiomyosarcoma will be h-caldesmon–positive, desmin–positive, and CD-10 variable (often positive). The main pitfalls to consider are that nearly 50% of leiomyosarcomas can be CD-10–positive, sometimes diffusely (see Fig. 20.29 ), some low-grade ESSs can be diffusely positive for desmin, and some ESSs can be CD-10–negative. Low-grade ESSs are typically not h-caldesmon–positive unless there are areas of smooth muscle differentiation. In the latter situation, interpretation of areas with classic morphology will facilitate the correct diagnosis. In the distinction from leiomyosarcoma, areas of smooth muscle differentiation in a low-grade ESS tend to be bland and will not exhibit the degree of cellularity and nuclear pleomorphism that can be present in a leiomyosarcoma.
A high-grade ESS is characterized by different patterns of staining in the morphologically low-grade versus high-grade component. The low-grade component is typically positive for CD-10, estrogen receptor (ER), and progesterone receptor (PR) and will show variable positivity for cyclin D1, whereas CD117 is negative. In contrast, the high-grade component is typically negative for CD-10, ER, and PR and shows strong and diffuse positivity (>70% tumor cell nuclei) for cyclin D1 ( Fig. 20.30 ); CD1-17 is often positive.
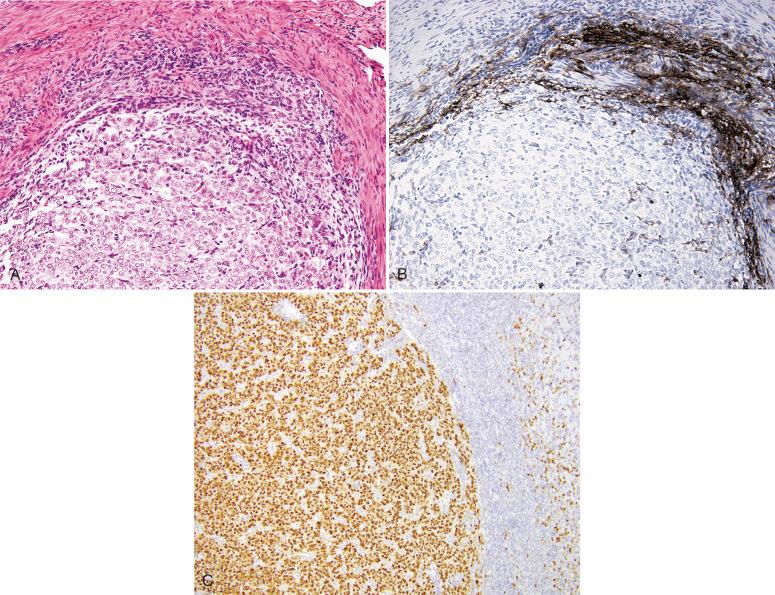
Low- and high-grade ESSs both show nodular permeative growth within the myometrium; however, the latter is more likely to present at an advanced stage, show destructive myometrial infiltration, and, in most cases, show areas of higher grade morphology, as described above. Moreover, the vascular pattern is different, with a high-grade ESS having numerous delicate and arborizing vessels as opposed to the spiral, arteriolar-like vascular network of a low-grade stromal sarcoma. Immunoperoxidase stains are helpful in this distinction, but correlation with morphology is key. The high-grade epithelioid areas are typically negative for CD-10, ER, and PR but strongly and diffusely positive for cyclin D1. In some cases, correlation with molecular testing may be useful in this distinction.
The morphologically high-grade component of a high-grade ESS has an epithelioid appearance and may be confused with an undifferentiated carcinoma, particularly in limited material, such as a biopsy or curettage. One pitfall to keep in mind is that undifferentiated carcinomas of the endometrium can also show strong and diffuse positivity for cyclin D1; however, they will usually show focal positivity for epithelial membrane antigen (EMA) and broad-spectrum cytokeratin or be positive for CD-10. In cases lacking positivity for these markers, molecular studies might be necessary for the diagnosis.
