Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
60,000 estimated new cases per year.
Approximately 10,400 deaths per year from the disease.
Endometrioid adenocarcinoma arises in the endometrium and accounts for approximately 90% of cases.
Median age is 63 years.
The most common presenting symptom is irregular vaginal bleeding.
Risk factors include unopposed estrogen therapy, obesity, chronic anovulation, tamoxifen use, diabetes, and nulliparity.
Staging is surgical according to International Federation of Gynecology and Obstetrics (FIGO) guidelines.
Uterine sarcomas and carcinomas are staged differently.
Primary surgery with hysterectomy, bilateral salpingo-oophorectomy, and lymphadenectomy is curative in the majority of cases.
Radiation alone may be used for inoperable patients or palliation.
Risk factors for recurrence or extrauterine spread include deep myometrial invasion, moderate- to high-grade histology, and lymphovascular space invasion.
Adjuvant radiation may be used in patients with high-intermediate and high-risk disease.
Adjuvant chemotherapy with or without radiation therapy may be administered in cases with aggressive histology, such as serous, clear cell, carcinosarcoma, or leiomyosarcoma and undifferentiated endometrial sarcoma.
Chemotherapy, radiation, or combination therapy is used in locally advanced, metastatic, or recurrent endometrial cancer.
Cytoreductive surgery is associated with improved overall survival.
Salvage radiotherapy is used for recurrences after surgery alone.
Endocrine therapy with progestins, tamoxifen, or both may be used when avoidance of cytotoxic chemotherapy is desired.
Tumor-directed radiation therapy may be used in cases of locally advanced, metastatic, or recurrent disease.
Survival is strongly correlated with surgical stage, tumor grade, and histology.
Uterine clear cell and serous carcinomas are associated with a worse prognosis than endometrioid adenocarcinomas.
Endometrial cancer, or uterine cancer, is a malignancy arising from the endometrium. Women have a 1 in 40 lifetime risk of being diagnosed with endometrial cancer, the fourth most common malignancy among women. Uterine cancer is the most common gynecologic malignancy in the United States. Stage I malignancies comprise the majority of endometrial cancers. Postmenopausal bleeding is the most common presentation. Cancer arising from endometrial glands is referred to as carcinoma compared with the less common uterine sarcoma that arises in mesenchymal elements such as smooth muscle or connective tissue.
An estimated 46,000 new diagnoses of uterine cancer and 8100 deaths from the disease occurred in 2011 in the United States. The median age at diagnosis is 63 years. The Surveillance, Epidemiology, and End Results program (SEER) reported that 70% of patients are diagnosed with localized disease, 17% with regional spread, 9% with distant disease, and 4% unstaged. Five-year survival rates are 80% to 90% for disease confined to the uterus ( Table 85.1 ).
| Stage | 5-Year Survival (%) |
|---|---|
| IA | 90 |
| IB | 78 |
| II | 74 |
| IIIA | 56 |
| IIIB | 36 |
| IIIC1 | 57 |
| IIIC2 | 49 |
| IVA | 22 |
| IVB | 21 |
Endometrial cancer is uncommon in premenopausal women, and genetic factors account for only 1% of newly diagnosed cases. Lynch syndrome, or hereditary nonpolyposis colorectal cancer syndrome (HNPCC), is associated with a relative risk of 1.5 for the development of endometrial cancer before menopause. Women with Lynch syndrome have a 27% to 71% risk of endometrial cancer. Mutations in the DNA mismatch repair genes impair the ability to maintain genomic integrity. Patients with Lynch syndrome have a germline mutation in one mismatch repair allele, and the second allele is inactivated through mutation, loss of heterozygosity, or epigenetic silencing by promoter hypermethylation. Inactivation of both genes leads to increased DNA mutations and alteration of microsatellite regions in the tumor compared with normal tissue. Microsatellite instability refers to the contraction or expansion of short repetitive DNA sequences and is caused by loss of DNA mismatch repair. Microsatellite instability is found in 90% of tumor tissue from patients with Lynch syndrome. Mutations in MSH2 or MLH1 account for approximately 90% of the identified mutations associated with Lynch syndrome. Mutations in PMS1, MSH6 , and MLH3 have also been described. The age of diagnosis in women with Lynch syndrome is 46 to 54 years. Endometrioid histology is the most common Lynch syndrome–associated endometrial cancer; however, nonendometrioid tumors have been reported. The majority of tumors are diagnosed with early-stage disease, similar to women with sporadic endometrial cancer. Women with Lynch syndrome are at risk for synchronous or metachronous cancers. The Amsterdam Criteria have been developed to identify families at risk for Lynch syndrome ( Table 85.2 ). The Society of Gynecologic Oncologists published a statement for genetic testing of individuals at risk for Lynch syndrome ( Table 85.3 ). Moreover, it released a practice statement suggesting that all women diagnosed with endometrial cancer should undergo a systematic clinical screening (including a review of person and family history), or molecular screening for Lynch syndrome, or both.
