Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
This work was supported by grants from the NIH DK59427 (G.J.G.) and K08CA236874 (S.H.R.). Support was also provided to Dr. Rizvi by a Pilot & Feasibility Award by the Center for Cell Signaling in Gastroenterology (P30DK084567), and an AGA Research Scholar Award.
Biliary malignancies comprise the vast majority of biliary neoplasms and are divided into 3 categories: (1) carcinomas of the intra- and extrahepatic bile ducts (cholangiocarcinomas), (2) carcinoma of the gallbladder, and (3) carcinoma of the ampulla of Vater. In the USA and other Western nations, biliary malignancies are rare. Cholangiocarcinomas are further classified into intrahepatic, perihilar, and distal cholangiocarcinoma on the basis of their anatomic location within the biliary treetract. In certain parts of the world, however, their prevalence rates are high, making them leading causes of cancer death in these regions. Biliary cancers are highly aggressive with dismal prognoses.
Cholangiocarcinoma is an epithelial carcinoma with differentiated features of biliary epithelium that arises from the intra- and extrahepatic biliary tract. It is the most common bile duct tumor and second most common primary hepatic malignancy (after HCC [see Chapter 96 ]). Since the 1970s, the incidence of cholangiocarcinoma has increased worldwide.
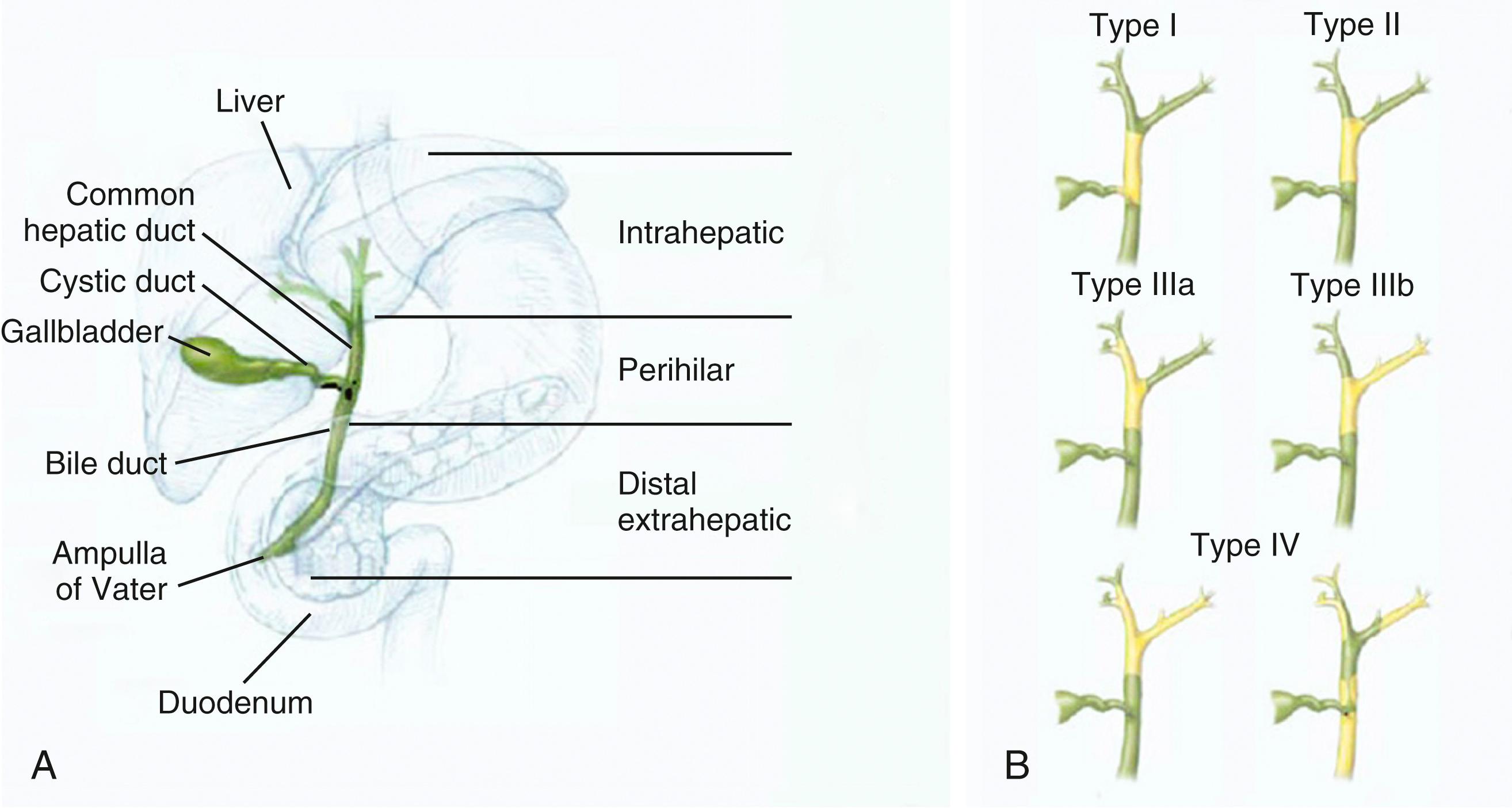
On the basis of their location within the biliary tract, cholangiocarcinomas are divided into intrahepatic, perihilar, and distal subtypes. Each anatomic subtype has a distinct epidemiology, pathogenesis, risk factors, management, and prognosis. 1 Intrahepatic cholangiocarcinomas arise within the liver parenchyma and above the second-order bile ducts (see Chapter 62 ). Perihilar or hilar cholangiocarcinomas arise between second-order bile ducts and the insertion of the cystic duct. Distal cholangiocarcinomas arise below the insertion of the cystic duct1 ( Fig. 69.1 A ). Perihilar cholangiocarcinomas, also referred to as Klatskin tumors , are described clinically according to the Bismuth-Corlette classification as types I to IV (see Fig. 69.1 B ). Type I cholangiocarcinomas involve the common hepatic duct distal to the union of the right and left hepatic ducts; type II tumors involve the union of the right and left hepatic ducts; type IIIa tumors involve the union of the right and left hepatic ducts and extend up the right hepatic duct; type IIIb tumors involve the union of the right and left hepatic ducts and extend up the left hepatic duct; and type IV tumors are multifocal or involve the biliary confluence and extend up the right and left hepatic ducts. The natural course of cholangiocarcinoma is aggressive, with a median survival of less than 24 months following diagnosis.