Benign and malignant uterine smooth muscle tumors occur throughout the female genital tract, from the vulva to the broad ligament and ovaries. The vast majority of these tumors, however, are located in the uterine corpus, where they are presumed to arise from benign myometrial cells. Their location in the uterus, in combination with their size, largely determines the resulting symptoms. For example, submucosal tumors often present with abnormal bleeding and may be associated with a poor reproductive outcome. In contrast, subserosal and intramural tumors typically present with pain or impingement on nearby pelvic organs. Subserosal leiomyomas are even thought by some to have the capacity to detach from the uterus after the development of a new blood supply, earning them the colorful term of parasitizing leiomyoma.
In most cases, classification of a smooth muscle neoplasm as benign or malignant is straightforward. Such determination rests entirely on histopathologic features, particularly the presence or absence of atypia, proliferative activity, and particular pattern of necrosis. As shall be discussed, uterine smooth muscle tumors are well known for their benign variants, which have one of the features of malignancy in isolation. Tumors with several features of malignancy—but that do not meet the criteria for the diagnosis of leiomyosarcoma—add further complexity to classification schemes. The prediction of clinical behavior of such morphologic intermediates is difficult at best. Superimposed on this spectrum of smooth muscle neoplasia is a number of morphologically benign smooth muscle proliferations with the biologic features of malignancy—namely, dissemination or distant metastasis, vascular invasion, or local infiltration. This morphologic and biologic diversity makes smooth muscle neoplasia a fascinating area of study.
Benign tumors of the muscular uterine wall are known variously as leiomyomata, myomas, fibromyomas, fibromas, or fibroids. Leiomyomata are observed in nearly 77% of hysterectomy specimens, regardless of the indication for surgery, and the average number of independent tumors per uterus has been estimated to be more than six. Consequently, they are the most common human tumor. Fortunately, only about 25% of women of reproductive age are symptomatic.
Women with symptomatic leiomyomata generally present after the age of 35 years. Symptoms may include abnormal uterine bleeding, pelvic pressure or pain, and reproductive dysfunction. Abnormal uterine bleeding as a result of leiomyomata may be characterized as menorrhagia or hypermenorrhea. Leiomyoma-induced menorrhagia may be so severe as to require changing sanitary napkins hourly for more than 5 days; such profound bleeding can result in significant anemia. Leiomyoma-associated bleeding also may become a source of social embarrassment and result in significant lost productivity. Pelvic pain or pressure is typically associated with tumors large enough to distort the uterine corpus. In a testament to the size that a uterus with leiomyomata may reach, these so-called fibroid uteri are clinically assessed by comparing them to a gravid uterus. Thus, a symptomatic uterus may be as large as a gestational uterus at 16 to 20 weeks. In addition to pain or pressure, large tumors may compress nearby structures and may occasionally cause constipation, urinary frequency, or ureteral obstruction. Although uncommon, the range of reproductive dysfunction disorders associated with leiomyomata includes infertility, spontaneous abortion, premature labor, and fetal malpresentation. Pseudo-Meigs syndrome (ascites and hydrothorax) rarely may be attributed to uterine leiomyoma.
The severity of symptoms associated with leiomyomata is broadly related to tumor size and location. The myometrium can conceptually be divided into three zones—submucosal, intramural, and subserosal. Submucosal leiomyomata, as well as larger intramural tumors that distort the endometrial cavity, may cause abnormal uterine bleeding ( Fig. 20.31 ). Although all the pathophysiologic details have yet to be elucidated, attenuation of the endometrium overlying leiomyomata is frequently found. Sampling of these attenuated areas produces strips of aglandular endometrium in curettings, which can be used to suggest the diagnosis of leiomyomata (see Fig. 20.25 ) and a cause for bleeding, provided that no other endometrial pathologies capable of causing bleeding are present. Subserosal or deeper leiomyomata are less likely to cause uterine bleeding but are more likely to be associated with pelvic pain or pressure.