| EACH OF THE FOLLOWING IS REQUIRED |
|
| PATIENTS WITH AN INCREASED LIKELIHOOD OF LYNCH SYNDROME AND FOR WHOM GENETIC ASSESSMENT IS RECOMMENDED: |
|
Table 85.4 lists many of the known risk factors for endometrial adenocarcinomas. Endogenous estrogen exposure associated with nulliparity, early menarche, late menopause, obesity, and estrogen-producing tumors are associated with an increased risk of endometrial cancer. Risk factors are associated with the unopposed estrogen effect, or lifetime estrogen exposure. Exposure to unopposed estrogen leads to increased endometrial cell proliferation, resulting in increased DNA replication errors and somatic mutations. Exogenous estrogen sources, such as hormone replacement therapy without progestins, increases the risk for endometrial cancer fivefold. The use of progestins with hormone replacement therapy may decrease the risk of uterine cancer through downregulation of hormone receptors. Cancers that occur in women on combined hormone replacement therapy tend to be of low stage and grade. Endogenous hormones, such as androstenedione, estrone, and estradiol, are associated with a threefold to fourfold increased risk.
| Risk Factor | Relative Risk |
|---|---|
| Unopposed estrogen therapy | 10–20 |
| Tamoxifen | 2.5 |
| Polycystic ovarian syndrome | 3–5 |
| Obesity | 2–5 |
| Diabetes | 2–3 |
| Nulliparity | 2–3 |
| Estrogen-producing ovarian tumors | 5 |
The National Surgical Adjuvant Breast Project (NSABP) and the Netherlands Cancer Institute clarified a causal link between the treatment of breast cancer with tamoxifen and endometrial cancer. Tamoxifen, a selective estrogen receptor (ER) modulator, was first described as a risk factor for uterine cancer in 1985. The NSABP evaluated 2834 women treated for breast cancer treated with tamoxifen. Twenty-four cases of endometrial cancer were diagnosed among the study patients. Nearly 96% ( n = 23) of endometrial cancer cases occurred in women taking tamoxifen. The annual hazard rates of endometrial cancer development were 1.6 per 1000 patient-years in the tamoxifen group and 0.2 per 1000 patient-years in the placebo group. In addition, 88% of endometrial cancers diagnosed were stage I, and 78% were of low or intermediate grade. The use of tamoxifen results in a 38% improvement in disease-free survival for breast cancer, which far outweighs the risk of endometrial cancer, from which there were only four deaths. A subsequent analysis by the NSABP showed the risk was predominately in women 50 years of age or older, with an incidence of approximately 2 per 1000. Regular gynecologic follow-up is recommended for patients on tamoxifen.
The Netherlands Cancer Institute performed a case-control study by identifying 98 patients in whom endometrial cancer developed after treatment for breast cancer compared with control participants in whom endometrial cancer did not develop. Twenty-four percent of the patients with endometrial cancer had taken tamoxifen compared with 20% of the control participants. Treatment with tamoxifen for 5 years or more was associated with an increased risk of 3.0-fold for endometrial cancer compared with control participants.
The etiology of uterine sarcomas is not well understood, but it may be related to prior exposure to pelvic irradiation. Uterine sarcomas and carcinomas have been reported after irradiation for cervix and rectal cancer. Radiation-associated uterine cancers are usually of higher grade and stage, with a more unfavorable histology.