Cholangiocarcinoma is the most common biliary and the second most common primary hepatic malignancy. It accounts for less than 2% of all malignancies but is the ninth most common GI malignancy. Hepatobiliary malignancies account for 13% and 3% of overall cancer-related mortality in the world and in the USA, respectively; 10% to 20% of these deaths are caused by cholangiocarcinoma.
Global incidence rates for cholangiocarcinoma are heterogeneous. The highest incidence is observed in Southeast Asia, with age-adjusted incidence rates up to 113 per 100,000 population, and the lowest incidence is observed in Australia, with rates as low as 0.1 per 100,000 population. , In the USA, ethnic differences in the age-adjusted incidence of cholangiocarcinoma have been observed, with the highest rates observed in patients of Asian and Hispanic descent (3.3/100,000 and 2.8/100,000, respectively) and the lowest in African Americans (2.1/100,000). The incidence of cholangiocarcinoma and age-adjusted mortality rates are higher for males than females. Worldwide, the average age at diagnosis is around 50 years. In Western industrialized nations, most cases are diagnosed in patients over the age of 65 years, and cholangiocarcinoma is uncommon before the age of 40 years, except in patients with PSC (see Chapter 68 ).
The majority of cholangiocarcinomas are either perihilar (50% to 60%) or distal (20% to 30%) tumors, whereas intrahepatic cholangiocarcinomas account for only approximately 20% of all cholangiocarcinomas. Since the 1980s, an increase in the incidence of intrahepatic cholangiocarcinomas and a concomitant decrease in the incidence of perihilar and distal cholangiocarcinomas has been reported in multiple studies. These trends may, in part, be explained by misclassification of perihilar tumors as intrahepatic. According to the Surveillance, Epidemiology, and End Results database in the USA, the age-adjusted incidence rate for intrahepatic cholangiocarcinomas increased between 1990 and 2001, whereas the incidence rate for perihilar and distal cholangiocarcinomas decreased over this time period. The second edition of the International Classification of Disease for Oncology classified Klatskin tumors as intrahepatic. In the third edition published in 2001, Klatskin tumors were reclassified as extrahepatic. ,
In the majority of cases, the etiology of cholangiocarcinoma is unknown. Several risk factors have been identified ( Box 69.1 ). These risk factors are characterized by their association with inflammation and cholestasis; however, studies exploring the association of risk factors with cholangiocarcinoma are frequently limited by inter- and intracontinental heterogeneity and inaccuracies of cancer registries, such as classification of cholangiocarcinoma with other biliary malignancies and HCC.
Although there are several known risk factors for cholangiocarcinoma, most cases are sporadic and occur in the absence of a known risk factor. PSC is one of the most common risk factors for cholangiocarcinoma (see Chapter 68 ); however, only 10% of cholangiocarcinomas are attributed to PSC. In patients with PSC, the annual incidence of cholangiocarcinoma is 0.6% to 1.5%, and the 30-year cumulative incidence is 20%. A new diagnosis of PSC should heighten suspicion for cholangiocarcinoma; 27% to 37% of biliary tract cancers are detected within the first year of the diagnosis of PSC. Other cholangiopathies have also been found to be risk factors for cholangiocarcinoma. Patients with Caroli disease and choledochal cysts, in particular types I and IV, have an up to 50-fold increased risk of cholangiocarcinoma, with lifetime incidence rates of 6% to 30% (see Chapter 62 ). Cyst excision reduces but does not eliminate the risk for the development of cholangiocarcinoma. Hepatolithiasis with recurrent pyogenic cholangitis carries a 10% risk of cholangiocarcinoma (see Chapter 68 ). Recurrent bacterial cholangitis in the setting of a biliary-enteric drainage procedure has also been associated with the development of cholangiocarcinoma. Other risk factors for cholangiocarcinoma include biliary infections with Opisthorchis viverrini and Clonorchis sinensis , which are endemic in East Asia (see Chapter 84 ). Carcinogens such as thorotrast (used as a radiologic contrast agent in the past) and dioxins have been associated with an increased risk of cholangiocarcinoma (see Chapter 89 ).