A fibroid uterus is the most common indication for hysterectomy, and 2.1 hysterectomies/1000 women are performed annually for this diagnosis in the United States. Despite the benign nature of leiomyomata, the impact of more than 200,000 major surgical procedures each year on public health and medical economics is considerable. A hysterectomy performed for leiomyomata is one of the most common procedures in the practice of surgical pathology.
Fibroids also may be managed expectantly if associated with minimal or no symptoms. For some time, gynecologists and patients have expressed a growing interest in avoiding hysterectomy by developing or selecting less invasive alternatives to hysterectomy. Myomectomy, resection with uterine conservation, is the most widely used alternative for women who wish to preserve fertility. Myomectomy may be performed using an open abdominal approach or by various closed techniques involving laparoscopy, hysteroscopy, or myolysis, which involves in situ coagulation using a laparoscopic probe. Tumor size and location play an important role in determining which technique is most appropriate. When compared with hysterectomy, these alternatives are associated with several unique risks—namely, the risks of tumor recurrence and uterine rupture during pregnancy following myomectomy. The risk of symptomatic recurrence is more likely due to the growth of a second crop of tumors and requires a second operation in 10% to 26% of cases. Morcellation with a powered device may result in peritoneal dissemination of benign and malignant smooth muscle tumors. Studies have shown that the rate of unexpected sarcoma after a laparoscopic morcellation procedure may be far higher than once thought, and that dissemination of leiomyosarcoma by morcellation occurs in nearly two-thirds of cases, with some resulting in mortality. Uterine artery embolization with particles of polyvinyl alcohol has been used to treat leiomyoma noninvasively. Although uterine artery embolization results in symptomatic improvement for most patients, this technique has been associated with adverse outcomes, ranging from postprocedure fevers to amenorrhea, uterine rupture, endomyometritis, and fatal sepsis. Uterine artery embolization also has been implicated as a factor delaying the diagnosis of uterine sarcomas. The latest noninvasive technique for the treatment of leiomyomata is MRI-guided focused ultrasound, which causes thermolysis of targeted smooth muscle cells (see later, Figs. 20.71 and 20.72 ).
Although surgery has been the mainstay of treatment for benign uterine smooth muscle tumors, medical therapy may be helpful in particular cases. Androgenic steroids, such as danazol and gestrinone, cause amenorrhea, which may be helpful in treating leiomyoma-associated anemia. Gestrinone also may reduce leiomyoma volume. Treatment with gonadotropin-releasing hormone (GnRH) agonists, the most widely used medical therapy for leiomyoma, also results in reduction of leiomyoma volume by 35% to 65% by producing a pseudomenopausal hypoestrogenic state. This therapy places the patient at risk for osteoporosis and other significant complications; consequently, GnRH agonists must be used only for a short time (e.g., until definitive surgery can be performed or while waiting for natural menopause to occur). Unlike gestrinone, the reduction in tumor volume associated with GnRH agonists is rapidly reversible and, in part, may reflect volume shifts in the extracellular matrix of leiomyomata rather than changes in the neoplastic cells. Volume changes in treated leiomyomata may be due to changes in apoptosis and insulin-like growth factor 1 (IGF-1) receptor activity as well. Mitigation of the risks associated with hypoestrogenism has been attempted by adding exogenous hormones after an initial period of complete suppression, but these protocols have not been widely adopted. Of note to pathologists, there are few or no histologic changes in leiomyomata after treatment with GnRH agonists. Finally, selective estrogen receptor modulators such as raloxifene may inhibit the growth of leiomyomatous smooth muscle cells and reduce leiomyoma volume. Similar to other noninvasive therapies, the medical treatment of leiomyomata may delay the diagnosis of leiomyosarcoma.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here