Systemic disease and lifestyle factors influence the risk of endometrial cancer. Hypertension, diabetes, and obesity all increase the risk. Nearly 40% of uterine cancers can be attributed to obesity. Increasing body mass indices (BMIs) are associated with higher relative risks of developing a uterine malignancy. Overweight women (BMI, 28 to 29.9) have a relative risk of 1.5 compared with women with a normal BMI. Obese women (BMI, 30 to 33.9) have a relative risk of 2.9, and markedly obese women (BMI >34) have a relative risk of 6.3. The increased risk of endometrial carcinoma associated with increasing BMI may be explained by higher levels of endogenous estrogen. The conversion of androstenedione to estrone and the aromatization of androgens to estradiol occurs in peripheral adipose tissue. Severely obese women are more likely than nonobese women to have a less aggressive histology and present with stage I disease.
Known protective factors against endometrial cancer include full-term pregnancy, multiparity, older age of menarche, and oral contraceptive use. Use of oral contraceptives for up to 5 years is associated with a relative risk of 0.2, and its use for at least 1 year reduces endometrial cancer risk by approximately 45%. Interestingly, cigarette smoking decreases the risk of endometrial cancer; however, the clear adverse risks of smoking greatly outweigh any potential benefit with respect to endometrial cancer.
Abnormal uterine bleeding is the most common presentation in women diagnosed with endometrial carcinoma ( Fig. 85.1 ). Approximately 80% of women with endometrial carcinoma present with abnormal uterine bleeding; however, the amount of bleeding does not correlate with the risk of cancer. Less commonly, abnormal cervical cytology may be the first indication of uterine cancer. The risk of endometrial carcinoma and the need for endometrial evaluation depend on age, symptoms, and the presence of risk factors. Postmenopausal bleeding, including spotting, carries a 3% to 20% risk of endometrial carcinoma. The diagnosis of endometrial carcinoma before age 45 years is uncommon; however, intermenstrual bleeding or prolonged periods of amenorrhea (≥6 months) after age 45 years should be evaluated. Nineteen percent of endometrial carcinomas occur between ages 45 and 64 years. The risk of uterine cancer before age 45 years is low and increases with advancing age. The majority of abnormal bleeding is caused by benign uterine pathology, but further evaluation is warranted and recommended by the American College of Obstetricians and Gynecologists.
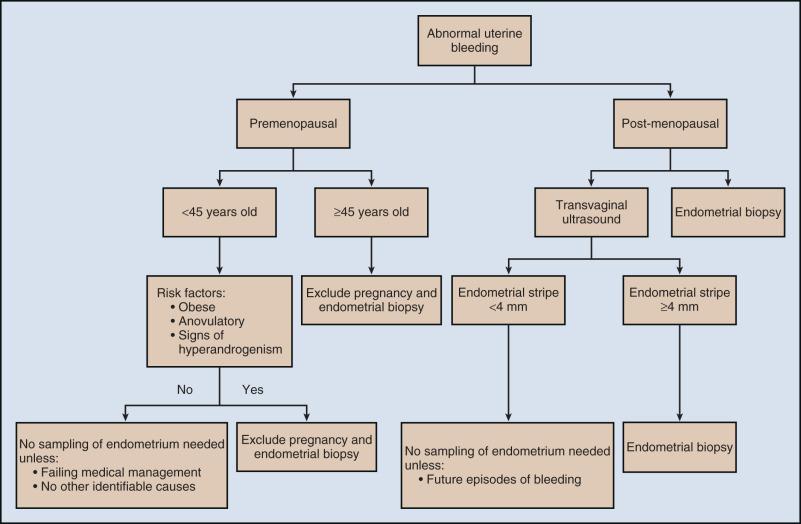
Women presenting with clinical signs suspicious for endometrial carcinoma should undergo a physical examination, including pelvic examination. Urine or serum human chorionic gonadotropin testing to exclude pregnancy should be performed on all reproductive-age women before any endometrial sampling. Pelvic ultrasonography is often ordered to assess the endometrial thickness. In postmenopausal women, transvaginal ultrasonography to evaluate the endometrial thickness may be used for endometrial neoplasia in selected women. In women with postmenopausal bleeding, an endometrial thickness less than 4 mm is associated with a low risk of endometrial carcinoma ; however, any focal endometrial lesion requires a biopsy. In contrast to postmenopausal women, the utility of transvaginal ultrasonography is not well established in premenopausal women. There is no standard threshold for endometrial thickness in premenopausal women. Transvaginal ultrasonography also does not appear to be an effective screening tool for women on hormone replacement therapy. Endometrial sampling is the gold standard for premenopausal women and women taking hormone replacement therapy.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here