The association between cholangiocarcinoma and IBD (UC or Crohn disease) is controversial and likely influenced by the presence and duration of PSC; therefore, it is unclear if the presence of IBD confers additional risk for cholangiocarcinoma in patients with PSC. Association studies between cholangiocarcinoma and viral hepatitis have shown intercontinental inconsistencies. Intrahepatic cholangiocarcinoma has been associated with HCV and HBV infection and cirrhosis, whereas perihilar cholangiocarcinoma has been associated only with cirrhosis (see Chapter 96 ). , , Obesity, diabetes mellitus, and NAFLD have been associated with an increased risk of cholangiocarcinoma, but the data are inconsistent. , The incidence of these conditions has been increasing in Western industrialized nations, a trend that could account for the increasing incidence of intrahepatic cholangiocarcinomas in these countries. Heavy alcohol consumption has been associated both with intrahepatic and with perihilar and distal cholangiocarcinoma and is thought to be a likely risk factor. , ,
Cholangiocarcinoma is a paucicellular, highly desmoplastic tumor. Macroscopically, intrahepatic cholangiocarcinomas can have 3 subtypes: mass-forming, periductal-infiltrating, and intraductal growth type. The mass-forming type is the most common type of intrahepatic cholangiocarcinoma, accounting for more than 85% of cases. ,
The mass-forming type tends to invade the hepatic parenchyma, with invasion of the lymphatics at advanced stages, whereas the periductal-infiltrating type spreads along the Glisson sheath via the lymphatics. Macroscopically, perihilar cholangiocarcinomas have a periductal-infiltrating, nodular, or intraductal growth pattern, with the periductal-infiltrating subtype being the most common. Because perihilar cholangiocarcinomas have a tropism for bile, the initial growth pattern is typically periductal, and with tumor progression, a mass lesion is formed resulting in a mass-forming and periductal-infiltrating lesion. , Intraductal-papillary adenocarcinomas spread superficially along the biliary mucosa without deep invasion of the fibromuscular wall layers and have a better prognosis than nonpapillary cancers ; metastases to regional and peripancreatic lymph nodes are frequently observed with this type. Histologically, 90% of cholangiocarcinomas are adenocarcinomas. Other histologic types include intestinal-type adenocarcinoma, clear cell adenocarcinoma, signet-ring cell carcinoma, adenosquamous carcinoma, squamous cell carcinoma, and small cell carcinoma.
The pathogenesis of cholangiocarcinoma is complex and likely related to inflammation in the microenvironment, environmental factors, and genetic aberrations of the tumor cells (both acquired and germline). Most available information relates to genetic aberrations, including analysis of the transcriptome, copy number variations, DNA methylation, microRNA profiling, and somatic mutations. The most comprehensive resource on genetic aberrations is The Cancer Genome Atlas integrated genomic analysis of cholangiocarcinomas that were predominantly intrahepatic, liver-fluke negative, and hepatitis virus negative. Inactivating mutations in tumor suppressor genes including ARID1A , ARID1B , BAP1 , tumor protein p53 ( TP53 ), and phosphatase and tensin homolog ( PTEN ) and gain-of-function mutations in the oncogenes isocitrate dehydrogenase ( IDH ) 1 and 2, BRAF , and KRAS were identified in these tumors. Discrete carcinogenic risk factors may impact the pattern of somatic aberrations in cholangiocarcinomas. For instance, liver fluke–related cholangiocarcinomas have a higher frequency of TP53 mutations, whereas IDH1 , IDH2 , and BAP1 mutations occur more frequently in nonliver fluke–related cholangiocarcinomas.
The repertoire of genomic alterations in cholangiocarcinoma varies by anatomic subtype. The frequencies of molecular aberrations in fibroblast growth factor receptor ( FGFR ) 1-3, IDH 1 and 2 , and BAP1 are higher in intrahepatic cholangiocarcinoma, whereas the frequencies of mutations of ARID1B , PBRM1 , and protein kinase cyclic AMP-activated catalytic subunit alpha ( PRKACA ) and beta ( PRKACB ) are higher in distal and perihilar cholangiocarcinomas. Inactivating mutations in E74-like ETS transcription factor 3 ( ELF3 ) occur preferentially in distal cholangiocarcinoma ( Table 69.1 ).
| Cholangiocarcinoma Subtype | Molecular Aberration |
|---|---|
| Intrahepatic | Fibroblast growth factor receptor ( FGFR ) 2 gene fusions Isocitrate dehydrogenase (IDH) 1/2 mutations Mcl-1 amplification MET-hepatocyte growth factor (HGF) overexpression BAP1 inactivating mutations ARID1A inactivating mutations PTPN3 activating mutations KRAS activating mutations PIK3CA activating mutations |
| Perihilar | HER2 amplifications ERBB2 genetic aberrations Protein kinase cyclic AMP-activated catalytic subunit alpha ( PRKACA ) and beta ( PRKACB ) gene fusions KRAS activating mutations PIK3CA activating mutations |
| Distal | E74-like ETS transcription factor 3 (ELF3) inactivating mutations ERBB3 mutations KRAS activating mutations PIK3CA activating mutations |
Deregulation of FGF signaling with consequent carcinogenesis occurs in a spectrum of malignancies, including cholangiocarcinoma. Gene fusions of FGFR2 , a receptor tyrosine kinase, result in its ligand-independent activation. FGFR2 gene fusions are present in 11% to 14% of intrahepatic cholangiocarcinomas. The discovery of these gene fusions is notable because they are often targetable driver mutations. IDH mutations are relatively common genetic aberrations in cholangiocarcinoma that are observed in 23% to 28% of intrahepatic cholangiocarcinomas. , IDH1 and IDH2 are enzymes that catalyze the reaction leading to generation of α-ketoglutarate. Point mutations of IDH and IDH2 result in an increase in 2-hydroxyglutarate, an oncometabolite that promotes extensive epigenetic aberrations in malignancy. , Clinical trials of small molecule inhibitors of FGFRs and of IDH1 and IDH2 for cholangiocarcinoma are being undertaken.
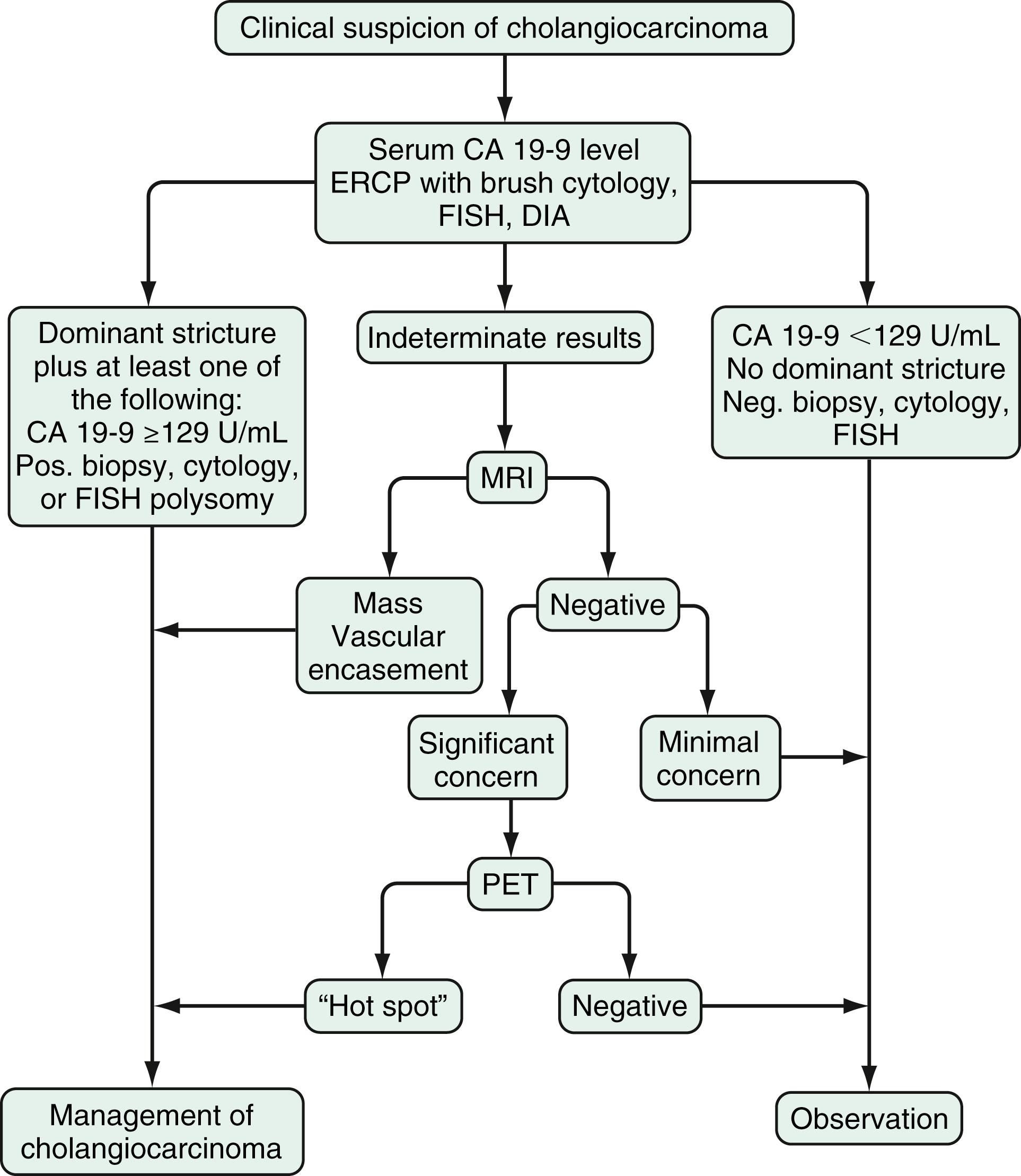
The diagnosis of cholangiocarcinoma is challenging because the presentation is often insidious, and the results of diagnostic studies are frequently nonspecific. A multidisciplinary approach that includes clinical evaluation and laboratory, endoscopic, and imaging studies is required ( Figs. 69.2 and 69.3 ).
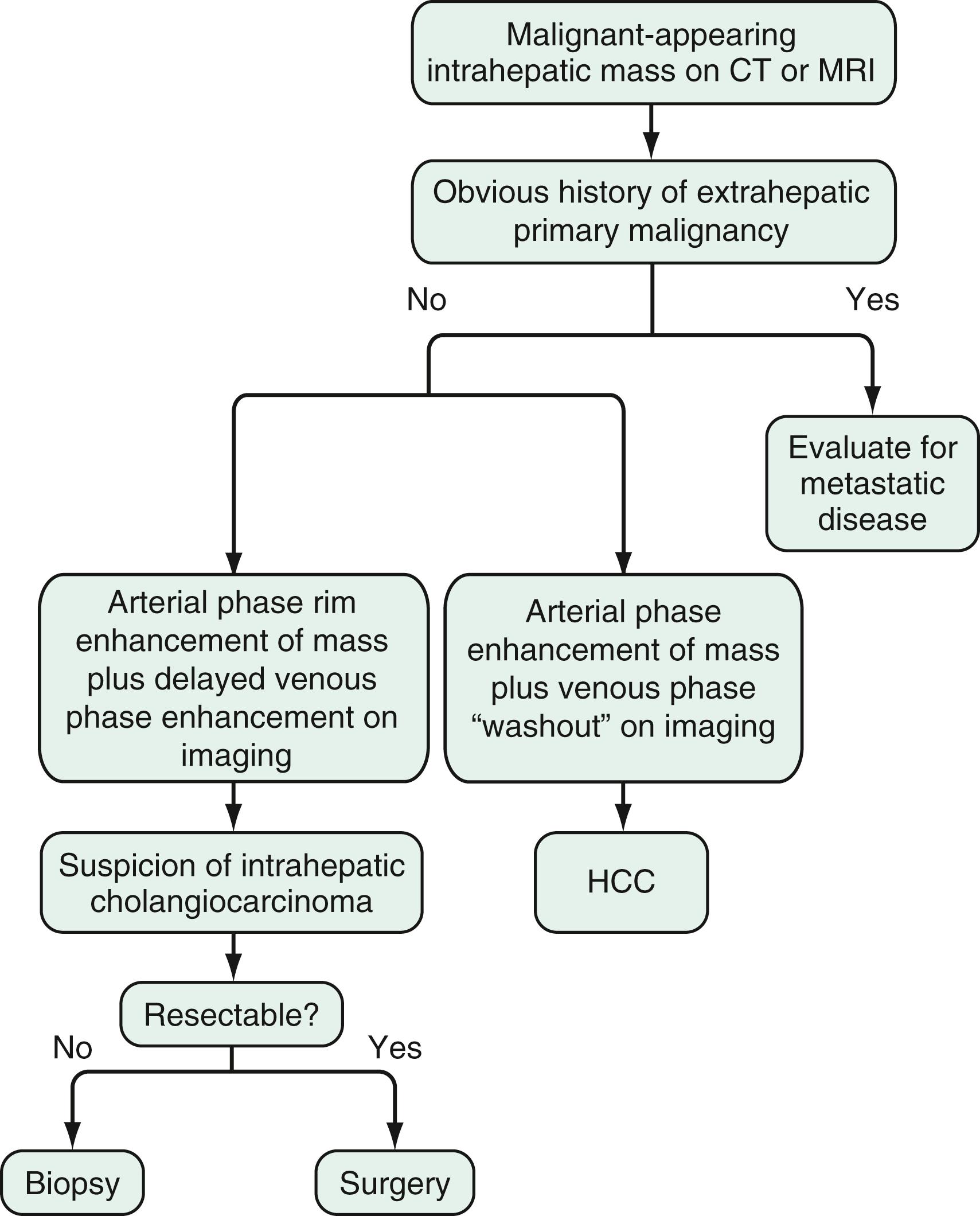
Intrahepatic cholangiocarcinomas manifest predominantly with abdominal pain and systemic symptoms such as cachexia, malaise, and fatigue. Perihilar and distal cholangiocarcinomas manifest in most cases with painless jaundice secondary to malignant biliary obstruction. In 10% of patients, bacterial cholangitis is the initial presenting symptom. An “atrophy-hypertrophy” complex can be documented by physical examination as palpable hypertrophy of the contralateral, unaffected lobe of the liver, with atrophy of the affected lobe as a result of vascular encasement and bile ductal obstruction.
Laboratory analysis may reveal evidence of obstructive cholestasis. In cases of indeterminate strictures, immunoglobulin G4 (IgG4) cholangiopathy should be ruled out serologically and with biliary samples (see Chapter 59, Chapter 68 ). The most commonly used serum tumor marker in cholangiocarcinoma is CA 19-9. In patients with PSC, the sensitivity and specificity of CA 19-9 for the diagnosis of cholangiocarcinoma are 79% and 98%, respectively, when the cutoff value is 129 U/mL. In patients without PSC, the sensitivity is 53% with a cutoff value of 100 U/mL. High levels of CA 19-9 levels (>1000 U/mL) have been associated with metastatic cholangiocarcinoma. It is important to note that patients who do not express Lewis antigen on red blood cells or in body fluids (approximately 5% to 10% of the general population) have undetectable serum CA 19-9 levels. Significant CA 19-9 elevations can occasionally be observed in patients with bacterial cholangitis and choledocholithiasis (see Chapter 65 ). Notably, up to one third of patients with an elevated serum CA 19-9 level do not have cholangiocarcinoma.
Intrahepatic cholangiocarcinoma often presents as a mass lesion in the liver. In the presence of cirrhosis, it can be challenging to differentiate intrahepatic cholangiocarcinoma from HCC. In the assessment of intrahepatic lesions more than 2 cm in diameter, dynamic CT and MRI ( Fig. 69.4 ) are helpful for distinguishing cholangiocarcinoma from HCC based on progressive contrast enhancement through the venous, arterial, and delayed venous phases in cholangiocarcinomas compared with the washout phenomenon observed in HCCs (see Chapter 96 ). An initial rim or peripheral arterial enhancement pattern in the early phases of dynamic CT or gadolinium-enhanced MRI with progressive centripetal enhancement in delayed phases is characteristic of intrahepatic cholangiocarcinoma. ,
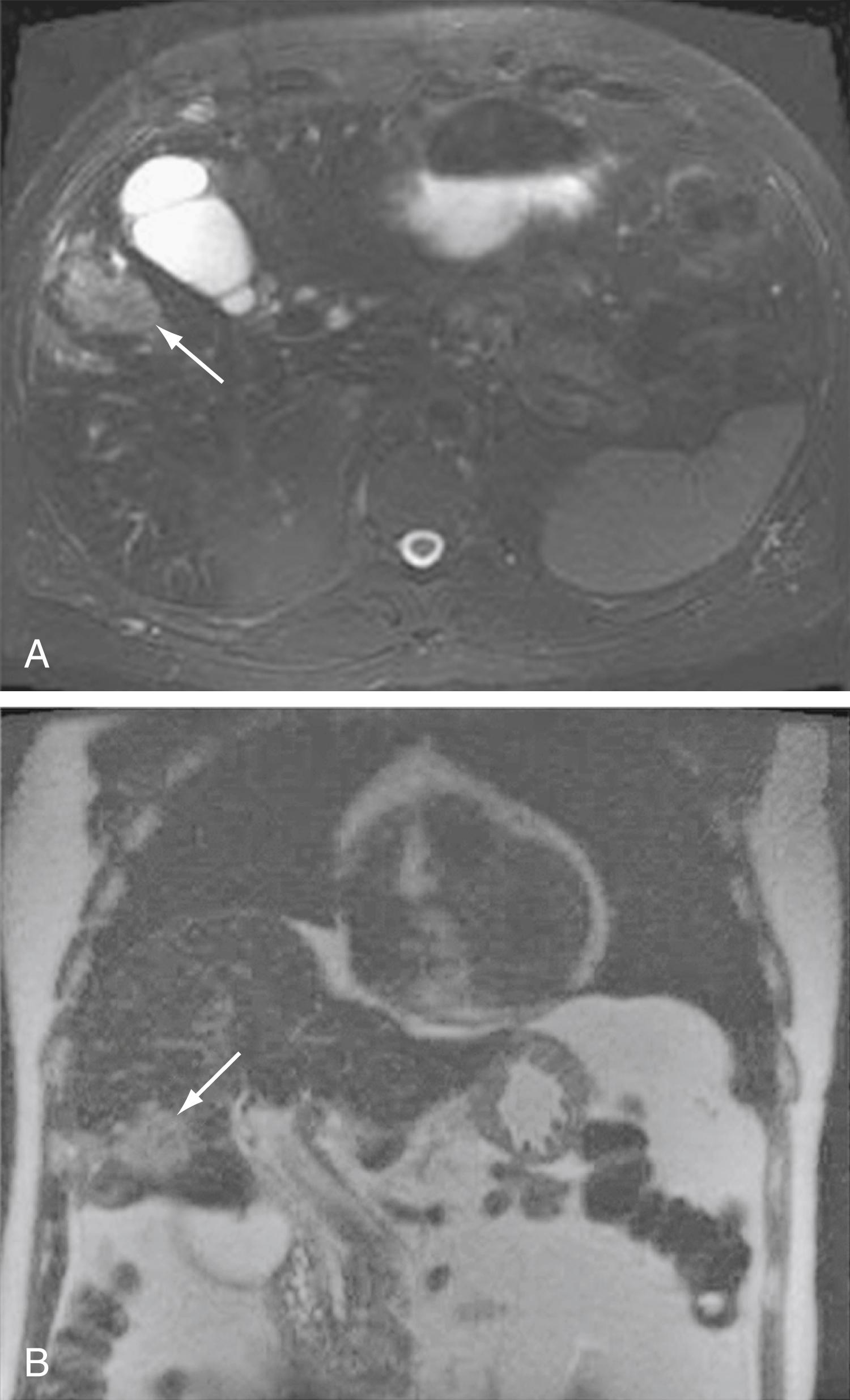
CT is used primarily for preoperative planning; it provides information on vascular and other anatomic structures, and its accuracy in the assessment of resectability is 60% to 88%. The sensitivity of CT for detection of nodal N2 metastases (see later) is only 54%. PET/CT has a sensitivity and specificity of 95% and 83%, respectively, in the assessment of intrahepatic cholangiocarcinomas. In intrahepatic cholangiocarcinoma, a tissue diagnosis should be limited to nonresectable cases because differentiation from HCC will affect the choice of therapy.
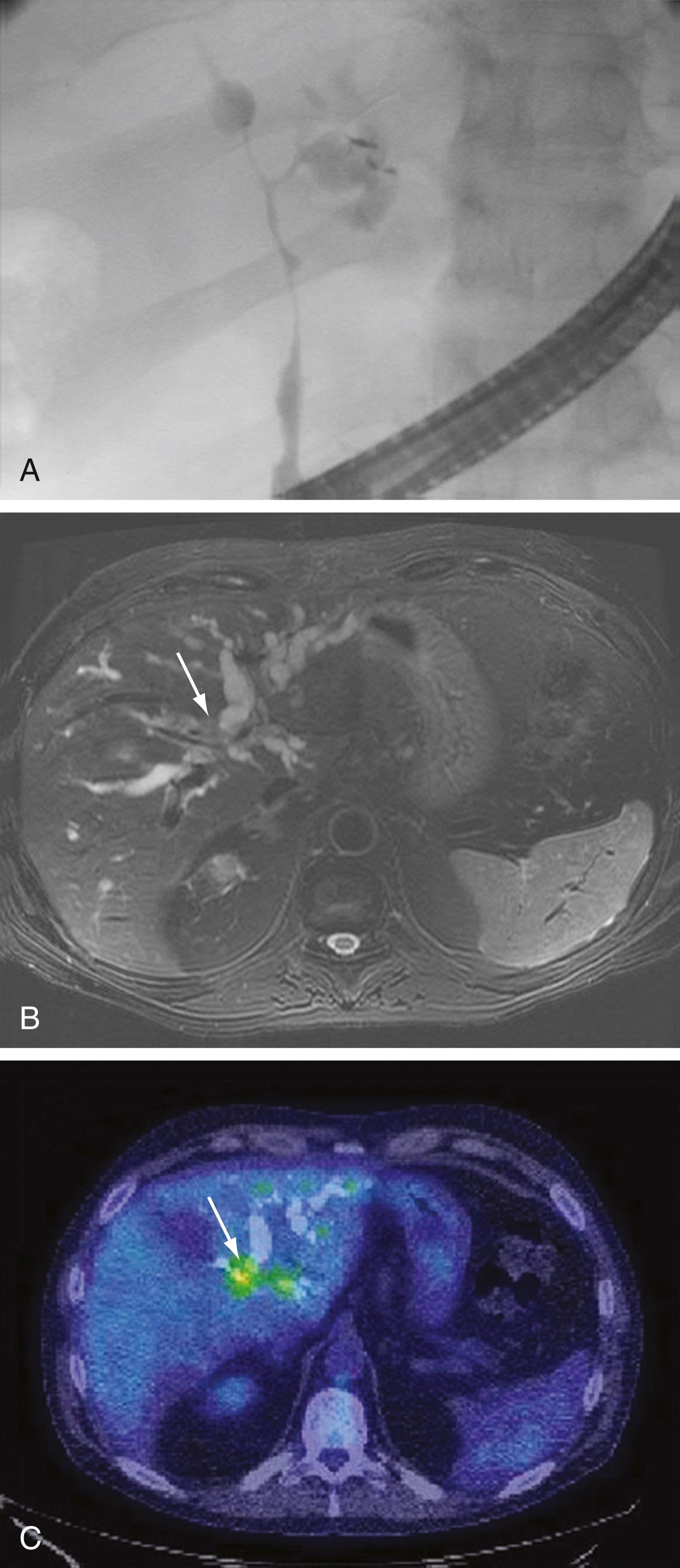
An essential diagnostic tool for perihilar and distal cholangiocarcinomas is cholangiography, which provides both anatomic information and material for a tissue diagnosis. Several approaches can be used, including ERCP, percutaneous transhepatic cholangiography (THC), and MRCP ( Fig. 69.5 ). The choice of ERCP or percutaneous THC depends on the location of a suspicious biliary stricture, local expertise, and accessibility of the stricture to the technique. Either ERCP or percutaneous THC provides information on intrabiliary tumor extension and allows cytologic sampling and therapeutic intervention for malignant biliary obstruction. Such interventions are not possible with MRCP, but this technique is noninvasive and provides additional information on the extent of the tumor, vascular encasement, the relation of the primary tumor to surrounding structures, and intra- as well as extrahepatic metastases. Cross-sectional imaging should be performed prior to cholangiography to help guide decisions regarding biliary drainage. MRI with MRCP is currently the best imaging technique for estimating the extent of biliary neoplastic invasion in perihilar cholangiocarcinoma, whereas CT is superior in detecting vascular enhancement and assessing resectability. For perihilar cholangiocarcinomas, the sensitivity and specificity of PET are only 69% and 67%, and the sensitivity for the detection of local lymph node metastases is only 13% to 38%. , False-positive PET results can be observed in the setting of inflammation. Therefore, its use in perihilar lesions should be limited to cases that are indeterminate with regard to extent and other features (including malignancy) after other studies have been performed. Mass lesions are unusual in distal cholangiocarcinomas, and frequently these tumors are grouped with perihilar cholangiocarcinomas in clinical studies; therefore, the same laboratory and imaging criteria should be applied as for perihilar cholangiocarcinoma.
EUS can contribute additional information about tumor dimension and anatomic location in relation to surrounding structures. Compared with CT or MRI, EUS has a higher tumor detection rate particularly in distal cholangiocarcinoma. In addition, EUS provides information about regional lymphadenopathy and allows sampling of lymph nodes by FNA. Biopsy of the primary lesion via EUS is highly discouraged, however, because of the potential for tumor cell spread, especially if LT is a therapeutic option.
As for most malignancies, a tissue diagnosis of cholangiocarcinoma is desirable; however, a pathologic diagnosis is challenging because of the tumor’s paucicellular character. Tissue is difficult to obtain, and cellular reactive changes resulting from inflammation often complicate the diagnosis. The most common technique for collecting tumor tissue in patients with perihilar and distal cholangiocarcinoma is brush cytology during ERCP. Although conventional cytology has a high specificity (100%), it is limited by low sensitivity (43%). Fluorescence in situ hybridization (FISH) has been demonstrated to increase the sensitivity and specificity of cytology for diagnosing cholangiocarcinoma in patients with and without PSC. Because chromosomal instability is a hallmark of cancer, the FISH assay is used for the assessment of chromosomal aneusomy (gains or losses of chromosomes) via the use of fluorescently labeled DNA probes. An optimized FISH probe set (targeting the 1q21, 7p12, 8q24, 9p21 loci) has enhanced sensitivity and specificity (93% and 100%, respectively) for the detection of pancreaticobiliary malignancies.
Newer techniques for diagnosing cholangiocarcinoma include cholangioscopy, intraductal US, and confocal laser endomicroscopy. A limited number of prospective studies in a small number of patients have demonstrated an improvement in diagnostic accuracy with cholangioscopy-directed biopsy compared with biliary brushings of indeterminate strictures. Therefore, these techniques are not yet established in routine practice and should be reserved for cases that remain unconfirmed after use of the aforementioned methods when the level of suspicion for cholangiocarcinoma is high.
Staging systems of malignancies aim to provide guidance in prognostication and treatment. Several staging systems exist for intrahepatic, perihilar, and distal cholangiocarcinomas; unfortunately, none of these has proved to be optimal. A modification was introduced in the American Joint Committee on Cancer (AJCC)/the Union for International Cancer Control (UICC) TNM system in 2009, by assigning a separate staging system to each cholangiocarcinoma type. , New and promising staging systems have been developed but require validation. ,
Independent prognostic factors for intrahepatic cholangiocarcinomas include the number of tumors, vascular invasion, and lymph node metastases. Three staging systems exist for intrahepatic cholangiocarcinoma and differ primarily in their T-staging approach: (1) the AJCC/UICC TNM staging system, (2) the Liver Cancer Study Group of Japan staging system, and (3) the National Cancer Center of Japan staging system. Of these 3 staging systems, only the AJCC/UICC TNM staging system ( Table 69.2 ) has shown a correlation between stage and survival, but the system is limited by its requirement for a tissue diagnosis for the Tis (carcinoma in situ) and T4 stages. ,
| TNM Stage | Criteria |
|---|---|
| Tx | Primary tumor cannot be assessed |
| T0 | No evidence of primary tumor |
| Tis | Carcinoma in situ (intraductal tumor) |
| T1a | Solitary tumor ≤5 cm without vascular invasion |
| T1b | Solitary tumor >5 cm without vascular invasion |
| T2 | Solitary tumor with vascular invasion OR multiple tumors, with or without vascular invasion |
| T3 | Tumor perforating the visceral peritoneum |
| T4 | Tumor involving the local extrahepatic structures by direct invasion |
| Nx | Regional lymph nodes cannot be assessed |
| N0 | No regional lymph node metastases |
| N1 | Regional lymph node metastases present |
| M0 | No distant metastases |
| M1 | Distant metastases |
| AJCC/UICC Stage | Tumor | Node | Metastasis |
|---|---|---|---|
| 0 | Tis | N0 | M0 |
| IA | T1a | N0 | M0 |
| IB | T1b | N0 | M0 |
| II | T2 | N0 | M0 |
| IIIA | T3 | N0 | M0 |
| IIIB | T4 | N0 | M0 |
| Any T | N1 | M0 | |
| IV | Any T | Any N | M1 |
Independent prognostic factors for perihilar cholangiocarcinomas include tumor differentiation, lymph node metastases, and negative (R0) surgical resection margins. Four major staging systems for perihilar cholangiocarcinoma are the Bismuth-Corlette classification, , the Memorial Sloan-Kettering Cancer Center (MSKCC) staging system, the AJCC/UICC TNM staging system, and the Mayo Clinic staging system. The Bismuth-Corlette classification was developed for guiding surgical treatment and is not a staging system; it does not provide prognostication and is not designed to correlate with surgical outcomes. Although the MSKCC staging system ( Table 69.3 ) showed a correlation between T stage and R0, N2, and M1 status, it was not able to distinguish resectable from unresectable disease reliably. Unlike the other 3 systems that use operative information, the Mayo Clinic staging system ( Table 69.4 ) is based on nonoperative information at the time of diagnosis. Therefore, the Mayo Clinic staging system can be used to predict survival in patients with unresectable disease. Moreover, this system had better prognostic performance, demonstrated by greater concordance statistics, compared with the AJCC/UICC system ( Table 69.5 ). Independent prognostic factors for distal cholangiocarcinomas include the depth of tumor invasion; lymph node metastasis; perineural, microscopic vascular, and pancreatic invasion; and R0 resection. Lymph node metastases are more commonly observed with this type of cholangiocarcinoma than with the others. The AJCC/UICC TNM staging system is the only staging system for distal cholangiocarcinomas ( Table 69.6 ). As outlined earlier, the separation of perihilar from distal cholangiocarcinomas is an improvement over previous versions, as is the redefinition of the T stages. This staging system provides prognostic information, which requires validation, but does not guide management.
| Stage | Criteria |
|---|---|
| T1 | Tumor involving biliary confluence ± unilateral extension to secondary radicles |
| T2 | Tumor involving biliary confluence ± unilateral extension to secondary radicles AND Ipsilateral portal vein involvement ± ipsilateral hepatic lobe atrophy |
| T3 | Tumor involving biliary confluence + bilateral extension to secondary radicles OR Unilateral extension to secondary radicles + contralateral portal vein involvement OR Main or bilateral portal vein involvement |
| Stage | Criteria |
|---|---|
| I | Unicentric mass ≤3 cm ECOG performance status 0 Serum CA 19-9 level (U/mL) <1000 No vascular encasement No metastasis |
| II | Unicentric mass ≤3 cm ECOG performance status 1-2 Serum CA 19-9 level (U/mL) <1000 Vascular encasement present No metastasis |
| III | Unicentric mass >3 cm or multicentric ECOG performance status 0-2 Serum CA 19-9 level (U/mL) ≥1000 Lymph node metastasis present |
| IV | ECOG performance status 3-4 Peritoneal (or other organ) metastasis present |
| TNM Stage | Criteria |
|---|---|
| Tx | Primary tumor cannot be assessed |
| T0 | No evidence of primary tumor |
| Tis | Carcinoma in situ |
| T1 | Tumor confined to the bile duct, with extension up to the muscle layer of fibrous tissue |
| T2a | Tumor invades beyond the wall of the bile duct to surrounding adipose tissue |
| T2b | Tumors invades adjacent hepatic parenchyma |
| T3 | Tumor invades unilateral branches of the portal vein or hepatic artery |
| T4 | Tumor invades main portal vein or its branches bilaterally OR Tumor invades the common hepatic artery OR Tumor invades unilateral second-order biliary radicles, with contralateral portal vein or hepatic artery involvement |
| Nx | Regional lymph nodes cannot be assessed |
| N0 | No regional lymph node metastases |
| N1 | Regional lymph node metastases (≤3 lymph nodes) (includes hilar, cystic duct, common bile duct, hepatic artery, posterior pancreatoduodenal, and portal vein lymph nodes) |
| N2 | Regional lymph node metastases (≥4 lymph nodes) |
| M0 | No distant metastases |
| M1 | Distant metastases |
| AJCC/UICC Stage | Tumor | Node | Metastasis |
|---|---|---|---|
| 0 | Tis | N0 | M0 |
| I | T1 | N0 | M0 |
| II | T2a-b | N0 | M0 |
| IIIA | T3 | N0 | M0 |
| IIIB | T4 | N0 | M0 |
| IIIC | Any T | N1 | M0 |
| IVA | Any T | N2 | M0 |
| IVB | Any T | Any N | M1 |
| TNM Stage | Criteria |
|---|---|
| Tx | Primary tumor cannot be assessed |
| Tis | Carcinoma in situ (intraductal tumor) |
| T1 | Tumor invades the bile duct wall with a depth <5 mm |
| T2 | Tumor invades the bile duct wall with a depth of 5-12 mm |
| T3 | Tumor invades the bile duct wall with a depth >12 mm |
| T4 | Tumor involves the celiac axis, superior mesenteric artery, and/or the common hepatic artery |
| Nx | Regional lymph nodes cannot be assessed |
| N0 | No regional lymph node metastases |
| N1 | Regional lymph node metastases (≤3 lymph nodes) |
| N2 | Regional lymph node metastases (≥4 lymph nodes) |
| M0 | No distant metastases |
| M1 | Distant metastases |
| AJCC/UICC Stage | Tumor | Node | Metastasis |
|---|---|---|---|
| 0 | Tis | N0 | M0 |
| I | T1 | N0 | M0 |
| IIA | T1 | N1 | M0 |
| IIA | T2 | N0 | M0 |
| IIB | T2 | N1 | M0 |
| IIB | T3 | N0-1 | M0 |
| IIIA | T1 | N2 | M0 |
| IIIA | T2-3 | N2 | M0 |
| IIIB | T4 | N0-2 | M0 |
| IV | Any T | Any N | M1 |
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here